Abstract
Background:
Gastrointestinal symptoms are a well-recognized and common premotor feature of Parkinson’s disease (PD). Moreover, multiple studies have assessed the value of colonic α-synuclein as a potential marker of prodromal PD. Recently, the International Parkinson and Movement Disorders Society (MDS) defined research criteria for prodromal PD.
Objective:
The aim of our study was to test the MDS research criteria in patients undergoing diagnostic colonoscopies as potential candidates for inclusion in prospective trials evaluating colonic biopsies as a potential biomarker of prodromal PD.
Methods:
We evaluated elderly patients without manifest parkinsonism undergoing diagnostic colonoscopies. During the study we assessed all risks and prodromal markers of the MDS research criteria, excluding radiotracer imaging and genetic testing.
Results:
The mean age of the 100 enrolled patients was 61.6±9.7 years; 42 were men. The most common prodromal marker in our cohort was constipation (40%), followed by MDS-UPDRS part III scores of >6 points, excluding action tremor items (39%) and hyposmia (37%). Substantia nigra hyperechogenicity was identified in 9%, and polysomnography confirmed REM sleep behavior disorder in 2% of the patients. Five of the 100 enrolled patients (5%) fulfilled the criteria for probable prodromal PD, while another 3 patients met the 50% probability threshold.
Conclusions:
Our findings suggest, that the prevalence of prodromal PD in patients undergoing diagnostic colonoscopies may be higher compared to the general elderly population, although this should be confirmed in further studies including also matched controls not undergoing colonoscopy. The real prevalence of prodromal PD in this cohort will have to be confirmed in longitudinal follow-up. Patients undergoing diagnostic colonoscopies may be good candidates for multistep screening and inclusion in prospective trials.
INTRODUCTION
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by motor, but also a number of non-motor manifestations [1]. Despite advances in our understanding of the disease, clinical diagnosis is still based on the presence of typical motor manifestations [1]. However, there are a number of well recognized non-motor symptoms, such as constipation, REM sleep behavior disorder (RBD) or hyposmia, which may precede the motor stage of PD by years [2]. Recently, the International Parkinson and Movement Disorder Society (MDS) Task Force on the Definition of Parkinson’s Disease published research criteria for prodromal PD [3]. These criteria use age-specific disease prevalence as a pre-test probability along with positive and negative likelihood ratios (LR) of different risk and prodromal markers, combining them for a final age-specific probability of a particular patient with prodromal PD. Despite covering a number of different markers, these criteria do not include any peripheral tissue biomarkers, as no prospective studies in this regard have yet been performed. There have been several studies published on PD showing the presence of α-synuclein aggregates in peripheral tissues, which are readily available for biopsies, such as the salivary glands [4], colon [5] or skin [6]. Increased α-synuclein staining has been found in early untreated patients with PD [7]. Shannon et al. [8] also found positive α-synuclein staining in the colonic biopsies of 3 patients with PD performed 2–5 years prior to onset of their first motor symptoms. Also, increased α-synuclein immunoreactivity was found in patients with REM sleep behavior disorder (RBD) compared to healthy control patients [9]. While the results of studies assessing the presence of colonic α-synuclein thus far vary, which is probably also due to different study designs and methodologies applied [10, 11], patients undergoing diagnostic colonoscopies are potential candidates for future prospective trials assessing the role of colonic α-synuclein as a potential biomarker of PD. Moreover, gastrointestinal (GI) symptoms, such as constipation, abdominal bloating and postprandial fullness, are well recognized premotor symptoms of PD [2], and thus it is possible, that these patients are more likely to undergo a colonoscopy compared with the general population. To date no information is available on the prevalence of risk and prodromal markers of PD or the prevalence of prodromal PD in the population of patients undergoing diagnostic colonoscopies. Our presented data are the first report from a prospective study assessing colonic biopsies as a potential biomarker of premotor PD (PARCAS study – PARkinson’s disease associated Colonic Alpha-Synuclein biomarker study). The aim of the study was to assess, based on the MDS research criteria, the prevalence of prodromal PD in elderly patients without manifest parkinsonism undergoing diagnostic colonoscopies and to evaluate the prevalence of risk and prodromal markers of PD in this population.
METHODS
Patients
Elderly patients (50–85 years) without clinically manifest motor Parkinson’s disease and undergoing diagnostic colonoscopy at two gastroenterology clinics were examined at a single University based Neurology department in Kosice, Slovakia, after agreeing with enrollment in the study. Patients were excluded if they had an active colonic cancer; inflammatory bowel disease (Crohn’s disease or Ulcerous colitis); an expected survival of <3 years based on other co-morbidities; or if they were not able to cooperate in the study, e.g. due to severe dementia. The study was performed according to the Declaration of Helsinki of 1975; it was approved by the local ethics committee and all patients signed written informed consent prior to enrollment.
Measures
Sociodemographic data, including age, gender and length of education, were collected, as well as information on family history of parkinsonism, exposure to environmental toxins, occupational solvent exposure, history of smoking and caffeine consumption, presence of severe erectile dysfunction and pharmacological therapy.
Scales
All patients were examined using the Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) [12]. The MDS-UPDRS is a four-subscale combined scale which comprehensively assesses the symptoms of PD. It consists of: Part I - non-motor experiences of daily living, including 13 items – 6 semi-structured interview, 7 self-reported items; Part II - motor experiences of daily living, including 13 self-reported items; Part III - motor examination, including 18 items (33 scores); and Part IV - motor complications, including 6 items assessed in a semi-structured interview. All items are scored on a scale from 0 (normal) to 4 (severe), and total scores are obtained from the sum of the corresponding item scores.
The REM sleep behaviour disorder screening questionnaire (RBDSQ) is a patient self-report questionnaire with 13 items rated as yes/no. The maximum total score of the RBDSQ is 13 points. Scores of ≥5 points have revealed a sensitivity of 96% and specificity of 56%, when also considering patients with other sleep abnormalities, while increasing specificity to 92% when comparing with healthy controls only [13].
Smell identification
Hyposmia was assessed using the 12-item Sniffin’ sticks odor identification test (SS-12) [14], which was culturally adapted to the Slovak population. The test involves multiple forced- choice identification of 12 odors from a list of 4 descriptors each and has a maximum score of 12 points.
Transcranial sonography
For evaluation of substantia nigra hyperechogenicity, all patients underwent a transcranial sonography with a 3-1 MHz sector array transducer probe (Phillips HD11XE, Phillips Ultrasound, Bothwell, WA, USA), adopting the previously published protocol of Walter et al. [15]. Patients were evaluated by a single experienced examiner (NL).
Polysomnography
Patients scoring ≥5 points on the RBDSQ were interviewed by a neurologist specialized in sleep disorders (EF) and subsequently underwent a single night polysomnography evaluation (PSG). Diagnosis of RBD was based on a polysomnographic recording demonstrating REM sleep without atonia (RWA) and repeated episodes of sleep related vocalization and/or complex motor behaviours, which were observed by sleep partners/family or recorded during PSG night [16, 17].
Ascertainment and definition of risk and prodromal markers
All prodromal and risk markers, except known genetic mutations and SPECT/PET imaging, were available for analyses [3]. LR for caffeine consumption and smoking were evaluated as in the study of Berg et al. [3]. In the case that age of onset <50 years in a sibling with PD was not clearly ascertained, a more conservative LR+ 2.5 was applied for positive family history. For SN hyperechogenicity a cut-off ≥0.25 cm2 for LR+ 4.7 was used based on the 90th percentile of normative data from our center; values of 0.20–0.24 cm2 were evaluated as LR 1 and values <0.20 cm2 as LR- 0.45. In the case of missing temporal bone window, LR 1 was applied. For patients scoring <5 points on the RBDSQ, LR- 0.76 was applied. Those who scored positive on the RBDSQ and refused to have a PSG performed were scored as LR+ 2.3. Patients who were examined by PSG were rated as LR+ 130 only in the case of an unequivocally positive RBD finding. For possible subthreshold parkinsonism, patients were rated as LR+ 10 if they had an MDS-UPDRS part III score, not including postural and kinetic tremor items, of >6 points as defined by the MDS research criteria. LR+ 4.0 for olfactory loss was considered in the case of an SS12 score <9 points. Constipation LR+ 2.2 was defined as ≤3 bowel movements per week. Information on excessive daytime somnolence was assessed by MDS-UPDRS item 1.8, symptomatic hypotension using MDS-UPDRS item 1.12 and urinary dysfunction using MDS-UPDRS item 1.10. For all three items scores of ≥2 were rated as LR+ (2.2, 2.1, and 1.9, respectively), a score of 1 was rated as LR 1 and a score of 0 was rated as LR- (0.88, 0.87, and 0.9 respectively). Severe erectile dysfunction was specifically addressed during the history taking and was considered as LR+ 2.0 if a patient complained about severe erectile dysfunction (ED). Complaints about mild ED were not considered as LR+. Depression was scored as LR+ 1.8 if a patient is currently or was previously treated for depression. When any of the values was missing, LR 1 was applied. For all likelihood ratios used please see Suppl. Table 1.
Statistical analyses
Statistical analyses were performed using the statistical software program PASW SPSS version 21.0 for Windows (SPSS Inc, Chicago IL). First, the demographic and clinical characteristics of our study cohort were described, including the prevalence of prodromal and risk markers of PD. Mean and interquartile range (IQR) scores were used to better describe the distribution of our sample. Total likelihood ratios for prodromal PD were calculated as previously published in the study of Berg et al. [3] by multiplying likelihood ratios of all prodromal and risk factors, excluding known genetic mutations and SPECT/PET studies, which were not performed in our study population. A lower threshold cut-off of 50% probability for prodromal PD was counted as well.
RESULTS
A total of 109 patients who fulfilled the initial inclusion and exclusion criteria and agreed with participation in this study were screened for the presence of risk and prodromal markers of PD. Nine patients were excluded after colonoscopy due to the presence of exclusion criteria (incidental colonic cancer or inflammatory bowel disease) and 100 remained for further analyses (see Fig. 1). Ten patients aged 40–49 years who fulfilled the remaining inclusion and exclusion criteria were additionally included in the study, in order to assess the prevalence of risk factors also in a younger population of patients. Seven patients were not included due to the presence of clinically manifest PD – all fulfilling the MDS clinical diagnostic criteria for Parkinson’s disease [18], no other form of parkinsonism has been identified during the study. The most common indications for colonoscopy in our sample were screening for colonic cancer (32.4%), non-specific gastrointestinal (GI) symptoms (25.5%), GI bleeding or sideropenic anemia of unknown origin (14.1%) and constipation (11.3%). The mean age of the patient sample was 61.6±9.7 years; 42 were men. The median MDS-UPDRS part III score, excluding action tremor items, was 4 points (IQR 1–9); the median RBDSQ score was 3 (IQR 1–4); and the median SS-12 score was 10 (IQR 8–11). The most common risk or prodromal marker of PD found in our sample was constipation (40%), followed by MDS-UPDRS III scores >6 points (39%) and hyposmia (37%). Out of 11 patients who screened positive on the RBDSQ, 2 refused a PSG examination and RBD was PSG confirmed in 2 out of 9 examined patients. The remaining 7 positively screened patients were diagnosed with other sleep-wake disorders: 2 restless legs syndrome, 1 insomnia, 1 sleepwalking, 1 sleep apnea, 1 post-traumatic stress disorder, 1 panic disorder. SN hyperechogenicity was identified in 9 patients. Detailed characteristics of the study sample, including the prevalence of risk and prodromal markers, are presented in Table 1. Out of the 100 patients evaluated, 5 met the criteria for probable prodromal PD. Three other patients met the 50% probability cut-off also mentioned in the MDS criteria (see Fig. 2). The most common risk and prodromal markers in these patients were constipation and hyposmia, followed by MDS-UPDRS part III scores >6 pts and non-smoking. Both of the patients with PSG-confirmed RBD fulfilled criteria for prodromal PD (see Table 2). To assess the prevalence of risk and prodromal markers in younger patients, 10 subjects aged 40–49 years were included. However, none of them had a total likelihood ratio higher than 6. Out of these patients, 4 screened positive on the RBDSQ, but PSG did not confirm RBD in any of them; SN hyperechogenicity was identified in 1 of these patients, hyposmia in 2, constipation in 8 and all had MDS-UPDRS part III scores <6 points.
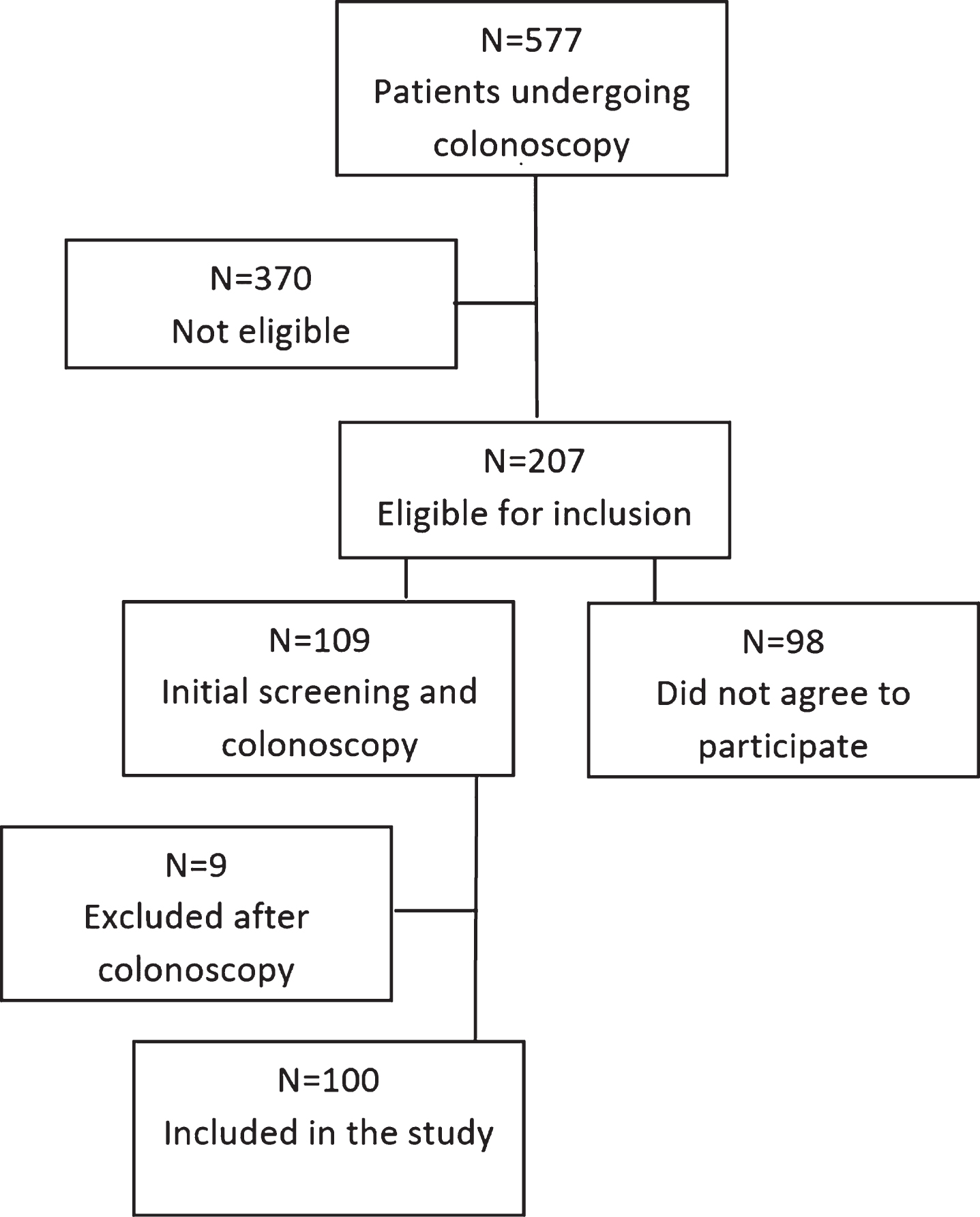
Flow diagram of ascertainment of study cohort.
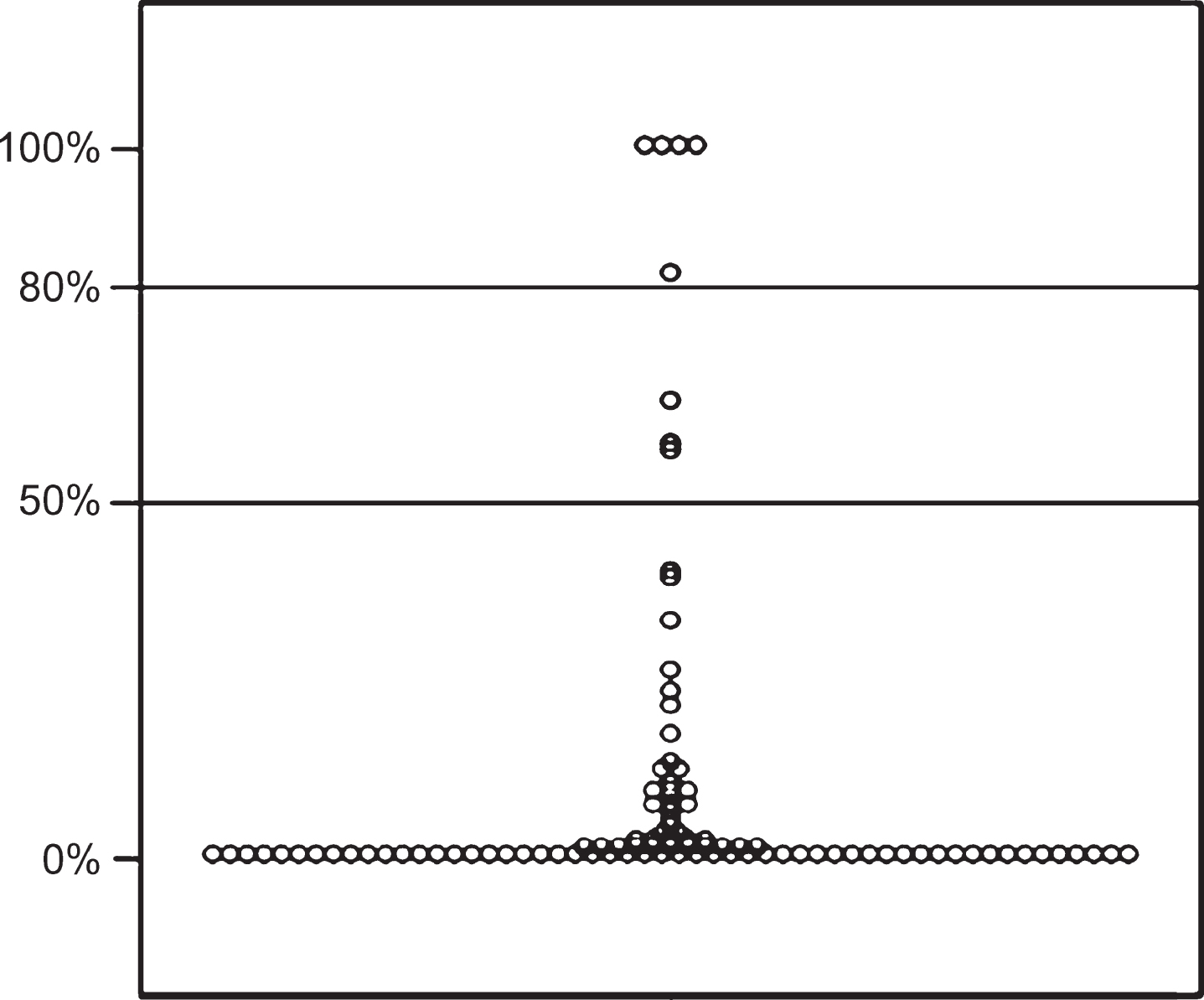
Posttest probability of prodromal PD.
Characteristics of the sample and prevalence of prodromal and risk factors
IQR – interquartile range, PD – Parkinson’s disease, SN – Substantia nigra, PSG – polysomnography, RBD – REM sleep behavior disorder; RBDSQ – REM sleep behavior disorder screening questionnaire; MDS-UPDRS – Movement Disorder Society-Unified Parkinson’s Disease Rating Scale; SS12 – Sniffin’ sticks 12-item identification test; EDS Excessive daytime sleepiness. *only patients who screened positive on the RBDSQ (>5 pts) underwent PSG, 2 patients refused PSG.
Presence of risk and prodromal markers in patients fulfilling prodromal PD criteria
PD –Parkinson’s disease, SN – substantia nigra, PSG – polysomnography, RBD – REM sleep behavior disorder, RBDSQ – REM sleep behavior screening questionnaire, MDS-UPDRS – Movement Disorder Society-Unified Parkinson’s Disease Rating Scale, SS12 – Sniffin sticks 12-item identification test, LR – likelihood ratio.
DISCUSSION
By prospective application of the MDS research criteria [3] to patients undergoing diagnostic colonoscopy, we found a prevalence of probable prodromal PD of 5%, with a further 3% of patients meeting the 50% probability threshold for prodromal PD. This seems higher than the prevalence of 2.2% found during retrospective analysis of the Bruneck cohort representing the general elderly population [19] and higher than expected in the general population by the MDS research criteria [3]. Despite higher prevalence of prodromal PD in our study population of patients having colonoscopy it is not possible to draw final conclusions in this regard, since a control sample of patients not having colonoscopy was not screened in our cohort. The major factor influencing the incidence of PD in a population is probably age, as defined also in the MDS research criteria pre-test probability calculations. In this regard the age of our population was lower (median 62, IQR 54–69) than that of the Bruneck cohort (median 67.2 IQR 61.4–76.2) [19], which is equivalent to an estimated pre-test probability of 1.3% (IQR 0.4–2.0) vs. 2.0% (IQR 1.3–3.5). Thus, an even lower prevalence of prodromal PD could have been expected in our cohort based on the age distribution of these cohorts. It is, however, important to note that the real prevalence of prodromal PD in our cohort is not known, as a longitudinal follow-up will be necessary to ascertain the true conversion into clinically manifest PD in our patient sample. Increased prevalence of prodromal PD in elderly patients undergoing diagnostic colonoscopies compared with the general elderly population is not fully unexpected, as GI symptoms, such as constipation, abdominal bloating, etc., are well-recognized premotor symptoms of PD [2] and are more likely to lead to colonoscopy examinations. In fact, constipation was the most common prodromal marker, found in 40% of our sample compared to only 9.3% of patients in the Bruneck cohort [19]. Similarly to the Bruneck study [19], we found a high prevalence of hyposmia in our sample (37% vs. 40%). In contrast to the Bruneck study [19], which found subthreshold parkinsonism in 6% of their sample using the original UPDRS part III scale threshold of >3 points, excluding action tremor, we found a prevalence of subthreshold parkinsonism of 39% using the MDS-UPDRS part III score threshold of >6 points, excluding postural and action tremor items. Our results are, however, consistent with another study of Keezer et al. [20], who evaluated the MDS-UPDRS scores in an otherwise normal elderly population and found 30% of patients having part III scores higher than 6 points, with the highest score found in the range 28–30 points, similar to highest score of 32 points found in our cohort. The MDS-UPDRS compared to the original UPDRS covers more items and better discriminates the slight and mild motor disturbances, which results in higher summary scores [12]. Moreover, by applying the official instruction of “rate what you see”, other disorders common among the elderly, such as stroke, arthritis or polyneuropathy, are likely to increase part III scores. In fact, 34 patients from our cohort and 18 of the 39 patients (46%) having MDS-UPDRS scores >6 points had another neurological comorbidity, which may have influenced the MDS-UPDRS part III scores. The most common neurological comorbidities included vertebrogenic problems and polyneuropathy; 4 patients had previously had a stroke and 2 patients had Essential tremor. Thus, prospective trials should help specify the most appropriate MDS-UPDRS part III cut-off score to be applied in the MDS research criteria, which may be in fact higher than the one recommended at the moment.
The SN hyperechogenicity prevalence of 9% in our sample was very similar to that reported in the general population [15]. However, it is interesting that it was found only in 1 out of 5 patients meeting the 80% and 1 out of 3 patients meeting the 50% probability cut-off for prodromal PD, compared to a sensitivity of 83% in clinically diagnosed PD patients, as published in a recent meta-analysis [21]. It is also lower than reported in a 5-year prospective follow-up study by Berg et al. [22], who found SN+ in 14/17 (82.4%) patients with incidental PD. On the other hand a prospective study of idiopathic RBD patients [23] found initial SN hyperechogenicity only in 4/9 (44.4%) of patients, which then converted into PD after a 5-year follow-up. It is necessary to emphasize that the time frame in which SN hyperechogenicity occurs before onset of motor symptoms of PD is not known exactly, and it is also not known whether all subjects fulfilling the prodromal PD criteria in our study will, in fact, convert to clinically manifest motor PD, which may underlie this discrepancy and a longitudinal follow-up will be necessary in this regard.
Out of 11 patients who screened positive on the RBDSQ, 2 refused to have PSG performed and RBD was confirmed in 2 of the remaining 9. This finding is similar to the multistep approach study of Postuma et al., who tried to identify patients with RBD by a similar procedure, and identified 111 patients screening positive on a single RBDSQ question, 40 patients screening positive on the Innsbruck RBD inventory and finally 19 patients having PSG confirmed RBD. In fact, a very recent study showed low reliability of a questionnaire-based RBD diagnosis based on RBDSQ, as out of 29 patients with questionnaire-based probable RBD during a first visit, only 8 (28%) still had probable RBD during a follow-up visit 2 years later, while another 15 patients also fulfilled the criteria at that time [25]. Screening for RBD may be a very effective approach for identifying individuals at high risk of prodromal PD. However, as PD is a disorder with highly heterogeneous clinical manifestations, especially in its early stages, and by recognizing different subtypes of PD, such as the brainstem, limbic, or cognitive dominant subtypes recently described by Marras et Chaudhuri [26], it is clear that by including strictly RBD positive patients in clinical trials, other PD subtypes may be omitted. Therefore, an effort should be made to also identify other cohorts of patients at high risk for developing PD and thus suitable for a more detailed screening.
Limitations of our study include its cross-sectional character and the necessity to confirm our results during a long term follow-up and preferably on a larger sample of patients, since not all patients meeting the MDS research criteria in our cohort must necessarily convert into manifest PD. Also, diagnosis of all patients was based on clinical evaluation, which would be strengthened by adding pathological data. Another limitation is the lack of a matched control group not undergoing colonoscopies, which would enable us to compare the prevalence of prodromal PD and draw firmer conclusions. Three of the markers, including EDS, symptomatic hypotension and urinary problems, were assessed by a single MDS-UPDRS item rather than by using a specific tool. However, the MDS research criteria do not specify a preferred method of marker assessment, and this will have to be better defined by future prospective studies. On the other hand, a comprehensive evaluation of the markers assessed in the MDS criteria, including highly specific PSG examinations, enabled us to properly define the studied population. Inclusion of the costly radiotracer imaging and genetic examinations could help even better define this population.
In conclusion, prevalence of prodromal PD, as defined by the MDS research criteria [3], seems to be higher in elderly patients undergoing diagnostic colonoscopies compared to the general elderly population, although this should be confirmed in further studies including also matched controls not undergoing colonoscopy. This may be useful especially for power calculations of future studies evaluating colonic biopsies as a premotor biomarker of PD. A number of studies have reported increased α-synuclein deposition in the peripheral nervous system of PD patients, including the colon [7, 10]. However, no study so far has assessed the value of peripheral α-synuclein examination in a prospective manner. In this regard, patients undergoing routine colonoscopies seem to have higher prevalence of prodromal PD and thus are a good candidate group for multistep screening and inclusion in such studies. As bioptic samples of the colon from the majority of the evaluated subjects were obtained, further enlargement and longitudinal follow-up of our cohort as well as evaluation of colonic a-synuclein in this population may shed more insight into the epidemiology and underlying mechanisms of prodromal PD.
CONFLICT OF INTEREST
The authors declare no conflict of interests in regards to this manuscript.
FULL FINANCIAL DISCLOSURES FOR THE PAST 12 MONTHS
MS received grants from the Slovak Research and Development Agency, the Slovak Scientific Grant Agency, speaker honoraria and compensations for consultations from Abbvie, Actelion, Egis, International Parkinson and Movement Disorder Society, Krka, Lundbeck, Medtronic, Sandoz, TEVA and UCB. ZL received grant from the Slovak Research and Development Agency and received travel grant from AbbVie. VH received grants from the Slovak Research and Development Agency, the Slovak Scientific Grant Agency, speaker honoraria and compensations for consultations from Abbvie, Actelion, Medtronic, Sandoz and UCB. NL received grants from the Slovak Research and Development Agency, speaker honoraria and compensations for consultations from Boehringer Ingelheim, Operatíva, and Pfizer. EF received grants from the Slovak Research and Development Agency, the Slovak Scientific Grant Agency, speaker honoraria and compensations for consultations from UCB, Eisai, and Phillips Respironics. ZG received grants from the Slovak Research and Development Agency, the Slovak Scientific Grant Agency, speaker honoraria and compensations for consultations from Actelion, Bayer, Biogen-Idec, Boehringer Ingelheim, Egis, Lundbeck, Medtronic, Novartis, Pfizer, and UCB. All other authors have no financial disclosures.
Footnotes
ACKNOWLEDGMENTS
The funding for research leading to this study was provided by the Slovak Research and Development Agency under contract no. APVV-14-0415 and by the Slovak Scientific Grant Agency under contract no. VEGA 1/0024/14. The authors wish to acknowledge Martina Sukenikova, Filip Kudelka, Lujza Strigacova and Angelika Turnova for their work during this study.
