Abstract
Background:
Gait disturbances in Parkinson’s disease (PD) are highly disabling and poorly responsive to drugs, especially in advanced stages. While the efficacy of a treadmill training based on external feedback and cues (treadmill-plus) on gait disturbances in early PD stages is demonstrated, no definitive evidence exists about advanced stages.
Objective:
We aimed to evaluate the feasibility and the effects of a treadmill-plus training on gait disturbances in advanced PD.
Methods:
Two hundred and six PD patients from medium to more advanced Hoehn & Yahr stage (stage 2, n = 79, stage 3 n = 74, and stage 4 = 53) who underwent a 4-week treadmill-plus training, were retrospectively identified. All patients were able to walk on a treadmill for one minute at 1.5 km/h, without support. Feasibility was evaluated by measuring safety, adverse events, and attrition rate. The effects of treatment were evaluated by assessing, both at enrolment and at the end of treatment, the on-land, self-paced 6-minute Walking Test (6MWT) and the gait parameters obtained from the treadmill during a 1.5 km/h trial.
Results:
All patients completed the treadmill-plus training and no adverse events were recorded, even among more disabled patients. After training, we observed a significant improvement in the 6MWT, an increase in step length and a reduction of cadence and step variability in the whole sample. After stratifying patients according to disease stage, we found that patients in more advanced stages experienced the same improvements in all gait parameters as patients in less advanced stages.
Conclusions:
Treadmill-plus training is well tolerated and may have a positive impact on many aspects of gait in more advanced PD stages.
INTRODUCTION
Gait dysfunction is one of the most disabling symptoms in Parkinson’s disease (PD) and it is associated with frequent falls, reduction of autonomy in activities of daily living and reduced quality of life [1–3].
In PD the dopaminergic system and other neurotransmitter pathways are involved in the neurodegenerative process [4–6]. This fact might explain the poor effectiveness of the dopamine replacement therapies for PD gait disturbances.
The alteration of the “internal cueing mechanism of the basal ganglia” is one of the possible hypotheses of gait dysfunction in PD [7]. The interplay between basal ganglia and cortical pre-frontal areas is important to switch from habitual to volitional motor control. Pre-frontal cortex controls the executive functions, which are essential for the planning and execution of movements. The alteration of dopaminergic transmission between the dorso-lateral striatum and the pre-frontal cortical areas could be the cause of the dysfunction of the executive functions [8, 9]. This fact might explain the relevant contribution of cortical areas to gait disorders in PD [10, 11].
It has been found that rehabilitation may improve motor performances in PD patients and recent large trials have demonstrated the positive effects of exercise on physical health and gait [12–14]. However, the optimal amount and setting of exercise is still debated [15, 16]. Among the proposed rehabilitative approaches, consistent evidence is available about the effectiveness of treadmill training and cueing techniques on gait disorder in PD [17, 18].
Gait in Parkinsonian patients is characterized by shorter stride length, higher cadence [7] and high stride-to-stride variability [19–22]. Treadmill training can improve gait performances in PD patients, acting as an external cue, which normalizes the spatiotemporal gait parameters [18, 23–25].
Cueing techniques (i.e. the use of external temporal or spatial stimuli to facilitate movement initiation and continuation), and feedback (the process of receiving input from the environment, based on the actions/outputs of the system) allow PD patients to generate a normal stride length with a positive impact on gait rhythmicity [26–29]. Different kinds of cues have been proposed and their specific action on gait parameters is related to the stimulated sensory system: while auditory cues seem to improve the cadence, visual stimuli act primarily on step length [26, 30].
As PD patients rely heavily on attention and executive processes to modulate their walking pattern [28], the use of a treadmill associated with visual and auditory cues can potentiate the effects on gait parameters, in comparison with “on-land” cueing or treadmill alone [25, 32].
To date, all available studies about this topic are referred to patients with mild to moderate PD stages [18, 33]. Nevertheless, with the progression of the disease [Hoehn & Yahr (HY) stage IV,] gait disturbances become even more invalidating [34]. Therefore, it could be useful to understand whether a specific gait training might be effective also in advanced PD stages.
In this study we aimed to evaluate the feasibility and the potential effects of treadmill training associated with visual and auditory cues and feedback (treadmill-plus) on gait dysfunction of Parkinsonian patients in more advanced disease stages.
METHODS
The study design and protocol were approved by the local Scientific Committee (Moriggia-Pelascini General Hospital, Gravedona ed Uniti, Como) and complied with the code of Ethics of the World Medical Association (Declaration of Helsinki, 1967). All patients included in the study had previously given consent to the use of their clinical data for scientific purposes. This study has been registered on ClinicalTrials.gov, with the Identifier NCT02731599.
Subjects and study procedures
PD patients were retrospectively identified among those hospitalized from January to December 2016 in our Department of Parkinson’s disease, Movement Disorders and Brain Injury Rehabilitation (“Moriggia-Pelascini” Hospital—Gravedona ed Uniti, Como, Italy) for a 4-week intensive gait rehabilitation.
Inclusion criteria were: (i) diagnosis of “probable” idiopathic Parkinson’s disease according to Gelb et al. [35]; (ii) mild to advanced stage of disease (stage 2-3-4) according to the HY scale [36]; (iii) ability to walk on a treadmill with no or minimal physical assistance; (iv) visual and hearing capacity sufficient to perceive cues and feedback; (v) no cognitive impairment (mini-mental state examination score >26) [37]; (vi) stable pharmacological treatment in the 4 weeks before admission and until the end of the training period. Exclusion criteria were: (i) diagnosis of atypical or vascular Parkinsonism; (ii) presence of cardiovascular, orthopedic, neuropathic, musculoskeletal and vestibular disorders affecting patient’s gait or balance; (iii) severe dyskinesia (iv) inability to walk on the treadmill for one minute at the belt-speed of 1.5 km/h without support.
Demographic and clinical data were derived from medical records and shown in Table 1. Data about the presence of Freezing of Gait (FOG) at baseline, evaluated by means of the Freezing of Gait Questionnaire (FOGQ) [38] were also available; the frequency of patients with FOG in our sample (patients with FOGQ score >0, indicated as FOG+ and expressed as percentage) is reported in Table 1.
A) Demographic and baseline clinical data for the sample as a whole and stratified in 3 groups according to HY stage. F / Chi-square: Test statistics (DF = 2), F value for continuous variables and Chi-square for discrete variables. p global: p value for the overall between groups comparison (3 groups). p HY2-HY3, p HY3-HY4, p HY2-HY4: p value from post hoc pairwise comparisons. FOG+: patients with FOGQ >0; NS: not significant at 0.05 level. B) Baseline values of primary and secondary outcome variables for the sample as a whole and stratified in 3 groups, according to HY stage. Chi-square: Test statistics (DF = 2). p global: p value for the overall between groups comparison (3 groups). p HY2-HY3, p HY3-HY4, p HY2-HY4: p value from post hoc pairwise comparisons
Abbreviations: 6MWT (Six-minutes walking test); L (Left); R (Right); CoV L (coefficient of variance of left step); CoV R (coefficient of variance of right step); HY (Hoehn & Yahr); UPDRS (Unified Parkinson’s Disease Rating Scale); FOG (Freezing of Gait).
On-going PD pharmacological treatments (dopamine agonists, levodopa and/or monoamine oxidases inhibitors) were converted in levodopa equivalent dose (LED), according to validated conversion tables [39]. LED was recorded for each patient at T0. No change in the total amount of dopaminergic therapy was made during the treadmill training.
Treadmill training
We used a motorized medical treadmill (Gait Trainer 3 Biodex, Biodex Medical System – 20 Ramsay Road, Shirley, New York, USA) for gait training. All patients underwent a 30-minute of treadmill training per day, divided in two 15-minute sessions, 6 days a week, for 4 weeks, with the supervision of a physiotherapist expert in movement disorders rehabilitation. Treadmill training was delivered independently from the assumption of medication. The belt speed was initially set at 1.5 km/h and progressively increased to a maximum of 3.5 km/h, depending on the physical ability of each patient. Patients were allowed to use handrails during the training, if needed. During the training visual and auditory cues and feedback were used and no weight support was provided. The visual cue was a target, delimited by two horizontal lines displayed on a screen which the patient had to reach with the stride. The software device calculated the space between the two lines for each patient, according to gender, height and age by means of an algorithm based on available normative data. The right and left footprints were shown alternatively on the screen: when they fell within the two lines, a “well done” message appeared; otherwise, patients were invited to take longer or shorter steps in order to adapt the stride length to the set target. The auditory cue and feedback consisted of musical beats with a frequency of 0.5 c/s synchronized with the visual cues. Figure 1 shows a treadmill-plus setting (A) and a detail of the screen, where visual cues and feedback are given (B).
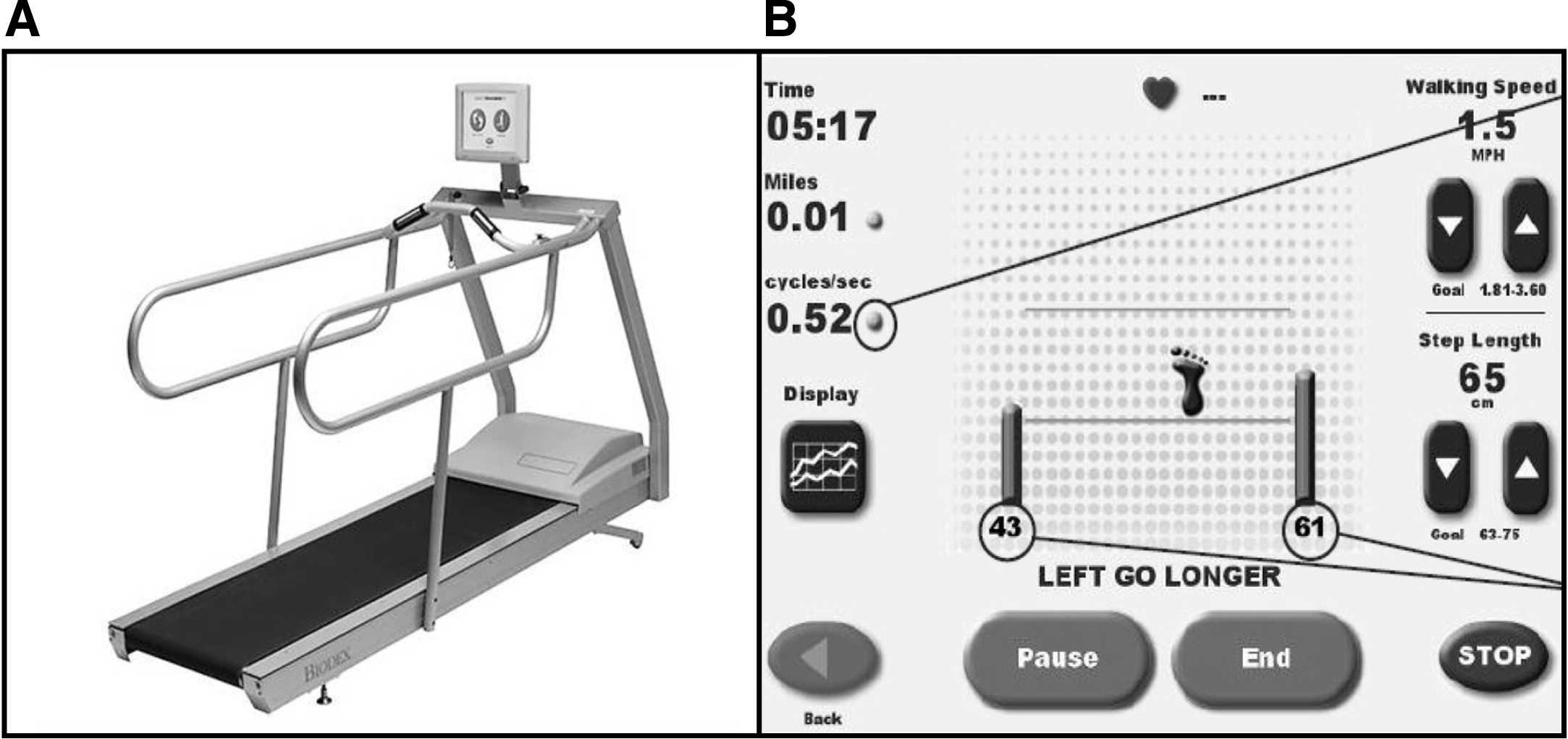
A representative image of the treadmill-plus setting, showing where the screen is situated respect to the scrolling belt (A), and a detail of the screen during a training session, in which the visual cues and feedback and the recorded parameters are displayed (B).
Besides treadmill-plus training, which was the only treatment proposed for the rehabilitation of gait, we also provided daily sessions of front-to-front physiotherapy, aiming at the improvement of joints range of motion, occupational and speech therapy, lasting one hour each.
Outcome measures
The feasibility of the treadmill-plus training intervention was assessed by recording adverse events (e.g., falls, extreme fatigue, changes in PD symptoms) throughout the 4-week intervention phase. We also quantified the levels of recruitment, attendance, attrition rates and therapist compliance.
In order to standardize data collection, at T0 and T1, all clinical and instrumental evaluations were performed in the morning, one hour after taking the first dopaminergic drug dose, in medication “on” state.
Clinical data and gait parameters were collected on the same day at baseline, before the beginning of the training (admission, T0), and at the end of the 4-week rehabilitation period (discharge, T1). While treadmill training was provided until the day before the discharge, the 6MWT at T1 was performed on the last day of hospitalization, just before the discharge, in order to avoid a short-term effect of the last training on the “on-land” performance.
Clinical assessment
A neurologist specialized in movement disorders performed neurological examination of all patients and assessed the Unified Parkinson’s Disease Rating Scale (UPDRS) score [40]. A specialized physiotherapist supervised and collected results of the 6-minute walking test (6MWT) [41].
The total distance (evaluated in meters) covered during the 6MWT was chosen as the primary outcome measure, as this has previously been shown to be a useful severity measure of patient’s self-paced gait [41, 42]. The 6MWT was conducted according to the following standardized procedure: the patient was first familiarized with the test by going once forward and backward along a straight 15-m line on the ground. Then, after a 15-minute rest, patients were instructed to walk from end to end of the line for 6 minutes and to cover as much distance as possible, independently selecting the most comfortable walking speed. No encouragement was offered during the test and no cues were used. Patients were allowed to use their habitual walking aids during the 6MWT, if needed. As secondary clinical outcome measure, total UPDRS and UPDRS III were taken into account.
Gait parameters
Gait parameters were collected using a motorized treadmill developed for gait training (Gait Trainer 3 Biodex, Biodex Medical System – 20 Ramsay Road, Shirley, New York, USA). The device is equipped with four strain gauges under the belt, sensitive to the bending of the belt caused by the weight applied by the patients’ walking. These gauges are connected to a software which enables quantification of several gait parameters. The gait evaluation was performed in the same setting conditions, both at T0 and T1, and it consisted in one-minute walking on the treadmill, without handrail support and without cues and feedback, at the scrolling belt speed of 1.5 km/h; this speed was expressively selected in order to highlight the spatiotemporal gait pattern alterations. Indeed, variables such as stride length and cadence are affected by gait velocity [43]; with lower velocities associated with shorter stride length and greater gait variability [31]. The following gait parameters were collected and included in the study as secondary outcome measures: the right and left step length (expressed in centimeters and calculated by averaging the length of all the corresponding steps), the coefficient of variance (CoV) of both right and left steps (expressing the standardized amount of variation occurring between footfalls, calculated as the ratio of the standard deviation to the mean value, in percent unit), and the step cycle (in cycle/s, average of all the step cycles of the exercise).
Statistical analysis
The Shapiro–Wilk statistic supported by visual inspection was used to test the normality of the distribution of all variables. Age, disease duration and amount of levodopa-equivalent dose did not violate the normality assumption. Accordingly, descriptive statistics are reported as mean±SD and parametric statistics (one-way analysis of variance) were used for between group comparison of these variables.
Conversely, the outcome measures violated more or less severely the normality assumption. Accordingly, descriptive statistics are reported as median (lower quartile, upper quartile) and between- and within-group comparisons were carried out by the Kruskal Wallis test and Wilcoxon signed-rank test, respectively.
Descriptive statistics for discrete variables are reported as number (percentage frequency) and comparisons were carried out by the Chi-square test or Fisher exact test, when appropriate.
A significant result for Kruskal Wallis test or parametric ANOVA, when comparing the three groups, was followed by post-hoc analysis (Dunn-Sidak and Tukey-Kramer, respectively), to compare pairs of groups.
When appropriate, false discovery rate was controlled at 5%, using the Benjamini-Hochberg method.
To assess the effectiveness of rehabilitation, the value of each outcome variable at discharge was compared with its value at admission in all patients and in the three HY groups.
To test whether rehabilitation effectiveness was dependent on disease severity, we considered the difference between values at discharge and values at admission in the outcome variables and we ran a one-factor non-parametric analysis of variance (Kruskal Wallis) on the HY factor (three groups).
The association between variables was assessed by the Spearman correlation coefficient. Since a high degree of association between HY stage, age and years of disease was expected, multivariable regression methods were used to investigate whether changes in outcome variables were associated with HY after adjustment for these covariates. All statistical tests were two-tailed and statistical significance was set at p < 0.05. All analyses were carried out using the SAS/STAT statistical package, release 9.2 (SAS Institute Inc., Cary, NC, U.S.A.).
RESULTS
Description of the sample
After being selected according to inclusion and exclusion criteria, the study population consisted of 206 patients, whose demographic and baseline clinical data are reported in Table 1.
Thirty-six patients were excluded because of a diagnosis of: i) vascular or atypical Parkinsonism (n = 12), ii) concurrent medical conditions for which treadmill training was contraindicated (n = 8), iii) cognitive impairment (n = 6), iv) severe dyskinesia (n = 5), v) inability to walk on treadmill for one minute at the belt-speed of 1.5 km/h without support (n = 5).
Of the 206 patients, 79 were in stage HY2, 74 in HY3 and 53 in HY4. As expected, the three groups differed for age, frequency of freezers (FOGQ >0), disease duration and amount of LED, with progressive worsening in more advanced stages. However, post-hoc analysis revealed that disease duration and LED were not significantly different between HY3 and HY4 patients. No differences in gender were observed.
Table 1 reports baseline values of primary and secondary outcome variables at baseline for the sample as a whole and stratified in 3 groups according to HY stage. As expected, a significant difference between groups at T0 was observed for all parameters (p < 0.0001 all), with worse values in higher HY stages; in particular, post-hoc comparison showed that comparing HY2 to HY3 and HY2 to HY4, all variables were significantly worse at a higher HY stage. Conversely, in the comparison between HY3 and HY4 several variables did not differ significantly (step cycle, left step length, left and right step CoV).
Age was significantly associated with the 6MWT and with treadmill-related gait parameters at baseline (spearman r: 6MWT = –0.51, step cycle = 0.30, left step length = –0.30, right step length = –0.29, left step CoV = 0.19, right step CoV = 0.18, p < 0.01 all). A slightly weaker association between disease duration and outcome measures was observed (spearman r: 6MWT = –0.31, step cycle = 0.16, left step length = –0.22, right step length = –0.13, left step CoV = 0.25, right step CoV = 0.26, p < 0.05 all).
No right-left side difference was recorded in step length and CoV in the whole sample (p = 0.21 and p = 0.26, respectively).
Treadmill training improves both self-paced and slow-speed gait in advanced PD
All subjects performed baseline treadmill evaluation and no dropouts occurred during the 4-week training, with an attrition rate of 0% and a 100% attendance recorded of all the scheduled training sessions; therefore, all 206 patients completed the study. No training-related adverse events were reported, such as severe fatigue or exacerbation of PD symptoms. Both FOG+ and FOG- patients could undergo treadmill training. No difficulties emerged regarding physiotherapist compliance to exercise supervision and data collection, which was complete for all patients. The study did not require any supplementary cost, as all the equipment was already available in our rehabilitation department.
Table 2 reports the difference of outcome variables (values at T1- values at T0) in the whole population and in the three groups.
Difference (values at T1-values at T0) of outcome variables as a whole and stratified in 3 groups according to HY stage. p ALL within, p HY2 within, p HY3 within, p HY4 within: p value for the within-group comparison (end of treatment vs admission) in all patients, HY2, HY3 and HY4 patients respectively. p between: p value for the overall between-group
Abbreviations: 6MWT (Six-minutes walking test); L (Left); R (Right); CoV L (coefficient of variance of left step); CoV R (coefficient of variance of right step); HY (Hoehn & Yahr); UPDRS (Unified Parkinson’s Disease Rating Scale).
After rehabilitation all outcome variables significantly improved, considering the whole population of the study (p < 0.0001 after adjustment for false discovery rate). The same result was obtained stratifying the patients according to the HY stage, with a significant improvement with respect to baseline in all disease stages (all p < 0.0001, except left CoV showing p = 0.003, p = 0.0004 and p = 0.003 for HY2, HY3 and HY4 respectively and right CoV showing p = 0.003, p = 0.029 and p = 0.003 for HY2, HY3 and HY4 respectively). Figure 2 shows the difference (values at T1- values at T0) of the gait parameters in the three groups, highlighting that a significant improvement was recorded after treadmill treatment both in gait endurance, evaluated with the 6MWT (Fig. 2A) and in gait regularity (expressed with the improvement of step length, cadence and step CoV, Fig. 2B-F), both in early and in more advanced disease stages.
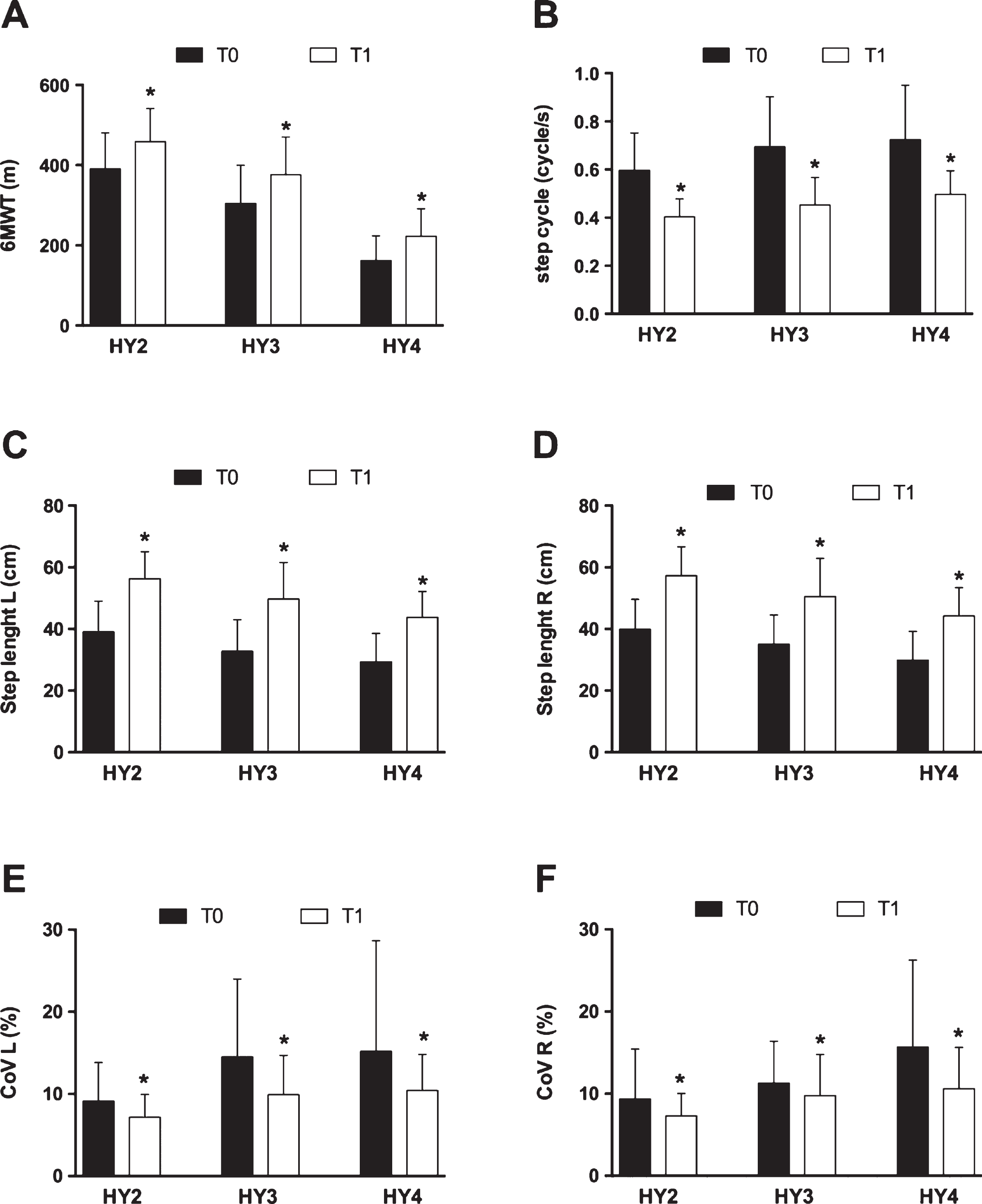
Baseline (T0) and discharge (T1) gait parameters demonstrate the effectiveness of treadmill training on gait disturbances in every disease stage. *p < 0.05 compared to the respective baseline value.
Finally, comparing the improvement after treatment among HY stages in outcome variables, Kruskal Wallis ANOVA revealed a significant difference only for total UPDRS (p = 0.01). Post hoc analysis revealed that the only significant difference in effectiveness was between HY2 and HY4 (p < 0.01), with better improvement in HY4 patients. No significant differences in treatment efficacy were observed in all remaining variables (p values ranging from 0.09 for changes in left step length to 0.66 for changes in the 6MWT).
Investigating the relationship between the improvement in outcome variables and HY stage, age, sex and disease duration, none of these variables was independently associated with the amount of improvement, with the exception of total UPDRS, where disease duration was the only independent predictor of improvement (p = 0.003, longer disease duration associated with better improvement), and HY stage became borderline significant.
DISCUSSION
In this study we have shown for the first time that treadmill training associated with visual and auditory cues and feedback is a feasible and safe approach for PD patients in more advanced stages of disease. Indeed, all patients included in the study completed the 4-week training without treadmill-related adverse events. Moreover, our results demonstrate that treadmill training is effective in improving gait parameters in more advanced PD patients (HY stages 3 and 4), and that the observed improvements are comparable to those obtained in early-moderate PD patients.
In our sample, the 6MWT score and the spatiotemporal gait parameters at baseline progressively worsened from HY stage 2 to 4. Nevertheless, a comparable improvement after the 4-week treadmill-plus training was found also in patients in advanced stages. Of note, this result was achieved not only in self-paced ambulation, as revealed by the 6MWT score, but also in the one-minute treadmill evaluation at 1.5 km/h. The improvement of all spatiotemporal gait parameters at slow speed is particularly relevant, since stride length and cadence are affected by gait velocity [43], with lower velocities associated with shorter stride length and greater gait variability [31].
Our findings are in line with those published in a recent study [32], in which a relevant improvement in gait parameters has been found after gait training with a treadmill plus with a visual cue, in PD patients. In both studies a significant increase in stride length has been documented, but, due to a different study design, Schlick et al. [31, 32] reported an increase in walking speed and no differences in cadence, while we observed a reduction in step cycle.
A relevant point of our study is that all spatiotemporal gait parameters were positively modified by treadmill-plus training. In fact, besides the effects on stride length and cadence, a significant reduction of the CoV after the 4-week training has been observed, highlighting an improvement in gait pattern. Parkinsonian patients in advanced stages of disease have a loss of basic locomotion rhythmicity with greater steps variability [44] due to the progressive sensory, motor and cognitive deficits (i.e., executive functions and attention) [11] associated with balance dysfunctions [45] and muscle weakness [46]. As recently outlined [47, 48], greater gait variability is associated with worse postural instability and occurrence of falls. It is therefore arguable that a reduction of the CoV might provide significant benefits in terms of rate of falls and balance disturbances.
The effectiveness of treadmill-plus training on gait may be due to different mechanisms. Firstly, the treadmill alone improves gait by reducing stride-to-stride variability [23], acting as an external pace and focusing the attention on gait. The effectiveness of this approach has already been described in patients in early-medium disease stage (HY 2-2,5) [23].
In addition, it has been demonstrated that the combined use of treadmill with visual-auditory cues and feedback is even more effective in improving gait parameters, such as gait speed, stride length and cadence, in patients with PD [25, 32]. The effectiveness of treadmill-plus training in improving gait could be related to the motor learning processes. In PD implicit learning is impaired due to the loss of dopamine in the dorsolateral striatum [49], but motor learning remains feasible [50]. The cognitive strategies based on explicit learning and requiring the use of the executive component of attention (such as cues, feedback and verbal instructions) allow PD patients to “by-pass” the dysfunctional basal ganglia-supplementary motor area circuit, performing voluntarily the defective movements through the goal-directed motor control. The use of treadmill-plus might optimize motor learning strategies in PD: explicitly by means of cues and feedback, implicitly by means of the repetitive movement of the treadmill belt. Cues and feedback play a synergistic role, while cues are system-independent signals to which patients have to adapt, feedback is affected by patients’ actions. Feedback learning requires greater cognitive engagement and strengthens the cues effects [51].
It is possible to hypothesize that the intensity of training, along with the repetitive use of cues and feedback, optimizes motor learning [52], allowing a generalization of the acquired information from the treadmill to the “on-land” environment. This consideration could explain why the changes in spatiotemporal gait parameters are associated with an improvement also in the 6MWT at the end of the training.
Moreover, it is possible to consider another mechanism responsible for gait improvement: the treadmill itself stimulates the spinal locomotor circuitry [53], stressing the functioning of the central pattern generator [54].
In our sample, HY4 patients took advantage from treadmill-plus training, and the improvement of gait parameters was comparable to that of patients in HY stage 2 and 3. This finding suggests that even in more advanced PD stages, patients may use the executive-volitional control strategies, getting significant benefit from specific rehabilitative strategies [55, 56].
Study limitations
This is a retrospective study analyzing a prospectively collected database. Even though the lack of a prospective randomized controlled trial design might constitute a potential limitation, data collection procedures were carefully designed and all relevant information was recorded on a structured protocol.
The lack of a direct comparison between traditional treadmill and treadmill-plus training does not allow quantifying the specific contribution of cueing techniques to the observed gait improvement; further studies will be planned to compare the two different strategies.
No follow-up data are available at the moment, thus limiting our knowledge about the duration of gait improvement. Another limitation arises from the analysis of gait parameters performed using the gait trainer belt sensors. Compared to over-ground walking, in fact, treadmill walking induces slightly shorter stride lengths and slightly higher cadence [57], particularly in PD patients [58]. However, all subjects were tested in the same experimental setting; the observed differences in spatiotemporal gait parameters among groups should not be affected by this bias.
Finally, another limitation concerns the speed of the belt scrolling that we set at 1.5 km/h: as already demonstrated [59], spatiotemporal gait variability depends on self-generated walking speeds. Thus, the selected speed of the belt scrolling could have influenced data about the coefficient of variation in this study.
Conclusions
In conclusion, our study supports the feasibility and the usefulness of a treadmill-plus training in patients at more advanced PD stages. The improvements in spatiotemporal gait parameters found using a slow-speed treadmill were recorded together with better performances in the 6MWT at the end of the training. Further studies will be needed to clarify the mechanism underlying the effectiveness of treadmill-plus training on gait disorders, to define its long-lasting effects and its role within a tailored rehabilitative protocol for advanced PD patients.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
