Abstract
We demonstrate the feasibility of estimating clinical tremor scores using an eating utensil with motion-sensing and tremor-cancellation technology in thirteen patients with tremor. Three experts scored hand tremor using the modified Fahn- Tolosa-Marin (FTM) scale. A linear model was trained to estimate tremor severity using the recorded motion signals. The average neurologist FTM score was 1.6±0.7 for PD and 2.6±0.7 for ET patients. The average model score was 1.6±0.7 for PD and 2.6±0.6 for ET. Correlation coefficient between the clinical and model tremor scores was 0.91 (
INTRODUCTION
Instrumented devices provide the opportunity to quantify disease symptoms in patients with essential tremor (ET) or Parkinson’s disease (PD). In current clinical practice tremor is assessed by rating scales and less often with calibrated sensors. Tremor assessment by a clinician is inherently subjective, depends on the experience, and it is relatively insensitive to small changes [1]. Quantitative tremor analysis using accelerometry, electromyography, and digital writing tablets has been shown to be useful in diagnosis and management of tremor [2]. However, this approach has not been widely accepted in clinical practice since diagnostic tools are not standardized and require expertise in data acquisition and analysis [3]. Furthermore, assessing tremor in the clinic provides extremely limited snapshot of patient’s daily function.
A handheld device called Liftware was developed as a more practical and effective alternative to treat tremor compared to other non-invasive tools such as weighted utensils and tremor-suppression orthoses [4–6]. Liftware utilizes active tremor-cancellation technology to sense the direction of tremor and move the attached implement (e.g. spoon) in the opposite direction (Figure 1S). In a recent clinical study, Liftware was shown to be effective in reducing tremor amplitude at the spoon tip during eating and transferring tasks in individuals with ET [7]. An eating utensil capable of quantifying tremor would provide a reliable, objective measurement during a fixed task that could be tracked daily.
The aim of this study was to demonstrate the feasibility and accuracy of estimating clinical tremor severity using a modified version of the Liftware device employing motion-cancelling technology and a new data-processing module with embedded inertial measurement capabilities.
MATERIALS AND METHODS
Patients
Patients were recruited from the UCSF Movement Disorders and Neuromodulation Center. Inclusion criteria were diagnosis of ET or PD with at least mild postural or action tremor. Exclusion criteria were symptoms other than tremor that would interfere with task performance (severe rigidity, bradykinesia or cognitive impairment). Patients were on their regular medications. Patients with deep brain stimulation (DBS) had the stimulator turned off for 30 minutes prior to testing. The study was approved by the Institutional Review Board and written, informed consent was obtained from all participants.
Tremor testing
Patients performed two tasks using the Liftware spoon device: 5 consecutive trials of moving dry cereal from bowl to mouth, and 5 trials of transferring cereal from bowl to bowl. Each task took about 30 seconds to perform. Each task was performed with the tremor cancellation feature turned on and off, in a randomized order and patients blinded to the condition (device off data was not collected for patients 1–4). Patients used dominant hand unless tremor was significantly more prominent in the non-dominant hand. Patient 6 had very severe dominant hand tremor and was unable to use the spoon so non-dominant hand was tested. Three movement disorder neurologists, blinded to tremor cancellation on or off, reviewed randomized video segments of each individual movement trial (20 videos, except 10 for patients 1–4) and scored the tremor according to the modified Fahn-Tolosa-Marin (FTM [8]) tremor rating scale (0 = no tremor, 1 = slight tremor, 2 = mild tremor with amplitude <2 cm, 3 = moderate tremor with amplitude 2–4 cm, 4 = severe tremor with amplitude >4 cm). Raters were instructed to focus on the patient’s hand and ignore tremor or movement at the attached spoon tip or food spillage, and to rate the most severe tremor observed. Scores from the 5 trials for each task were averaged for further analysis. Each patient was also scored on the complete clinical motor FTM scale by a single rater (SM).
Signal processing
Motion data was recorded at 50-Hz sampling rate using a tri-axial accelerometer and a tri-axial gyroscope housed in the device handle, and downloaded through the device’s cradle charger via a USB for off-line analysis using custom Matlab (MathWorks, Natick, MA) scripts (version R2015b). For each trial, the spectrogram of each of the six motion signals was computed separately with a 5-second analysis, 0.5 second shifts, and a 1024-point Fast Fourier Transform. The spectrograms were used to calculate the dominant power and frequency for each motion signal. These were then averaged across all motion signals to arrive at the average power and frequency for each task. An ordinary least-squares linear regression model was built using average neurologist score as dependent variable, and average tremor power and frequency as independent variables.
Statistical analysis
Tremor scores and power with spoon cancellation feature on vs. off were compared using Wilcoxon rank-sum test. A leave-one-subject-out cross validation approach was used for estimating model tremor scores, meaning that the model was trained on all the patients except the one for which it was estimating the score. Pearson correlation coefficient was used to compare model scores with average neurologists’ scores, and to compare spoon task scores with the standard FTM rating scale. A
RESULTS
Fifteen patients were recruited for the study, and 13 included in the analysis with clinical characteristics and tremor ratings detailed in Table 1 (two patients were excluded due to technical issues with data collection).
Clinical characteristics and tremor severity scores
1Fahn-Tolosa-Marin tremor rating scale: sum of motor scores (items 1–14). 2Fahn-Tolosa-Marin tremor rating scale for hand used in spoon testing: average of postural, action and activity scores (items 5, 6, 11–14). 3Average eating and transfer task score for all 3 neurologists, for both device conditions. 4Average eating and transfer task score for the model, for both device conditions. ET: essential tremor; PD: Parkinson’s disease; C/L: carbidopa/levodopa; DA: dopamine agonist; DBS: deep brain stimulation; FTM: Fahn-Tolosa-Marin.
The average neurologist score for the spoon tasks (eating and transfer) was 1.6±0.7 for PD and 2.6±0.7 for ET (mean±std). The neurologist task scores were unaffected by the tremor cancellation feature being on or off (
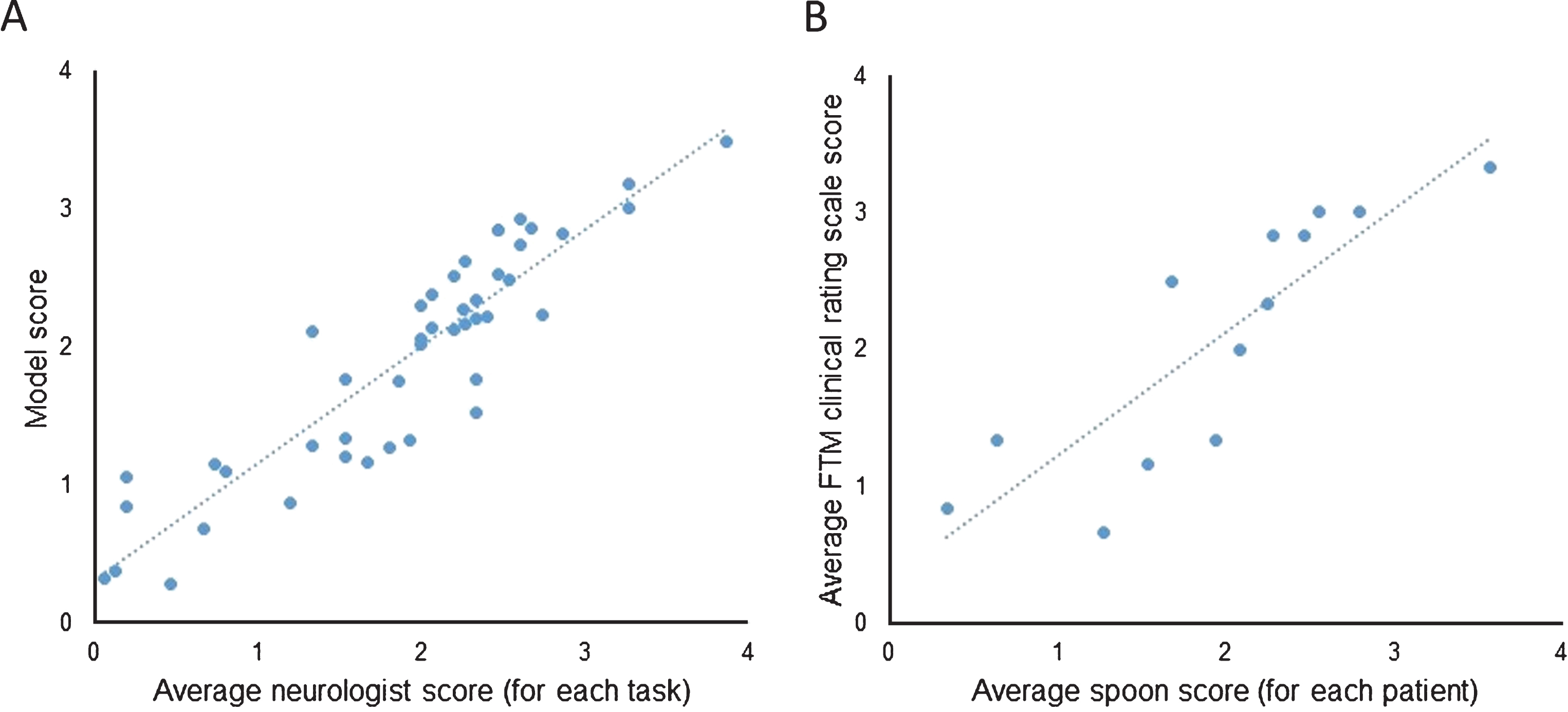
(A) Correlation between the average neurologist scores and Liftware’s linear regression model scores using leave-one-subject-out approach (
DISCUSSION
Our results demonstrate that Liftware can accurately estimate clinical tremor severity scores during eating and food transfer tasks using motion data recorded from embedded sensors. The model was developed to report tremor scores on a 0–4 point scale, similar to the commonly used FTM scale. Tremor severity during the spoon tasks correlated well with standardized FTM clinical tremor scale. Therefore, device results could be easily incorporated into routine clinical assessment and would complement existing clinical information.
There is a growing interest in the development of tools for assessing movement disorder symptoms outside the clinic. Several devices have been developed to monitor tremor in the home environment [9–12]. Some require specialized equipment and most instruct patients to perform standardized motor tasks at specified time intervals. Liftware is unique among these devices since it provides direct benefit to the user and places no additional demands on the patient, facilitating reliable collection of data. Furthermore, data gathering is performed during a fixed, well-defined eating event allowing meaningful comparisons over time. The ability to feed oneself is very important for quality of life so this type of monitoring would be particularly well suited for patient-centered outcomes. In practice, the use of this device in the home environment could more accurately capture patient’s typical clinical state and provide longitudinal monitoring to assist the clinician with therapy adjustments. The data would be available to the clinician through a web portal and would include metrics such as tremor amplitude, frequency, and estimated score at several time points throughout the day (typically during meal times, but a more precise protocol could be set for research studies). Additionally, this device could be used in conjunction with other wearable or non-wearable sensors that passively assess symptoms and behavior (such as gait and medication usage) to better inform a patient’s condition and guide therapy.
The limitations of the study include a small sample size and limited range of tremor severity. The spoon works best with mild to moderate tremors [7], and patients with severe tremor are less likely to use the tool limiting the information that could be gathered from this population. None of the PD patients exhibited dyskinesias, and although severe dyskinesia may prevent the use of the spoon, tremor score could theoretically be extracted since signal analysis is limited to peak tremor frequency which is higher than the typical 0.2–4 Hz dyskinesia activity [13], but this should be directly tested.
In summary, the Liftware device measures tremor during a task that is meaningful for quality of life, and it was previously shown to be provide direct benefit to the user making it potentially suitable for in-home monitoring. Daily evaluation of tremor has the potential to enhance clinical care and research study assessments which should be evaluated in future studies.
CONFLICT OF INTEREST
UCSF Movement Disorders and Neuromodulation Center received grant support from Verily Life Sciences to conduct this study. Authors Shoeb and Pathak are employees of Verily Life Sciences.
