Abstract
Background:
Ambulatory individuals with spinal muscular atrophy experience weakness and impairments of speed and endurance. This leads to decreased motor skill performance required for daily living including transitioning from floor to stand, climbing stairs, and traversing short and community distances. Motor function improvements have been reported in individuals receiving nusinersen, but changes in timed functional tests (TFTs) which assess shorter distance walking and transitions have not been well documented.
Objective:
To evaluate changes in TFT performance over the course of nusinersen treatment in ambulatory individuals with SMA and identify potential factors [age, SMN2 copy number, BMI, Hammersmith Functional Motor Scale Expanded (HFMSE score), Peroneal Compound Motor Action Potential (CMAP) amplitude] associated with TFT performance.
Methods:
Nineteen ambulatory participants receiving nusinersen were followed from 2017 through 2019 (range: 0–900 days, mean 624.7 days, median 780 days); thirteen of 19 (mean age = 11.5 years) completed TFTs. The 10-meter walk/run test, time-to-rise from supine, time-to-rise from sitting, 4-stair climb, 6-minute walk test (6MWT), Hammersmith Expanded and peroneal CMAP were assessed at each visit. Linear mixed-effects models were used to evaluate unadjusted and adjusted changes in these outcomes over time.
Results:
Apart from time to rise from sitting and from supine, all TFTs were found to improve over the course of treatment after adjusting for baseline age and BMI.
Conclusions:
Improvement in TFTs over time in patients with SMA treated with nusinersen suggests that shorter TFTs may have value to assess individuals with SMA who have or later gain ambulatory function during treatment.
Keywords
INTRODUCTION
Spinal Muscular Atrophy (SMA) is a recessively inherited neuromuscular disorder caused by homozygous deletion or mutation in the survival motor neuron 1 (SMN1) gene with concurrent retention of a nearly identical copy gene, SMN2, on chromosome 5q13 [1]. A silent nucleotide transition in SMN2 exon 7 impedes splicing of the full-length SMN2 transcript, yielding very low functional SMN protein levels in the absence of SMN1 and selective motor neuron dysfunction [1–5]. Concomitant degeneration of anterior horn cells results in progressive weakness and subsequent impairment of motor function. Disease severity and clinical prognosis appear proportional to the number of copies of the paralogous SMN2 gene, with age of symptom onset and maximum motor milestone achievement utilized to describe the clinical spectrum [6–8]. Individuals with SMA at the weakest end of the phenotypic spectrum are typically unable to achieve sitting (type 1 SMA), while patients who are stronger may sit (type 2) or stand and walk (type 3 and 4). Without treatment, progressive loss of motor function is observed.
While there is currently no complete cure for SMA, rapidly evolving drug development and innovative gene therapies have led to promising therapeutic strategies available through clinical trials as well as FDA approved commercial therapies [9–11]. In clinical trials, a broad range of symptomatic and pre-symptomatic study participants have demonstrated significant and clinically meaningful benefits, as evidenced by increased survival, improved motor function, and acquisition of new motor milestones, and in those treated presymptomatically on target achievement of motor milestones has been observed [9–15]. These changes have been measured by a plethora of functional outcomes developed and adapted specifically for the SMA population [16–21]. While each of these outcomes has effectively monitored disease progression in both natural history studies and trials of disease modifying therapies, it is imperative that we continue to explore outcomes that are meaningful to our patients and families as the natural history of treated SMA evolves. Shorter TFTs were used in the distant past but discontinued as primary outcomes as the disease process made the activities difficult and not effective at quantifying positive progress [22, 23]. As more individuals with SMA gain or regain the ability for household ambulation or transitional milestones, such as rising to stand and climbing stairs, there is a need to re-consider use of shorter timed function tests (TFTs) to evaluate these improvements in rising [time to rise from supine on floor to standing (TTR-supine)], climbing [time to climb 4 stairs (TTC-4 stairs)], and short distance ambulation [10-meter walk-run test (10MWRT)] [17, 22–25]. In conjunction with the validated 6MWT for SMA, change in time to complete shorter TFTs can provide an additional metric to assess the newly evolving phenotypic spectrum of individuals affected by SMA who have or later gain ambulatory function and transitional skills during treatment.
The primary aim of this study was to characterize change in TFT performance over the course of nusinersen treatment in a cohort of ambulatory individuals with SMA receiving disease modifying therapy at a comprehensive multidisciplinary neuromuscular clinic. A secondary aim was to identify potential factors (age, SMN2 copy number, BMI, HFMSE score, peroneal CMAP amplitude) associated with TFT performance.
MATERIALS and METHODS
Ethics Statement
The natural history study was approved by the Institutional Review Board (IRB) at Ann and Robert H. Lurie Children’s Hospital of Chicago (LCH). Before inclusion in the natural history study, all individuals or their parent(s)/legal guardian(s) provided written informed consent and/or assent in accordance with local IRB guidelines. Research was done in accord with the Helsinki Declaration of 1975. Data was de-identified for analysis.
Participants
All ambulatory individuals with type 3 SMA who participated in the SMA natural history study at LCH, received commercial nusinersen starting prior to 2020 (between 2017 and 2019) and completed TFTs for any visits after initiating treatment, were included in this study. All except one participant had confirmed homozygous deletion of the SMN1 gene and were classified as SMA type 3; one participant had a point mutation within SMN1 exon 1 (compound heterozygote). The participant that was a compound heterozygote harbored one deleted SMN1 allele and an A2 G point mutation within exon 1 of the other SMN1 allele and presented with 2 copies of SMN2.
Procedures and outcomes
Individual’s height, weight, age, type of SMA, maximal motor function, and survival motor neuron 2 (SMN2) copy number were recorded for each visit. Data for all patients was collected prospectively within the 2-week window prior to baseline and at each follow up visit (Day 60, 180, 300, 420, 540, 660 and 780, 900). As the study had rolling enrollment, we used all visits for ambulatory patients that started on nusinersen at any point within the study period between 2017 and 2019 and who successfully completed at least two TFT assessments for any one TFT or for all TFTs. Some patients could not complete all TFTs at their initial visits and therefore had missing data for those TFTs until they could successfully complete them. Therefore, not all subjects had TFTs completed at all visits over the study time period and not all subjects had the same number of visits for each TFT during the study period despite being followed every 4 months after loading doses were completed. The mean number of visits per ambulatory subject enrolled was 5.6, and the median was 6. Data was collected using standardized protocols for the: 10-meter walk/run (10MWRT), time-to-rise [from supine (TTR-supine) and from sitting on a bench (TTR-sitting)], 4-stair climb (TTC-4 stairs), and the 6-minute walk test (6MWT). Additionally, Hammersmith Functional Motor Scale Expanded (HFMSE), and peroneal CMAP amplitude were completed at each visit.
Outcome measures
While standing, height was measured to the nearest centimeter using a stadiometer. Body weight was collected to the nearest kilogram (kg) on a standard balance scale. Body mass index (BMI; weight in kg/height in m2) was calculated from measured weight and height. Age and gender-specific BMI reference values (CDC, NHANES) were used to assign BMI to study participants [26].
As part of a comprehensive motor testing battery, all TFTs and motor function tests were administered by experienced physical therapists (PTs) with SMA expertise. Tests were administered in standardized order with rest periods between tests as needed. PTs (n = 3; 2 primary, 1 back-up) were trained in all motor function measures and timed function tests and reliability was assessed bi-annually. No physical prompts or cues were provided during testing, however verbal encouragement was allowed. No orthoses or assistive devices were allowed for any testing. Shoes were allowed for the 6MWT, and verbal prompts were per ATS guidelines [26]. All shorter TFTs were completed without shoes.
10MWRT
This test effectively measures short distance walking ability with minimal participant burden. It can effectively discriminate between younger and older ambulant individuals with SMA and correlates with both knee flexor and extensor strength and with longer distance walking [22, 29] Participants were asked to walk/run as fast as possible over a fixed 10-meter distance. The fixed 10-meter distance was measured and marked with tape and included a cone at the start line and the therapist standing at the finish point. The participant was instructed to walk/run as fast and as safely as possible. The clinical evaluator stood at the end of the distance with parents/caregivers providing safety measures from behind. To rule out the effects of deceleration on gait velocity the participant was allowed to continue several feet past the finish line before stopping. The added ramp down distance at the end also minimized falls and loss of balance with rapid deceleration at the end of the 10MWRT. A stopwatch was turned on as the lead limb crossed the start line and clicked off when the trail limb crossed the finish line. Time required to walk/run the fixed distance was recorded in seconds.
Time to climb four 6” stairs (TTC-4 stairs)
TTC-4 stairs has been shown to discriminate between younger and older ambulant individuals with SMA, and correlate with knee flexor and extensor strength [22]. For this test the child started in front of and at the base of the stairs with arms at sides and was instructed to climb up the stairs in an upright position, as safely and quickly as possible, using rails only as needed. The timer was turned on at the “go” signal and stopped once both feet were on the top stair and the participant was facing forward away from the evaluator. Time was recorded in seconds. Pattern of climbing (reciprocating or step-to) and use of rails (none, one or two, or two hands on one rail) was also recorded.
Time to rise from supine on floor to standing (TTR-supine)
TTR-supine discriminates between younger and older ambulant individuals with SMA and correlates with knee flexor and extensor strength [22]. Participants started this task lying supine on the floor with arms at sides and feet in a comfortable resting position, no greater than hip width apart. They were instructed to get up to stand as safely and quickly as possible without use of an external surface. Timer was started at the “go” signal and stopped once the participant was in upright with both hands by sides. Time to complete was recorded in seconds and use of Gower’s maneuver (no hands, 1 hand or 2 hands) was recorded.
Time to rise from a bench (TTR-bench)
For this test, participants sat on a bench/plinth placed on a tile or carpeted surface with hips and knees at 90/90 and feet in contact with the floor. They were instructed, if able, to get up from the bench without use of arms. Timer was started at the “go” signal and stopped once the participant was in upright and standing still. If they required hands on bench for balance, they were instructed to use hands only for balance and not to push-off using momentum. Time to complete was recorded in seconds and use of hands on the legs or bench was recorded.
6-Minute Walk Test (6MWT) and Children’s Omni Rating of Perceived Exertion (OMNI-RPE)
Six-minute walk test (6MWT) guidelines were adapted from ATS guidelines and SMA study specific procedures as previously reported [27, 31]. Participants were instructed to walk along a linear 25-meter course, turning at each end around a cone marker, repeating the course loop for 6-minutes, or for as long as they could. They were instructed to walk, without running, and standardized, ATS verbal cues and encouragement were provided at regular intervals. If a participant needed to rest, they were allowed to do so but had to remain standing during the rest. Distance walked for the 6-minute period and distance walked for each minute was recorded. Time to complete each 25-meter interval, distance per minute, and number of falls, if any, was recorded. Participants were asked to complete the Children’s OMNI Rating of Perceived Exertion (OMNI-RPE) scale for walking and running immediately after completing the 6-minute walk. The OMNI-RPE scale allowed participants to self-report general level of physical effort and fatigue after completion of the 6MWT using a pictorial scale with perceived exertion responses that ranged from 0–10 [32]. Fatigue was calculated as the difference between the distance walked in the 1st and the 6th minute by subtracting the 6th minute distance from the 1st minute distance. The difference was divided by the distance walked in the 1st minute and expressed as a percentage, with a positive value indicative of fatigue [25, 33].
Hammersmith Functional Motor Scale Expanded (HFMSE)
The HFMSE is a disease specific gross motor function measure that consists of 33 items assessing gross motor function in individuals with type 2 and 3 SMA. The HFMSE has demonstrated reliability and validity, is widely used in clinical trials of SMA to document natural history and disease trajectories, and has demonstrated reliability, validity, and sensitivity in clinical trials of nusinersen [14, 34–37]. On average, the HFMSE takes less than 15 minutes to complete, requires minimal participant burden, and uses standardized equipment. For the purposes of our study, the HMFSE was administered using previously reported standardized procedures [35].
Peroneal Compound Muscle Action Potential Amplitude
Peroneal CMAP amplitude measurements were obtained from the tibialis anterior (TA) muscle. The low-frequency and high-frequency filter settings were set to 10 Hz and 10 kHz, respectively. Skin temperature was maintained at > 31°C. The G1, G2 and ground electrodes were placed on the TA muscle using standard measures based on anatomical landmarks. Using an age-appropriate bipolar probe, the peroneal nerve was supramaximally stimulated just proximal to the popliteal fossa (behind the knee). Maximum values for negative peak amplitude and negative peak area were obtained in millivolts (mV). Studies were either performed without anesthesia or post anesthesia (local or general), prior to scheduled nusinersen dosing.
Statistical Methods
Descriptive statistics were used to summarize SMA type, SMN copy number, height, weight, baseline BMI, TFT times, 6MWT, 6MWT-fatigue, HFMSE, and peroneal CMAP amplitude. Frequency and percentage were recorded for all categorical variables. For numeric variables, mean, standard deviation, median, interquartile ranges and range were calculated.
A linear mixed effects model was used to model time function tests and peroneal CMAP amplitude over time. Time to rise from supine, 10MWRT time, time to climb 4 stairs, and time to rise from bench were log transformed. The Kenward-Roger approximation was used to estimate denominator degrees of freedom. A random intercept for the subject was included to account for repeated measurements. All models included baseline age, BMI, and time as fixed effects, where time was defined as the number of years since initial nusinersen treatment (i.e., baseline).
Associations between TFTs (10MWRT, TTC-4 stairs, TTR-supine, TTR-bench) and HFMSE, and peroneal CMAP negative peak amplitude were explored by adding each variable as a fixed effect, above one at a time, in the models specified. All analyses were conducted using R (version 3.5.3, 2019, The R Foundation) or SAS (SAS Institute Inc., Cary, NC, USA) and assumed a two-sided, 5% level of significance. No adjustment was made for multiple hypothesis tests.
RESULTS
Nineteen participants with type 3 SMA treated with commercial nusinersen were followed between 2017 and 2019 at our institution. At initiation of drug intervention (start of this study) 6/19 were no longer ambulatory which left us with n = 13 patients diagnosed as type 3 who were still ambulatory. We only collected and reported TFT data for those that were independently ambulatory. Because the study was an ongoing clinical study and anyone with type 3 could potentially become ambulatory over time and complete TFTs all 19 were initially included in case they achieved the ability to ambulate again during the study follow-up period. During the time period under observation (2017–2019) none of the 6 who received treatment and were non-ambulatory at the start of the study achieved ambulation post treatment. Therefore, TFT analyses only included thirteen of the original nineteen[mean age = 11.5(7.05), median age 7] who were ambulatory at baseline and completed any TFTs. See Table 1 for participant demographics. Thirteen participants completed at least one TTR-sitting, 10MWRT, and TTC-4 stairs. Eleven completed TTR-supine and 6MWT.
Summary of participants at baseline
SMN: spinal motor neuron; HFMSE: Hammersmith Functional Motor Scale Expanded; CMAP: Compound Motor Action Potential.
Correlations between TFTs and covariates of interest (age, BMI, HFMSE, peroneal CMAP and SMN2 copy number at baseline) are pictured in Fig. 1. After adjusting for baseline age and BMI, 10MWRT, TTC-4 stairs, and 6MWT distance and fatigue were associated with time on treatment (Table 2, Fig. 2, and Fig. 3). Specifically, for each subsequent year, 10MWRT was 0.85 times or 15% faster [eβ: 0.85 (0.810 0.90), p-value < 0.0001], and TTC-4 stairs is 0.80 times or 20% faster [eβ: 0.80 (0.69, 0.92); p-value: 0.0037]. On average, 6MWT distance increased 48.48 meters [β: 48.48 (36.77, 60.20), p-value < 0.0001] for every year from initial treatment (Table 2). TTR-supine, TTR-bench, and 6MWT fatigue showed no significant change over time on treatment [TTR-supine eβ: 0.92 (0.83, 1.02), p-value = 0.1229; TTR-bench eβ: 1.14 (0.90, 1.44), p-value = 0.2646; 6MWT fatigue: –5.03 (–11.0, 0.88), p-value = 0.0933].
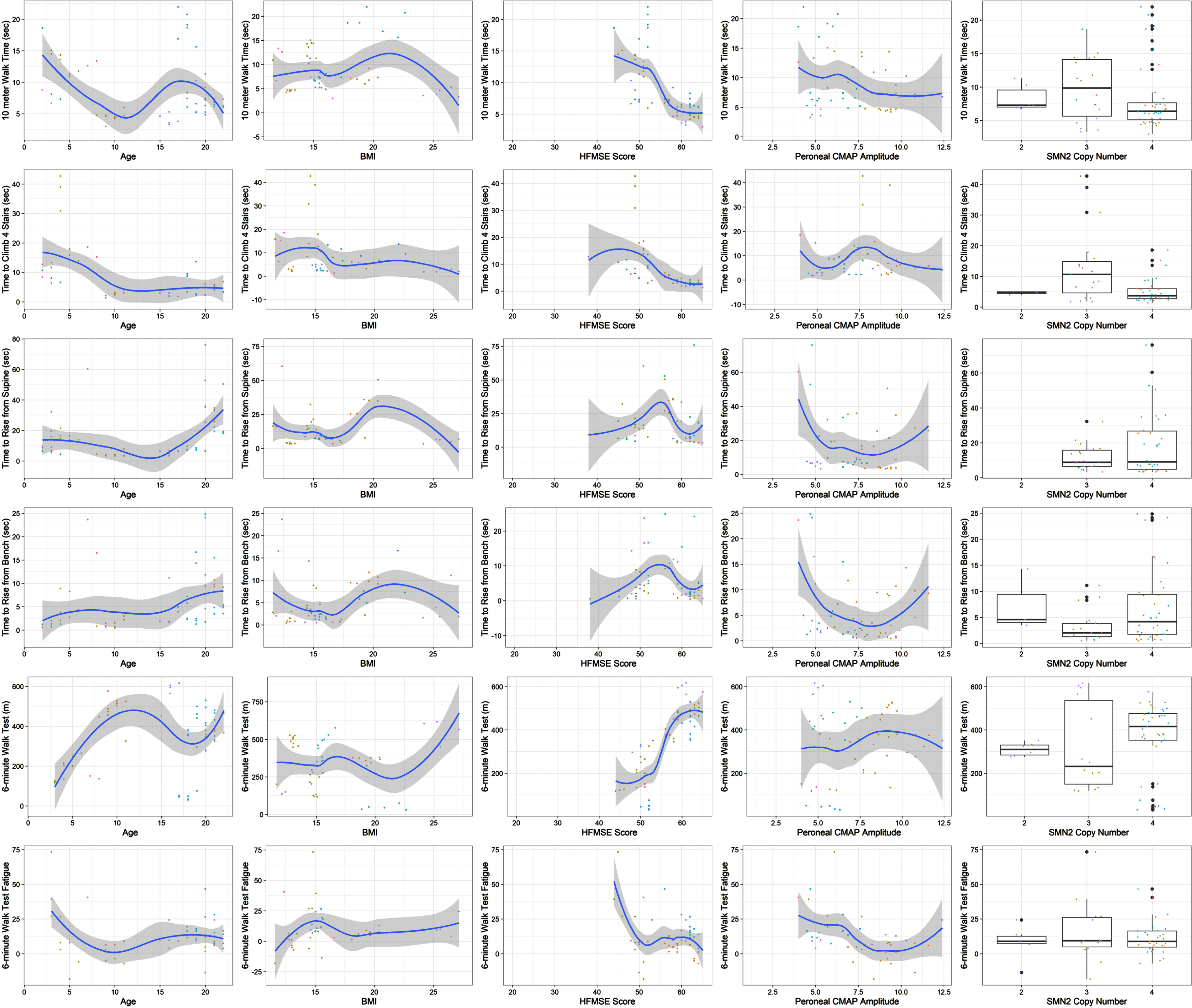
Covariates Associated with Timed Function Tests. Relationship of covariates [age, body mass index, Hammersmith Functional Motor Scale expanded (HFMSE), peroneal compound motor action potential (CMAP) and spinal motor neuron 2 (SMN2) copy number] with each of the TFTs (10-meter walk/run test, time to climb 4 stairs, time to rise from supine, time to rise from a bench, 6-minute walk test distance, 6-minute walk test fatigue).
Changes in Timed Function Test after adjustments for age and BMI
NS = non-significant; *Significant at p < 0.05; 6MWT: 6-minute walk test.
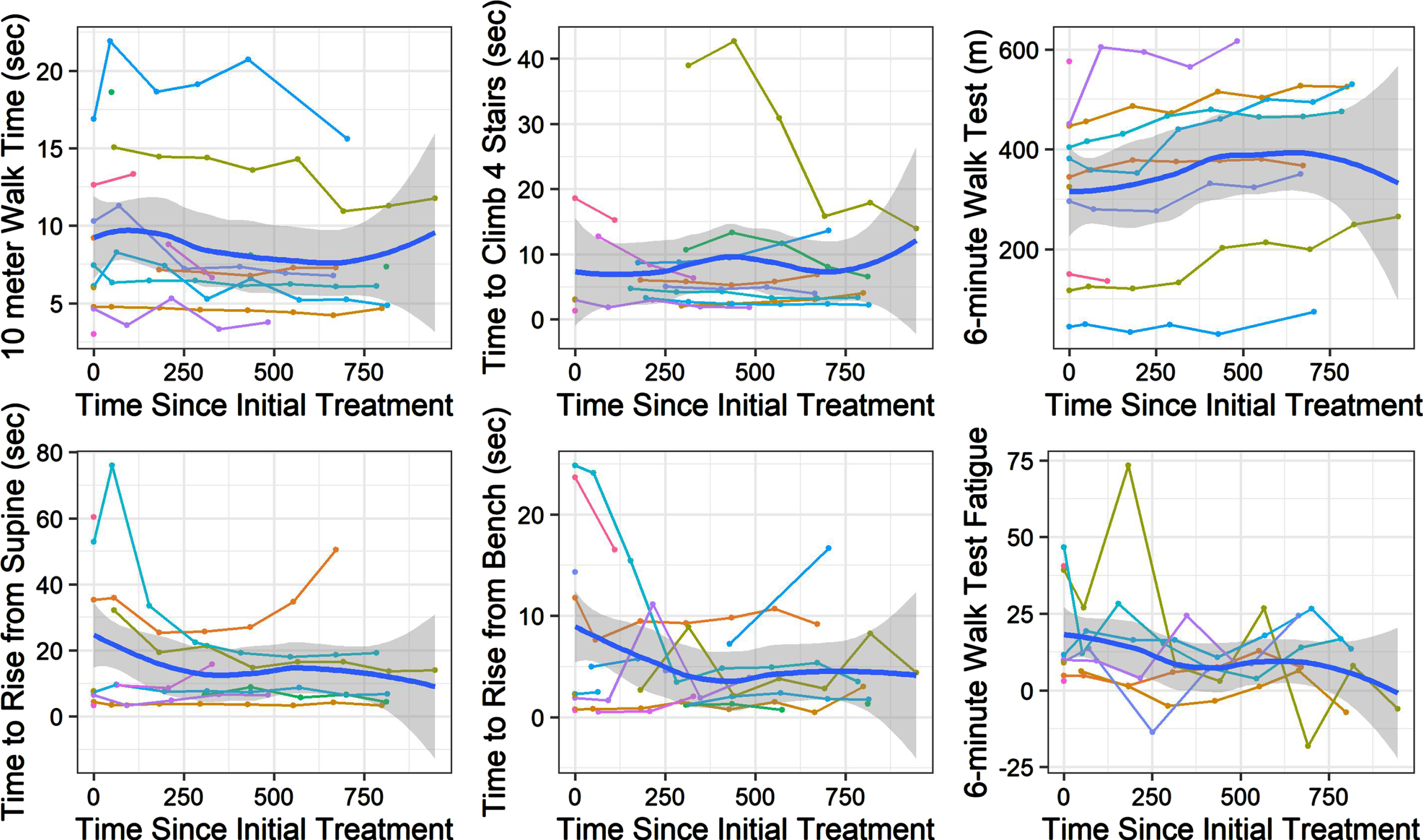
Longitudinal change over time in timed function tests. Change in time across days in study depicted for 10-meter walk run, time to rise from supine, time to climb 4 stairs and time to rise from a bench. Change in distance depicted for 6-minute walk test and change in fatigue (calculated as the difference between the distance walked in the 1st and the 6th minute divided by the distance walked in the 1st minute and expressed as a percentage, with a positive value indicative of fatigue). Polynomial line (ribbon = 95% confidence interval) describes progression over time.
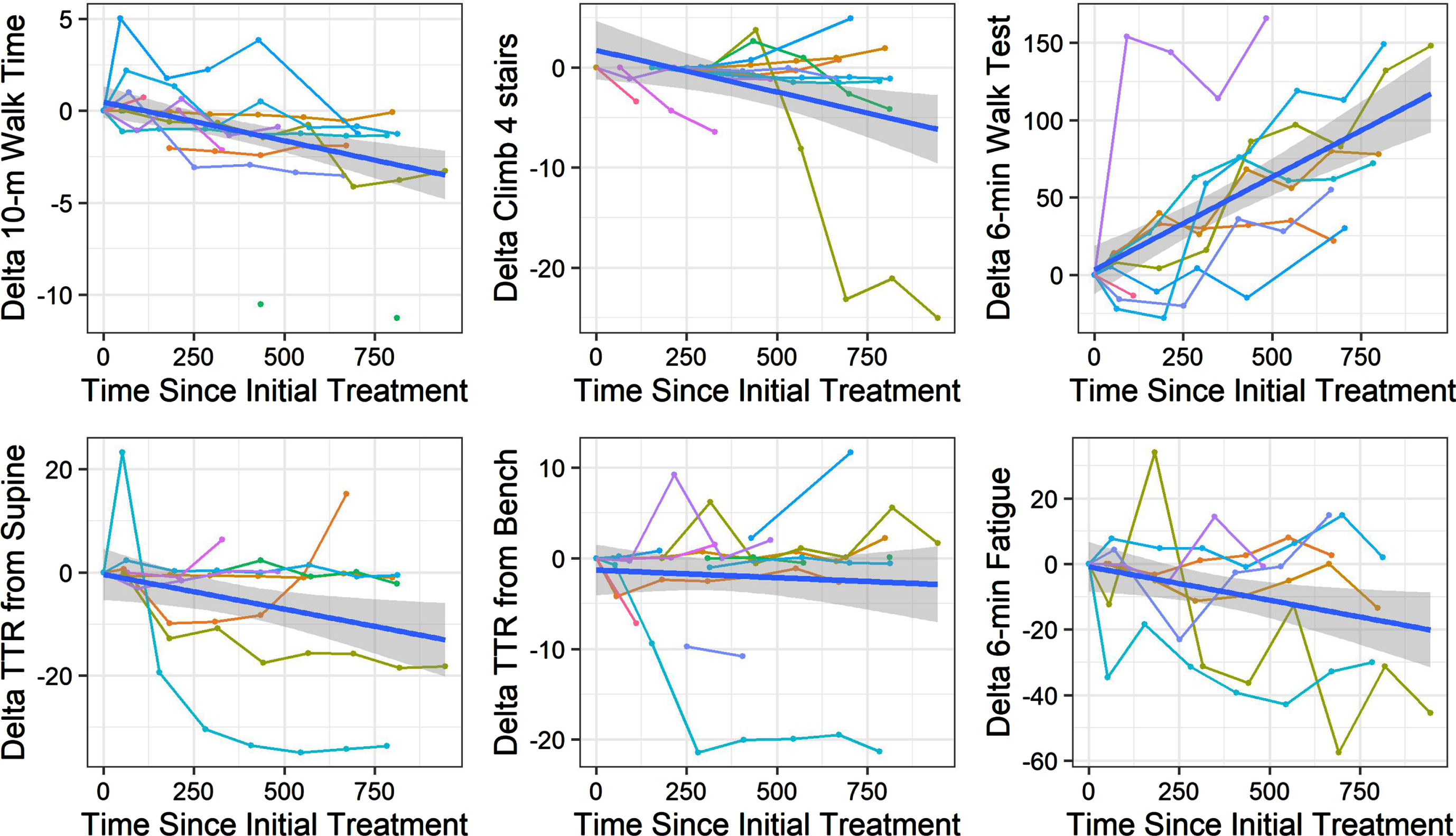
Delta change in timed function tests over time in days. Longitudinal delta change in time in seconds (10 meter walk/run test, climb 4 stairs, rise from supine, rise from a bench) and distance in meters (6MWT) on timed tests of function.
An increase in HFMSE of 1 point was associated with a 7% decrease in time to climb 4 stairs (eβ: 0.93, 95% CI: (0.89, 0.97); p-value = 0.0020). The HFMSE showed no association with other TFTs. Due to the small cell sizes, no formal comparison was made between copy numbers.
Finally, after adjusting for baseline age and BMI, the data suggested that peroneal CMAP amplitude increased over time. Specifically, peroneal CMAP amplitude increased 0.63 mV each year on treatment [β peroneal: 0.68 (0.37, 0.98), p < 0.0001]. There was no significant association between change in CMAP and change in TFTs.
DISCUSSION
In the last decade extensive efforts have been undertaken to develop or revise outcome measures that are valid, robust and capture meaningful aspects of disease course in people affected by SMA, in natural history studies, clinical trials and in “real-world” clinical settings. As treatment availability increases it is paramount to continue efforts to utilize and apply measurement tools that can assess clinically meaningful changes that lead to improvements in daily life. This will allow clinicians to effectively capture changes in phenotype with treatment. Emerging improvements in shorter TFT trajectories in ambulatory individuals treated with nusinersen may effectively translate to an improved ability for individuals affected by SMA to complete functional transitions related to daily living and independence, and thus should be considered as important and meaningful additions to our outcomes’ toolbox.
In this study, all TFTs, except time to rise from the bench and from supine, were observed to be responsive to nusinersen treatment with individuals exhibiting improved walking and stair climbing speed as time on treatment increased. Therefore, pertinent TFTs should be considered for individuals with SMA that are ambulant as secondary endpoints in both research and clinical settings. Although limited natural history data in SMA are available for the 10MWRT, TTR-supine, TTC-4 stairs and TTR-sit [22, 38], these easily administered clinical measures of function have held up as clinically meaningful and valuable outcome measures used for prognostication, assessment of disease progression, and evaluation of response to therapy in other neuromuscular disorders such as DMD [39, 40]. Our data demonstrate that individuals with SMA treated with nusinersen do exhibit improvement in some TFTs overtime.
One could hypothesize that the lack of significance for delta change in TTR-bench and TTR-supine was reflective of the complexity of those two tasks for persons with SMA as compared to the 10MWRT, 6MWT and TTC-4 stairs. Each TFT utilizes different movement patterns, biomechanics, levels of strength and motor control. It is speculated that the 6MWRT, 10MWT and TTC-4 stairs all start in upright and as the patients are already balanced in upright prior to initiating and completing the task they really only need to gain increased power for forward or upward motion which might come from a gain in strength from treatment. Time to rise from the bench and the floor require somewhat more sophisticated motor planning, control, strength, and balance and likely call into play a larger variety of muscle groups as they must transition into standing and then manage to stand and balance at task completion. Additionally, the transitions within these tasks break up their compensatory patterns and would likely require significant strength gains over a longer period of time to change/improve scores significantly. For the TTR-bench most participants who could stand up easily had very fast baseline times (<1 second) and really could not improve speed over time. Participants that found the task harder actually used a much more complex pattern as they shifted their weight and turned to face the bench prior to rising using a Gower’s maneuver. For those participants the task seemed more arduous and they thus moved slowly and may need more power and control to improve their time and efficiency. Both of these may explain the lack of improvement in speed over time in this task. Similarly, TTR- supine is a complex multipart task that often requires several transitions (ie, roll to prone, push up onto all fours or plantigrade and then use of a bilateral Gower’s to rise). This task also requires far more proximal power and control than the others so was hypothesized to be less likely to change during the time period of this study, especially in the older patients leading to a group delta change that was not significant. These two tasks could also be more impacted by any scoliosis or contractures that patients may have to start with. Further study may better delineate if change can occur or why change does not occur in these tasks.
Intrathecal nusineren therapy resulted in improvement in motor function and peroneal CMAP amplitude [41]. It has previously been demonstrated that disease severity in SMA is correlated to the degree of innervation as determined by CMAP amplitude [42]. In our study change in peroneal CMAP amplitude showed no significant association with motor function, however we demonstrate that peroneal CMAP amplitude trends in the same direction as expected with improvements in TFTs. (Fig. 1). This might suggest that potential reinnervation, as evidenced by peroneal CMAP amplitude increase, contributed to improved speed on most TFTs in this study cohort.
In SMA natural history studies there is limited published data on shorter TFTs in untreated individuals. Merlini et al (2004) compared TFT speed (10MWRT, time to climb 6 stairs, time to rise from supine) in individuals who were younger (5–17 years) vs adults in a cross-sectional study cohort [22]. Findings supported faster performance in participants 5–17 years of age vs adults. In another study, a significant improvement was demonstrated at 12 months, but not at 6 months, for the 10MWRT in patients with SMA treated with gabapentin compared to untreated patients [23]. However, there were no changes for other timed tasks (time to rise or time to climb stairs). Our study demonstrates change over time with nusinersen treatment on all TFTs except for time to rise (from bench or supine). In a more recent SMA longitudinal natural history study, the 10MWRT demonstrated increased speed from ages 3 to 8 years, albeit at a slower rate than observed in age typical peers, and then a plateau or continued decrease in speed was noted through age 20 [38, 44]. Therefore, continued longitudinal improvement in subjects with a median age of 11 (7.0–18.0), treated with nusinersen suggests this observed improvement is an effect of treatment.
While this data provides a unique insight into how shorter TFTs change over time in ambulatory individuals with SMA treated with nusinersen, we recognize that the small number of participants and visits per TFT, as well as a broad range of abilities amongst participants, limits generalization of these findings. In addition, rolling enrollment into natural history studies leads to variation in number of participants in various age cohorts. Age and time are confounding, and as all participants were on nusinersen, we cannot distinguish between age and time on drug as an explanation for observed changes. These limitations can be addressed by a continued longitudinal study of larger groups and across multiple sites. Based on their responsiveness to SMA treatment in this study, multisite studies with a larger participant pool would also allow for validation of responsiveness of TFTs with treatment. Additionally, to fully appreciate the sensitivity of TFTs further studies comparing change in TFTs in treated versus untreated individuals using a concurrent randomized placebo control group would be of value, however, such is not realistic in the US with most patients now opting for treatment. In our study all the individuals with SMA were on disease modifying treatment (DMT) i.e nusinersen. The outcome of the DMT in this population was therefore the objective of the study. The placebo effect that could occur in treated populations is always a possibility, but our study used TFTs rather than patient questionnaires only, to address change over time. Also, without treatment one would not expect a delta change over time but rather a plateau or lack of response. We therefore believe that a placebo effect was mitigated. One might also consider that observed change over time could have been secondary to a placebo effect or learning curve, with improved strategies of control over time. Thus, in any follow-up study a longer follow-up period might be needed to assess durability of response (boiler vs placebo effects). Finally, ongoing studies are needed to provide greater understanding of trajectories of change in ambulatory patients with SMA (improvement vs stabilization) and to determine effects of add on therapies as patients continue nusinersen over time, and/or choose alternate treatments or dual therapies.
Dysfunction in gait and transitional activities of daily living have important implications for function in everyday life. Ambulant children with neuromuscular disorders, including SMA, commonly report problems with walking, poor balance, dependence for transfers and transitions, frequent trips and falls, increased fatigue and difficulty keeping up with peers [28, 46]. Therefore, consideration of gait assessment and functional transition abilities in ambulant children with SMA in clinic and research settings, is useful. There is no single assessment tool of functional ambulation or transfers that encompasses all factors. However, there are several clinical assessments that may collectively illustrate the effect of disease and disability on gait and function in children with SMA. While the 6MWT is the gold standard for assessing endurance and physiologic fatigue in SMA, the shorter TFTs may assess power during single short-duration tasks that are meaningful to individuals in daily life. This is a current limitation of our study as the correlation between the 6MWT and other TFTs were not fully explored and could provide support for complementary findings that using two sets of tests together can offer. As the direction of change in TFTs, like the 10MWRT, varies by age it would be important to consider such, in any therapeutic trial where TFTs were utilized [38]. Blending age groups could wash out effects of a therapeutic intervention, especially in children under 8, where developmental change is expected in both typically developing children and untreated children with SMA.
Early “real world” studies of nusinersen in persons with SMA who are ambulatory are limited to date, but do note that improvements are slow and primarily significant in the 6MWT and the Revised Upper Limb Module [47–51]. Therefore, the changes our study noted in these shorter TFTs is an important observation. For persons affected by SMA, the ability to climb stairs, get off the floor and walk are important milestones that are clinically meaningful and relevant to regulatory authorities. Improvement in TFTs over two years, noted in this study, suggests that nusinersen is slowing disease progression and stabilizing and/or improving function.
Footnotes
ACKNOWLEDGMENTS
This work was supported in part by:
The Mazza Foundation Neuromuscular Disorders Program, Anne & Robert H. Lurie Children’s Hospital and The National Institutes of Health’s National Center for Advancing Translational Sciences, Grant Number UL1TR001422. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
This work was prepared while CJD was employed at Ann & Robert H. Lurie Children’s Hospital and Northwestern University. The opinions expressed in this article are the author’s own and do not reflect the view of the National Institutes of Health, the Department of Health and Human Services, or the United States government.
CONFLICT OF INTEREST
KJK has received personal fees for advising, consulting or speaking from Biogen, Genentech-Roche, ASPA Therapeutics, Columbia and Stanford University, and Cure SMA and her institution receives subcontracted support from Lurie Children’s for Biogen clinical trials. All are outside of the submitted work.
LB has received personal compensation as a speaker for Biogen and Sarepta unrelated to this study.
KH has received personal compensation as an Advisory Board member Genentech-Roche outside of the submitted work.
CJD was employed at Ann & Robert H. Lurie Children’s Hospital and Northwestern University when this work was prepared. The opinions expressed in this article are the author’s own and do not reflect the view of the National Institutes of Health, the Department of Health and Human Services, or the United States government.
NLK serves on Medical Advisory Boards for Argenx, Biogen, Novartis, Roche and Sarepta. Her institution receives research funds from Biogen, Novartis, Roche and Sarepta.
VKR has received personal fees for advising, consulting, or speaking from Avexis, Biogen, Genentech-Roche, Scholar Rock, PTC therapeutics, NSPharma, Regenxbio, Sarepta therapeutics, France foundation, Cure SMA, and MDA outside of the submitted work.
KBZ, HM, JB, DPS, ANS, AND ANB have nothing to disclose.
