Abstract
Background:
Spinal muscular atrophy (SMA) is caused by bi-allelic, recessive mutations of the survival motor neuron 1 (SMN1) gene and reduced expression levels of the survival motor neuron (SMN) protein. Degeneration of alpha motor neurons in the spinal cord causes progressive skeletal muscle weakness. The wide range of disease severities, variable rates of decline, and heterogenous clinical responses to approved disease-modifying treatment remain poorly understood and limit the ability to optimize treatment for patients. Validation of a reliable biomarker(s) with the potential to support early diagnosis, inform disease prognosis and therapeutic suitability, and/or confirm response to treatment(s) represents a significant unmet need in SMA.
Objectives:
The SMA Multidisciplinary Biomarkers Working Group, comprising 11 experts in a variety of relevant fields, sought to determine the most promising candidate biomarker currently available, determine key knowledge gaps, and recommend next steps toward validating that biomarker for SMA.
Methods:
The Working Group engaged in a modified Delphi process to answer questions about candidate SMA biomarkers. Members participated in six rounds of reiterative surveys that were designed to build upon previous discussions.
Results:
The Working Group reached a consensus that neurofilament (NF) is the candidate biomarker best poised for further development. Several important knowledge gaps were identified, and the next steps toward filling these gaps were proposed.
Conclusions:
NF is a promising SMA biomarker with the potential for prognostic, predictive, and pharmacodynamic capabilities. The Working Group has identified needed information to continue efforts toward the validation of NF as a biomarker for SMA.
INTRODUCTION
Spinal muscular atrophy (SMA) is an autosomal recessive neurodegenerative disorder that has historically been the most common monogenic cause of death in infancy [1], affecting 1 in 11,000 live births [2, 3]. SMA is most often caused by homozygous deletions of the survival motor neuron gene (SMN1), and less often, by missense, nonsense, or frameshift mutations of SMN1 [4–6]. Both the SMN1 and SMN2 genes generate the survival motor neuron (SMN) protein, but the transcripts that arise from SMN1 are full-length and generate a functional protein, whereas the vast majority of those transcribed from SMN2 are truncated and generate a less stable version of the SMN protein that cannot interact optimally with binding partners [7]. Although the SMN2 gene is therefore unable to fully compensate for the loss of a functional SMN1 gene, SMN2 copy number is inversely correlated with disease severity [8–16].
SMA is characterized by reduced expression levels of the SMN protein and subsequent degeneration of spinal cord and brainstem motor neurons [17–19]. The disease phenotype ranges widely in severity and rate of progression. If untreated, reduced SMN protein levels can cause a range of debilitating complications such as functional motor deficits, limb and truncal muscle weakness, difficulty breathing, and eventual respiratory failure [1, 20–28]. Historically, SMA patients have been segmented into Types 0–4 depending on severity and clinical disease onset (e.g., “Type 0” refers to fetal onset and severe muscle weakness, whereas “Type 4” describes the small proportion of patients who do not display symptoms until adulthood) [8, 11, 20, 21, 29]. However, disease severity is understood to comprise a wide gradient, and this nomenclature is becoming less relevant as newborn screening and pre-symptomatic treatments are more widely utilized [30–32]. Since 2016, three disease-modifying SMA treatments have been approved by the United States Food and Drug Administration (FDA). All three therapies act by increasing SMN protein levels [33–35]. Nusinersen (SPINRAZA®) [36–38] and risdiplam (Evrysdi®) [39, 40] each modulate SMN2 mRNA splicing through different mechanisms to increase the amount of full-length, functional protein generated from the SMN2 gene. Both are indicated for use in pediatric patients, including newborns, and adults [41, 42]. Onasemnogene abeparvovec-xioi (ZOLGENSMA®) is an adeno-associated virus 9 (AAV9)-mediated, SMN1 gene replacement therapy approved for children under two years old and administered intravenously [43, 44]. Responses to nusinersen [45–54], onasemnogene abeparvovec [55–62], and risdiplam [63–67] vary widely between individuals and are affected by a number of factors including, but not limited to SMN2 copy number, age, and disease severity at the time of treatment. Because of this variability, SMA biomarkers to guide treatment decisions (prognostic biomarkers) and predict therapeutic responses (predictive biomarkers) are urgently needed. In addition, several therapeutics that do not target the SMN protein but focus on preventing or reversing motor neuron loss, improving function at the neuromuscular junction, or enhancing muscle performance are in various stages of development and clinical trials [35, 68–71]. A combination of SMN-dependent and SMN-independent therapeutics may be the most effective way to treat SMA [35, 69, 72, 73]. Furthermore, because SMA is a progressive disease that changes over the life of a patient, the specific combination of drugs used may change with time [74]. As such, new pharmacodynamic SMA biomarkers would be extremely useful as outcome measures in clinical trials and in tracking responses to evolving therapeutic regimens over time.
A great deal of research into putative biomarkers for SMA has occurred (Table 1; Fig. 1) [75–78]. Although SMN mRNA and protein are natural candidate biomarkers for SMA and are readily measurable in blood, SMN blood levels may not correlate with levels in the CNS, disease severity, or motor function gains [75–79]. For these reasons, SMN quantification has not emerged as a leading SMA biomarker thus far. However, a recent study quantified full length SMN transcript levels within the extracellular vesicles (EVs) found in blood serum of individuals with Type 2 SMA [80]. This technique was developed on the premise that EVs play a critical role in cell-to-cell communication across tissue types and cross the blood brain barrier in both directions, thereby potentially reflecting motor neuron health status. Study results indicated that full length SMN transcript levels in EVs increase with nusinersen treatment. Notably, the investigators chose to compare SMN transcript levels against levels of neurofilament-heavy chain (NF-H) in blood serum and CSF, which decreased with nusinersen treatment.
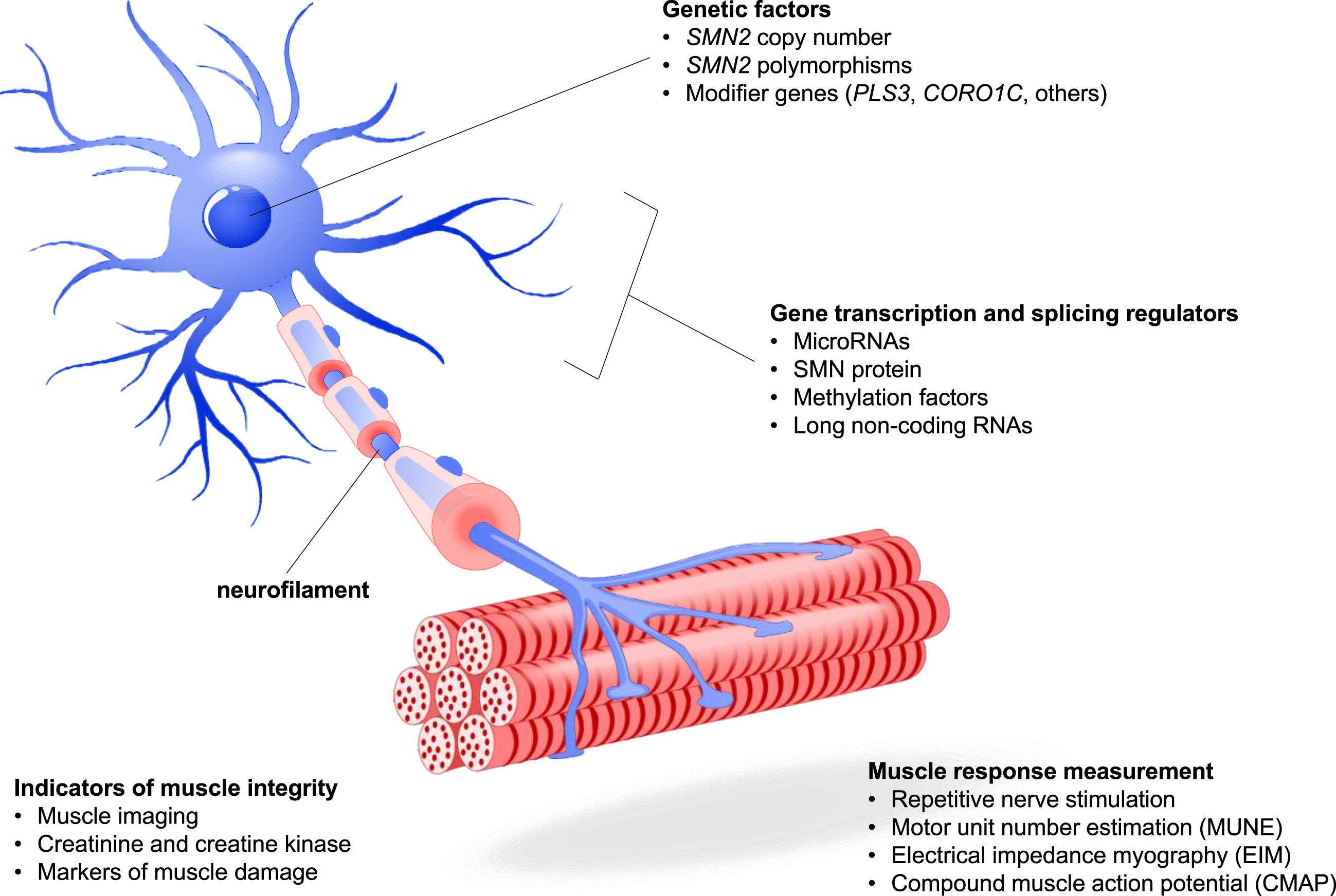
SMA Pathophysiology and Candidate Biomarkers. A number of candidate biomarkers have been proposed based on the pathophysiology of SMA. Genetic factors impacting the expression of SMN protein include SMN2 copy number and polymorphisms, the presence of modifier genes that can improve downstream neuronal and motor functions associated with SMA disease state, and gene transcription factors that affect the expression of SMN2 and other genes. Muscle presence and function can be measured through a variety of techniques including imaging, action potential and electrical response following motor nerve stimulation, and quantification of molecular factors that relate to muscle metabolism or damage. The presence of neurofilament in serum and CSF has been the subject of extensive research.
Candidate biomarkers for SMA
Other recent candidate biomarkers include genetic factors such as SMN2 copy number [10, 13–15, 81, 82], SMN2 polymorphisms [10, 13–16, 81–83], and modifier genes [73, 84–89]; transcription and splicing regulators, such as microRNAs [90–94], methylation factors [95–99], and long non-coding RNAs [100–103]; biomolecular factors such as SMN protein [18, 39, 48, 57, 81, 104–106], neurofilament (NF) [78, 107–118], tau protein [112, 119–121], and muscle damage indicators [122–124]; muscle imaging parameters including magnetic resonance imaging (MRI) [122–124] and electrical impedance myography (EIM) [75, 125–127]; and electrophysiological parameters including compound muscle action potential (CMAP) [128–133], motor unit number estimation (MUNE) [75, 125–127], and repetitive nerve stimulation [45, 75, 134–138].
Despite this abundance of research, there remains a need for consensus among the SMA research and clinical care community as to which prognostic, predictive, and pharmacodynamic biomarkers for SMA are the most promising candidates for widespread use [139–141]. To address this need and to advance the field of SMA biomarker research, Cure SMA convened a working group of SMA experts. The SMA Biomarkers Multidisciplinary Working Group reviewed evidence in support of lead candidate biomarkers and engaged in a modified Delphi process consisting of a series of surveys designed to build consensus and determine best next steps to advance the field of SMA biomarker research.
METHODS
The SMA Biomarkers Multidisciplinary Working Group mission
The SMA Biomarkers Multidisciplinary Working Group comprised 11 physicians, scientists, and other healthcare professionals with expertise in the treatment and research of SMA. These individuals were selected for their expertise in SMA clinical care, roles in SMA clinical trials, and their contributions to the understanding of the natural history of SMA or SMA biomarkers. For logistical simplicity, Working Group advisors were recruited from within the U.S. Therefore, discussion was held under the assumption that group members were working within the scientific and regulatory framework of the U.S. The aims were to review the existing candidate biomarkers for SMA (Table 1), select the candidate SMA biomarker best poised for further development, generate a list of data needed for the advancement of this candidate biomarker, and specify the validation studies necessary to generate those data. Cure SMA acted as a neutral moderator for this study. Employees of Cure SMA are neither invested in nor biased toward SMA biomarkers or therapies.
Achieving consensus through the Delphi technique
A modified Delphi technique was used to gain consensus and work towards selection of a biomarker for further development [142, 143]. This process consisted of surveys and phone calls to discuss the questions posed. Surveys and phone calls were reiterative in order to gain increasing consensus with each discussion [144]; initial, broad questions were designed to evoke more specific follow-up questions. Each of the six discussions was designed to build on consensus derived from the previous discussions. Surveys were distributed prior to each phone call. The questions were answered anonymously either through a multiple-choice selection or in an open-ended manner. Most multiple-choice questions also contained a comment field for working group members to utilize if they felt the choices given were insufficient, or if they wanted to add more context to their choice.
The surveys and phone calls took place between October 2020 and April of 2021. The first seven phone calls were proceeded by an online survey and occurred at a frequency of one call per month. A final survey was issued after the last working group call. Survey responses were shared with the wider Working Group prior to each round of discussions, and each voting member was given the opportunity to maintain the response they had given or to modify their response after considering the opinions of their colleagues. This approach balanced the need for voting members to provide opinions anonymously and without influence from colleagues initially while considering the group’s outlook on each question before moving to the next stage. In this manner, it was possible to determine which topics were areas of consensus and which topics necessitated more discussion. From the outset of the study, consensus was defined as a majority; however, the group achieved at least 80% consensus around most decisions. A neutral, non-voting member with expertise in the Delphi process and no conflicts of interest was present to moderate the discussion. The objectives of this modified Delphi process were presented to the Working Group as follows: “The goals of this process are 1) the selection of the biomarker most poised for further development for SMA, and 2) the recommendation of an experimental plan for the purposed validation studies. The objective for the first three calls will be to select a biomarker(s) for further development. The first three calls will be directed towards understanding which potential biomarkers exist and what data is available on those biomarkers. These calls will include guest speakers presenting relevant data, as well as data summaries from the Cure SMA moderator. The goal for the last three calls will be to determine the exact data that exists for the selected biomarker(s) and the gaps that remain for validation. The last three calls will also focus on developing recommendations for a validation study to generate missing data for the selected biomarker(s). The group will select the type of study (i.e., basic research, clinical) and map out the recommended experiments/data to be collected within the determined scope, desired cohort to study (including sample size), parameters to be assessed, assays to be utilized, etc.”
Prior to the first Working Group call, a briefing packet containing a reference list was distributed to all Working Group advisors. The reference list contained 64 peer-reviewed articles intended to provide basic background on each candidate biomarker. The articles were compiled from a literature search performed on PubMed using the following search terms: “biomarker” OR “biomarkers” AND “SMA” OR “spinal muscular atrophy.” Search limits were used to restrict the search to primary research articles and omit reviews and case studies. Working Group members were invited to suggest additions to the reference list, and an amended reference list was recirculated. The briefing packet also contained an initial/pre-call survey in which advisors were asked to rank their interest in five types of biomarkers (biomolecular, electrophysiological, genetic factors, muscle imaging, and transcription and splicing regulators) within the context of the group’s objectives (Table 2; initial/pre-call survey). Biomarker types were ranked on a scale of 1 to 5, where 1 was least interested and 5 was most interested. Scores were averaged to obtain a group score. Having ranked biomolecular and electrophysiological biomarkers of most interest, advisors were asked to indicate which specific candidate biomarkers of these types they were most interested in pursuing (Table 2; initial/pre-call survey). Working Group members could choose more than one biomarker in each category. The next three surveys and calls were directed toward the discussion of specific potential biomarkers and the data that are available about those biomarkers (Table 2). The goal for this phase of the project was to select the biomarker(s) most poised for further development. After the advisors viewed presentations by guest subject matter experts, and reviewed relevant data and data summaries from the Cure SMA moderator, three surveys followed by discussions were administered. Survey one asked questions about prognostic, predictive, and pharmacodynamic biomarkers 1 [145] in infants, children and teens, and adults to narrow the discussion to a small number of candidate biomarkers (Table 2; survey one). Working Group members were asked to rank their top three candidate biomarkers in order of interest on a scale of 1 to 3, where 1 was of the most and 3 is of the least interest. Survey two asked about perceived gaps in understanding in the field of the consensus candidate biomarker (ranked 1 to 5, where 1 was least pressing and 5 was most pressing), and which data collections were of the highest priority (ranked 1 to 6, where 1 was the highest and 6 the lowest priority) (Table 2; survey two). Survey three asked Working Group members whether they favored revisiting the second and third biomarker candidates identified, as well as to priority rank the consensus biomarker candidate data that remained outstanding (Table 2; survey three).
Interview questions and advisor responses
1Rankings of 1–5 (1 = least interested and 5 = most interested) were averaged to obtain group scores. 2Working Group advisors could choose more than one biomarker. 3Results represent the percentage of Working Group advisors who ranked these biomarkers among the top three candidates for stated age group and function. 4Rankings of 1–5 (1 = least pressing and 5 = most pressing) were averaged to obtain group scores. 5Rankings of 1–6 (1 = highest priority and 6 = lowest priority) were averaged to obtain group scores. 6Working Group advisors chose one biomarker.
The final three surveys and discussions were tailored to determine the precise data that exists for the selected biomarker(s) (Table 3). The goal of these final interviews was to identify the gaps that need to be filled to validate the candidate biomarker. Once priority knowledge gaps were agreed upon, the Working Group members tailored a list of recommendations for validation studies to generate missing data for the selected biomarker(s). Details of these experiments included 1) type of study (i.e., basic research, clinical); 2) specific experiments; 3) data to be collected within the determined size and scope; 4) desired cohort to study, including sample size; 5) parameters to be assessed; and 6) assays to be utilized.
NF assay validation studies: Group questions and answers from surveys four-six
RESULTS
Building consensus around a priority candidate biomarker
When asked to rank categories of potential biomarkers for SMA, Working Group advisors favored biomolecular entities, followed by electrophysiological parameters, over genetic factors such as transcription factors and splicing regulators, as well as over muscle imaging (Table 2; initial/pre-call survey). When next asked which of several biomolecular candidates the Working Group favored, 100% of members selected NF, followed by 75% who also wanted to explore SMN protein, and 50% of members who also wished to follow up on creatinine, or creatine kinase and other markers of muscle damage. The electrophysiological biomarkers of highest interest were CMAP and MUNE.
Working Group advisors unanimously selected NF as the best biomarker to use to determine pharmacodynamic proof of concept and dose size and frequency optimization (Table 2; survey one). Second-priority candidate biomarkers differed depending on patient age; Working Group members favored CMAP for use in adult and infant patients, but SMN in children and teens. The advisors believed NF would be the best predictive biomarker to monitor disease progression and determine the timing of drug initiation in infants, children and teens, and adults. Second to NF in the predictive biomarker category were CMAP (for infants and children/teens) and blood markers of muscle damage (for adults). Furthermore, advisors felt that NF would be the best prognostic biomarker for clinical monitoring (i.e., for safety, efficacy, and stratification of phenotypic severities) for all age groups, followed by CMAP for all age groups (Table 2).
Identifying and filling gaps in knowledge about NF
The Working Group members identified several knowledge gaps in the field’s understanding of NF as a biomarker for SMA (Table 2; survey two). In healthy individuals, NF levels fluctuate during both in early childhood development and during aging [146], but it is currently unclear how age-related changes in NF levels in healthy individuals compare to changes in SMA-affected individuals [109, 111, 115, 116, 119, 121, 147–149]. It is also unknown how NF levels differ across severities of SMA and whether NF can be used to detect disease onset or track disease progression in patients with three or more copies of SMN2. Finally, it is not currently understood whether a deviation from a standardized baseline value, or a change from an individual’s own baseline, is the relevant factor when tracking NF levels alongside disease progression and treatment response [140, 151]. These unknowns were weighted by the Working Group as being of equal importance.
The advisors next ranked the additional data that should be collected to fill the identified knowledge gaps (Table 2). The information most needed to move forward with biomarker validation was deemed to be the selection of an appropriate assay, followed by data on NF in healthy control subjects, insights about differences between NF heavy (NF-H) and light (NF-L), NF natural history data in patients with SMA, and data regarding changes following treatment. The Working Group indicated that comparison of natural history data from SMA patients to normative controls could elucidate how NF changes with age and in which scenarios a threshold value (e.g., to initiate therapy) or a change from baseline (e.g., when monitoring individual disease progression) would be most relevant, as well as help establish those values. While most advisors also favored revisiting SMN protein and CMAP as additional candidate biomarkers, the group favored waiting until more information about SMN expression is made available and instead moving ahead with CMAP research in adults, for whom NF may be less useful (Table 2; survey three) (See Discussion).
Designing a pilot study to work toward validation
The Working Group concluded that the first step towards validating NF as a biomarker for SMA would be to validate an assay to measure NF in the context of SMA (i.e., to understand reference values relevant to the SMA patient population and corresponding healthy individuals). The advisors discussed the usefulness of conducting a small pilot study to this end. Several key data elements were identified as priorities for the pilot study (Table 3). Members agreed that to validate a candidate NF assay for SMA, it would be beneficial to determine the degree of biologic and measurement variability that exists and to determine the sample volume needed for accurate readout. Once these parameters of the assay were determined, the study could then outline the natural history of NF in SMA patients compared to healthy individuals to determine pathological thresholds and normal age ranges. The reproducibility of the assay (i.e., the intra-individual variation over multiple measurements taken over time) would also be an important consideration, and it would be advantageous to compare the study participants’ NF levels to their SMN2 copy numbers, levels of SMN protein, and measurements of other candidate biomarkers such as CMAP. The group discussed the availability of assay platforms for NF-L and NF-H, as well as in which populations an NF assay would be most useful. Advisors favored quantifying NF in blood rather than CSF. Potential challenges, such as performing repeated blood draws and obtaining an adequate volume of blood from infants, were also discussed. Finally, the Working Group discussed specific parameters for such a pilot study, including ideal subject age ranges and sample sizes. While no broad agreement was reached at this level of detail, the input may serve as a valuable foundation for the planning of this validation study in the future.
DISCUSSION
As of February 2023, 99% of all U.S. infants were being screened for SMA at birth [152], enabling early treatment with three FDA-approved, SMN-dependent, disease-modifying SMA therapies. Widespread SMA treatment will alter the natural history of the disease as it is currently understood, creating new sub-populations of treated SMA-affected individuals with unique responses to SMN-dependent therapies. In addition, several SMN-independent therapies are currently in the clinical trial pipeline [112, 140, 153, 154], and a combination of therapies from these two categories may be the most effective way to treat SMA [35, 69, 72, 73]. Combination treatment regimens will require new biomarkers to help predict and measure treatment responses and disease progression. This rapidly evolving SMA treatment landscape, combined with the heterogeneity of SMA phenotypes and the progressive nature of the disease, makes the need for SMA biomarkers especially urgent [140, 141, 153, 154]. It is likely that combinations of prognostic, predictive, and pharmacodynamic biomarkers will be needed [140, 153].
The SMA Multidisciplinary Biomarkers Working Group successfully reached a consensus on the suitability of NF as a candidate biomarker for SMA (Table 2), particularly at younger ages, and outlined next steps for its further development (Table 3). NFs are significant structural components of the neuronal cytoskeleton and are abundant in large, myelinated axons, such as those of motor neurons [146]. Their importance to the structural integrity of axons is not limited to their role as cellular scaffolding [155–157]. NFs also impact microtubule and actin dynamics, as well as synaptic function. When neurons degenerate or experience damage to axons, in which NFs are enriched, NFs are degraded and released into the interstitial fluid, cerebrospinal fluid (CSF), and blood in significant quantities. This process underlies the observed correlation between NF levels in blood serum or CSF and axonal injury, axonal loss, and neuronal death.
The NF protein consists of four subunits in varying states of phosphorylation, which strengthens them and confers additional resistance to protein degradation. When NF is utilized as a biomarker, researchers typically measure the NF-heavy chain (NF-H) and/or NF-light chain (NF-L) subunits, which are the most and least phosphorylated subunits, respectively [146]. NF has been considered as a biomarker in a number of neurodegenerative conditions [151, 157, 158], including multiple sclerosis [159], Parkinson’s disease [160, 161], Charcot-Marie-Tooth disease [162], peripheral neuropathy [163], and amyotrophic lateral sclerosis (ALS) [164].
NF has also been studied as a potential biomarker for SMA [110, 116]. Research has shown that NF is elevated in SMA-affected infants and young children compared to healthy controls, and it rapidly declines with treatment by nusinersen before stabilizing above normal [107, 113, 118, 165]. For example, Finkel et al. (2022) analyzed data from the CS3A and ENDEAR studies and found that in infants with SMA, lower NF-H levels in CSF were associated with increased nusinersen dosage, which in turn correlated with better treatment outcomes [165]. Studies that have evaluated the effects of nusinersen treatment on NF levels in SMA-affected adolescents and adults have yielded conflicting results, suggesting a need for further, standardized studies on these populations [109, 111, 115, 121, 147–149, 166, 167]. Factors that may have contributed to these variable results include whether NF was measured in blood plasma, serum, or CSF, and whether the NF-H and/or NF-L subunit was measured. Because NF may not be as informative a biomarker in adolescents and adults, the Working Group advisors favored CMAP as a candidate biomarker for further development for those age groups [45, 75, 134–138] (Table 2; survey one). The majority of working group members also rated SMN protein as a top biomolecular candidate, but the Working Group opted to wait for forthcoming data about SMN protein expression during risdiplam treatment. In July 2022, Mercuri et al. [168] published the results of the SUNFISH Part 1 clinical trial, a dose-finding study for risdiplam that demonstrated a dose-dependent increase in SMN protein in treated SMA-affected individuals ages 2–25 years.
The SMA Multidisciplinary Biomarkers Working Group identified additional knowledge gaps that needed to be addressed to validate NF as a biomarker for SMA (Table 2; survey two). At the time of this study, it was unclear whether NF levels change over the lifetime of healthy individuals, and how such changes vary with disease severity and disease progression [109, 111, 115, 116, 121, 147–150]. Furthermore, the working group stated that it must be determined whether the relevant metric is an increase beyond a universal NF threshold level or a deviation from each individual’s own NF baseline [140, 151, 169]. Specific parameters for NF assay validation studies were outlined, including experiments to determine biofluids volumes needed, variations in NF measurement results from within the same sample, and ages and numbers of study candidates needed to draw meaningful conclusions (Table 3). A pilot study to examine these unknown variables was determined to be of great benefit to the field and is the likely next step towards validation of NF as a biomarker for SMA.
Since the SMA Multidisciplinary Biomarkers Working Group convened in 2020, a handful of studies seeking to establish NF-L reference intervals in healthy individuals over a range of ages have been published [170–173]. These studies have added valuable information to the knowledge base about NF-L levels in individuals who are not affected by neurological disease. However, data gaps remain regarding NF-L levels in healthy infants and young children, and more studies are needed among more diverse populations. Furthermore, establishment of a standardized assay for measuring NF-L levels—as well as a consensus as to whether to measure the protein in blood plasma, serum, or CSF—would allow for better comparison of results across studies.
Limitations of the study
NF is expressed exclusively in the nervous system, and the protein is released in response to a wide range of neurologic diseases and traumatic events that result in axonal damage. Therefore, NF has been studied extensively as a biomarker, and the peer-reviewed literature on the subject is robust. This abundance of information had the potential to bias the Working Group in favor of selecting NF as the best SMA biomarker on which to focus future research. However, because the group evaluated all candidate biomarkers on the basis of multiple criteria, any bias imparted by an imbalance in published research was likely corrected. A second limitation of the study is that the Working Group consists exclusively of researchers from the U.S., which had the potential to influence the members to limit their due diligence to biomarker studies performed there. However, the group balanced this potential bias by reviewing literature published by SMA biomarker researchers from around the world. In addition, most of the Working Group members regularly participate in international collaborations on SMA research. Finally, the recent FDA approval of three disease-modifying SMA therapies, along with a dramatic increase in U.S. newborn screening, has enabled approximately 70% of SMA-affected individuals to receive treatment for the disease [174]. As the population of untreated individuals living with SMA grows smaller, it will become more difficult to establish reference values for SMA biomarkers like NF.
CONCLUSION
SMA clinical trial pipelines in the U.S. and worldwide are burgeoning with a variety of potential treatment options, many of which may be used in combination with currently approved SMN-dependent therapies [35, 68–71]. As such, future SMA treatment regimens are likely to involve the use of multiple, often concurrent therapies and evolve over the lifetime of the individual. For these reasons, the need for validated biomarkers in multiple areas of SMA clinical care and research—including predicting treatment response, monitoring disease progression, and measuring pharmacologic response—has never been more urgent.
Footnotes
ACKNOWLEDGMENTS
We would like to thank Biogen and the Miller McNeil Woodruff Foundation for funding this project. We also thank Hilary Scheler, PhD, for writing the early drafts of this manuscript.
CONFLICTS OF INTEREST
Thomas Crawford has served as a paid advisor, consultant, and/or speaker to commercial entities Biogen, AveXis/Novartis, Roche/Genentech, Scholar Rock Pfizer and Cytokinetics, and has received research funding/study site investigation support from Biogen, Avexis, Cytokinetics, Catalyst, and Erydel. He advises several patient organizations: Cure SMA, SMA Foundation, Muscular Dystrophy Foundation, Ataxia Telangiectasia Children’s Project.
Basil T. Darras, MD, has served as an ad hoc scientific advisory board member for AveXis/Novartis Gene Therapies, Biogen, Pfizer, Sarepta, Vertex and Roche/Genentech; Steering Committee Chair for Roche FIREFISH and MANATEE studies and DSMB member for Amicus Inc. and Lexeo Therapeutics; he has no financial interests in these companies. BTD has received research support from the National Institutes of Health/National Institute of Neurological Disorders and Stroke, the Slaney Family Fund for SMA, the Spinal Muscular Atrophy Foundation, Cure SMA, and Working on Walking Fund and has received grants from Ionis Pharmaceuticals, Inc., for the ENDEAR, CHERISH, CS2/CS12 studies; from Biogen for CS11; and from AveXis, Sarepta Pharmaceuticals, Novartis (AveXis), PTC Therapeutics, Roche, Scholar Rock, and Fibrogen. BTD has also received royalties for books and online publications from Elsevier and UpToDate, Inc.
This work was prepared while Christine J. DiDonato was employed at Ann & Robert H. Lurie Children’s Hospital and Northwestern University. The opinions expressed in this article are the author’s own and do not reflect the view of the National Institutes of Health, the Department of Health and Human Services, or the United States government.
Bakri Elsheikh has received compensation for consulting from Biogen, Genentech, Argenx, and Stealth Bio-therapeutics.
Wildon R. Farwell was an employee of Biogen during the period of time this manuscript was written.
Jacqueline Glascock is an employee of Cure SMA and has no financial stake in any SMA drug or company involved in developing SMA drugs.
Kelly Howell has no conflicts to disclose.
Jill Jarecki was an employee of Cure SMA at the time of writing and is currently an employee of Biomarin.
Stephen Kolb receives grant support from Novartis and has served as a paid advisor for Novartis, Biogen and Genentech.
Marco Petrillo is a former employee at Biogen Inc. All conflict statement: Marco Petrillo has received Biogen stocks during his employment period.
Charlotte J. Sumner receives or has received grant support from Roche Ltd., Ionis Pharmaceuticals, Biogen, and Argenx and has served as a paid advisor, consultant, and/or speaker to Biogen, Ionis Pharmaceuticals, PTC Therapeutics, AveXis, Novartis, Roche/Genentech, Cytokinetics and Biomarin. All conflict statement: Charlotte J. Sumner receives grant support from Roche Ltd., Biogen, Argenx, and Actio Bio and has served as a paid advisor, consultant, and/or speaker to Biogen, Ionis Pharmaceuticals, PTC Therapeutics, AveXis, Novartis, Roche/Genentech, Cytokinetics, Sarepta, Atlanta, and Biomarin.
Jessica Tingey is an employee of Cure SMA and has no financial stake in any SMA drug or company involved in developing SMA drugs.
At the time of this work, Marta Valente was a full-time employee of and held stock/stock options in Biogen.
