Abstract
INTRODUCTION
Parkinson’s disease (PD) is a chronic and progressive extrapyramidal disorder characterized by a decline in spontaneous movement, tremor, and rigidity [1]. The diagnosis of PD is currently based on the application of specific diagnostic criteria, which increases accuracy but reduces sensitivity. In addition, diagnosis can be difficult in early stages when symptoms may be mild, atypical, or lack a robust response to medications. In fact, approximately one in five patients with Parkinson symptoms are classified with an uncertain disorder [2] and eventual neuropathologic confirmation in PD patients with early, untreated symptoms has been demonstrated to be as low as 26% [3]. An early and accurate diagnosis of PD is critical for patient care and counseling as well as research into biomarker discovery and disease-modifying therapies. In addition, misdiagnosis can lead to disability if effective treatments are delayed, and inappropriate medical examinations and therapies may expose patients to unnecessary risks.
While the definitive diagnosis of PD requires histological demonstration of intraneuronal Lewy body inclusions in the substantia nigra pars compacta and loss of nigrostriatal dopaminergic neurons, demonstrating these changes is impractical during life. In this context, new neuroimaging techniques have been developed to aid diagnostic accuracy. [123I]-FP-CIT (DaTSCAN) is a radiopharmaceutical that binds with high affinity to the dopamine transporter, a presynaptic protein located on the membrane of dopaminergic neuron terminals in the striatum. Thus, DaTSCAN serves as a surrogate marker for nigrostriatal projections by estimating the density of dopamine transporter sites. Indeed, studies have demonstrated sensitivity for identifying dopamine cell loss in 92–95% of patients with clinically certain PD [4–6].
Given the ability to identify dopamine cell loss, DaTSCAN has demonstrated utility in differentiating between PD and non-neurodegenerative tremor disorders in situations where diagnostic confusion exists. In these instances, DaTSCAN has been shown to significantly impact clinical management, while improving diagnostic accuracy and confidence [2, 7]. However, as imaging has become more available, the role of DaTSCAN has expanded to encompass diagnostic confirmation in patients with early suspected PD. Recent reviews have challenged this approach [8], arguing that functional neuro-imaging has added no advantage over clinical diagnostic accuracy. To address this question, we conducted a randomized, prospective study of initial vs. delayed DaTSCAN in de novo PD patients. The goal of this investigation was to assess the impact of in vivo dopaminergic imaging on clinical management, motor function, and patient expectations in subjects with early,probable PD.
METHODS
Trial design
This was a randomized, prospective, 54-week, comparative clinical trial to assess the impact of DaTSCAN SPECT imaging on clinical management, motor function, patient expectations, and safety in patients with early, de novo PD. To assess the impact of DaTSCAN on clinical management and motor outcomes, subjects were randomly assigned to either initial or delayed imaging (Supplementary Figure 1). The initial imaging group underwent a total of three DaTSCANs throughout the study (at months 1, 6 and 12), while those in the delayed group had two imaging sessions (at months 6 and 12). As imaging has been shown to increase diagnostic confidence in certain Parkinson populations, a scripted interaction was used to describe the study rationale, design, and procedures to avoid adding individual physician confidence to the suspected PD diagnosis.
Subjects
This prospective study included twenty subjects recruited from the Parkinson Disease and Movement Disorder Center at Duke University Medical Center, Durham, NC. All subjects met diagnostic criteria for early, probable PD (presence of at least 2 of the cardinal signs, without evidence of atypical features, and disease severity not greater than Hoehn and Yahr stage III [9]). Any patient with current or previous exposure to anti-Parkinson therapy >8 weeks, significant cognitive impairment (Montreal Cognitive Assessment (MOCA) <26 [10]), or evidence of secondary Parkinsonism was excluded. Unified Parkinson’s Disease Rating Scale (UPDRS) [11] was used as a measure of disease severity. The clinical characteristics of participants are summarized in Table 1. Briefly, patients were matched for mean age of PD onset: initial imaging group = 56.33 years (10.59) vs delayed imaging group = 64.67 years (11.15), p = 0.132; and disease severity: initial imaging group = UPDRS III at study entry 19.6 (7.26) vs. delayed imaging group = 24.8 (9.07), p = 0.306.
Baseline patient characteristics. Patients were equally matched for age at PD onset, disease duration, and disease severity at baseline
The study was approved by the Duke University Medical Center IRB, and all subjects gave written informed consent prior to performing any study-related procedures.
Study treatments and assessments
All eligible subjects were evaluated by a movement disorders specialist to enhance diagnostic accuracy and fulfillment of enrollment criteria. The baseline/randomization visit included assessments of demographics, medical history, neurological evaluation, UPDRS [11], Hoehn and Yahr staging [9], MOCA [10], and establishment of PD diagnosis. Patients were randomized to undergo either immediate or delayed DaTSCAN imaging. All subjects were seen within 30 days of randomization, and for those randomized to the initial imaging group, DaTSCAN was completed prior to this visit. During treatment initiation, an assessment of planned clinical management was discussed with a movement disorders specialist. When appropriate, the results of the most recent DaTSCAN were reviewed with the participants prior to any discussion of potential interventions. All patients received pertinent clinical information and a written summary of planned treatment interventions. Medical therapy included anticholinergics, MAO-B inhibitors, dopamine agonists, amantadine or levodopa [12] and followed a tiered treatment schedule (Supplementary Table 1). Non-pharmacologic interventions included physical therapy, speech therapy, occupational therapy, and regimented exercise programs. Subjects returned at weeks 16, 28, 40, 54 for a comprehensive clinical evaluation, assessment of the treatment plan, and to review completed imaging (performed at weeks 28 and 54). The need for any additional medication, withdrawal of medication, or additional diagnostic evaluation was discussed. Any changes from the initial treatment plan were recorded.
Clinical evaluations were performed by a separate, blinded neurologist, and included UPDRS, H&Y, vital signs, and review of any adverse events.
Imaging techniques
Each imaging session consisted of intravenous administration of a single dose of DaTSCAN via the antecubital vein while in a supine or recumbent position, followed by SPECT imaging of the head 3 to 6 h later. Thyroid blocking was performed according to hospital routine. Images were assessed visually by an expert in neuroimaging who was blinded to clinical history and examination. There was an option to have a second reader evaluate the imaging if the primary reader felt that the image interpretation was ambiguous. Images were classified as normal, abnormal type 1, abnormal type 2, or abnormal type 3, as per convention (Fig. 1). The normal or abnormal determination was made by assessing the extent (as indicated by shape) and intensity of the striatal signal. The treating neurologist reviewed each SPECT image in detail with the individual subject scanned.
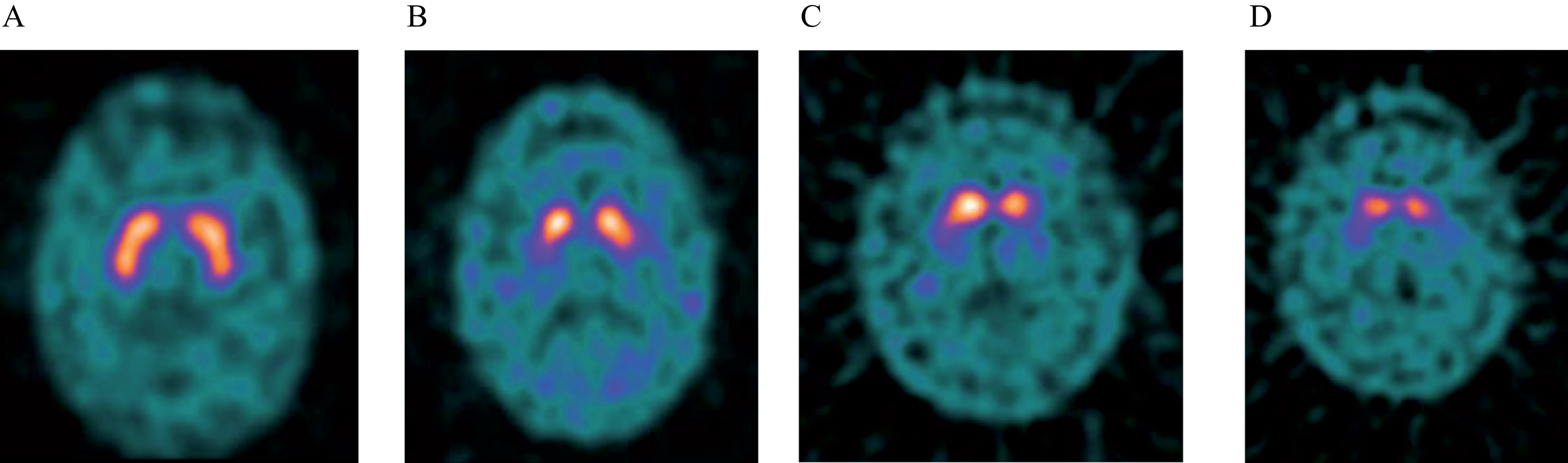
Example of DaTSCAN images in normal and PD cases. A) Normal tracer uptake bilaterally in putamen and caudate nuclei. B) Abnormal image grade 1: asymmetric uptake with normal/near normal putamen activity in one hemisphere and with a more marked reduction in the contralateral putamen. C) Abnormal image grade 2: significant bilateral reduction in putamen uptake with activity confined in the caudate nuclei. D) Abnormal image grade 3: virtually absent uptake bilaterally affecting both putamen and caudate nuclei.
Statistical analysis
The primary outcome measure was the frequency of deviation from the initial treatment plan from baseline to 54-weeks between the two groups. Secondary endpoints included a targeted evaluation of treatment changes at 6 months, coinciding with the first DaTSCAN for the delayed group. Further comparisons also spanned the duration of the study and included UPDRS part III, disease related expectations and optimism (as measured by Life Orientation Test – Revised (LOT-R)), and physician prescribing practices. Summary statistics means/standard deviations/median/range for continuous variables (age, scales of UPDRS, LOT-R, etc.) and counts/percents for categorical variables respectively were used to summarize data. Group comparisons assumed intention-to treat. Comparison between early vs. delayed treatment and continuous variables were conducted using Wilcoxon-test. Association between treatments and primary outcome of repeated measures of UPDRS were examined using mixed models. For both groups, safety parameters, including adverse events (AEs) and serious adverse events, were collected from enrollment until completion of the study. Statistical significance was examined at alpha = 0.05. Statistical analyses were performed with the SAS 9.4 statistical software (SAS Institute Inc.,Cary, NC).
RESULTS
To evaluate the impact of [123I]-FP-CIT (DaTSCAN) on the clinical management of patients with early, probable PD, we conducted a novel prospective study in 20 de novo subjects randomly assigned to initial or delayed imaging. The baseline characteristics of the randomized patients were matched in each group and are detailed in Table 1.
Treatment
An initial treatment plan was agreed upon between the treating physician and patient, and all changes in treatment were tracked throughout the study. For subjects randomized to the initial DaTSCAN group, imaging results were discussed prior to the initiation of any treatment. Thus, any added diagnostic confidence gained from in vivo dopaminergic imaging was incorporated into decisions on treatment. There was no significant difference in the number of treatment changes between the two groups: initial imaging group = 4.2 (SD:2.74) vs. delayed imaging group = 2.3 (SD:2.0, p = 0.107) (Table 2). Furthermore, we completed a targeted comparison of treatment changes specifically coinciding with the first imaging visit for the delayed group and the second scan for the initial imaging group (week 16): 50% in the delayed group and 66.67% in the initial group had medication changes, which was not significantly different (p = 0.6372 using Fishers Exact Test).
Treatment outcomes in patients randomized to initial or delayed imaging with Ioflupane [123I]-Injection (DaTSCAN). There was no difference in outcomes between the two groups in terms of motor function, clinical management, treatment decisions, and patient expectations of outcomes
aFrom initial screening visit to week 54; bThroughout the entirety of the study; cRank sums.
In addition, there was no difference between groups in medication requirements or decision-making. For example, six patients in the initial imaging group and seven in delayed imaging group required levodopa at some time during the study (p = 0.639). Further analysis showed no differences between groups in the number treated with a dopamine agonist: initial imaging group n = 2 (20%) vs. delayed imaging group n = 5 (50%), (p = 0.16), or final levodopa equivalent daily dose (LEDD): initial imaging group mean = 211(SD:204.96) vs. delayed imaging group mean = 172 (SD:176.43, p = 0.731). On a qualitative level, there was no indication that early vs delayed DaTSCAN impacted decision making. The most common treatment change in both groups was an adjustment of medication dose, for example: titrating ropinirole from 3 mg three times per day to 4 mg three times per day.
Motor function
Motor function was assessed at each study visit by a blinded movement disorders neurologist. At the end of 12 months, there was no difference in motor function, as measured by UPDRS III, between the two groups: initial imaging group = 17.5 (SD:9.35) and delayed imaging group = 19.1 (SD:9.07, p = 0.91). Analyzing repeated values throughout the 12-month study using a mixed model, there was no significant treatment by time interaction on summed scales of UPDRS III items (p = 0.90), suggesting no acute impact on motor function following imaging review with the treating clinician (Fig. 2). In addition, the change in UPDRS III score from baseline to end-of-study was greater in the delayed group (mean = –2.1, SD:7.46) vs. delayed (mean = –5.7, SD:6.55), but did not reach statistical significance (p-value = 0.343).
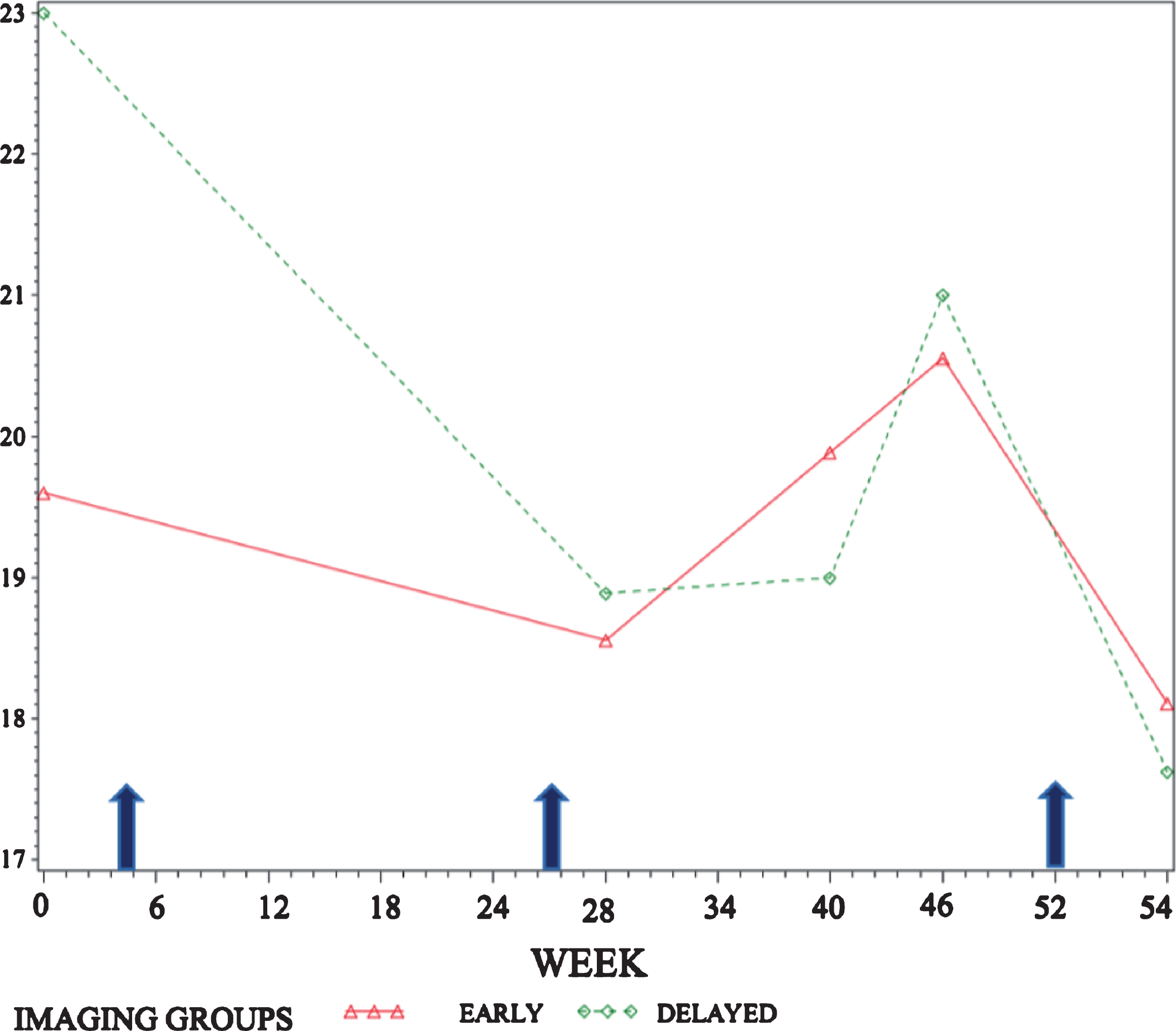
Mean repeated values of UPDRS III from screening visit through week 54 (visit 10) by treatment group (early vs. delayed imaging). There is no significant treatment by group interaction on summed scales of UPDRS III (p-value of visit by treatment interaction p-value = 0.90). Patients in the initial imaging group underwent DaTSCAN at diagnosis and weeks 26, and 52; patients in the delayed imaging group underwent DaTSCAN at weeks 26 and 52 respectively (indicated by arrows). All patients were evaluated by an independent, blinded rater at weeks 0, 28, 40, 46, and 54.
Patient expectations and optimism
Despite any added time of diagnostic confidence in the initial imaging group, there was no difference in patient expectations and optimism as measured by LOT-R between groups: initial imaging group = 16.3 (SD:3.3) and delayed imaging group = 15.2 (SD:2.3), Wilcoxon Scores (Rank Sums) p = 0.350.
Safety
No adverse events occurred during the course of this study.
DISCUSSION
In this study we explored the impact of DaTSCAN added to the diagnostic algorithm in subjects with probable PD, where no clinical uncertainty exists. To evaluate this, we utilized a delayed-start design where participants were randomized to undergo in vivo dopaminergic imaging immediately upon initial PD diagnosis (with two additional scans at 6 and 12 months), or imaging delayed to 6 months after initial PD diagnosis (with one additional scan at 12 months). Subjects with early, untreated PD were eligible for this study and all participants were required to be free of any Parkinsonian treatment at the time of enrollment. In this controlled and consistent patient population, any differences in treatment approach or functional outcome could be attributed to the additional diagnostic confidence added by dopaminergic imaging. We found that in patients with early, probable PD, DaTSCAN affords no added benefit above clinical diagnosis alone in terms of clinical management, motor function, progression, or patient expectations of their disease.
Previous investigations have demonstrated the utility of DaTSCAN in patients with early, suspected Parkinson symptoms, but have focused on subjects where diagnostic uncertainty exists [2, 14]. This has been a valuable area of exploration given the breadth of clinical findings in Parkinson disorders and the large number of potential underlying etiologies, which often makes accurate diagnosis a challenge. Since the course of disease progression, success of therapy, and prognosis may differ substantially between disorders, improved diagnostic accuracy is extremely important. Indeed, dopaminergic imaging results frequently provide a more accurate diagnosis than clinical evaluation alone [15] and DaTSCAN is FDA-approved to aid in the differentiation between Parkinson syndromes and movement disorders that are not associated with nigrostriatal neuronal loss. Furthermore, DaT imaging has also proven to significantly impact clinical management in this patient population, most frequently influencing medication decision-making [7].
However, DaTSCAN is also commonly used as a confirmatory test in support of the clinical diagnosis of suspected PD. As availability has become more widespread, some have supported its incorporation into the standard diagnostic algorithm [16]. This approach has also been opposed, and in the absence of neuropathological validation of disease, mathematical models have demonstrated equivalent diagnostic accuracy between a clinical exam and DaTSCAN in patients with early disease symptoms [8]. As PD is a relatively common neurodegenerative disorder, affecting some 1%–2% of the older population, the large-scale use of dopaminergic imaging as a diagnostic tool may have substantial implications in terms of patient safety, payer costs, and clinical management. To help address these concerns, we sought to explore the influence of DaTSCAN on clinically meaningful measures in patients with early, probable PD, where there is no diagnosticuncertainty.
To our knowledge, this is the first controlled, prospective trial using DaTSCAN with selected endpoints in patients with early, suspected PD. Our results demonstrate that the addition of dopaminergic imaging to the diagnostic algorithm does not alter the clinical management of patients with early PD, where no diagnostic uncertainty exists. These results are extremely important given the potential for widespread use in this patient population. Antonini et al previously demonstrated potential cost savings associated with DaTSCAN imaging employed in patients with clinically uncertain syndromes. In these patients, the costs of testing are recouped through improved diagnostic accuracy, thus reducing the need for further diagnostic procedures, and increasing the time of potentially beneficial therapy [18]. However, in patients without diagnostic uncertainty, DaTSCAN imaging, and its associated costs, appear to be redundant and unnecessary for optimization of clinical management.
Furthermore, we found no impact of DaTSCAN imaging on motor outcomes in subjects with de novo PD throughout the study. Subjects underwent regular motor evaluation by an independent blinded rater, demonstrating no significant difference in motor function at any time point during the 12-month study period (Fig. 2). Presynaptic DaT imaging in clinical trials is an increasingly common practice for confirmation of PD diagnosis, and includes investigations of disease-modifying therapies in patients with early, probable PD [19, 20]. For these studies, it is imperative to understand how imaging may impact the clinical management of patients and their functional outcomes. Our results show that DaTSCAN imaging has no impact on potentially planned interventions and will not confound motor evaluations.
Finally, objective imaging data and further diagnostic confidence with DaTSCAN afforded no impact on patient expectations as measured by LOT-R (Table 2). The uncertainty associated with the current diagnosis of PD may lead to problems with adherence to medical therapy, which is an increasing concern when treating PD patients [21]. Suboptimal adherence to PD medication regimens has been identified in 60 – 70% of individuals [16, 21] and can result in decreased motor function, the development of motor fluctuations, a decline in quality of life, and potentially higher mortality [22, 23]. Poor medication compliance impacts PD symptoms, and likely influences health care costs, long-term side effects, and hospitalizations [21, 22]. In addition, expectations have been shown to affect clinical motor performance in PD patients, [24–26], possibly related to endogenous dopamine release and changes in subthalamic nucleus neuronal firing [27–29]. However, our results show that imaging analysis does not have a meaningful effect on patient-reported expectations, and is unlikely to influence patient behaviors.
The small number of patients evaluated limits this study; with a total of 10 subjects randomized to both the immediate and delayed imaging groups. To illustrate this point, based on the change in UPDRS III in the two treatment groups (mean/SD of UPDRS in (early scan group: (–2.1 (7.46)) vs delayed scan group –5.7 (6.55)), with 10 subjects in each group for alpha = 0.05, the power of this study (with high and unequal variances in the 2 groups) is only 19.2%. However, this investigation was meant as an exploratory pilot study to probe for differences in outcome measures. Perhaps a larger cohort of patients would demonstrate an impact on clinical decision-making or functional outcomes with DaTSCAN imaging. Two strengths of this study support our findings. First, all motor and safety assessments were completed by an independent rater who was blinded to group assignment and treatment plan, thus avoiding potential bias when evaluating motor function and progression. Next, the unique design of this trial allowed analysis of individual outcome measures at multiple meaningful time points throughout the study. For example, differences in patient expectations could be compared in both groups as a whole, or contrasted at the time when the initial imaging group underwent their first DaTSCAN imaging and the delayed group had no imaging. This methodology provided close analysis of the impact of imaging over the course of thestudy.
Conclusion
Parkinson’s disease (PD) is a progressive movement disorder characterized by the loss of dopaminergic neurons in the substantia nigra pars compacta, leading to striatal dopamine depletion. The diagnosis of PD requires the demonstration of bradykinesia, along with tremor, rigidity, and/or gait disturbance on clinical evaluation. [123I]FP-CIT (DaTSCAN) imaging has been developed to evaluate adult patients with PD signs and provides utility when there is diagnostic uncertainty. Studies suggest that uncertainty exists in up to 20% of patients with Parkinson symptoms [30] and DaTSCAN imaging can improve diagnostic accuracy and influence treatment approach in these patients. However, whether presynaptic dopaminergic imaging is useful in patients without diagnostic uncertainty is less clear. This pilot study suggests limited utility of DaTSCAN imaging in patients meeting criteria for early, probable PD. In this patient population, where low diagnostic uncertainty exists, our exploratory findings demonstrate no impact on the clinical management or functional outcomes with the addition of DaTSCAN to the diagnosticalgorithm.
While our findings may help guide the diagnostic approach to patients with de novo PD, further research is needed. First, large clinicopathological studies are necessary to determine the true diagnostic accuracy of DaTSCAN imaging. In addition, close analysis of cost-effectiveness would assist in informed decision making for clinicians and patients. In the meantime, the current data does not support the expansive use of DaTSCAN in routine clinical practice.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This study was funded by a generous grant from GE Healthcare Worldwide. The sponsor played no role in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
