Abstract
Background and objectives:
Parkinson’s disease (PD) patients experience several non-motor symptoms from the gastrointestinal tract that may partly be caused by parasympathetic deficiency. The pancreas is densely innervated by the vagus nerve, which mediates early meal-induced secretion of pancreatic polypeptide (PP). Early secretion after sham feeding has been validated as a marker of vagal integrity. Thus, the aim was to evaluate the ratio of increased PP plasma levels after sham feeding in PD and correlate findings with gastrointestinal transit time (GITT).
Methods:
Twenty-five PD patients and 17 controls were included. PP, insulin, and blood glucose levels were measured before, during, and after sham feeding with white bread and chocolate spread. GITT was measured using radiopaque markers. Furthermore, faeces samples were analyzed for pancreatic elastase enzyme as a marker of exocrine pancreatic function.
Results:
PD patients showed significantly lower PP ratio levels after sham feeding, which was most pronounced at 10 minutes. No significant association was seen between attenuated PP response and GITT in PD patients. No between-group differences were seen in glucose or insulin levels over time, but PD patients showed generally lower insulin levels compared to controls. No difference was found in faeces pancreatic elastase.
Conclusions:
Early-to-moderate stage PD patients demonstrated significantly decreased PP response after sham feeding suggestive of vagal denervation.
INTRODUCTION
Most patients with Parkinson’s disease (PD) experience a range of non-motor symptoms (NMS) from the gastrointestinal (GI) tract, which are in part caused by decreased parasympathetic function [1, 2]. Lewy pathology has been detected in the vagus nerve, the dorsal motor nucleus of the vagus, and the gastrointestinal tract of PD patients [3]. The disorder has been proposed to initiate in the olfactory epithelium and autonomic nerve endings of the GI tract with subsequent spreading via the vagus nerve to the dorsal motor nucleus of the vagus [4].
The pancreas is densely innervated by the parasympathetic vagus nerve and Lewy pathology has been detected in the pancreas [3]. Using the acetylcholinesterase PET ligand 11C-donepezil, we recently demonstrated that early-to-moderate disease stage PD patients displayed decreased pancreatic PET signal, which may be suggestive of parasympathetic denervation [5]. The early secretion of the hormone pancreatic polypeptide (PP) after meal ingestion is mediated by the vagus nerve (= cephalic phase), as opposed to the subsequent gastrointestinal phase, which is only partially mediated by vagal activity [6, 7]. Thus, early cephalic phase PP secretion after a chew-and-spit sham-feeding procedure has been validated as a marker of vagal integrity. This early PP response is markedly attenuated in vagotomized patients [6–8]. Since the dorsal motor nucleus of the vagus is known to be significantly damaged in PD, we hypothesized that PD patients would have an attenuated cephalic phase PP peak in response to sham feeding.
The study aim was to evaluate plasma PP concentration during sham feeding in PD patients and healthy controls (HC), as a marker of pancreatic parasympathetic denervation. In the PD group, correlation analyses were performed between PP levels and subjective constipation as defined by the non-motor symptoms questionnaire (NMSQuest), and with gastrointestinal transit time (GITT) measured by radiopaque markers. The exocrine pancreatic function in PD was evaluated by measurements of faeces-elastase
MATERIALS AND METHODS
Ethics statement
The study was approved by the Central Denmark Region Committee on Health Research Ethics (No. 1-10-72-405-14). All participants provided written informed consent.
Subjects
Twenty-five PD patients and 17 healthy control subjects without neurological disorders were included. PD patients were diagnosed according to UK Brain Bank criteria [9]. Clinical disease stage and motor function were evaluated using the Hoehn and Yahr scale (H&Y) [10] and MDS Unified Parkinson’s Disease Rating Scale part III (UPDRS III) [11]. Study exclusion criteria were: diabetes and other endocrine disorders, prior gastrointestinal and pancreatic surgery, significant medical conditions, including gastrointestinal disorders, and renal, heart, or liver failure, psychiatric disease, and substance abuse. Constipation was not an exclusion criterion. The NMSQuest was used to evaluate NMS in general, including constipation defined as <3 bowel movements per week and/or straining during the past month [12]. Objective intestinal function was evaluated with the radio opaque marker (ROM) technique in 24 of 25 patients and 15 of 17 controls, and a cut-off score of 23 retained markers was used for separating subjects with and without delayed intestinal transit time. The methodology and a subset of ROM data were published recently [13].
All PD patients received anti-parkinsonian medication (see online supplementary information for details). Median levodopa equivalent daily dose (LEDD) was 600 mg (range 180–1750 mg) [14].
The UPDRS-III was performed after >12 hours of parkinsonian medication abstinence, and the study protocol on usual parkinsonian medication.
Test meal
A test meal consisting of white bread with chocolate spread (Nutella™) was used for sham feeding after an overnight fast of >12 hours. A chew-and-spit procedure was performed during the initial 5 minutes of the study protocol (i.e. from 0 to 5 min.), and participants were carefully instructed to avoid swallowing. During and after sham feeding the participants rinsed the oral cavity with water.
Blood and faeces analyses
Blood samples were collected in chilled EDTA tubes and centrifuged at 2013G for 10 min. Plasma was stored at –20°C for PP analyses. Serum PP and insulin levels were determined at 0 min. (baseline), 2.5, 5, 7.5, 10, 15, and 20 min., and plasma glucose at 0, 5, 10, 15, and 20 min. Furthermore, faeces samples were collected and analyzed for pancreatic elastase enzyme levels as a marker of exocrine pancreatic function. Plasma HbA1c baseline concentration was determined as a marker of mean long-term blood glucose level.
PP analysis
Plasma samples were extracted with ethanol before analysis (70% final concentration). PP was measured by radioimmunoassay using a mid-region specific antibody, code no HYB 347-07 (Statens Serum Institut, Denmark) as described previously [15]. Human PP was used for standard, and the tracer was 125I-labeled human PP, cat. no. NEX315 (Perkin Elmer, Boston, USA).
Statistics
Prism 6 (GraphPad Software, La Jolla, USA) was used for statistical analyses. Unpaired two-way t-tests or equivalent non-parametric tests were used to test demographic and clinical data. Differences in gender and percentage of constipation presence were evaluated by Fisher’s exact test. Between-group differences in plasma PP concentration ratio (each time point divided by 0 min. baseline concentration), plasma glucose, and serum insulin concentrations at 0–20 min. as well as PP ratios in constipated versus non-constipated PD patients were analyzed by two-way repeated measures ANOVA with post hoc analyses (Sidak). The cumulative PP increase (delta) was calculated as area under the curve (AUC) from 5 to 15 min. [16]. A post hoc receiver-operating characteristic (ROC) analysis was performed to determine the optimal PP ratio cut-off threshold for separating PD patients from controls.
RESULTS
Demographic and clinical data are shown in Table 1. No between-group differences were seen in HbA1c and faeces elastase concentration (Table 1). PD patients experienced significantly more NMS including constipation, evaluated by NMSQuest (Table 1).
Demographic and clinical data of patients with Parkinson’s disease (PD) and healthy controls
Data listed as mean ± SD or median (range). Abbreviations: BMI = Body Mass Index, MDS-UPDRS = Movement Disorder Society Unified Parkinson’s Disease Rating Scale, HbA1c = HaemoglobinA1C, NMSQuest = Non-Motor Symptoms Questionnaire.
PP
Compared to controls, PD patients showed significantly attenuated PP fractional increase after sham feeding (p = 0.015; Fig. 1A), and significantly reduced delta PP AUC (5–15 min.) (Table 1). The between-group mean difference was most pronounced at 10 minutes (p = 0.040; Fig. 1B). Based on ROC analyses, a cut-off PP ratio of 1.05 at 10 minutes yielded a sensitivity of 68%, and specificity of 71% for separating PD patients and controls (Fig. 1B; dashed line). No between-group differences were seen in glucose levels over time (p = 0.084), or in insulin levels over time (p = 0.072; Fig. 1C), although PD patients showed generally lower insulin levels compared to controls (p = 0.012).
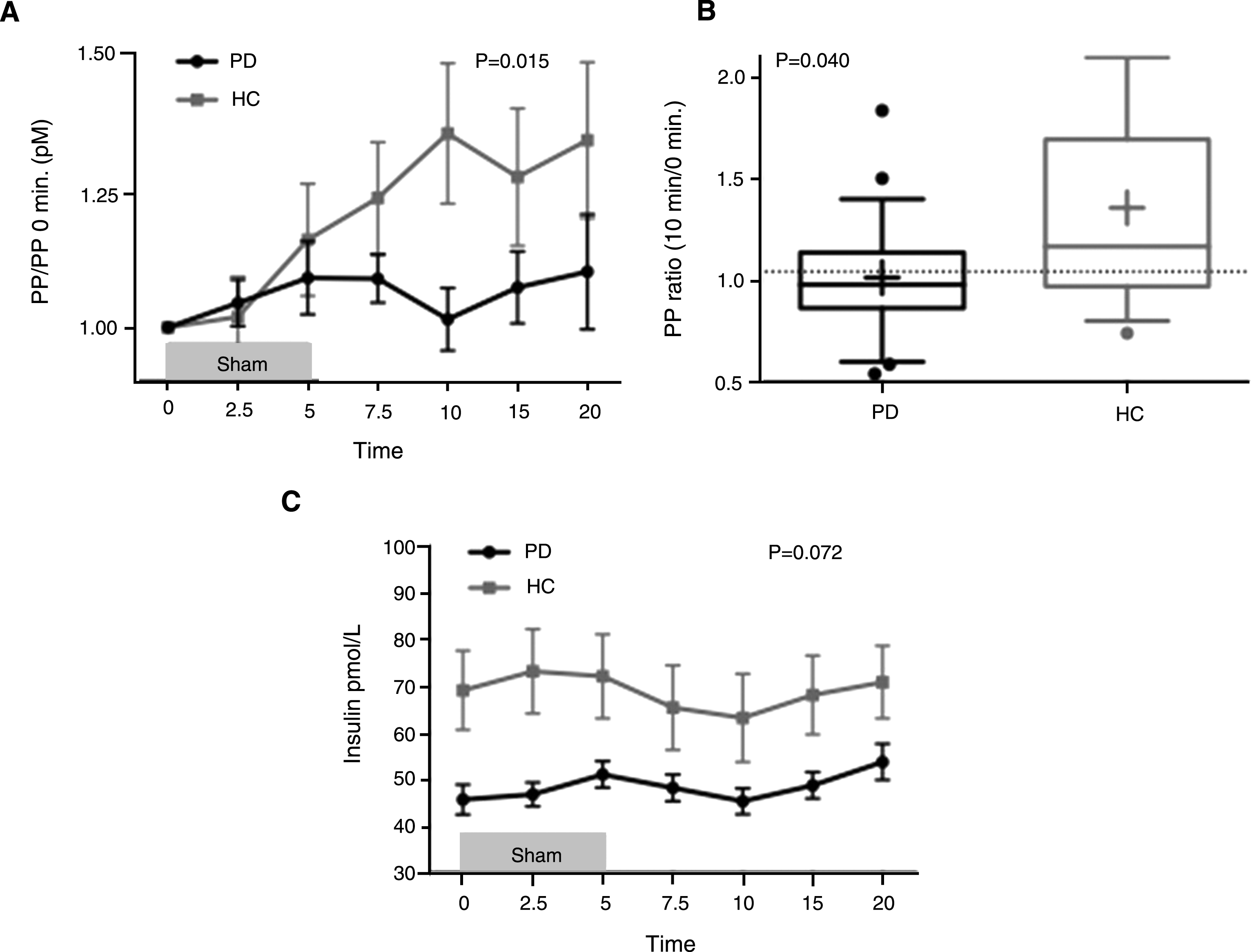
(A) Mean±SEM pancreatic polypeptide (PP) concentration ratio at time 0–20 min. during and after sham feeding 0–5 min. (each time point divided by 0 min. concentration) in PD patients and healthy controls (HC). (B) PP at 10 min. /PP baseline 0 min. Box-and-whiskers plot (median, interquartile range; + denotes mean). Dashed line signifies cut-off value of 1.05 (sens: 68%, spec: 71%). (C) Mean±SEM insulin concentration at time 0–20 min. during and after sham-feeding 0–5 min.
No difference was seen in PP ratio levels when comparing PD patients with and without subjective constipation (p = 0.873). Also, no significant difference in PP ratio levels was seen between PD patients with and without prolonged GITT. However, patients with prolonged GITT, as defined by >23 retained ROM, showed a slight tendency towards a more pronounced attenuated PP response (Fig. 2A). The difference in PP ratio was most notable at 10 min. (Fig. 2B, uncorrected two-way t-test p = 0.011; ANOVA with Sidak’s correction p = 0.186). Although this result was based on small subgroups of patients and should be interpreted with caution, it would nevertheless be interesting to further explore this potential relationship in future studies.
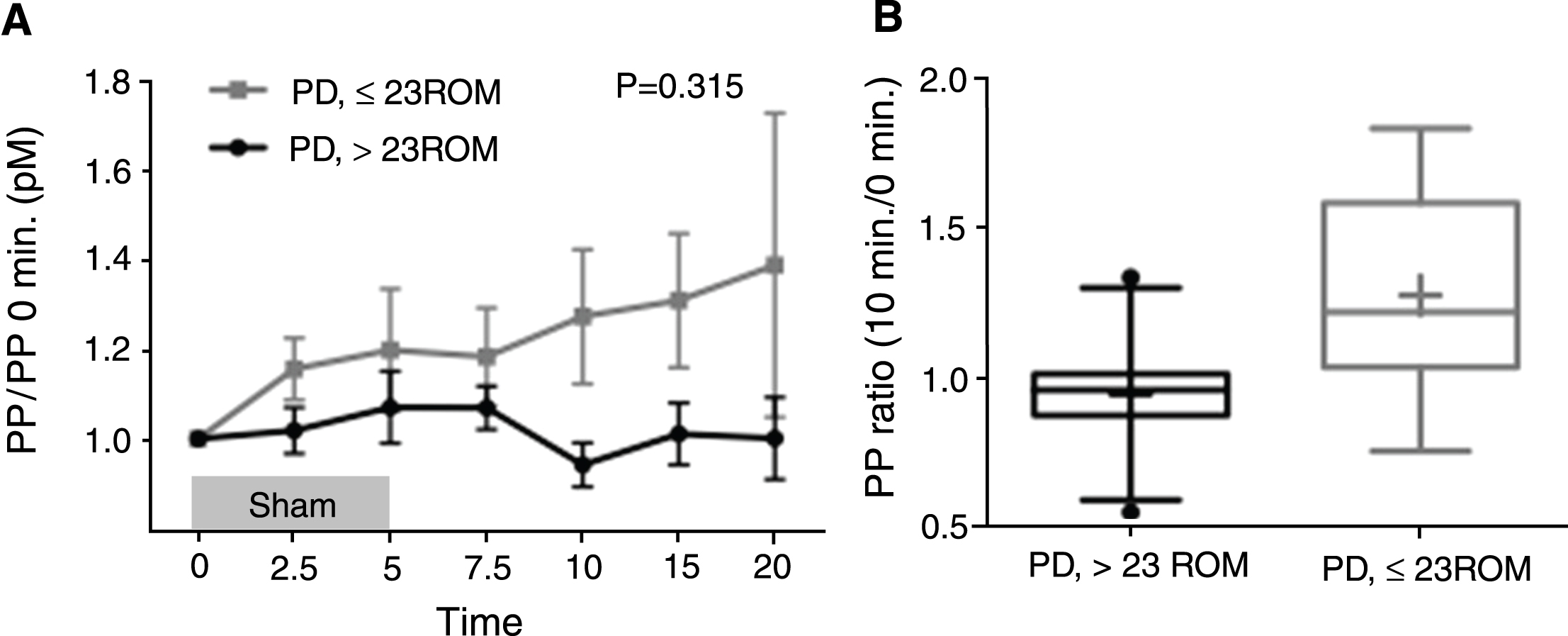
(A) Mean±SEM Pancreatic Polypeptide (PP) concentration ratio at time 0–20 min. during and after sham-feeding 0–5 min. (each time point divided by 0 min. concentration) in PD patients with and without delayed gastrointestinal transit time defined by a retention cut-off of 23 radio opaque markers (ROM). (B) PP at 10 min. /PP baseline 0 min. Box-and-whiskers plot (median, interquartile range; + is mean).
There was no difference in LEDD in PD subgroups with and without prolonged GITT (740±452 mg vs. 422±264 mg; p = 0.151). The PP response at 10 min. showed no correlation with disease duration (p = 0.896) or UPDRS-III score (p = 0.221).
DISCUSSION
In the present study, significantly lower cephalic phase PP ratio levels were detected in PD patients after sham feeding, suggestive of compromised pancreatic parasympathetic innervation. Also, at all time points lower insulin levels were seen in the PD group. No group-difference was seen in faeces-elastase levels, indicating that exocrine pancreatic function was not significantly affected in this sample of PD patients.
PP
It has been reported that 82% of vagotomized patients showed markedly attenuated PP response after sham feeding, whereas 93% of healthy controls had a normal response [7]. These findings support PP measurements as a useful marker of vagal integrity. A previous study investigated postprandial PP levels in PD patients and patients with REM sleep behavior disorder (RBD), and found no deficit in PP response compared to controls [17]. However, several aspects of that study differed from the current study protocol. First, PP was evaluated subsequent to meal ingestion and not sham feeding. An earlier study of vagotomized patients reported significant decreases in cephalic phase PP levels after sham feeding, but not after food intake [18]. This underscores that sham feeding probably provides the most robust stimulus to elicit a well-defined vagus-derived cephalic PP response. Second, the test meal in the study by Unger et al. was consumed over 10 minutes, and blood samples obtained before, immediately after, and 15 minutes after meal consumption [17]. Given this relatively poor temporal resolution of PP measurements, the early cephalic phase peak may have been missed or perhaps escaped detection, as this peak is of significantly lower magnitude compared to the subsequent gastrointestinal response, and also the two phases are known to overlap [16, 19]. These points may explain the discrepant results of the present study and Unger et al.
A difference was seen in baseline PP plasma concentration between PD patients and controls (Table 1). The significance of this finding is unclear, and to our knowledge baseline values have not been validated as a measure of vagal function. Also, the data exhibit a wide within-group range indicating presence of a large biological variation. However, a previous study found somewhat decreased median baseline PP levels in vagotomized patients as compared to control subjects, although not significant (p = 0.13) [7]. Also, Tong et al showed that gender and body fat composition can influence baseline PP levels [20], which however does not explain the findings in the present study, as no between-group differences were seen in gender and BMI.
It is known that gustatory stimuli of the oral cavity is carried via cranial nerve afferents (VII and IX) to the nucleus tractus solitarius (NTS), and subsequently to the dorsal motor nucleus of the vagus (DMV) [21]. Thus, in theory PP secretion could be decreased due to damage to these upstream systems rather than damage to the DMV per se. It has been suggested that gustatory function may be affected even at early stage PD [22], but only few studies have published data on objective gustatory testing, and the results are somewhat contradictory [23]. Moreover, the DMV consistently shows more Lewy type pathology compared to the NTS [24, 25], suggesting that damage to the efferent system is the main contributing factor to the attenuated PP response.
Parasympathetic denervation can also be investigated by means of heart rate variability (HRV) measures. Several HRV studies in PD patients have been performed, but the results have been somewhat unclear [5, 27]. In the present proof-of-concept study, HRV data was not obtained, but it would be relevant to correlate PP ratio levels with HRV measurements in future studies to establish a more firm indication of parasympathetic involvement.
The dorsal motor nucleus of the vagus, the vagus nerve itself, and the parasympathetic sacral part of the intermediolateral cell column all show marked pathology in autopsy studies of PD patients [3, 29]. However, it remains unclear how this neuropathological evidence of parasympathetic damage relates to the presence and severity of non-motor symptoms including constipation. Objective markers of colonic transit time and subjective constipation symptoms are only moderately correlated, and different constipation questionnaires do not define the same patients as being constipated [13]. In the present study, we detected no difference in PP response levels, when comparing subjectively constipated and non-constipated patients, as defined by the NMSQuest. Interestingly, a minor trend towards more pathological PP response was seen in PD patients with prolonged gastrointestinal transit time, although the finding was not significant. It should be noted, however, that GI motility is mediated not only by the vagus nerve but also the enteric nervous system. Thus, decreased motility is probably related to disruption of both the vagal and sacral parasympathetic as well as the enteric innervation. Nevertheless, this observation suggests that the degree of parasympathetic dysfunction, as defined by the validated PP-test, could be one of several underlying causes of colonic dysmotility in PD. Interestingly, a previous heart rate variability study reported that subjectively constipated PD patients displayed significantly attenuated high-frequency variation in heart rate in response to deep breathing compared to non-constipated PD patients, which also seems to support that parasympathetic dysfunction may be an underlying cause of colonic dysmotility [30].
We have recently studied 11C-donepezil PET/CT as another potential objective marker of parasympathetic denervation, and found that the majority of early-to-moderate stage PD patients (disease duration 4.5 years) display decreased 11C-donepezil PET signal in the pancreas [5]. In contrast, a group of newly diagnosed PD patients (duration 1.5 years) exhibited intact pancreatic 11C-donepezil PET signal at the level of matched controls [31]. This suggests that vagal denervation occurs gradually after motor symptoms appear. However, it is possible that 11C-donepezil PET is a less sensitive marker of vagal integrity as compared to the sham feeding PP test. Future studies are needed to investigate the correlation between PP response and 11C-donepezil PET signal in the pancreas in PD patients at different disease stages.
There is some evidence to suggest that treatment with dopaminergic drugs may influence the PP response [32]. In a case study of a single patient with an islet cell tumor and prolactinoma, decreased PP levels were detected subsequent to dopamine agonist treatment. In contrast, Witteman et al. reported PP decrease after insulin-induced hypoglycemia in PD patients both with and without levodopa treatment [33]. Other studies found increased fasting PP levels after dopamine agonist treatment and decreased PP meal-response after dopamine infusion in normal individuals [34, 35]. In general, due to methodological differences in these studies and the absence of PP data in PD patients obtained with the sham-feeding technology, the potential influence of dopaminergic treatment on PP responses in PD remains unclear. Interestingly, the current PD subgroup with prolonged GITT also tended towards a more severe reduction in cephalic PP peak, but this PD subgroup did not differ in LEDD compared to the PD subgroup without prolonged GITT. This preliminary finding suggests that the cephalic PP response is not markedly attenuated by dopaminergic medication.
Some studies have reported higher magnitude of PP ratio levels and percentage increase compared to the present data [7, 33]. This could be caused by methodological differences in the type of food stimulus used and especially in the PP analysis methodology. In the present study, we used bread with chocolate spread, since sweet, palatable foods rich in both fat and sugar provide the most potent stimulus for eliciting robust PP responses in a sham-feeding paradigm [16]. Yet, it cannot be ruled out that other food stimuli might have resulted in a cleaner separation between the PD patients and control subjects. Methodological studies with varying food stimuli in PD patients are needed to address this question. Regarding assay methodology our assay includes a plasma extraction step, which was included in order to avoid non-specific matrix effects, and this may explain the lower values reported here.
Insulin and glucose
Several studies have shown that a large fraction of PD patients exhibit decreased glucose tolerance and increased prevalence of diabetes mellitus compared to control subjects [36]. It has been proposed that diabetes may be associated with an increased risk of PD development [36], but it could also be speculated that disturbances in glucose and insulin regulation are early signs of vagal denervation in the prodromal stage of PD.
In the present study, no significant difference was seen in plasma glucose levels between PD and controls. The insulin levels were generally lower in PD patients at all time points, but no difference was seen in the insulin curve shape between the groups. The underlying cause for lower insulin levels in PD patients is unclear, but a previous study reported attenuated insulin response to a glucose tolerance test after truncal vagotomy compared to selective and highly selective vagotomy [37]. Those authors speculated that the decrease in insulin response may be caused by decreased vagal innervation of both the pancreas and the small intestine, the latter leading to decreased secretion of intestinal hormones regulating the insulin response. It has been reported that efferent vagal stimulation increases glucagon levels, whereas atropine decreases glucagon levels [38, 39]. Thus, it is possible that decreased glucagon levels may contribute to an increase in insulin sensitivity in PD patients, although this was not tested in the present study.
Limitations
First, a vagotomized control group was not included. However, the currently used PP assay has already been validated in a cohort of vagotomized control subjects, most of whom showed markedly attenuated cephalic PP response to sham feeding [8]. Second, dopaminergic treatment was not withdrawn in the PD group. Future studies are needed to address the influence of dopaminergic medication on PP secretion in PD. Third, data on gustatory function and presence of Lewy body pathology in oral cavity tissue and cranial nerve VII and IX are scarce. Nevertheless, it cannot be ruled out that decreased PP response in PD may be caused partly by afferent nerve damage from the tongue to the NTS. Fourth, faeces-elastase level is only a moderately sensitive marker of exocrine pancreatic function, so the present results do not prove that exocrine pancreatic function is normal in PD. Fifth, most of the PD patients were H&Y II, and it would be interesting to examine PP responses in both later and earlier stages, including the prodromal phase.
Conclusion
Significantly decreased cephalic phase PP response after sham feeding was demonstrated in PD patients compared to controls. This finding suggests that functionally important vagal denervation is present in early-to-moderate stage PD patients. No group differences were seen in glucose- and insulin responses to sham feeding, but PD patients showed overall lower levels of plasma insulin. Further longitudinal studies of cephalic phase PP responses in PD patients are required to follow vagal denervation of the pancreas over time.
CONFLICT OF INTEREST
Karen Østergaard serves as a consultant to Medtronic Inc. and has received honoraria from Medtronic Inc. and AbbVie outside the submitted work. Dr. Østergaard also has grants from the Lundbeck Foundation, the Danish Research council for Independent Research, and the Danish Parkinson Association during the conduct of the study. Klaus Krogh serves on the advisory boards of Coloplast, Denmark; Wellspect, Sweden; Nordic Lifesciences, Canada; and Almirall, Denmark. Per Borghammer serves as a consultant to F. Hoffman-La Roche and receives Aarhus University PhD salaries. All other authors have no conflicts of interest to report.
