Abstract
Keywords
BACKGROUND
Walking difficulties are among the earliest signs of disability in people with Parkinson’s disease (PD) [1] and include, for example, reduced gait speed, step length and arm swing as well as gait asymmetry [2, 3]. Freezing of gait (FOG) is also common and is experienced as “if the feet were glued to the floor” [4]. FOG most frequently occurs in the home environment and is provoked by certain activities (e.g., turning around while walking) and environmental factors such as being in a confined space [5]. Several PD studies have identified contributing factors to objectively measured walking difficulties (e.g., assessed by using an electronic walkway system) [6–9]. To the best of our knowledge, no prior study has considered a broad diversity of factors (e.g., personal, social environmental, and disease related factors) to identify those that are independently associated with perceived walking difficulties among people with PD.
In studies that used objective gait measures, FOG has been shown to contribute to impaired step length and increased variability of step duration in persons with PD [6, 10]. Moreover, reduced gait speed has been associated with fear of falling [11], postural instability [7], disease severity [11, 12], bradykinesia [8], cognitive impairments, physical fatigue [9, 13], depressive symptoms [11, 13], and muscle weakness in people with PD [14]. In addition to reduced gait speed, shorter strides as well as an increased stride variability have been associated with postural instability [7]. Reduced arm swing while walking has been associated with bradykinesia [15]. Using objective measures of walking difficulties may not capture perception of walking difficulties in the complexity of daily life circumstances. Especially so if the collection of data using objective measures was conducted during a short time period and/or in a standardized setting that mimics capacity more than actual performance in authentic daily life settings.
Several qualitative PD studies have described factors that are perceived as negatively associated with walking difficulties such as FOG [19–23], fatigue [19, 22], anxiety [22], FOF [19], pain, orthostatic hypotension [24], ineffective dose of medication [22] and environmental hazards (e.g., crowds, inclement weather, and uneven/slippery surfaces) [19, 24]. On the other hand, informational support (e.g., advice/knowledge provided by other people) may influence that people with PD participate in physical activity, and social as well as emotional support can facilitate that they engage in taking a walk [25]. It would be of interest to investigate whether some of these qualitative findings could be verified in a larger quantitative study. When following large cohorts, qualitative data collections and analyses are not feasible, but survey data that includes a patient-reported outcome measure would make it possible to identify factors that explain perceived walking difficulties in daily life. A better understanding of the factors associated with perceived walking difficulties may facilitate to develop individually targeted rehabilitation and may result in more efficient physical activity prescriptions for people with PD. Accordingly, this study aimed to identify factors that independently contribute to perceived walking difficulties in people with PD.
MATERIALS AND METHOD
This study was based on a cross sectional study design. It was based on baseline data collected for the project “Home and Health in People Ageing with PD”, which aimed to generate knowledge on home and health dynamics in people with PD, with an explicit attention to PD-specific symptomatology. The project design, inclusion and exclusion criteria, recruitment process, ethical considerations, procedure and data collection have been described in detail in the study protocol [26].
The data collection included a self-administered postal survey and a subsequent home visit that involved interview-administered questions and questionnaires, observations and clinical assessments. The home visits were scheduled during the time of day when the participant in question stated that he/she usually feels best (“on” state). Two trained project assistants (experienced reg. occupational therapists) conducted the data collection.
The project was conducted in accordance with the Helsinki Declaration and was approved by the Regional Ethical Review Board in Lund, Sweden (No. 2012/558). All participants provided their written informed consent.
Participants and recruitment
Participants were recruited from three hospitals (outpatient registers) in Region Skåne in southern Sweden; 653 participants met the inclusion criterion of being diagnosed with PD (G20.9) for at least one year. Out of these, 216 individuals were not eligible due to the exclusion criteria: difficulties in understanding/speaking Swedish (n = 10), severe cognitive difficulties (n = 91), living outside Skåne (n = 58) or other reasons (n = 57) (e.g., severe hallucinations, recent stroke). That is, a potential participant was excluded if not deemed to be able to give an informed consent or partake in the majority of the data collection. The remaining 437 persons were invited to participate. However, 22 were impossible to reach and two had their PD diagnosis revised. That is, 413 participants that had a PD diagnosis were contacted whereof 157 (38%) declined to participate.
One participant was excluded due to extensive missing data. In the present study, four additional participants were excluded since they did not respond to any of the self-administered questionnaires, stated that someone else had responded or had severe delays in responding. Yet another eight were excluded since they had no total score (i.e., had not responded to all items) of the generic Walk-12 (Walk-12G), i.e. the used PROM and the dependent variable in the present study. Accordingly, the final sample consisted of 243 (62% men) participants. Their mean (min-max) age was 70 (45–93) years, and the PD duration was 8 (1–43) years. When comparing the final sample to those who that declined to participate (n = 157), there was a statistically significant difference in age (p = 0.016, Independent T-Test), but not in relation to sex (p = 0.066, Chi-squared test) or PD-duration (p = 0.487, Independent T-Test), see Table 1 for details. Figure 1 illustrates the recruitment process of the participants.
Participant characteristics, n = 243
SD, standard deviation; q1-q3, first-third quartile; Walk-12G = Generic Walk-12; GSE = General Self-Efficacy Scale; PD = Parkinson’s disease; H&Y = Hoehn & Yahr; UPDRS = Unified Parkinson’s Disease Rating Scale (part III = motor examination); FOGQsa = self-administered version of the Freezing of Gait Questionnaire; GDS-15 = the 15-item Geriatric Depression Scale; NMSQuest = Nonmotor Symptoms Questionnaire; NHP-EN = Energy subscale of the Nottingham Health Profile; MoCA = Montreal Cognitive Assessment. Possible scoring range, scoring direction: a0–42, higher = worse; b10–40, higher = better; c1–5, higher = worse; d - f0–4, higher = worse; g0–15, higher = worse; h0–30, higher = better. iMissing data due to they were unable to perform or complete the timed Chair Stand Test. Those who declined to participate (n = 157) had a mean (SD) age of 72 (9.8) years, PD duration (n = 129) of 9.2 (6.4) years, and were 52 % men.
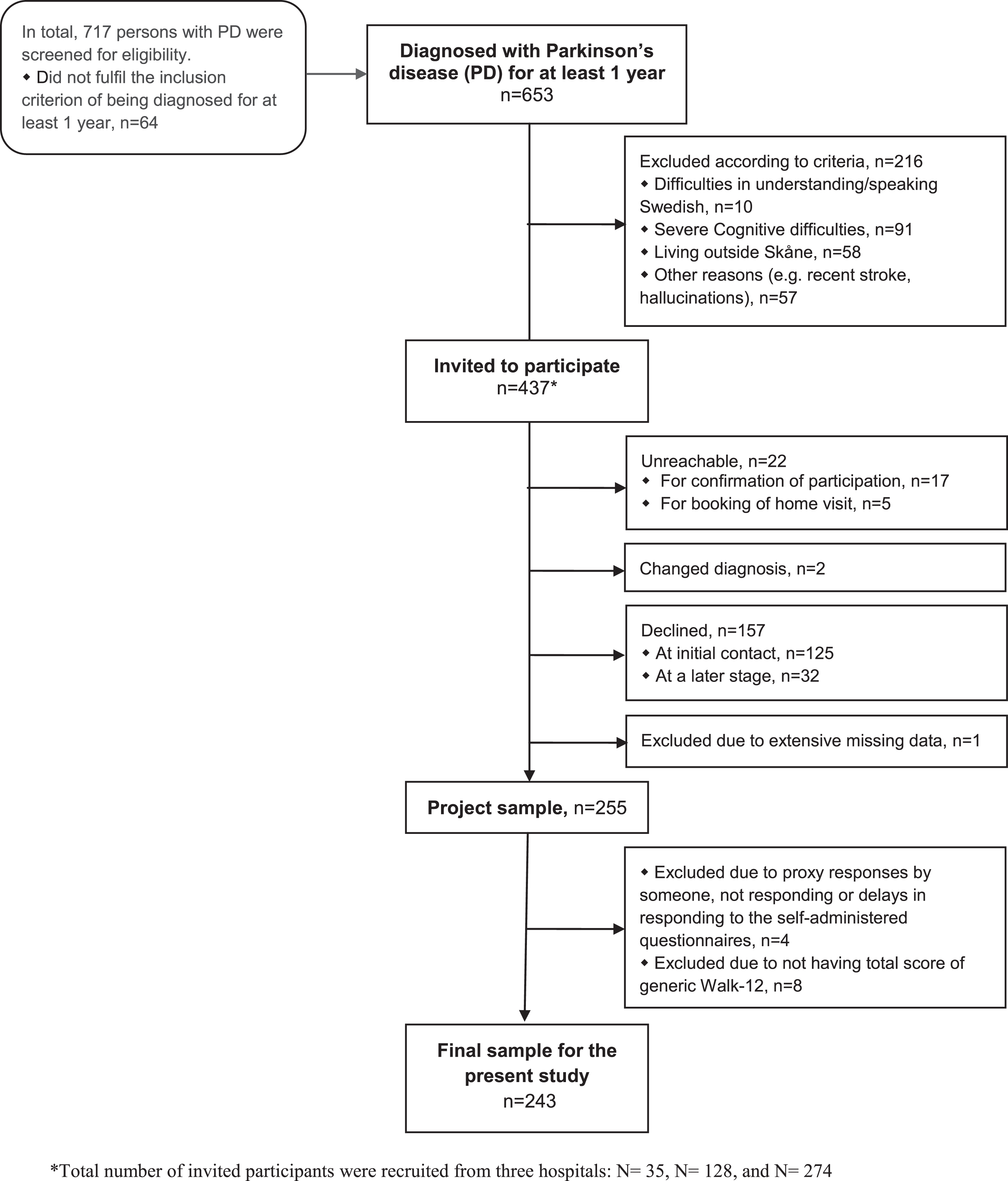
Flow diagram: the recruitment process of participants.
Data collection
Variables and Instruments
In addition to the instrument descriptions below, details regarding the self-administered questionnaires, interview questions, observations and clinical assessments are presented as footnotes in Table 1 and in the study protocol [26].
Perceived walking difficulties
Perceived walking difficulties was assessed by using the Walk-12G [27]. This instrument includes 12 items that concern perceived walking difficulties during the past two weeks in relation to, for example, the need for support when walking (indoors and outdoors), stair climbing, maintaining balance, distance, slowness, effort, and the need for concentration. Items 1–3 have three response categories (scored 0–2) whereas items 4–12 have five (scored 0–4). The possible total score ranges from 0 to 42 (higher = worse). The Walk-12G has been shown to be reliable and valid in people with PD [27].
Independent variables
The independent variables represented personal, social environmental and disease-related factors. They were selected based on findings from prior research [6–8, 25] and/or their clinical relevance for rehabilitation.
Personal and social environmental factors
Data on personal factors included age (years), sex (man/woman) and general self-efficacy. The General Self-Efficacy Scale (GSE) was used, which is scored 10–40 (higher = better/stronger general self-efficacy) [28]. Data on social environmental factors were collected with structured questions on social support and living situation. Social support was addressed by the question: “Is there someone around, who could assist you in case you would need some help and support?” If responding yes, the relationship to the assisting person/s was specified. The three response categories were recoded as social support from partner, other than partner or none. A dichotomous question targeted the living situation (living alone/not alone).
Disease related factors- severity, motor and non-motor symptoms
Disease severity was assessed according to Hoehn and Yahr (HY) [29], which ranges from stage I (unilateral involvement) to stage V (confinement to bed or wheelchair unless aided). The postural response in relation to an external perturbation (postural instability, item 30) as well as bradykinesia (item 31) were assessed according to the motor examination (part III) of the Unified PD Rating Scale (UPDRS) [30]. These two items (scored 1–4, higher = worse) were dichotomized; those with scores≥1 on item 30 were categorized as having postural instability whereas those with scores≥1 on item 31 were categorized as having bradykinesia. FOG was assessed according to item 3 (scored 0–4, higher = worse) of the self-administered version [31] of the FOG questionnaire [32], i.e. FOGQsa. Those scoring≥1 were categorized as “freezers” [33]. Lower extremity function was assessed with the timed Chair-Stand Test [34, 35]; the time (seconds) for completing five repetitions as fast as possible was registered.
Non-motor symptoms included depressive symptoms, anxiety, symptoms of orthostatic hypotension, fatigue, cognitive function and pain. Depressive symptoms were assessed with the 15-item Geriatric Depression Scale (GDS-15, interview-administered), scored 0–15 (higher = worse) [36]. Anxiety and orthostatic hypotension were assessed with two dichotomous (No/Yes) items (nos. 17 and 20) of the self-administered Nonmotor Symptoms Questionnaire (NMSQuest) [37]. Fatigue was assessed with the self-administered Energy subscale of the Nottingham Health Profile (NHP-EN) [38]; those who affirmed at least one out of three dichotomous (Yes/No) questions (tired all the time, everything is an effort, soon out of energy) were classified as having fatigue [39]. Cognitive functioning was assessed by using the Montreal Cognitive Assessment (MoCA), scored 0–30 (higher = better) [40]. Pain was assessed by the dichotomous (No/Yes) question “Are you bothered by pain?”
Statistical analyses
Categorical variables are described by number of participants (percentage), while ordinal and continuous variables are expressed by medians (first and third quartiles, q1-q3), or means (SD). Pearson (r) or Spearman (rs) correlations were used to assess relationships among independent variables (i.e., personal, social environmental, PD-related factors) in order to identify any multi-collinearity. Because the results from both correlation matrices were almost the same, we have used Pearson (r) correlations throughout. There was a sign of multi-collinearity (r >0.7) between ‘Postural response (item 30, UPDRS)’ and ‘Disease severity’ as well as between ‘Social support’ and ‘Living alone’. Disease severity (HY) was omitted since it is not a modifiable factor, whereas social support was omitted due to a skewed distribution of data (only two participants did not receive any social support).
Univariable linear regression analyses were used to investigate the unadjusted relationship of each independent variable and the dependent variable (Walk-12G scores). In order to avoid leaving out a confounding variable, we decided to include all variables with a p-value < 0.3 in the multivariable analysis. Probability values (P) for all independent variables were inspected and the variable with the highest p-value was manually removed. This procedure continued until all independent variables in the final model had p-values < 0.1, which became the final model. The strength of the relationship between each independent variable and the dependent variable was assessed by the standardized regression coefficient (β).
In a second multivariable model, the timed Chair Stand Test was excluded. This since 31 participants were unable to perform or complete the test. That is, the second model was computed due to the concern that the results might not be possible to generalize to people with poor lower extremity function.
The significance level applied was <0.05. All statistical analyses were performed using SPSS Windows 23.0 (IBM SPSS Inc., Chicago, IL, USA, Released 2015).
RESULTS
The mean (SD) Walk-12G score was 15.8 (11.0). The results from the univariable analyses are presented in Table 2. A total of 15 variables of interest were included in univariable analyses and all these variables turned out as significant (p < 0.05). Of all 15 variables, FOG explained the largest amount of variability (β= 0.505, p < 0.001) of perceived walking difficulties, whereas sex (women) explained the least (β= 0.157, p = 0.014). All these 15 variables were entered into the multivariable linear regression model.
Simple linear regression analyses with Generic Walk-12 scores as the dependent variable in people with Parkinson’s disease, n = 243
GSE = General Self-Efficacy Scale; PD = Parkinson’s disease; UPDRS = Unified Parkinson’s Disease Rating Scale (part III = motor examination), FOGQsa = self-administered version of the Freezing of Gait Questionnaire; GDS-15 = the 15-item Geriatric Depression Scale; NMSQuest = Nonmotor Symptoms Questionnaire; NHP-EN = Energy subscale of the Nottingham Health Profile; MoCA = Montreal Cognitive Assessment. 1Higher scores = better, 2Higher scores = worse. Missing data is described in Table 1.
The multivariable linear regression analysis resulted in eight statistically significant independent variables that explained 56.3% of the variance in perceived walking difficulties (Table 3). The strongest independent variable was FOG (β= 0.265, p < 0.001). It was followed by general self-efficacy (β= –0.242, p < 0.001), fatigue (β= 0.204, p < 0.001), PD duration, (β= 0.178, p < 0.001), lower extremity function (β= 0.130, p = 0.013), orthostatic hypotension (β= 0.126, p = 0.014), bradykinesia (β= 0.120, p < 0.017), and postural instability (β= 0.112, p = 0.024) (Table 3).
Multiple linear regression analysis with Generic Walk-12 scores as the dependent variable in people with Parkinson’s disease, n = 212a
FOGQsa = self-administered version of the Freezing of Gait Questionnaire; GSE = General Self-Efficacy Scale; NHP-EN = Energy subscale of the Nottingham Health Profile; PD = Parkinson’s disease; NMSQuest = Nonmotor Symptoms Questionnaire; UPDRS = Unified Parkinson’s Disease Rating Scale (part III = motor examination); 1The following 15 independent variables were included: Sex, age, living alone, general self-efficacy, PD duration, postural instability, bradykinesia, freezing of gait, lower extremity function, depressive symptoms, anxiety, orthostatic hypotension, fatigue, cognitive function, pain. aThe final model included the participants who had data for lower extremity function (whereof 36 participants in HY stage IV and none in stage V). bStepwise linear regression was conducted with all the independent variables included in the final model to get the change in R2 values.
After excluding the chair-stand test and rerunning the analysis, seven statistically significant independent variables explained 53.4% of the variance in perceived walking difficulties. The strongest independent variable was FOG (β= 0.275, p < 0.001). It was followed by fatigue (β= 0.236, p < 0.001), general self-efficacy (β= –0.225, p < 0.001), PD duration, (β= 0.173, p < 0.001), bradykinesia (β= 0.164, p = 0.001), postural instability (β= 0.107, p = 0.025) and orthostatic hypotension (β= 0.103, p = 0.036) (Table 4) None of the final multivariable models included any participant in HY stage V.
Multiple linear regression analysis with Generic Walk-12 scores as the dependent variable in people with Parkinson’s disease after excluding the variable chair stand test, n = 243
FOGQsa = self-administered version of the Freezing of Gait Questionnaire; NHP-EN = Energy subscale of the Nottingham Health Profile; GSE = General Self-Efficacy Scale; PD = Parkinson’s disease; UPDRS = Unified Parkinson’s Disease Rating Scale (part III = motor examination); NMSQuest = Nonmotor Symptoms Questionnaire; 1The following 14 independent variables were included: Sex, age, living alone, general self-efficacy, PD duration, postural instability, bradykinesia, freezing of gait, depressive symptoms, anxiety, orthostatic hypotension, fatigue, cognitive function, pain. aStepwise linear regression was conducted with all the independent variables included in the final model to get the change in R2 values.
DISCUSSION
Our study aimed to identify factors that independently contribute to perceived walking difficulties in people with PD. We identified FOG as the strongest independent variable in relation to perceived walking difficulties in people with PD, followed by general self-efficacy, fatigue, PD duration, lower extremity function, orthostatic hypotension, bradykinesia and postural instability. To the best of our knowledge, this is the first study that investigated factors that independently contribute to perceived walking difficulties in people with PD.
The findings indicate that FOG should be the primary target when addressing perceived walking difficulties in people with PD. That FOG is of importance for walking difficulties corroborates previous research [6, 19–23]. For example, persons with PD have described that FOG negatively influences community walking and perceived participation [22, 23]. Our findings further underline the importance of addressing FOG to improve walking in people with PD. For example, cueing strategies may facilitate walking if the person has FOG episodes [41, 42].
General self-efficacy was the second strongest contributing factor to perceived walking difficulties in people with PD, which to the best of our knowledge is a novel finding in PD-research. Based on data from the larger project [26] that this study is part of, general self-efficacy has been shown to independently contribute to life satisfaction [43], but not to concerns about falling [44] in people with PD. Other PD-studies have reported that self-efficacy is of importance for engagement in exercise [45] and self-management [46]. Moreover, support by family, healthcare professionals and others has been reported as important for both self- efficacy and self-management in PD [41, 46]. One PD intervention study that included a self-management approach, reported no statistically significant improvements in walking activity and endurance [47]. Further studies are needed to investigate whether a self-management approach is beneficial for walking ability among people with PD. A narrative literature review [48] described that the Chronic Disease Self-Management Program developed by Lorig et al., [49] has a positive impact on self-efficacy. According to Bandura self-efficacy is “the belief in one’s capabilities to organize and execute the courses of action required to manage prospective situations” [50, p. 2] and is a predictor of behavior that influences the choice of activities and motivation. Persons with high self- efficacy are more likely to pursue an active role in goal setting and coping as well as adhere to prescribed regimens [51]. All considered, our findings add to the current body of literature and suggest that general self-efficacy is an important aspect to consider in PD care and rehabilitation.
In the present study, fatigue was the third strongest contributing factors to perceived walking difficulties. Previous PD-studies have shown that fatigue is associated with walking economy [52], and with lower levels of self-reported [53, 54] as well as objectively measured [55] physical activity. Moreover, lower limb muscle fatigue (i.e., physical fatigue) is associated with objectively measured gait parameters in people with PD [9]. Although further studies are needed to understand the association between fatigue and walking difficulties, one explanation may be that fatigue induces difficulties in maintaining attention [56]. This as attention has been shown to be of importance for walking in people with PD [57, 58]. Although attention is a cognitive function, global cognitive functioning (as assessed with MOCA) did not contribute to perceived walking difficulties in this study. Rerunning the analyses and substituting the MOCA total score by its domain scores (results not shown but available on request) yielded largely similar results. This applied for both models and when using the original domain scores [59] (i.e. Visuospatial/Executive, Naming, Attention, Concentration and Calculation, Language, Abstraction, Delayed recall and Orientation) as well as when using more recent suggested domain scores [60].
One factor that independently contributed to perceived walking difficulties needs specific attention, that is, lower extremity function as assessed with the chair stand test [34, 35]. It should be noted that 31 participants (whereof 19 in HY stage IV and 3 in stage V) were unable to complete this test. After excluding this variable in the multivariable analysis, the results remained largely similar. This consistency indicates that all the identified factors independently contribute to perceived walking difficulties regardless of their interaction with lower extremity function. That lower extremity function turned out as a significant contributing factor highlights the need of promoting lower extremity strength [61, 62]. According to recommendations [63, 64], strength training should be combined with training that includes other components such as balance. This is further underlined by the fact that postural instability contributed independently to perceived walking difficulties in this study.
The postural response was assessed in relation to an external perturbation. Several studies showed that training in responding to external perturbations [65–67] has some effect on gait, balance and balance-related activity performance. Balance training should challenge the person with PD [68] and home based training on postural instability seems to have no beneficial effect in people with PD [63].
Strengths, limitations and future perspectives
We consider it a strength that we included the full spectrum of PD severity although some readers might argue that we should have excluded those in HY stage V. It should be noted that none of those in HY stage V (n = 3) were included in any of the two final models since they had missing data on some of the independent variables. To clarify, the final model of any regression analysis includes only those that have complete data on all the included independent variables.
Another strength is that we used multivariable analyses and that the sample size allowed us to consider a broad variety of explanatory factors. Even if the regression model explained 56.3% of the variance in the dependent variable, there are additional independent variables of interest for perceived walking difficulties in people with PD. For example, the social environmental factors included represent a limited portion of the wide range of possible environmental factors that might be of importance for perceived walking difficulties, for example, crowds, inclement weather and uneven/slippery surfaces [19, 24]. In addition, although balance problems are complex in people with PD and may incorporate several aspects, the present study only addressed the ability to counteract an external perturbation. Future studies should preferably also incorporate additional aspects of balance control. We used a rather coarse indicator of fatigue and it might be of interest to incorporate assessments of different types (i.e., mental and physical) of fatigue in future studies [69, 70].
The Walk 12-G is a PRO instrument. According to the Food and Drug Administration (FDA), “use of a PRO instrument is advised when measuring a concept best known by the patient or best measured from the patient perspective” [71]. The responses may vary due to cultural differences in relation to the targeted construct [72] and using a PRO instrument may be disadvantageous for people with disabilities and low literacy [73]. A previous psychometric study of Walk-12G did, however, report satisfactory data completeness in PD as well as multiple sclerosis samples [27].
The cross-sectional design makes it impossible to infer any causal relations. Longitudinal studies are needed to identify predictive factors but also to gain an increased understanding of how perceived walking difficulties evolve over time. Since this is the first study that used multivariable analysis to identify contributing factors to perceived walking difficulties in people with PD, the findings need to be replicated in other PD-samples as well as in different national contexts.
CONCLUSIONS
This study identified eight contributing factors for perceived walking difficulties in people with PD. FOG was the most important factor, followed by general self-efficacy, fatigue, PD duration, lower extremity function, orthostatic hypotension, bradykinesia and postural instability. That is, motor and non-motor symptoms as well as personal factors (i.e., general self-efficacy) seem to be of importance when addressing perceived walking difficulties among people with PD. Longitudinal studies are needed to identify predictive factors and understand how perceived walking difficulties evolve over time. With such knowledge at hand, interventions addressing modifiable factors could be developed, ultimately enhancing walking ability in people with PD.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The authors wish to thank reg. nurses Jan Reimer, Susanne Lindskov and Eva Aronsson who were involved in the selection of participants, and reg. occupational therapists Maya Kylén and Malin Mejstad for the data collection effort. This project was funded by the Strategic Research Area in neuroscience at Lund University, Sweden (MultiPark), the Swedish Research Council, the Ribbingska Foundation in Lund, the Greta and Johan Kock Foundation, Sweden, the Swedish Association of Persons with Neurological Disabilities (NHR), Sweden; Norrbacka-Eugenia Foundation Stockholm, Sweden; NEURO Sweden, and The Swedish Parkinson Foundation. The study was conducted within the context of Centre for Ageing and Supportive Environments (CASE) at Lund University, financed by the Swedish Council for Working Life, Public Health and Welfare (Forte).
