Abstract
Background:
Serum neurofilament light chain (sNfL) levels are an increasingly employed tool for the assessment of active axonal injury in a variety of neurological disorders including polyneuropathy. Injury to the small nerve fibers can lead to small fiber neuropathy (SFN), a neurological condition which is clinically manifested by combination of burning pain, various sensory disturbances and symptoms of autonomic dysfunction. SFN is evident by identifying reduced epidermal nerve fiber density (ENFD) in skin biopsy.
Objective:
To assess the utility of sNfL measurement as a marker for active axonal injury in a population of patients who were referred for skin biopsy as part of the evaluation of chronic sensory symptoms.
Methods:
sNfL levels were measured in 94 patients at the time of skin biopsy. Clinical, electrodiagnostic, and imaging data were collected and neurological comorbidities including central nervous system disorders, large-fiber polyneuropathy, and radiculopathy which may increase sNfL levels were reviewed.
Results:
Eighty-six patients had pathological skin biopsy result supporting the diagnosis of SFN. sNfL was increased in 9 (10%) SFN patients. Seven of them had neurological comorbidities which may explain the elevated sNfL. The other two patients had history of hypothyroidism and endometriosis.
Conclusions:
sNfL levels are typically normal in patients with SFN. Furthermore, when sNfL are elevated, evaluation for another neurological disorder should be considered.
Keywords
Introduction
Sensory disturbances are common neurological complaints that may be due to central nervous system (CNS) or peripheral nervous system (PNS) disorders. The clinical neurological examination may indicate the anatomical localization of the disorder to one of these systems and direct the appropriate investigation. CNS imaging is used in cases where brain and spinal cord disorders are suspected and electrodiagnostic studies which include nerve conduction tests and electromyography are used to detect large-fiber polyneuropathy or radiculopathy. When these tests are normal and the clinical presentation is of sensory symptoms, frequently combined with burning pain and dysautonomic features, the diagnosis of small fiber neuropathy (SFN) should be taken into account and skin biopsy from the distal leg is employed for quantification of the epidermal nerve fiber density (ENFD
Numerous disorders may cause SFN and its diagnosis calls for an early search for their identification and treatment when appropriate. Diabetes mellitus is the most common cause of SFN. Other causes include immune-mediated, toxic, metabolic, elemental, and hereditary disorders. 5 When the systemic cause continues to evolve, clinical exacerbation in SFN symptoms as well as the development of large-fiber polyneuropathy can be identified. 6 Nevertheless, when the cause for the neuropathy has resolved, clinical improvement is reported 5 and although nerve damage might remain long standing, still possible axonal regeneration may occur.7,8 Examples include chemotherapy-induced neuropathy, post-infection or post-vaccination responses, treated vitamin deficiency, and immune-mediated or genetic disorders in remission due to treatment.5,9 Those findings collectively prompt the question of whether active axonal injury persists in SFN patients who continue to experience chronic neuropathic symptoms.
Blood neurofilament light-chain (NfL) measurements have been extensively used in recent years to identify axonal injury, and multiple studies examined the reference intervals in adults, with similar results that show age-dependent increasing values during normal aging.10–13 An abnormal increase in serum NfL (sNfL) was identified following CNS damage due to vascular, immune, or degenerative disorders. 14 Similarly, PNS disorders showed increased sNfL.15,16 It was recently shown that sNfL levels increase three weeks following initiation of chemotherapy-induced polyneuropathy, with a longitudinal increase up to 12 weeks. 17 High sNfL levels have also been detected in patients with Guillain-Barre syndrome (GBS), which decline to normal limits within a year. 18
In this study, we aimed to determine if sNfL levels are increased in SFN patients. We employed this test on patients with chronic sensory symptoms who were referred for a skin biopsy for evaluation of SFN.
Methods
Study design and participants
This is a cross-sectional observational study conducted in June-December of 2021 at the Sheba Medical Center neuromuscular clinic in Israel. The institutional review board approved the study (protocol SMC 6525-19). Informed consent was obtained from all patients.
Medical records were reviewed to collect demographic, clinical, electrodiagnostic, radiological, and laboratory data. Data was designated as complete if the main clinical complaint, past medical history, neurological examination, and electrodiagnostic studies of the lower extremities were documented. CNS imaging documentation was available for patients whose symptoms and/or signs suggested a possible CNS disorder. Follow-up interviews were conducted for assessment of symptom duration when required. Inclusion criteria were: (1) age older than 18 years, (2) Clinical presentation of pain, sensory, and/or autonomic disturbances and clinical signs of either thermal, pinprick hypoesthesia or hyperalgesia on neurological examination19,20 (3) Abnormal result of skin biopsy which was performed on the day of blood sampling for measurement of sNfL levels. Exclusion criteria were: (1) acute or subacute onset of symptoms (less than two months), (2) normal skin biopsy result (3) patients older than 85 years (3) patients with renal insufficiency.
Skin biopsies
A 3-mm punch skin biopsy was performed at the distal leg 10 cm above the lateral malleolus, fixed in Zamboni solution, and immuno-labeled with anti–protein gene product (PGP) 9.5 (Bio-Rad) according to standard procedures and as previously described for the determination of the ENFD.4,21 The ENFD was determined by immunofluorescence and compared with internationally recognized age and gender-matched normal values. 22 An ENFD below the 5th percentile was considered abnormal. A Congo red staining was used for identification of amyloidosis.
SNfL measurement
Venous blood samples were incubated at room temperature for 30 min to enable clot formation and centrifuged at 1500 g for 10 min at room temperature. The serum was frozen and stored at −80 °C until analysis. sNfL concentrations were measured in duplicates by a single molecule array (Simoa) assay 23 employing a commercial bead-conjugated immunocomplex kit (NF-light Advantage kit for SR-X, Uman Diagnostics Umea, Sweden). sNfL pg/ml measurements were compared to established upper normal limits per age, defined as the 97.5 percentile. 11 To indicate the magnitude of sNfL abnormality with adjustment to age, results were expressed as the ratio of the test result and the upper normal limit (test/UNL), i.e., sNfL magnitude. sNfL values that exceeded the age-determined upper normal limit (test/UNL >1) were considered abnormal.
Statistical analysis
Demographic patient characteristics were expressed by calculating mean and proportions (%). Comparisons between patient groups were performed by t-test which employed normality distributed data sets. Data sets that failed normality tests were analyzed by Mann-Whitney tests. Chi or Fisher's exact tests were performed for categorical variables. Statistical analyses were performed with GraphPad Prism (version 8.0.1 for Windows, GraphPad Software, La Jolla California, USA). Results are expressed as mean ± SD,
Results
Sample characteristics
A total of 94 patients with sensory symptoms were evaluated by sNfL measurements and skin biopsy. Eight patients who had normal skin biopsy results were excluded. Skin biopsy showed reduced ENFD in 86 patients, supporting the diagnosis of SFN. The mean age of patients was 48 ± 15.7 years (range 21–85), and 57/86 (66%) were females as summarized in Table 1. Over half of the patients (56/86, 65%) had length-dependent sensory symptoms, of them 17/56 (30%) had autonomic symptoms. The other 30/86 (35%) had non-length dependent sensory symptoms, 9/30 (30%) experienced also autonomic symptoms. Autonomic manifestations included most frequently orthostatic complaints, motility-related gastrointestinal symptoms, dry eye and mouth, urinary disturbances or to lesser extent abnormal sweating and erectile dysfunction. sNfL was increased in 9/86 (10%) of these cases (Figure 1).
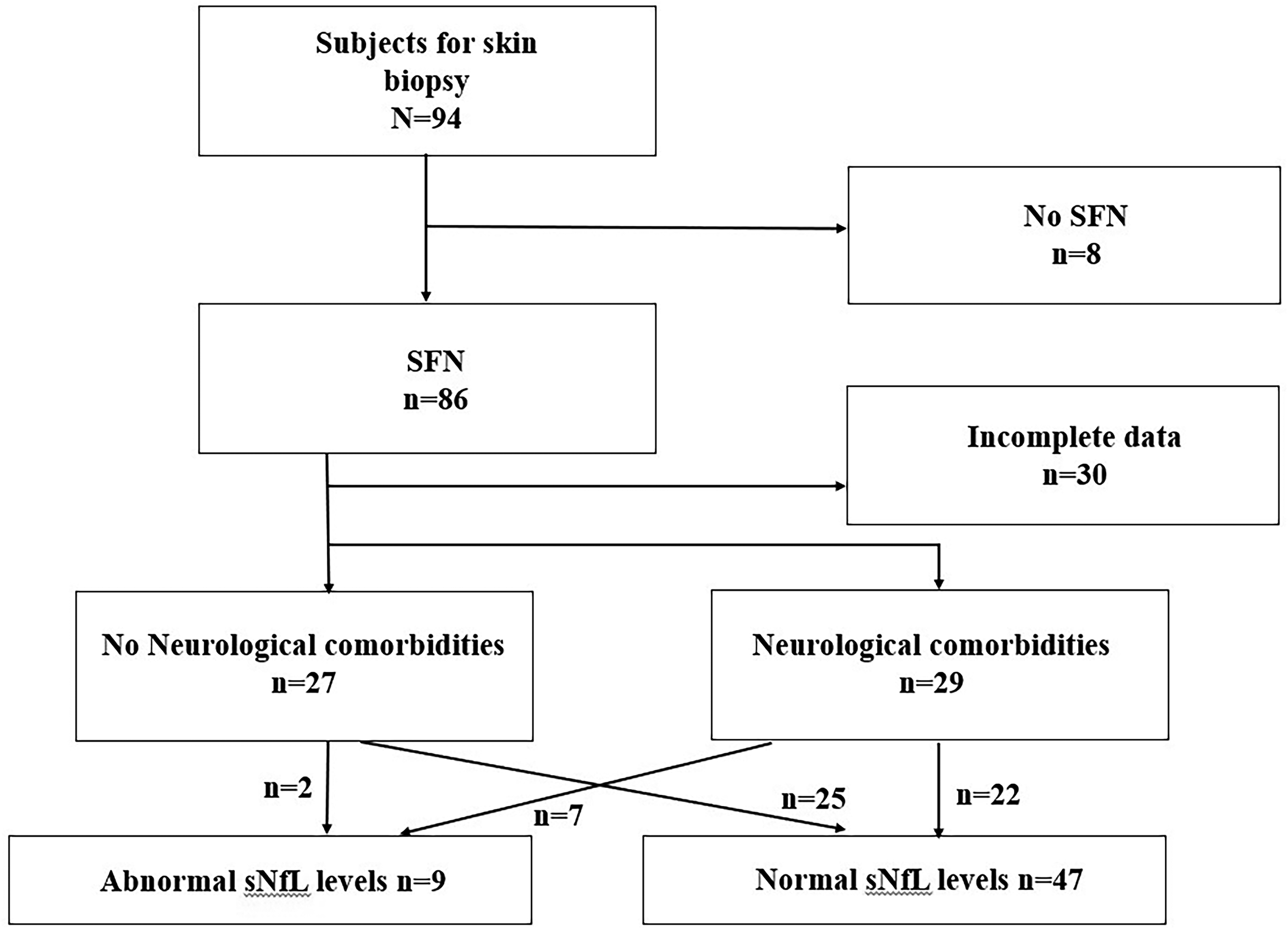
Flow chart – patients with small fiber neuropathy. Abbreviations: SFN, small fiber neuropathy; sNfL, serum neurofilament light chain; CNS, Central Nervous System; PNS, Peripheral Nervous System.
Demographic characteristics of SFN patients.
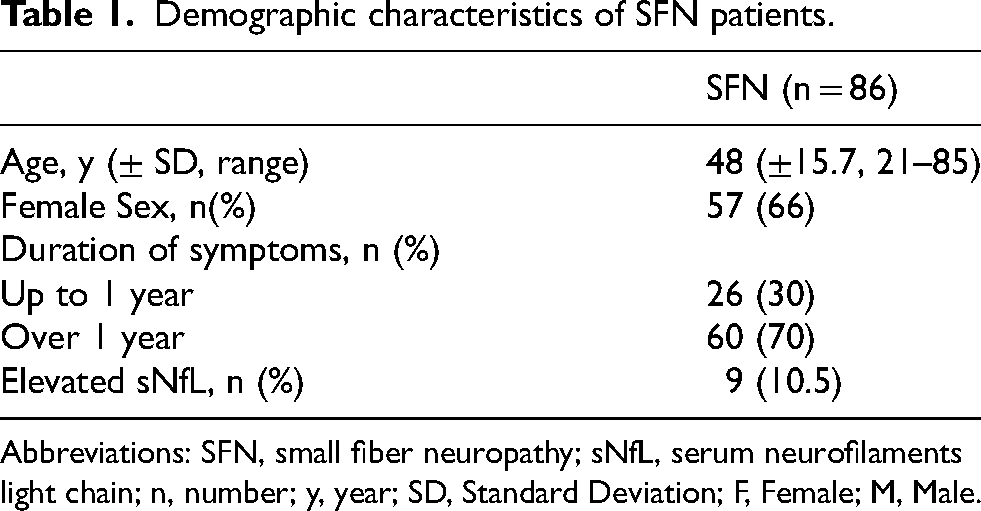
Abbreviations: SFN, small fiber neuropathy; sNfL, serum neurofilaments light chain; n, number; y, year; SD, Standard Deviation; F, Female; M, Male.
Complete medical data including neurological examination was available for 56 patients with SFN and indicated neurological comorbidities in 29/56 (52%) of cases. Of these patients, 33/56 (59%) had paresthesia or hyperalgesia without other focal neurological deficits, 23/56 (41%) had thermal and/or pinprick hypoesthesia in socks and glove distribution on neurological examination. Of them, 8/23 (35%) had additional focal neurological deficits due to associated neurological comorbidities.
Characteristics of patients with increased sNfL levels
All nine patients with elevated sNfL had reduced ENFD on skin biopsy. SFN patients with elevated sNfL more commonly had neurological comorbidities, identified in 7/29 (24%) cases with, versus 2/27 (7%) cases without an additional neurological disorder, however not in a statistically significant manner, p = 0.14 (Figure 2(a)). These included a CNS disorder in six patients and a large fiber polyneuropathy in one (Table 2). No significant difference was found in sNfL magnitudes in SFN patients with or without neurological comorbidities (Figure 2(b)).
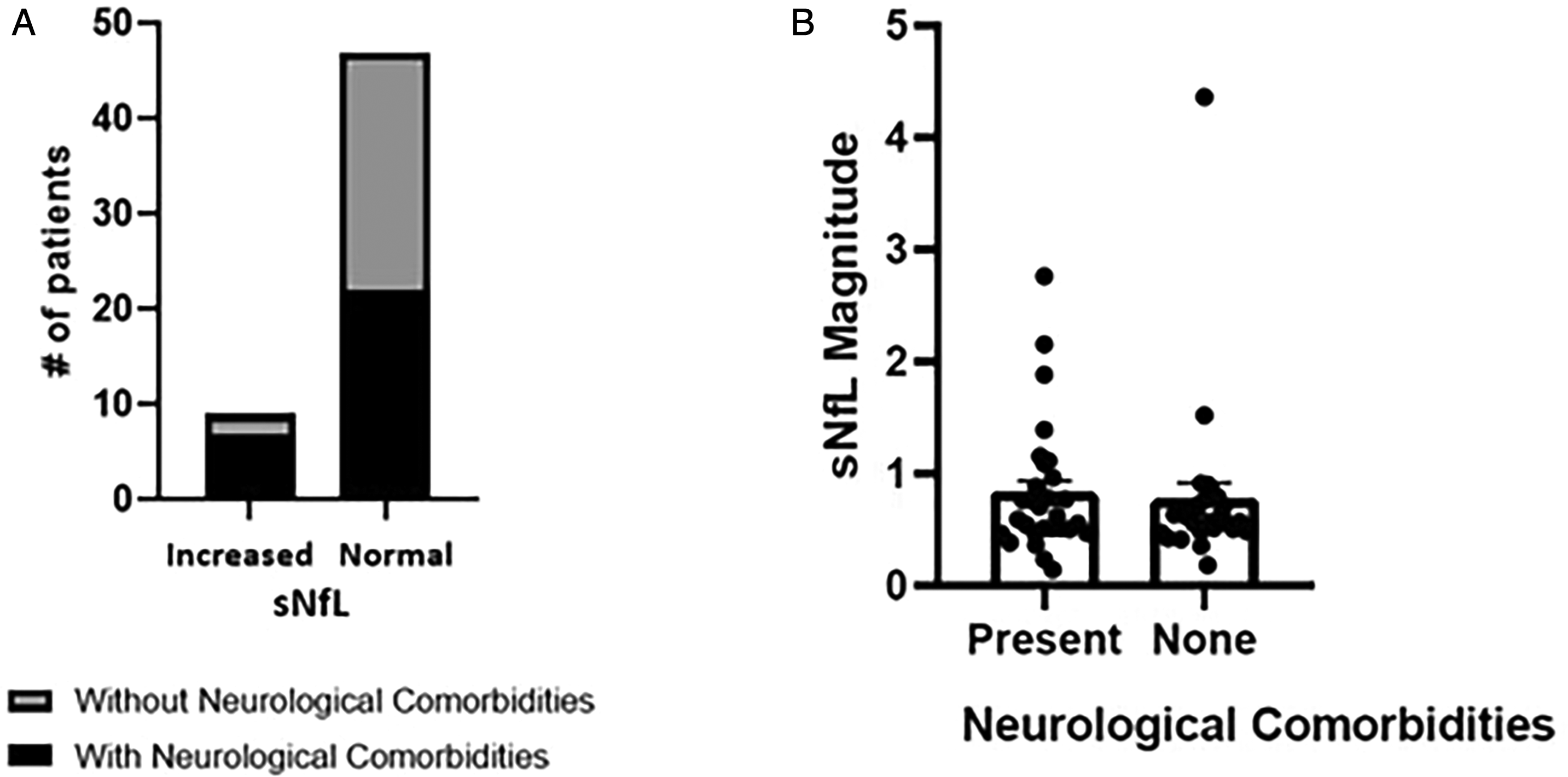
Neurological comorbidities and sNfL ratio in patients with SFN. A Fisher exact test, p = 0.14 . B Mann Whitney test, one tail, p = 0.29. Abbreviation: sNfL, serum neurofilament light chain.
Clinical, imaging, and electrodiagnostic characteristics of patients with elevated sNfL.

Abbreviations: sNfL, serum neurofilaments light chain; UNL, Upper Normal Limit; y, years; Female; M, Male; ENFD, Epidermal Nerve Fiber Density; EMG, electromyography; NCS, nerve conduction studies; PN, Polyneuropathy; RP, Radiculopathy; TPO, Thyroid Peroxidase; FVL, Factor V Leiden; IVF, In Vitro Fertilization; PTSD, Post-traumatic Stress Disorder.
Characteristics of patients with normal sNfL levels
Normal sNfL levels were detected in 77/86 (90%) of patients with SFN. In this group, 22/47 (47%) patients with complete data documentation had a medical history of neurological disorders which included demyelinating CNS disease, subarachnoid hemorrhage, hydrocephalus with ventricular shunt, myelopathy, radiculopathy, and polyneuropathy. A family history and/or Congo red findings indicate further genetic evaluation for a transthyretin (TTR) pathogenic variant/mutation. In two patients we identified a TTR variant, one of them with diagnosis of cardiac amyloidosis, and the other was considered to be a carrier without current manifestation of amyloidosis.
sNfL in SFN patients without neurological comorbidities
In 27/56 (48%) SFN patients with complete data documentation no indication of a coexisting neurological disorder was identified. All of these patients had a clinical phenotype consistent with the diagnosis of SFN,19,24 and normal electrodiagnostic tests. Brain imaging was performed in 19/27 (70%) and spinal imaging in 11/27 (41%) patients and in all cases, was without evidence of a neurological disorder. Hence, in these patients, neuropathy-related symptoms were considered to be due to SFN alone. The mean age of these SFN patients was 42 ± 15.13 years, 18/27 (67%) were females and their symptom duration was more than a year in 19/27 (70%). Systemic disorders were documented in 20/27 (74%) patients, which in 12/20 (60%) may be related to the development of SFN including diabetes mellitus, hypothyroidism, monoclonal gammopathy of undetermined significance, and chemotherapy-induced neuropathy.
Both patients with increased sNfL (2/27) and no neurological comorbidities had a medical history of endometriosis and hypothyroidism, with associated autoimmune antibodies in one (case 5). The other had a combination of psychiatric disorders (case 9). Both patients reported symptoms for over a year, similar to most cases in this cohort.
Comparison of duration of symptoms and sNFL
sNfL levels were similar in patients with symptoms duration of less than or more than a year (p = 0.43).
Discussion
In this study, elevated sNfL were not associated directly with SFN. High sNfL levels were found in about 10% of patients who had chronic neuropathic symptoms and confirmed SFN by skin biopsy. However, SFN patients with elevated sNfL had significanly more neurological comorbidities. Those few SFN patients without neuroloical comorbidities that had elevated sNfL, did have significant systemic disorders. These findings indicate that in most patients with chronic, protracted SFN symptoms, sNfL levels are normal indicating the lack of widespread active axonal damage. This is in line with a recent study which reported normal sNfL levels in patients with SFN. 25 However, while this study compared SFN patients to healthy control, 25 our study highlights that while sNfL is sometimes increased in SFN patients, this suggests mainly other neurological disorders that warrant further investigations.
In our study, 70% of SFN patients had symptoms for over a year, according to a recent study, over half of SFN patients continue to experience ongoing neuropathic pain at least two years following the diagnosis of SFN. 26 Another study explored the mechanism of chronic pain and allodynia development following nerve injury in an animal model. This study showed that neuropathic pain was actually induced by reinnervation, associated with sprouting of nociceptors into denervated territories. 27 Consistent with this, painful diabetic neuropathy was associated with increased small nerve fiber regeneration following intensive glycemic control. 28 Therefore, worsening chronic neuropathic pain may actually indicate resolution of active axonal disruption.
Blood NfL has been reported as a biomarker of peripheral nerve axonal damage in polyneuropathy due to various systemic disorders which include diabetes mellitus, 29 amyloid light chain (AL), and transthyretin (ATTR) amyloidosis, 30 and in inflammatory demyelinating polyneuropathies including the GBS and chronic inflammatory demyelinating polyneuropathy.18,29 In these conditions, the level of neurofilaments declines following treatment or resolution of the active disease.18,31 This elicites the idea that, as occurs with CNS disorders, in certain PNS disorders, normal sNfL levels may represent the resolution of active axonal damage. In the adult human sural nerve, small unmyelinated axons are approximately 5 times more abundant in number and 4 times in density in comparison to myelinated axons. 32 Therefore, damage to these small but numerous and widely spread fibers may result in substantial axonal degeneration which may be detectable in blood. In fact, recent study demonstrated the elevation of serum phosphorylated high molecular weight neurofilament subunit in SFN patients with glucose intolerance in comparison to healthy controls and patients with glucose intolerance without SFN. 33 However, small nerve fibers contain only small amounts of NfL due to their small caliber, and accordingly, the magnitude of blood NfL derived from small axons may, perhaps, be too low for detection. This may explain why in our study sNfL levels were kept normal in most SFN cases.
In our study, we identified only two SFN patients with increased sNfL and no other neurological disorders, both were female with history of thyroid disease. One patient had also history of post-traumatic disorder, which may explain the elevated sNfL, as indicated by recent study comparing sNfL in patients with post-traumatic disorder and healthy control. 34 The other patient had unbalanced Hashimoto thyroiditis, which is a common autoimmune cause of hypothyroidism that may be associated with the development of polyneuropathy. 35 Polyneuropathy and autonomic manifestations are fairly common among individuals with hypothyroidism 36 and reduced ENFD was described in 60% of patients with overt hypothyroidism and 12.5–25% of patients with subclinical hypothyroidism.37,38 Furthermore, hypothyroidism was also associated with CNS alternations, and patients with hypothyroidism were found to have significant prolongations of visual and brainstem evoked potentials compared to controls. 39 Thus, evidance of active axonal injury expressed by elevated sNfL in these patients may perhaps be associated with this syndrome, whether as a result of hypothyroid CNS or PNS pathology is undetermined. Still our subanalysis included an SFN patient with Hashimoto thyroiditis and normal sNfL.
Both patients with elevated sNfL additionally had a history of endometriosis, and in total, 4/27 (15%) in the cohort had endometriosis. The association between SFN and endometriosis has been evaluated previously and, interestingly, PGP 9.5 antibody staining of endometrioma biopsies identified small nerve fibers within this tissue.40,41 Currently, to the best of our knowledge, no data regarding measurements of sNfL among patients with hypothyroidism or endometriosis is available.
This study is limited by the lack of data regarding associated comorbidities in about one-third of SFN patients. With regard to patients with neurological comorbidities, we could not determine if they had active or inactive disorders due to the absence of follow-up imaging or electrodiagnostic studies. Our high rate of positive skin biopsy results may be explained firstly by the selection of patients which was based on chronic sensory disturbances with a pattern suggesting SFN. Secondly, our local availability of skin biopsy is limited and therefore this increases the proportion of positive SFN results. In a prior study the specificity of skin biopsy for evaluation of SFN was about 90% which suggest false positive rate of approximately 10%, 20 We did not test patients with acute SFN to establish a direct association between active small fiber loss and the gradual elevation of sNfL. This may have provided key findings but requires a different setup and multiple blood and skin biopsy measurements to provide the temporal profile of sNfL increase in SFN. The strength of our study is that it employed patients who had clinical assessments which included electrodiagnostic tests for evaluation of large fiber polyneuropathy and skin biopsy for all.
In conclusion, in this cohort, sNfL levels were not increased in most SFN patients with chronic sensory symptoms. When sNfL levels were increased, neurological comorbidities were commonly identified, which may assist in determining the need for further investigation.
Footnotes
Abbreviations
Acknowledgments
The authors have no acknowledments to report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data avilability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
