Abstract
Several successful clinical trials have been conducted in spinal muscular atrophy (SMA) over recent years which have led to the approval of splicing modifiers and gene transfer therapies. With an increasing number of other agents progressing through pre-clinical and clinical development, increasing worldwide clinical trial readiness is becoming essential.
SMA Europe initiated a clinical trial readiness project, which included the development of a pilot face-to-face educational-training initiative for clinical specialists and physiotherapists involved in SMA, with an emphasis on the patient perspective. Participants were selected through two surveys and, ahead of the meeting, a mock protocol with specific questions was provided. The initiative involved a series of presentations, role-play and interactive exercises. We describe here our experience and evaluation of this educational-training initiative, emphasising scientific aspects, psychosocial implications and level of satisfaction.
From a participant, patient and industry perspective, such training was considered successful and met the objective, which was to improve clinical trial readiness in emerging sites. Resource planning, ethical considerations and communication with patients were identified as three important topics for future training. This initiative highlights the need to develop a training programme to achieve clinical trial readiness across Europe and showcases a collaborative effort with different stakeholders, clinicians, patient advocacy groups and sponsors to address an important issue.
The present landscape of SMA therapeutic is evolving with new treatments and clinical trials Neuromuscular centres in different countries need to acquire experience to conduct clinical trials A unique cross-cutting educative experience to prepare centres for clinical trials is presented A full training programme should now be developed to achieve clinical trial readiness across Europe
ABBREVIATIONS
Adverse Events
Competent Authorities
confidential disclosure agreement
Clinical research associate
Clinical Research Coordinator
Clinical Research Facility
Contract research organisation
Clinical trials
European Commission
Electrocardiogram
European Medicines Agency
European Reference Networks
Good clinical practice
Investigator Brochure
Industry Consortium
Magnetic resonance imaging
Neuromuscular disease
Principal Investigators
Physiotherapists
Research Ethics Committee
Serious Adverse Events
Site Initiation Visit
Spinal muscular atrophy
Source Data Verification
Source Document Review
Suspected Unexpected Serious Adverse Reactions
INTRODUCTION
Spinal muscular atrophy (SMA) is a neuromuscular disorder with an incidence of approximately 1:10,000 live births and a worldwide carrier frequency of around 1:50 [1]. The primary manifestation of the disease is the weakening of voluntary muscles whereas the experience of patients is dominated by the downstream complications including limited functional ability, deformities, respiratory problems and impaired nutrition. SMA clinical manifestations range from serious congenital forms to minimal manifestations in adulthood [2]. Although the disease manifests itself as a continuum, three main types with specific subtypes are recognised based on age at onset and maximum milestones achieved. In the absence of disease modifying treatment, type I patients never sit, type II never walk independently, and type III patients walk but may lose this ability later in life. All SMA types are associated with significant disability and burden for caregivers and health insurances [3].
Several successful clinical trials (CT) have been conducted over recent years which have confirmed the safety and efficacy of nusinersen, an intrathecally administered antisense oligonucleotide in SMA Types 1 and 2, [4,5, 4,5], onasmnogene abeparvovec, an AAV9-mediated gene transfer in Type 1 [6, 7] and risdiplam, an oral compound [8]. A review of currently available data on new treatments was published by Ramdas & Servais [9]. Other compounds, including anti-myostatin, are currently in pre-clinical or clinical development.
In this context, increasing worldwide clinical trial readiness, through providing multidisciplinary teams with specialist training, preparing centres for SMA clinical complexity and developing communication skills, has not only the potential to increase site requirements and offer opportunities to recruit patients but also to increase disease awareness, hands-on experience worldwide and decrease patient burden. In addition, increasing clinical trial readiness may benefit the development of therapy in less common neuromuscular diseases.
To provide better basic knowledge and improve information concerning clinical trials in SMA and more generally in neuromuscular disease (NMD), with an emphasis on the patient perspective, SMA Europe initiated a pilot clinical trial readiness project in Europe, in collaboration with Cure SMA Industry Consortium [10]. As part of this project, we developed a pilot face-to-face educational-training initiative for clinical specialists and physiotherapists involved in SMA. Target professionals were initially selected from European countries where clinical trials in SMA have not yet been implemented. The experience focused on three main aspects: interactive evaluation of a simulated protocol; theoretical training in clinical research topics to prepare participants for the common difficulties of SMA and communication skills and role-play situations to recruit and involve patients. We describe here our experience and evaluation of the initiative, emphasising scientific aspects, psychosocial implications and level of satisfaction.
MATERIALS AND METHODS: STUDY DESIGN AND PROCEDURE
The organisation of the Clinical Trial Readiness educational workshop included four phases (see Figs. 1 and 2), the identification of centres, evaluation of their current knowledge on how to conduct SMA clinical trials, the workshop and feedback. Its overarching goal was to help ensure that the centres were equipped to carry out trials that meet the requirements of healthcare professionals, sponsors and people living with SMA.

The outline of the intiative and program content at a glance. Preliminary work in each Center was assigned. During the face-to face meeting 8 main topics were developed as a theoric part whilst a second role-play section with different situations was organized. Patient's perspective and industry interaction was also included in the final debate. Recollection of a second survey on opinions, suggestions and satisfaction was performed.
Phase 1 –Identification of participants
Survey of trial centres in Europe
Participants were selected from results obtained from two surveys carried out in Autumn 2018 and Autumn 2019, which set out to better understand the current clinical trial landscape in Europe to identify and pursue opportunities to increase trial site capacity. This formed part of an overall initiative to meet the needs of trial sponsors and the SMA patient community as the number of SMA clinical trials in Europe increases. This initiative was set-up by SMA Europe and the Cure SMA Industry Consortium in the US.
Clinical trial centres in Europe were identified through patient groups, Treat-NMD,
European Reference Networks (ERNs) and online resources. The surveys were designed using
Survey Monkeytrademark and consisted of 31 questions (Supplemental material 1) grouped in
4 main topics: General information The
centre’s SMA population Studies
taking place at the site Site
capacity for clinical trials
The surveys were sent to the centres identified which met the following criteria: Those conducting an
SMA clinical trial or had conducted an SMA trial in the past 5
years Those highlighted by SMA
patient organisations Those
administering nusinersen (Spinraza)trademark Those publicly acknowledged as a reference for SMA in a
particular country Those with
resident expert SMA clinicians
Countries selected and invited
An educational pilot programme to educate Principal Investigators (PIs), Clinical Research Coordinators (CRCs) and Physiotherapists (PTs) on best practice for effective trial conduct was developed for centres interested in conducting clinical trials in SMA in the near future.
Participants were sent a mock protocol with specific questions to carry out ahead of the face-to face meeting. This consisted of the aforementioned study design and procedure for a multi-centre, phase 1, open-label, single-dose peptide injection clinical trial to determine the safety and tolerability of delivering a fictitious gene therapy product, named Scottine-75/007, by intravenous infusion in patients with SMA Type 1 already on treatment with nusinersen. A deadline of one calendar month was given. Several organisational (huge burden for sites) and ethical (quantity of blood/ number of patients, safety) issues were inserted on purpose.
Phase 2 - pre-workshop evaluation of participants
Participants were asked to prepare a budget for the mock protocol. They also had to prepare a list of resources needed and anticipate potential organisational, regulatory or ethical issues. This preparatory work was sent to the trainers ahead of the meeting for evaluation (the preparatory work is available in supplementary material).
Phase 3 –educational workshop (Formal presentation and main topics)
A total of 8 main interactive educational lectures related to clinical trials in general, and SMA in particular, were provided (See Table 1 and Fig. 1).
Topics developed in the formal presentations
1. Setting-up a clinical trial: Pitfalls & tips
To introduce the complexity of clinical trial organisation, a series of concepts from regulation [European Medicines Agency (EMA), Clinical Trial Regulation (Regulation (EU) No 536/2014), EU Clinical Trial Directive (EC) No. 2001/20/EC and national legislations] to infrastructure (space, facilities and equipment as well as the personnel needed) were initially established. Discussion included required documentation needed to register the physician as a clinical trial investigator and to track and evaluate the ethical and procedural conduct of trials. Specific highlights were the study visit: a) the pre-study visit to evaluate the research staff members, facilities and departments, equipment, potential patient population and competing studies information; b) the site initiation visit after completion of signatures and ethical committee approval.
2. Ready to go: The ideal CRF
The ideal Clinical Research Facility (CRF) and the setting-up of a clinical trial in the context of SMA were presented. The most important part, as with any ward in a hospital, is patient safety. All CRF staff must be up to date with their clinical training, including life support and emergency scenario training. They must also have access to other units which carry out study procedures (such as X-ray, ophthalmology, magnetic resonance imaging (MRIs), theatres), to dose a patient by intrathecal injection of intravenous gene therapy in particular. Ideally CRFs will be able to facilitate patients’ stay overnight, which is often required for early phase studies or for observation post dosing or pharmacokinetics blood tests. Among the practical considerations to consider besides storage, it is important to ensure that patient rooms suitable for those with disabilities (wheelchair accessible) and if the environment is child and family friendly, making the study visits as pleasant as possible. The trainers shared their experience on how over the years they have encountered delays and issues, which they have learnt from. To ensure everything is ready for the site initiation visit (when the study opens), it is important that every relevant department is involved and that all equipment is onsite and checked before booking patients in for visits. Everyone must be prepared for adverse reactions, especially anaphylaxis, which is always a risk when trialling new medication. Medication to treat anaphylaxis must be close-by and all staff should be made aware when a first or second dose is to be administered. It was pointed out that clinical trials can be stopped at any time by the pharmaceutical company, leading to extreme disappointment and frustration for the families/patients and health professionals involved. It is therefore best practice to inform the family of this risk during the consent process and involve psychological support to the family if this happens, as it can be heart breaking forthem.
3. Networking, patient selection and approaching families
An overview of the issues related to patient selection, based on the experience gathered in different recent SMA trials, was given. One of these issues is the problem that the number of available slots is far below the number of candidates. The potential criteria for patient selection were also discussed: age, best clinical conditions, randomisation, patients living near the centres, parent commitment and level of understanding. The ethical and practical considerations of these different criteria weredebated.
Discussion was also focused on trials that are conducted in a site located in a country that has an approved drug. This leads to international recruitment, which involves patient relocation, and which could potentially lead to social, financial, administrative, and psychological issues for some parents. Some trials also have a very limited period of time for recruitment. Opportunity for early intervention, based on the possibility to confirm SMA genetically in the first weeks of life, made the development of pre-symptomatic trials possible. To ensure speedy recruitment, extensive networking must be done and plans for rapid parental information and patient inclusion must be made. Finally, the difficulties facing a trial that include a sham-procedure or a placebo when an alternative treatment is available, were also discussed. Randomisation is a fundamental element of clinical trial quality that ensures comparable groups and avoids bias. Other critical issues include facilitating a controlled setting in these trials, comparing new interventions to placebo or known alternative therapies in particular.
A special emphasis was put on issues of communication and patient (family) perception of the importance of participation and opportunities to receive the drug at some point during the trial. Communication skills and transparency to help in patient engagement are becoming crucial for best capture of the patient voice in clinical trials.
4. Informed consent and Good Clinical Practice (GCP)
The main principles of GCP which guide the design, conduct, performance, monitoring, auditing, recording, analyses and reporting of clinical trials were reviewed. The session focused on the GCP two main functions in particular: ensuring the rights, safety and wellbeing of trial subjects and the quality of data generated by the study.
The delicate process of informed consent/assent was extensively discussed with particular focus on ensuring that the patient/parents have ample time to read, discuss and ask questions to allow them to decide whether or not to participate in that specific trial. The informed consent/assent process should then be detailed in the source documentation and an informed consent log maintained for each subject and each version of consent approved by the EC. Copies of the signed version should be provided to the parents/patient.
Finally, the PI’s safety reporting responsibilities were discussed, drawing attention to the importance of declaring all Serious Adverse Events (SAEs) to the sponsor/CRO via the eCRF within 24 hours of the study site being aware of them. These should also be recorded in the eCRF, including concomitant medications and/or treatments.
5. Clinical endpoints and outcome measures
The aims and pitfalls encountered in outcome measures in SMA by a physiotherapist,
based on experience gathered in clinical trials and clinical follow-up of patients with
SMA, were presented: The aim of an outcome measure and how an outcome helps to understand the
disease trajectory of a patient The
development of different outcomes in SMA, by giving an overview of the origins of
the different outcomes and scales currently used in clinical trials and how they
have been designed. This included an overview of the most common scales and
outcomes depending on age and motor ability used in previous and current clinical
trials The rationale and the
importance of determining the most appropriate and accurate outcome for a specific
patient The need to adapt existing
outcomes to changing phenotypes was presented as an open question, which led to a
group-discussion.
6. Safety management: AEs, SAEs and SUSARs
An overview of the definitions of Adverse Events (AEs), Serious Adverse Events (SAEs) and Suspected Unexpected Serious Adverse Reactions (SUSARs) was presented. The importance of recording clinically significant laboratory abnormalities, such as AEs, was also discussed. Planned surgical procedures, for example spine surgery because of scoliosis, are not considered AEs if the conditions were known before study inclusion. Pre-existing medical conditions, present at the initial study visits that worsen in severity or frequency during the study, should be recorded as AEs. The classification of AEs by intensity was discussed and the Common Terminology Criteria for AEs presented, along with the way AEs can be classified based on their relationship to the study drug. The concept of study drug overdose, and other situations putting the subject at risk of an adverse reaction, and actions to take in the occurrence of these events, were also discussed. The concept of Expedited Safety Report was presented. The importance of SAEs and SUSARs collection and reporting timeframes was highlighted. Extensive discussion with the audience, considering the presence of potential principal investigators among the participants, was devoted to the investigators’ responsibilities, as these are crucial to maintaining patient safety during the study.
7. Audit & monitoring, protocol deviation
This presentation consisted of four main parts; the listing of PI and sub-investigator responsibilities; the differences between monitoring and auditing; the importance of adhering to principles for optimal data entry; preparation for regulatory inspections (see Table 2 for details). Deviations from the protocol, or non-compliances, are inevitable across multi-centre studies, which are very often the case in SMA. It is crucial not to deviate from the protocol, except when it is necessary to eliminate immediate hazard to patients. If this occurs, they should be noted in source and eCRF. Some deviations can be prevented, but others, such as missed visits due to patient's schedule, are challenging.
Investigator responsibilities, monitoring, auditory and data entry in SMA clinical trials
8. Patient retention
The importance and impact of retention were reviewed, factors related to dropout of patients during a CT discussed and strategies to avoid or improve retention of patients shared.
Retention is a crucial component of the study workload when conducting a clinical trial. Keeping patients on protocols from start to finish can be just as challenging as recruiting enough patients. There are serious consequences to dropouts, ranging from costly delays to missing data that can compromise the results and integrity of a study.
It is important to understand that patients have the right to discontinue participation
in research at any time and that a percentage of dropouts is usually considered normal.
However, some actions can be taken to prevent these withdrawals: Establishing follow-up mechanisms that
promote participant retention. Understanding why a patient chooses to discontinue
a study is valuable information to obtain, as it may provide insight into what
changes can be implemented to improve the patient
experience. Establishing a good
channel of communication and interaction, to instil confidence in the CT team,
that should be carefully maintained during the study. In this respect, the
therapeutic scenario of SMA has pros and cons that should be specifically
considered (see Table 3).
Expectations, trust, conviction, and final decisions should be balanced to warrant
good retention. In particular, the approval of a given medication may tempt
susceptible families to abandon a CT with one specific therapy in order to be
treated with one newly approved. Conduct all activities related to retention within the framework of ethics and
privacy regulations. The principle of respect for autonomy should support the
patient’s decision to withdraw from a CT. Good communication and keeping the
family informed every step of the way during a clinical trial, is important for
them to feel involved, as is regular feedback from the study progress when
possible.
Representative pros and cons for retention of patients in SMA trials
9. Role-play scenarios (brief summary of interactive outcomes)
This section was mainly organised as a role play session, which is considered an
effective strategy for developing multiple perspectives fostering empathy in different
hypothetical situations. Participants were invited to communicate with families about
the possibility of participating in the trial. The roles of the parents were played by
two trainers. Pre-established scenarios were proposed: Scenario 1: The patient does not meet
the inclusion criteria, but has extremely committed parents pushing for
inclusion Scenario 2: The parents
and patient live abroad with a willingness to relocate, but with poor
understanding of all practical considerations Scenario 3: The parents disagree with the aim of the
protocol Scenario 4: The parents
have limited understanding of trial implications, but are willing “to get the
drug”
All role plays were observed by the participants, who had to note all positive and negative aspects of the activity.
Phase 4 - post-workshop evaluation
Feedback surveys were distributed to all participants to gain insights on how beneficial participants found the workshop to be and to help plan future activities. Their opinion was sought on the quality and the relevance of the event, as well as the suitability of the formats used. In addition, we aimed to understand the ways in which the workshop might affect the running of clinical trials in the participants’ own hospitals. The diagram chart of the initiative is represented in Fig. 2 whereas the main programme and agenda is depicted in Fig. 1. Besides faculty members and professional trainers, patients' representatives also participated in this workshop. Industry representatives, however, were only present at the end to participate in a common debate.
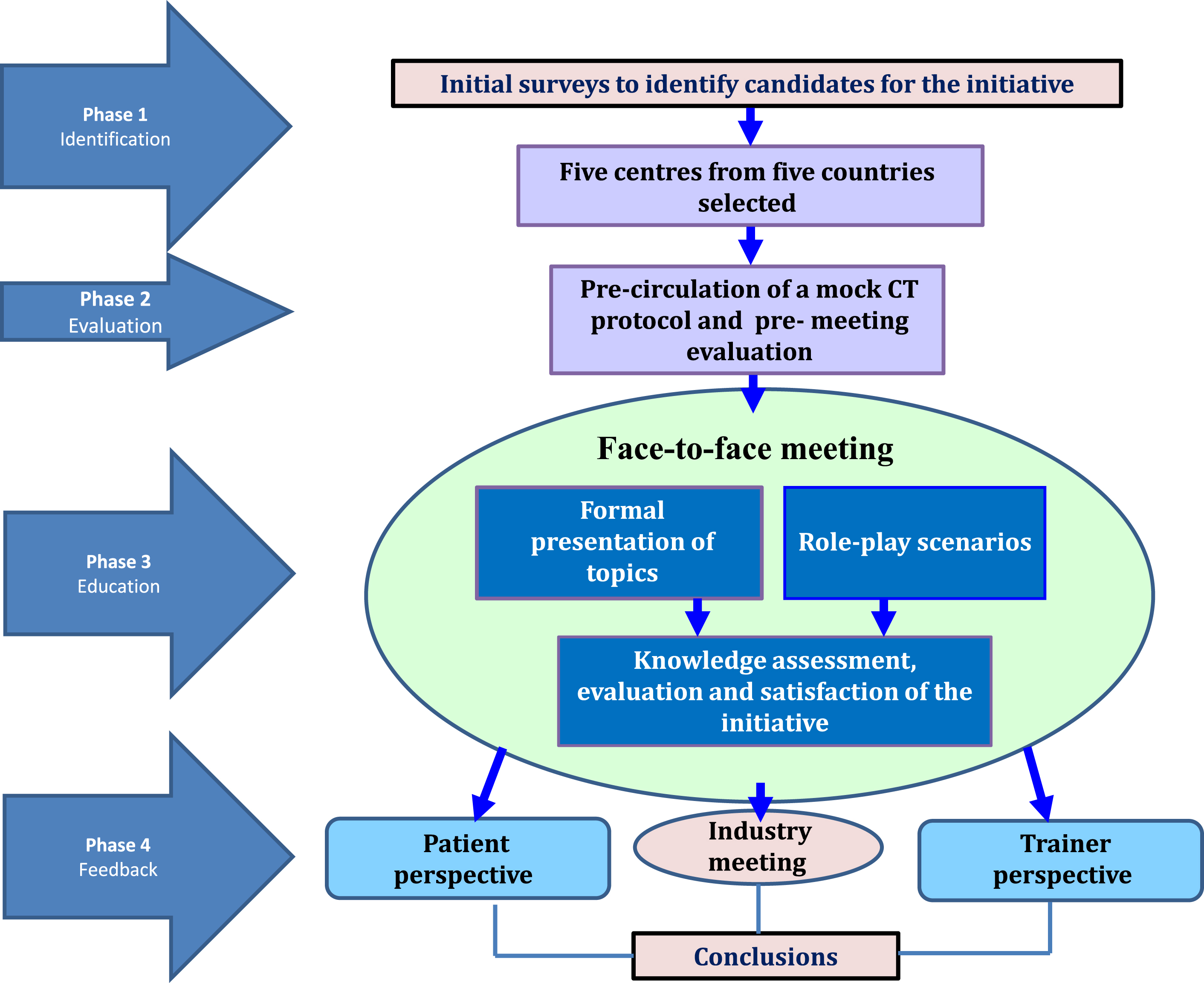
Diagram chart of the educational initiative. A total of 226 initial surveys to identify candidates to participate were disseminated. Five centres were selected from five countries without previous experience in SMA clinical trials. Precirculation of material such a mock protocol and comments and suggestion of changes to the protocol were indicated. The face-to face- meeting allowed a cross cutting experience between faculty members, participants and patient's representatives. A conclusion with perspectives from all sides including a spot for industry representatives (that were not part during the sessions but at the end when we have a common debate) was accomplished.
RESULTS
Phase 1 - Survey to select participants
A total of 226 clinical trials centres with experience and knowledge of SMA were identified in 31 countries: Austria, Belgium, Bulgaria, Croatia, Czech Republic, Denmark, Finland, France, Germany, Greece, Ireland, Iceland, Israel, Italy, Lithuania, Netherlands, North Macedonia, Norway, Poland, Portugal, Romania, Russia, Serbia, Slovakia, Slovenia, Spain, Sweden, Switzerland, Turkey, Ukraine and the UK. A preliminary analysis revealed that 84 hospitals in 25 countries responded. Of these: 56 centres could conduct new trials; 37 currently run SMA trials; just 3 lack Good Clinical Practice (GCP) certificates; all centres have space to conduct outcome measurements; some centres lack resources (staff or experience); training is required.
Invitations were sent to 17 of these centres, selected in collaboration with the countries’ patient organisations and according to the following criteria: if they had capacity to run clinical trials or if they were willing to start trials for SMA. Centres were limited to 1 or 2 per country.
From the responses received, five centres based in five different countries (Portugal, Russia, Serbia, Switzerland and Ukraine) were selected to attend, based on their neurologist(s) and physiotherapist(s)’ commitment to undertake the work set ahead of this pilot workshop and on availability on the day prior to SMA Europe’s 2nd International Scientific and Clinical Congress on SMA. Just 1 hospital had some experience of running a clinical trial for SMA (Table 4).
Participants CT experience
Phase 2 - pre-workshop evaluation of participants
Participants were asked to prepare a budget for a mock protocol, prepare a list of resources needed and anticipate potential organisational, regulatory or ethical issues.
It appeared that for most issues, sites tended to grossly under-estimate the burden, the staff or the resources needed. This would be largely due to the participants’ lack of experience in clinical trials; in order to provide practical guidance and advice, both the principal investigators and the trial coordinators, were involved in an open discussion sharing the process that each centre follows in budgetary preparation.
Interestingly, ethical issues were not raised, the main reason was that if the trial was approved by regulators and an ethical committee, no further ethical considerations were needed. Detailed results of the evaluation are available upon request.
Phase 4 - Knowledge assessment, evaluation and satisfaction of the workshop
Surveys were conducted in four main aspects: usefulness, quality of the programme and the best and worst aspects of the event and reports on trainer, patient and industry perspectives about the event.
Surveys
Usefulness of the event. 67% of Neurologists found the event to be extremely useful compared to 33% who found it to be useful. 100% of Physiotherapists found the event useful. All Physiotherapists and 83% of Neurologists thought the workshop very much fulfilled their educational goals and expected learning outcomes, compared to 17% of Neurologists who thought that it “somewhat” did.
Quality of the programme. 100% of Neurologists and 33% of Physiotherapists thought the quality of the programme was excellent compared to 67% of Physiotherapists found it to be good. The most useful session for all Neurologists was the one which tackled audit, monitoring, protocol deviation and patient retention whereas for Physiotherapists, it was the session which covered endpoints, outcome measures and safety. Sessions on setting up a trial and patient recruitment were either extremely useful or useful for both categories of participants.
The best aspects of the workshop. Some participants thought it was the role play to different scenarios. Others named meeting opinion leaders, patient advocates, pharmaceutical industry representatives and colleagues from other countries allowing for a stakeholder overview of clinical trials (to understand the problems from different standpoints and thereby allowing ideas of things to change or develop in the future), the homework and its analysis and atmosphere and specificity. In each session besides sharing opinions with other participants, they were able to interact with other professionals in a relaxed and confident setting.
The worst aspect of the workshop. These were minimal, ranging from wishing they could have changed rooms for lunch or thinking the font on some of the slides was too small. However, one of the Physiotherapists thought the workshop was “too medical” for them, although it was found to be interesting, there were no topics they would use in their practice. This is in fact reflected in the first question, where 100% of Physiotherapists found the event to be “useful” rather than “extremely useful”.
Most participants thought some of the information learned might be implemented in their centres such as how to organise, plan and conduct trials but some of it will be dependent on national legislations. Participants indicated that they picked-up very helpful tips on how to organise the space needed and how to be prepared for regulators’ inspections. They also learned helpful tips on how to communicate with sponsors, CROs and patients and picked up useful information from our patient representatives.
Trainer perspective
Trainers noted the participants’ willingness to learn and to improve. They also noted the gross under-estimation of resources and budget needed, which could lead to a real pitfall for the organisation and the conduct of trials at their site. In addition, ethical consideration and communication with patients was certainly a topic to be included in future training. Overall, trainers took on board the interest for such workshops but felt that it was far more operative to ensure clinical trial readiness in sites with little previous experience. They advocated for a much more structured and formal programme, with on-site training that would allow site-specific discussion, and twinning of experienced sites with emerging sites through a mentorship programme. They also advocated for the addition of training in standards of care, since standards of care and clinical trial readiness should not be dissociated.
Altogether, it appeared that participants were quite confident when explaining the protocol, but experienced difficulties in dealing with parent expectations and unrealistic plans.
Patient perspective
Patient representatives thought this workshop was an excellent first step towards the education of trial centres, as it allowed to assess the level of the participants and ensure a common level of knowledge. Participants were found to have varying levels of experience and understanding of how to lead CTs and in what the role of CRCs is in conductingthem.
On that basis, they thought the concept of the workshop was very good, especially the communication training with the use of role-play on scenarios. These allowed a comprehensive overview, with input from both sides and brought a patient-oriented approach to communication on the different aspects of CT participation such as the moderation of the language used by CT Centre staff to make it more understandable and better fulfil the need for information that individual families may have regarding CT participation to understand each step of the study and what is expected of them.
The trainers explained, emphasised and advised on all relevant aspects of clinical trials from the patients’ perspective and from the perspective of clinical doctors and physiotherapists. Patient’ representatives appreciated how the trainers put patients at the heart of the workshop. They spent time on the recruitment and retention of patients, how doctors should speak to them, the problems encountered during real-life processes and how to manage them. Similarly, participants’ willingness to learn and improve their communication skills was noted.
Furthermore, they highlighted the need for physiotherapy training in current outcomes used in SMA, which should be beneficial for the site to be selected to run CTs. Finally, patient advocates felt that future workshops should also include sessions on how to communicate with the adult population and other people involved in the decision-making process and communication, and to extend this training to a broader audience, including the whole multidisciplinary team.
Industry perspective on such events
At the end of the workshop, an hour discussion was planned with participants and industry representatives (Roche, Cytokinetics, Biogen and Avexis). Participants underlined the need for formal training, especially for physiotherapists, that could help their site become qualified, the industry representative expressed the need for similar workshops and training sessions to support clinical trial readiness and expansion of clinical trials in more countries. They all highlighted their wish to expand clinical trials in a larger number of countries and stressed the benefit of such training to support this plan.
Limitations
The number of participating centres was small. Seventeen centres out of eighty-four were invited, selected in collaboration with the countries’ patient organisations and according to the following criteria: if they had capacity to run clinical trials or if they were willing to start trials for SMA. Centres were limited to 1 or 2 per country with five out of the seventeen centres invited took part. This is because the workshop was designed as a pilot initiative, for which it was decided to include few interested centres, based on a first-come, first-served basis, aiming to inform with its results a larger training initiative.
CONCLUSIONS
From a participant, patient and industry perspective, such training was successful and met the objective to improve SMA clinical trial readiness in emerging sites. However, there is still a substantial amount of work to be done in order to accomplish clinical trial readiness and to ensure that trials are conducted in each site with the same policy and standards. Resources planning, ethical considerations and communication with patients were identified as three important topics for future training.
In conclusion, the programme of activities carried out by SMA Europe to expand Europe’s capacity to undertake clinical trials for SMA, which include this pilot experience, showcases how a collaborative effort with different stakeholders, clinicians, patient’ advocacy groups and sponsors, can address an important issue and may serve as a model for organisations in other regions and diseases.
Footnotes
ACKNOWLEDGMENTS
This work was initiated and driven by SMA Europe as part of its initiative to ready centres across Europe to either run or expand SMA clinical trials. We would like to acknowledge the individuals and organisations that participated in this pilot workshop.
FUNDING
This work was supported by a grant from the Cure SMA Industry Consortium (IC) in the US. The members of the SMA IC at the time the work was undertaken were AveXis, Inc, Biogen, Genentech/Roche Pharmaceuticals, Novartis, Astellas Pharmaceuticals, Cytokinetics Inc, Ionis Pharmaceuticals, Inc., and Scholar Rock. Each contributed financially to the overall programme of the Industry Consortium, which includes the work described in this paper.
