Abstract
Background:
SMA1 natural history is characterized by early development of chronic respiratory failure. Respiratory interventions in type 1 SMA infants are subject to great practice variability. Nusinersen, has been recently approved in Argentina. The advent of novel treatments has highlighted the need for natural history studies reporting disease progression in type 1 SMA.
Objective:
To analyze the progression, respiratory interventions and survival based on the type of respiratory support in type 1SMA patients, in a third level pediatric hospital in Argentina.
Methods:
Cohort of SMA1 patients followed at the Interdisciplinary Program for the Study and Care of Neuromuscular Patients (IPNM). Patient survival was analyzed by using the Kaplan-Meier method. Log-rank test was performed to compare the survival curve for three respiratory intervention groups.
Results:
59 patients. Mean age of symptom onset was 2.19 (±1.4) months, age at diagnosis was 3.9 (±2.1) months. Patients developed respiratory failure at 5.82 months (±2.32) and 13.8 months (±5.6) in Type 1B and Type 1C, respectively (p < 0.001) 53 p were SMA1B. Three copies were found in 1/6 SMA1C. Respiratory interventions: SRC 23 p (56.1%); SRC + NIV 8 p (19.5%); SRC + IV 10 p (24.4%). 8 patients were already on invasive ventilation when included in the IPNM. Patients with invasive ventilation showed longer survival.
Conclusions:
This series provides valuable information on respiratory intervention requirements and life expectancy in children with SMA1 before the implementation of novel treatments that increase the expression of the SMA protein.
Keywords
INTRODUCTION
Spinal muscular atrophy (SMA) is a devastating genetic disorder with an incidence of about 1 in 10,000 [1]. It is the second most common autosomal recessive disorder in childhood after cystic fibrosis. Pediatric SMA is classified based on age of onset and maximum motor function achieved [2] SMA type 1(SMA1) is the most frequent and severe form; it is characterized by profound global weakness, which may be evident at birth. Children never acquire the ability to roll and sit unsupported. Early respiratory failure is the major cause of morbidity and mortality and most patients do not survive beyond 2 years of age without medical intervention and respiratory support [3, 4]. SMA1 is further subdivided into 3 subtypes: 1A with clinical presentation since birth; 1B with onset of symptoms before 3 months of age, and 1C, the milder form, with onset between 3 and 6 months and head control achievement [5].
Deletions or subtle pathogenic variants in the survival of motor neuron 1, telomeric gene (SMN1) cause SMA. The absence of the SMN1 results in a reduced amount of functional full length survival motoneuron (SMN) protein. There is a nearly identical SMN2 centromeric gene, that differs from the SMN1 gene in a single substitution that dictates alternative splicing of exon 7, resulting in a truncated non-functional protein [6]. About 10– 15% of SMN2 protein is alternatively spliced to include exon 7. SMN2 gene copy number modifies the clinical presentation of the disease [7].
Respiratory management and interventions in infants with SMA1 are still today subject to great practice variability among countries and physicians and this leads to ethical concerns. Different levels of care are possible regarding respiratory support: tracheostomy and assisted mechanical ventilation, noninvasive ventilation (NIV) and palliative care without any assisted respiratory support [8, 9].
Treatment decisions are based on the child underlying condition and parental choice. The 2007 Consensus Statement for Standard of Care in Spinal Muscular Atrophy did not encourage the use of tracheostomy in SMA1 [10]. Experience, ethical and philosophical issues have always influenced physician recommendations for respiratory management in SMA1 [11].
In 2017 a novel treatment strategy, Nusinersen, a 2-O-methoxyethyl phosphorothioate-modified antisense drug, was approved by the FDA and has been recently approved in Argentina. Nusinersen is specifically designed to alter splicing of SMN2 pre-mRNA and thus increase the amount of functional SMN protein [4, 12]. Significant improvement in motor function has been observed with Nusinersen treatment, modifying the clinical course in children with SMA [2, 13]. Other therapeutic drugs under investigation may alter as well, in the near future, the disease progression [14, 15].
New therapeutic strategies have an impact on treatment decisions and respiratory support indications. The advent of these novel treatments has highlighted the need for natural history studies reporting disease progression in type 1 SMA. On the other hand, unfortunately, many patients (p) do not and most probably will not have access to these expensive pharmacologic treatments, depending on country-specific access criteria worldwide.
Data on natural history of SMA1 patients from Argentina have never been published. Hospital de Pediatría Dr. Juan P. Garrahan(HG) is the largest third level pediatric referral center, in Buenos Aires, Argentina, with 588 beds. An Interdisciplinary Program for the Study and Care of Neuromuscular Patients (IPNM) was created in 2007 to optimize diagnosis process, prevention of complications and care of these patients. It involves the coordinated effort of several specialists, including a neurologist, pulmonologist, pediatrician, respiratory and physical therapists, molecular biologists, pathologist, orthopedist, psychologist, cardiologist, sleep and palliative caregivers, among others. Since its creation, 220 SMA patients have been included in the Program.
The aim of this study is to analyze the progression, respiratory interventions and survival based on the type of respiratory support in type 1 SMA patients, in a third level pediatric hospital in Argentina.
MATERIALS AND METHODS
This is a retrospective study from a cohort of SMA1 patients followed at the Interdisciplinary Program for the Study and Care of Neuromuscular Patients (IPNM) in HG, between 02/2007 and 02/2017. All consecutive patients with a diagnosis of SMA 1 admitted to the IPNM were recruited in the study (Table 1). Prospectively collected data from the IPNM medical records were considered for the analysis.
Patients admitted to the IPNM. Baseline characteristics
∗Results expressed in months as mean±SD. ∗∗Median range. aFor seven patients there was no sample available for SMN2 copy numbers studies.
The primary outcome was defined as the age at death. The secondary outcomes were: respiratory interventions and survival according to the type of respiratory support.
SMA diagnosis was performed by molecular test (PCR-RFLP) in exon 7 and 8 of SMN1 gene [16]. SMAI was diagnosed on the basis of homozygous deletion in SMN1 gene, and the inability to roll or sit unsupported at any time. The copy number of SMN2 was established using SALSA MLPA probemix P021-A2 SMA(MRC-Holland). Classification of SMA1: SMA Type 1A: Onset of symptoms at birth or perinatally, with respiratory failure. SMA Type 1B: Onset of symptoms before 3 months of age. Head support not achieved. SMA Type 1C: Onset of symptoms after 3 months of age. Head support achieved. Demographics, age at symptom onset (by parental recollection), age at diagnosis confirmation, age at admission in the IPNM, home distance from HG, family history of SMA (siblings/1st grade cousins), SMA 1 subtype, SMN2 copy number, age at respiratory failure (RF), respiratory interventions and survival time were analyzed. Respiratory failure was defined as the inability to maintain normal oxygen saturation (measured by pulse oximetry) under spontaneous ventilation. Respiratory interventions: Since the creation of the IPNM, the different options to treat respiratory failure were thoroughly discussed with the parents, who had the time to think and consider which was the best option for their child and family.
Treatment options: Supportive respiratory care (SRC), which included intensive physiotherapy to clear respiratory secretions, training in hyperinflation maneuvers and cough assistance, use of nasogastric tube (NG) to avoid aspiration into the respiratory system and to provide appropriate nutritional support, immunizations and palliative care. SRC was indicated in all patients since diagnosis, according to their progression and needs. Supportive respiratory care plus noninvasive ventilation (SRC + NIV), once recurrent respiratory infections and hospitalizations, acute or chronic respiratory failure appeared. Supportive respiratory care plus tracheostomy and invasive ventilation (SRC + IV), once chronic respiratory failure appeared or NIV failed in face of an acute respiratory infection.
Patients were followed up by the IPNM team, whichever the accorded management was.
The present study was approved by Hospital Garrahan Ethical Committee.
Statistics
Continuous variables were represented by means and SD or medians and interquartile range and analyzed by t test and analysis of variance or Mann-Whitney tests, as needed.
Patient survival was analyzed by using the Kaplan-Meier method. A comparison of the survival curve estimates for each of the three respiratory intervention groups was performed using log-rank test.
Statistical analysis was performed using STATA 13 [17].
RESULTS
Between 02/2007 and 02/2017, 59 patients with SMA1 were admitted for follow up to the IPNM. Fifty-one patients had constant follow up since diagnosis; 8 p were older and already on invasive ventilation when included. The baseline subject characteristics are summarized in Table 1.
The mean age of symptom onset of the 59 p was 2.19 (±1.4) months, while the age at diagnosis was 3.9 (±2.1) months. In this series of patients there were no infants with SMA type 1A.
SMN2 copy number was available in 52 of the 59 infants. Three copies were found in only 1 of the 6 children with the milder phenotype who achieved head control.
Patients came from allover Argentina and neighboring countries: 6 p from Buenos Aires city; 28 p from cities <200 km away; 25 p (42%) from distances greater than 200 km, including 4 p from Paraguay and 2 p from Bolivia. Seven patients were referred for treatment and follow up closer to their home. Three patients deserted follow up after diagnosis confirmation.
Seven patients (12%) had a previous family history of SMA1. In six cases there was a sibling with SMA1. In one case, a 1st grade cousin had recently died from SMA1 and respiratory failure.
Patients developed respiratory failure at 5.82 months (±2.32) and 13.8 months (±5.6) in Type 1B and Type 1C, respectively (p < 0.001).
The respiratory interventions agreed between families and the IPNM team once RF arouse were as follows: SRC 23 p (56.1%); SRC + NIV 8 p (19.5%); SRC + IV 10 p (24.4%). As previously mentioned, 8 patients were already on invasive ventilation when included in the IPNM.
SRC+NIV patients were started on NIV as follows: In 4 p, during acute respiratory failure, in an intensive care unit. In the other 4 p, it was started proactively, during night and daytime sleep, before the development of respiratory failure.
Patients who received invasive ventilation showed longer survival, compared with the other two groups. Comparison of mean survival between patients who received SRC and SRC + NIV showed no statistical significance (p 0.32) (Table 2 and Fig. 1).
Respiratory interventions and Survival
∗1 patient survived 64 m. Both patients with longer survival were type 1C. NIV: Noninvasive ventilation, IV: Invasive ventilation through tracheostomy.
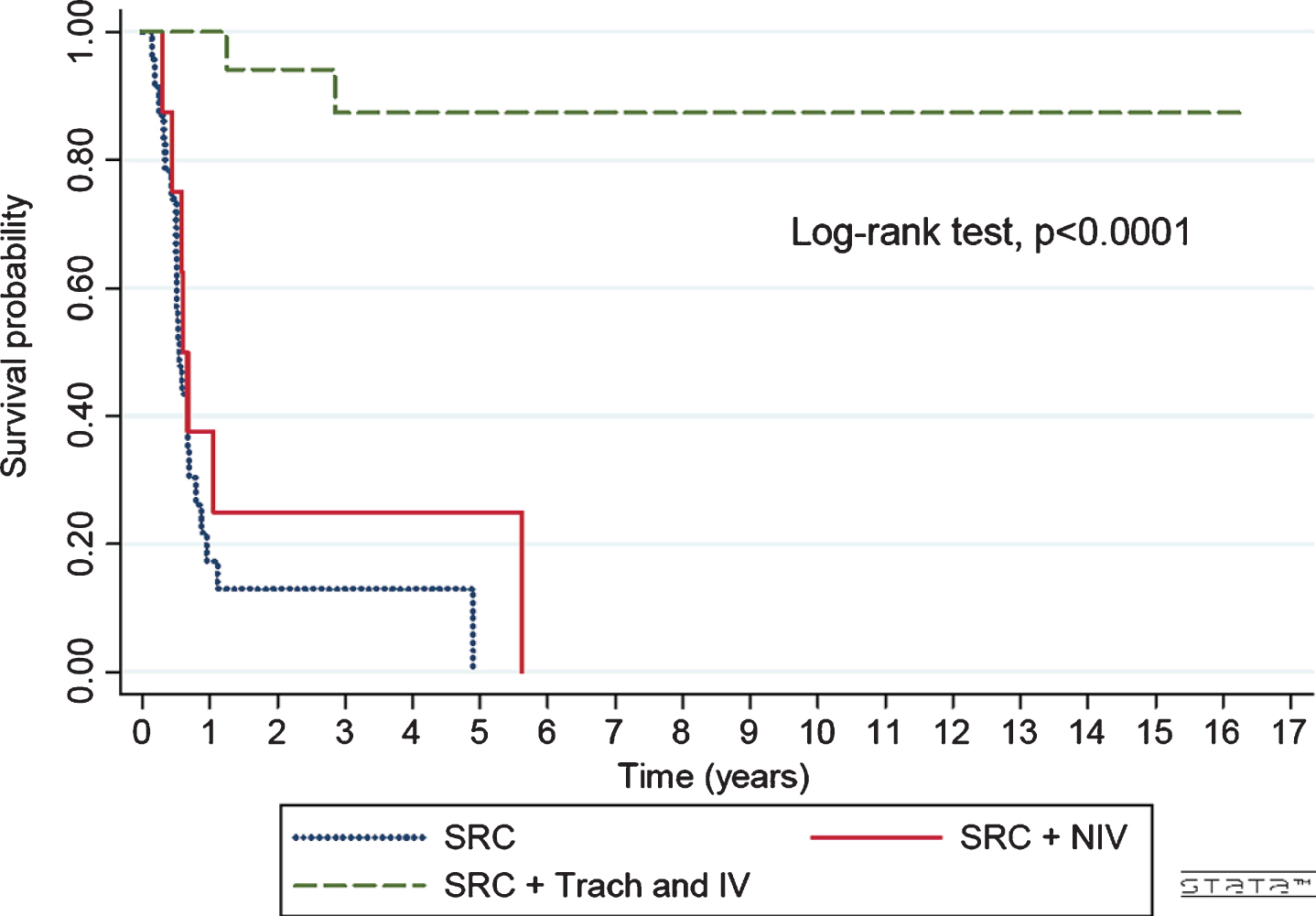
Survival probability based on respiratory intervention. n: 49. SRC (Support respiratory care): 23p, SRC + NIV (Support respiratory care plus noninvasive ventilation) n: 8p, SRC + IV (Support respiratory care plus invasive ventilation): 18p.
Those SMA1 children who were tracheostomized and invasively ventilated in our hospital (n = 10) required hospitalizations between 210 and 2,370 days long. Median hospital stay was 605 days.
DISCUSSION
Clinical studies regarding natural history of SMA1 in LatinAmerica are scarce [18]. The possibility of prompt molecular diagnosis, early respiratory and nutritional support and the indication of SMA new treatments is very variable in this region [19]. Argentina is considered an emergent country, in which 27% of the total population live below the national poverty line.
With new treatments for SMA becoming available and Nusinersen recently approved in our country, it is crucial to describe the natural progression of the disease in order to objectively assess changes in treated infants and to support regional treatment recommendations. The present study generates data on the natural history of type 1 SMA in a national pediatric referral hospital, where more than 70% of Argentinian pediatric SMA patients are followed up.
Our data revealed that since the creation of IPNM there has been consistency in SMA1 management, in terms of clinical, respiratory, nutritional and symptomatic care. Molecular tests were performed in the Molecular Biology Laboratory of the Department of Genetics, with results available within a week after diagnosis suspicion. The delay between symptom onset and diagnostic confirmation is probably related to a low suspicion index in primary pediatric care.
In this series, most SMA1 patients were type 1B and they had a lower age at symptom onset than type 1C patients, as expected. All SMA type 1B patients had 2 SMN2 copies. Only one of the six type C patients had 3 copies of SMN2. The SMN2 copy number is inversely related to clinical severity; however, the correlation is not absolute [7].
As described before SMA1 natural history is characterized by early development of chronic respiratory failure [5, 20]. Respiratory failure developed around 6 months in patients with type 1B SMA, in accordance with was has been previously described in other international series [20]. Type 1C patients developed respiratory failure later than type 1B infants (13.8±5.6 months) Under such circumstances, medical team and families had to take difficult decisions. Choices in respiratory interventions were mostly due to differences in the severity of the disease and in parents’ desires, considering cultural and ethical aspects, religion, personal values, family structure and support.
Most families rejected the option of IV. The perception of a low quality of life without changes in the disease progression and severity, the image of an intellectually normal child in a state of complete paralysis with very limited possibilities of communication in addition to the financial and the family psychological burden might be some of the reasons for those decisions.
The IPNM team assisted the decision-making process before progression to respiratory failure and gave support to the family throughout the disease. A high adherence to the follow-up program was found.
As in other series, in our population the probability of 2 years’ survival without respiratory support was below 20% [5, 20]. We could not demonstrate an advantage in terms of survival with the introduction of NIV once chronic respiratory failure developed, compared to the use of supportive care only; however, the sample size was small for this purpose. Those patients with invasive ventilation lived significantly longer, but long hospitalizations were the rule in our population. The absence of an established homecare organization in the public health system arises with great difficulties to provide the patients and families with suitable in-homecare services.
Over the last years, our approach to respiratory treatment has changed. Airway clearance techniques, pulmonary hyperinsuflation and pressure support ventilation have been introduced in a proactive way, following the latest international recommendations [13]. In May 2019 Nusinersen was approved for SMA type 1, 2 and 3A in Argentina. Local guidelines for the treatment of SMA have been recently published [21]. We are aware that conflicts concerning ethics of economic resource allocation and difficulties with financial coverage may arise [22]. However, there is evidence that the irreversible loss of motor neurons in humans with SMA type 1 begins in the perinatal period [23], and early treatment is critical to modulate this rapid and progressive degeneration. Best results have been associated with early treatment, before significant motor weakness or loss is present [24].
Limitations to the interpretation of these data include the relatively small sample size that precludes the possibility of comparisons. Furthermore, some data were missing in clinical charts.
CONCLUSION
This series provides valuable information on respiratory intervention requirements and life expectancy in children with SMA1 before the implementation of novel treatments that modify SMA protein expression. Treatment and respiratory interventions in SMA1 patients were variable, reflecting the lack of local recommendations at that time. We still do not know whether the possibility of treatment will change the family and medical perceptions about tracheostomy and invasive ventilation. It remains likely that clinical decisions regarding the extent of respiratory support will remain difficult in many patients with SMA.
We hope that in the near future all SMA patients could have access to diagnosis, care and specific treatments in due time.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
