Abstract
Background and Objectives:
Thymic pathology is common in Myasthenia Gravis(MG) and plays a crucial role in its pathogenesis and clinical outcome. This study aims to discuss the clinicohistopathological spectrum of thymic lesions in MG.
Methods:
In this retrospective study, MG patients who underwent thymectomy from 2011 to 2020 were included. Clinical, radiological, serological, and histopathological details are described.
Results:
Of 83 patients(F = 45; M = 38), 7(8%) had ocular myasthenia, and the remaining 76(92%) had the generalized form. At onset, the median age was 36 years(M = 44; F = 31). AChR antibody was positive in 71/79 patients. RNST showed decrement response in 68/78 patients. The histopathological study demonstrated thymoma in 44(53%), thymic hyperplasias [32(38%)], involuted thymus [5(6%)], thymic cyst (1) and thymic lipoma (1). WHO grading of thymoma: B2- 48%, AB-18%, B-18%, B3-14%, A-2.3%. In these, capsular infiltration was noted in 11/44, 9 had focal and 2 had diffuse infiltration. Active germinal centers were present in 20/32 patients with thymic hyperplasia and 4/44 with thymoma. Thymomas were predominant in males and thymic hyperplasia in females. The age of onset and antibody positivity rate was higher in thymoma patients.
Conclusion:
In our cohort, there is a female preponderance. Thymoma was the commonest pathology followed by hyperplasia. We observed earlier onset of myasthenia in females. AChR antibody positivity rate was more frequent in thymomas. This study indicates that clinico-radiological evaluation adequately supported by serology and histopathology can effectively recognize the type of thymic pathology that can guide these patients’ treatment planning, management, prognosis and follow-up.
INTRODUCTION
Myasthenia gravis (MG) is a relatively rare acquired autoimmune disorder caused by an antibody-mediated blockade of neuromuscular transmission resulting in fluctuating skeletal muscle weakness and rapid muscle fatigue [1]. The overall global annual incidence rate is thought to be 8 to 10 cases per 1 million persons and a prevalence of 150 to 250 cases per 1 million [2]. The exact epidemiological data of MG in India is not known. There is a bimodal age of onset, with a predominance in females below 40 years and males over 50 years [3]. The autoimmune attack occurs when autoantibodies form against the nicotinic acetylcholine postsynaptic receptors (nAChR)at the neuromuscular junction (NMJ) [4, 5]. The most common subtype of autoantibody-mediated MG (approximately 85%) is nAChR. In the remaining 15%, autoantibodies targeting muscle-specific kinase (MuSK), lipoprotein receptor-related protein 4 (LRP4), voltage-gated calcium channel (VGCC), ryanodine receptor (RyR), and titin muscle protein are found [6]. The small fraction of patients with no detectable autoantibodies are called seronegative MG. MG may be restricted to the eyes (ocular myasthenia gravis- OMG) or maybe more generalized (generalized myasthenia gravis- GMG) [7].
It is well established that there is an association between thymic pathology and MG. The thymic abnormalities are known to trigger autoimmunity against the NMJ proteins [5]. Up to approximately 75 percent of individuals with MG have distinctive thymus abnormalities, which is a peculiar feature of the disease [8, 9]. Of these patients, 75% have thymic hyperplasia, 15–20% thymoma and a small percentage possess rare lesions, including thymolipoma and thymic carcinoma [10]. Many novel methods have been developed in the management of MG [11]. But an effective thymectomy in MG patients presenting with thymic mass can alleviate the symptoms and improve the prognosis.
The first known successful thymectomy in an MG patient was reported by Blalock et al., in 1939, where they removed a cystic tumor of the thymus followed by near-total resolution of symptoms in the patient [12]. Most of the studies in the literature have not included combined comprehensive clinical, radiological, serological and histopathological features of thymic lesions associated with MG. Hence, the current study aims to discuss these characteristics associated with MG in a large cohort of patients referred to a quaternary center providing neurological services.
MATERIALS AND METHODS
Study design
This retrospective study attempts to describe thymic pathology in patients with MG who had thymic abnormalities on radiography and had undergone thymectomy. Institutional ethics committee approval has been obtained to collect all the relevant data from the medical records. The study period extended over ten years from 2011 to 2020. The patients who were clinically diagnosed with myasthenia gravis and got operated on for thymic lesions were included in this study.
The patient details were collected from revaluation and review notes in the medical records. The clinical information mainly included demographics of the patients, presenting symptoms, distribution of muscle weakness, presence of myasthenic crisis and radiological findings. The laboratory findings and serological (antibody) profile were noted wherever available. The histopathological sections (from formalin-fixed paraffin-embedded blocks) of the resected thymus were reviewed in the department of neuropathology. The findings were recorded and correlated with the clinical and other ancillary results.
Investigations
Routine investigations like complete blood count (CBC), serum electrolytes, blood glucose, HBA1c, creatine kinase (CK), liver / renal / thyroid tests, lipid profile, HIV, Hepatitis -B, and C, erythrocyte sedimentation rate (ESR), urinalysis, prothrombin time and activated partial thromboplastin time (PT/aPTT), electrocardiography (ECG) and 2D ECHO were done preoperatively. Other special investigations like antinuclear antibody profile, antineutrophil cytoplasmic antibodies (ANCA), anti-phospholipid antibodies, and paraneoplastic neuronal antibodies were done to rule out other associated autoimmune disorders and internal malignancy.
A repetitive nerve stimulation test (RNST) was done at a slow rate of 3 Hz to look for decremental responses in the rested and post exercised muscles [13]. Abductor digiti minimi, abductor pollicis brevis, deltoid, trapezius, nasalis and orbicularis oculi muscles were examined. Decrement of greater than 10% in the baseline to peak amplitudes between first and fifth stimulation was taken as clinically significant. Nerve conduction studies and electromyography were done in some patients to rule out other muscle disorders and associated peripheral neuropathy.
The AChR autoantibodies (AChRAb) were measured by the ELISA or Radioimmunoassay (RIA) method. Titres less than 0.25 nanomoles per liter (nmol/l) were considered negative, between 0.25–0.40 nmol/l as equivocal and greater than 0.40 nmol/l as positive. The ELISA/RIA technique measured the MuSK antibody (MuSKAb) in some seronegative patients. Titres of more than 0.05 nmol/l were considered positive [6].
Indications for thymectomy were thymic enlargement/tumor documented on contrast high-resolution computed tomography (HRCT) [14]. All patients underwent transsternal thymectomy (total or sub-total) depending on the condition of the thymic mass regardless of the CT diagnosis [15, 16]. Some patients with normal CT underwent exploratory surgery/CT-guided biopsy in suspicion of a thymic mass, and the resected tissues were subjected to histopathological examination (HPE).
HPE
The sections from the formalin-fixed paraffin-embedded blocks were reviewed. These were studied on routine hematoxylin and eosin. Special stains such as Masson’s Trichrome (for connective tissue), reticulin and PAS stains were carried out wherever required and when histologically indicated. Immunohistochemistry (IHC) was generally not performed; however, a proliferative marker (MIB1) was performed occasionally. The histological analysis included recognizing the lesion, cellularity, components, participating cells, and their arrangement/architecture pattern, cell characteristics (nucleus and cytoplasm), stroma (septa/interstitium) and associated cells. Cystic change and additional features, if any, were recorded. A definitive diagnosis was arrived at after considering the histological features. The thymomas were classified according to the recent WHO classification [17].
Statistical analysis
The statistical analysis was done by Statistical Package for Social Sciences (SPSS) version 23 (IBM, Armonk, NY, USA). Fisher exact or X2 tests were done for categorical/qualitative variables and Student t-test or ANOVA (Analysis of Variance) for continuous/quantitative variables. A variable with a two-tailed P-value of < 0.05 was considered significant at a 95% confidence interval.
RESULTS
The study included 83 patients evaluated over the last decade (2011 to 2020). There were 45 females and 38 males, including three children. Clinical and investigative features between males and females are tabulated in Supplementary Table 1. The M:F is 0.8:1. The mean age of onset is 37±16 years (range- 3–80). In males, it is 43±15, and in females, it is 32±15. The median age of onset is 36±16 years; in males, it is 44±15 years and for females, it is 31±15 years. The mean age of onset in males is significantly higher than in females (P = 0.003). M:F in young adults (< 30 years) is 0.36:1, whereas, for older adults (> 30 years), it is 1.3:1. The mean age of onset in seropositive patients (37 years) was higher than in seronegative patients (27 years) with no significant difference (P = 0.069). The age of the children at presentation ranged from 9 to 12 years.
Of the 83 patients, we found thymoma in 44 (53%), thymic hyperplasia in 32 (38%) and 5 (6%) had involuted thymus. Thymoma was predominantly found in males with M:F of 1.3:1 and thymic hyperplasia had a female preponderance with M:F of 0.45:1 (Fig. 1b). This association between gender and thymic pathology is significant (P = 0.036). The mean age of onset is significantly higher in thymoma (45 years) than in thymic hyperplasia (26 years) patients (P = < 0.001). The clinical characteristics and findings among various thymic pathologies are tabulated in Supplementary Table 2. The most common initial presentation overall was generalized fatigable weakness (23%), ptosis (23%), and bulbar symptoms (13%) (Fig. 1a). In males it was ptosis (29%), whereas, in females it was generalized fatigable weakness (31%). The median duration of illness at presentation was 6±22 months (F > M). The overall mean duration of illness at presentation was 15±23 months with no gender difference. Restricted ocular movements were significantly higher in thymic hyperplasia than in thymoma patients (P = 0.041). Bulbar palsy was more common in thymoma than in thymic hyperplasia patients with P = 0.016. Thyroid abnormality (hypothyroidism) was found in eight patients (seven females and one male). Of these, four had thymic hyperplasia, and the remaining had thymoma. All of them are AChRAb positive and asymptomatic (without any symptoms of hypothyroidism).
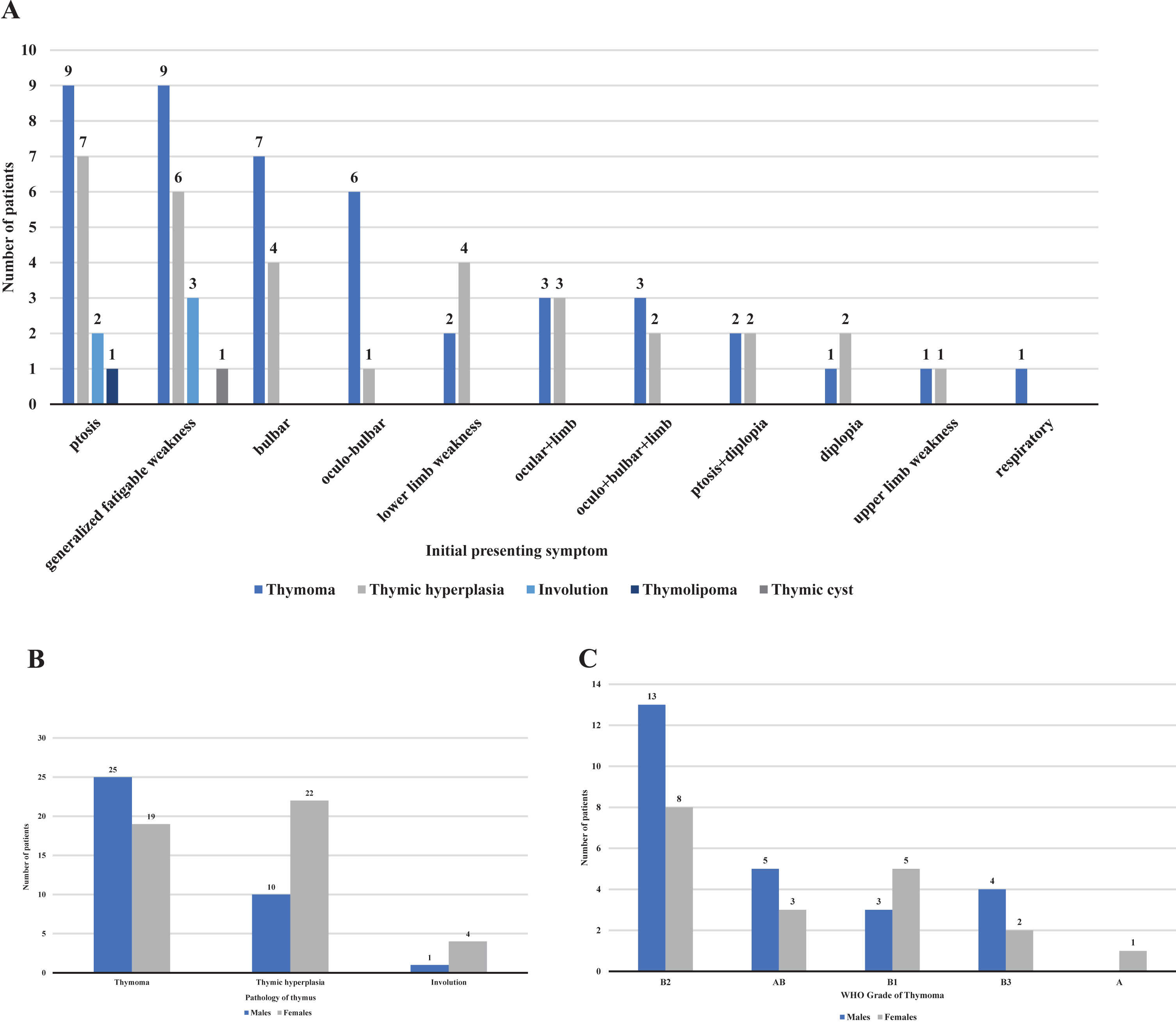
Showing clinical presentation and gender distribution among different thymic pathologies; A. Initial symptoms among different thymic pathologies; B. Distribution of the pathology of the thymus with respect to gender; C. Distribution of thymomas with respect to gender.
We found that only 8% (n = 7) of our patients had OMG, and the remaining 92% (n = 76) had GMG. M:F in OMG and GMG patients was 0.75:1 and 0.85:1, respectively. RNST was done in 78 patients, of which 87% (n = 68) showed a decrement response. No gender difference was noted. Serological studies for AChRAb were done in 79 patients, and 90% (n = 71) of them tested positive. In eight patients with absent AChRAb, we found that one patient had a MuSKAb. Of these seronegative patients, five had thymic hyperplasia, one had thymoma and two had normal thymus. Only one patient was double-seropositive with both AChRAb and MuSKAb. AChRAb was present in 86% (n = 6) of OMG and 90% (n = 65) of GMG patients, and the positivity rate was significantly more common in thymoma (39/40) compared to thymic hyperplasia (27/32) patients (P = 0.044).
Contrast high-resolution computed tomography (HRCT) of the thorax was performed on all the patients. The details of radiographic findings were available for 79 patients. Thymic enlargement was found in 69 (86%), (89% of thymoma and 81% of thymic hyperplasia patients, P = 0.025). Of these, 53 were diagnosed with thymoma and 16 with thymic hyperplasia. Figure 2 depicts a well enhanced anterior mediastinal mass on HRCT, which was later diagnosed as thymoma. Only one of these lesions was present in the middle mediastinum, whereas, the remaining were found in the anterior mediastinum (pre-vascular). Calcification was present in 2 patients. Contrast enhancement was present in 25 patients (36%) and the difference in this finding was significant between thymoma (43%, 23/53) and thymic hyperplasia (12.5%, 2/16) types diagnosed on CT (P = 0.007). Sixteen patients (64%) had homogeneous enhancement, while 9 (36%) had heterogeneous enhancement. These lesions on CT ranged in size from 1 cm to well over 9 cm in their greatest dimension. In 45 patients the size of thymus was recorded and the overall mean size was 4±2 cm, with no gender difference. The sensitivity and specificity for thymoma were 95% (39/41 true positives) and 64% (25/39 true-negatives) respectively. The sensitivity and specificity for thymic hyperplasia were 44% (14/32 true positives) and 96% (46/48 true negatives) respectively. On HRCT, two patients with thymic hyperplasia showed normal thymic morphology on HPE and in one with thymoma normal thymic morphology was noted. Twelve patients were misclassified as thymoma on imaging, whereas HPE showed thymic hyperplasia.
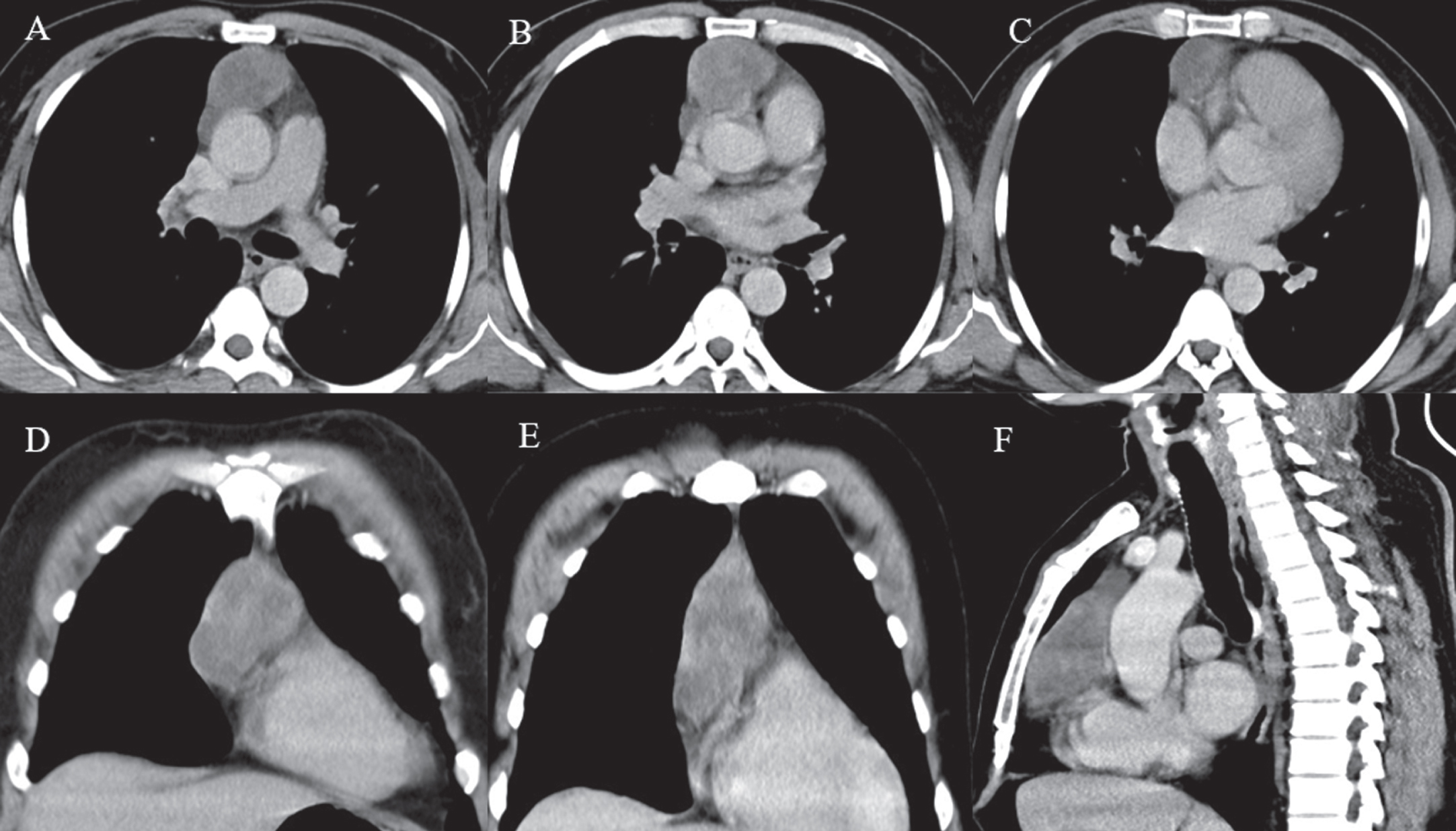
Contrast-enhanced CT chest of a 41-year-old male MG patient with WHO type B thymoma; A-C: Axial reformatted images of contrast-enhanced chest CT scan showing a well-circumscribed, lobulated, heterogeneously enhancing anterior mediastinal lesion; D-E: Coronal and (F) Sagittal reformatted images of chest CT scan in the same patient depict the anterior mediastinal lesion with a lobulated contour and relatively maintained fat planes with the adjacent structures. (CT, computed tomography; MG, myasthenia gravis; WHO, world health organization).
Histopathology
The histopathological study of the resected thymic tissue showed thymic hyperplasia in 38% (32), thymomas in 53% (44), normal looking/involuted thymus in 6% (5) and one each of a thymic cyst and thymic lipoma (Fig. 3). The tumors ranged from 1 to 12 cm in their greatest dimension. Both mean and median size of these tumors were 6±2 cm with no gender difference in 57 patients in whom these details were available. The distribution of thymomas among males and females is depicted in Fig. 1c.
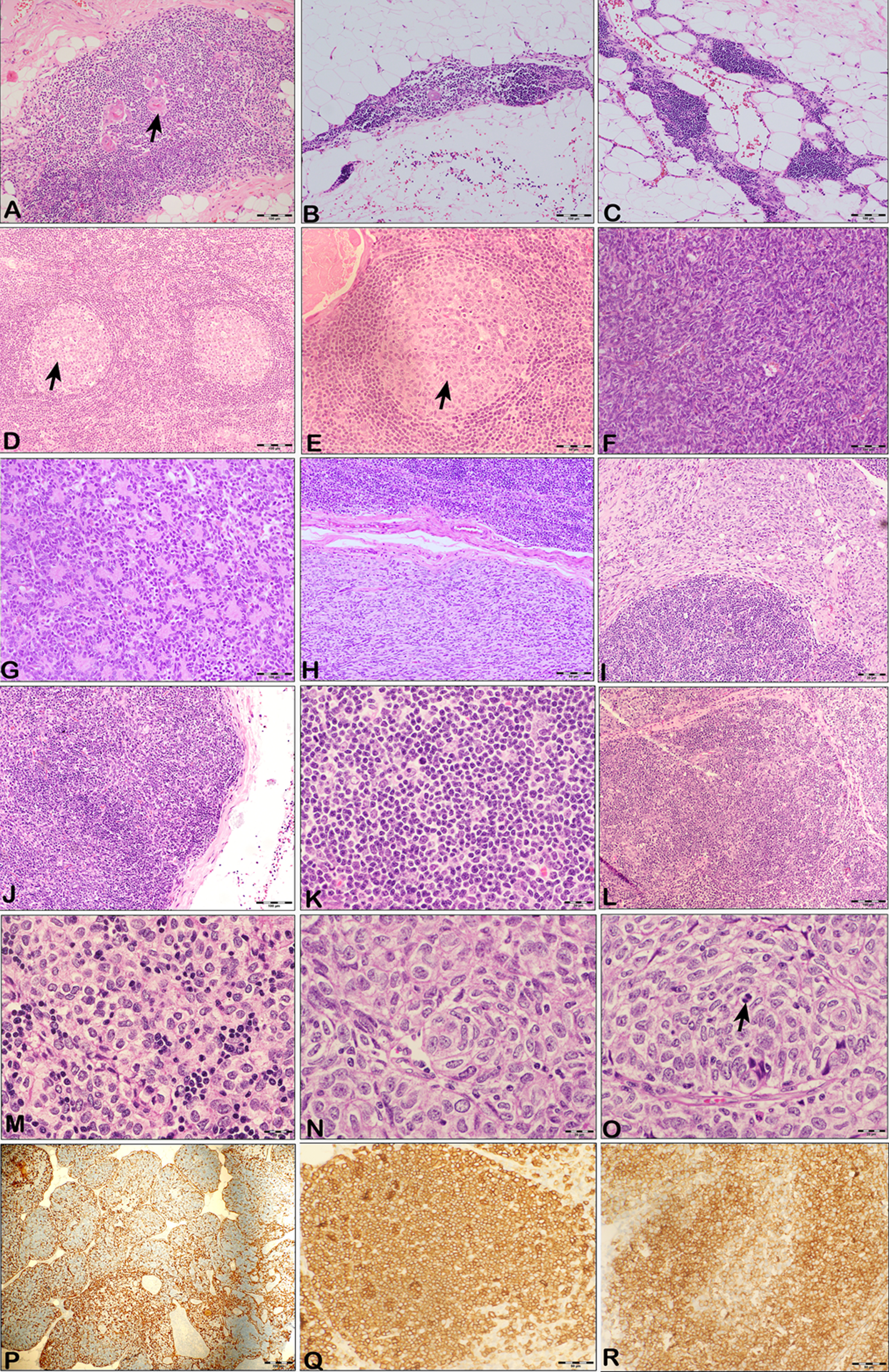
A: Microphotograph showing normal thymus with Hassall’s corpuscles (arrowhead) with its perithymic tissue. H & E×100; B: Microphotograph showing thymic tissue with features of involution. H & E×100. Note the small aggregate of lymphocytes with very scant epithelial cells; C: Microphotograph showing the histology of a case of Thymolipoma with lobules of mature adipocytes admixed with thymic parenchymal tissue. H & E×100; D & E: Microphotograph showing thymic hyperplasia with reactive germinal center (arrowhead). Note the expanded thymic parenchyma with a normal distribution of cells but with a marked increase in the lymphocyte population forming reactive germinal centers. H & E. D –× 100; E –× 200; F & G: Microphotograph showing Type A thymoma. Note the spindle nature of the epithelial cells arranged in broadsheets and vague fascicles with no lymphocytes. Figure G –showing the rosette pattern of arrangement of neoplastic cells. H & E × 200; H & I: Microphotograph showing Type AB thymoma. H & E × 200. Note the spindle cell component no lymphocyte (type A-like) distinctly seen with the well-demarcated adjacent lymphocyte-rich (type B-like with polygonal epithelioid cells) component; J & K: Microphotograph showing Type B1 thymoma. H & E; J –× 100; K - × 200. Note the scant, scattered epithelial cells admixed with a dense sheet of lymphocytic population giving a bluish color to the stained tissue on low power examination; L & M: Microphotograph showing Type B2 thymoma. H & E; L –× 100; M- × 400. Note the aggregates of large polygonal epithelial cells admixed with a moderately dense infiltrate of lymphocytes; N & O: Microphotograph showing Type B3 thymoma. The neoplasm consisted of sheets of epithelial cells with very sparse lymphocytes of varying configurations (phenotype). Note the mitosis in figure O (arrowhead) H & E × 400; P: Cytokeratin Immunostaining highlighting the epithelial cells admixed with unstained lymphocytic cells in a case of Thymoma B2. H & E × 100; Q & R: Immunostains staining the lymphocytes in a case of Thymoma B1. Q –CD99 and R –CD1a suggest immature lymphoid cells. H & E × 200.
Thymic hyperplasia
Thymic hyperplasia showed an increase in the volume of the thymus with normal appearance of the gland or with expanded thymic follicles along with or without reactive germinal centers (Fig. 3D-E). The presence of such lymphoid follicles without the proliferation of the epithelial component constituted “thymic follicular hyperplasia”. This hyperplasia may not account for the increase in the weight of the gland. The lymphoid follicles with the germinal centers represent a secondary form and are mainly filled by B lymphocytes.
Thymoma
According to the recent WHO classification, thymomas were classified based on the number and proportion of different cell types on HPE. Of the 44 thymomas, Type B2 constituted 48%, AB (18%), B1(18%), B3(14%) and type A (2.3%) (Fig. 3F-O). No type C tumor (thymic carcinoma) was identified. The characteristics (clinical, radiological, serological) of different types of thymomas are tabulated in Table 3. The thymomas on gross were nodular and greyish to pink white. The nodules were of varying size and exhibited a pushing border abutting the resected margin. Type A thymoma, also referred to as spindle cell thymoma was composed of spindle-shaped epithelial cells arranged in long and short fascicles and swirls along with solid sheets. One of the cases in the current series also showed many rosettes and vague whorls. Type AB thymoma consisted of spindled areas with sparse or no lymphocyte (type A-like) and a lymphocyte-rich (type B-like with polygonal epithelioid cells) component. These components were seen both as intimately admixed with each other and also as sharply demarcated from each other. The lymphocyte-rich areas displayed a broad range of morphology in the epithelial cells resembling those seen in type B1 or B2 thymomas.
Type B1 thymoma (Lymphocyte rich / Lymphocytic thymoma) consisted of dispersed epithelial cells admixed with a dense lymphocytic population. The neoplasm appeared blue on low power examination. The neoplasm resembled the thymic cortex in most of the areas but without lobularity. Small foci displayed medullary type morphology. The polygonal and dendritic epithelial cells were sparse, and no epithelial clusters were seen. Type B2 thymoma (Cortical thymoma/Mixed lymphocytic and epithelial thymoma) consisted of aggregates of large polygonal epithelial cells admixed with a moderately dense infiltrate (almost equal density) of lymphocytes. There was a significant increase in epithelial cells as compared to type B1 thymoma. The epithelial cells appeared as singly scattered cells and as clusters (≥3). The epithelial cells were highlighted by cytokeratin immunostaining (Fig. 3P).
Type B3 Thymoma (Atypical / Epithelial thymoma/Well-differentiated thymic carcinoma) consisted of sheets of epithelial cells that exhibited atypia, admixed with a scant infiltrate of lymphocytes. Hence the neoplasm appeared pink on low power examination. The epithelial cell nuclei were round to oval and irregular with variably prominent nucleoli and pale to clear cytoplasm. Distinct perivascular and periseptal palisading of the epithelial cells were observed accompanied by a prominent fibrous capsule, septa, and perivascular spaces. Mitosis was distinctly seen. The lymphocytes in these thymomas appeared bland but were positively stained for CD99 and CD1a, suggesting the immature nature of the lymphocytes (Fig. 3Q-R).
In thymomas, capsular infiltration was noted in 11 (25%), with 9 having focal and 2 showing diffuse infiltration. Active germinal centers were present in 20 (63%) patients with thymic hyperplasia and 4 (9%) with thymoma. Thymic lipoma was a benign lesion (neoplasm) that consisted mainly of mature adipocytic lobules separated and traversed by fibrous septa and small components of thymic epithelial cells (Fig. 3C). A thymic cyst was a benign cystic lesion, with the cyst wall formed by delicate fibro-collagenous tissue and often lined by columnar cells. The wall also showed thymic tissue.
DISCUSSION
Thymic pathology is reportedly found in almost three-fourths of patients with autoimmune MG and is thought to play a crucial role in its pathogenesis and outcome [8, 9]. Thymectomy is a well-established treatment modality for MG and is associated with favorable clinical outcomes [16]. Thus, histopathological characterization is the gold standard in thymic lesions associated with MG and also guides treatment strategies.
Most studies on MG from India primarily focus on epidemiology, clinical aspects, serological analysis and its management [18–23]. However, very few studies have been carried out that deal with pathological aspects without much clinical correlation [24–27]. The current report is an extensive clinicopathological correlation study from a quaternary care referral center in India.
Two studies on a large western Indian population (Maharashtra) cohort reported male predominant MG with a mean age of onset in the 4th decade of life [9, 26]. Other studies on the south Indian population also show that males are predominantly affected [20, 28]. In contrast, our study has a female (54%) predominance, but the average age of onset of 36 years (44 in men and 31 in women) is similar to other Indian studies [19, 26]. The majority of the patients in our cohort belonged to the southern part of India (Karnataka and Tamil Nadu). This type of prevalence is also reported in some studies on Australian, Chinese and western populations [29–32]. So, the data suggests that it is a “disease of young women and relatively older men" [33]. Approximately three-fourths of the patients present with ocular symptoms and almost all of the affected patients will have ocular features at some point [34]. The most common initial presentation in our study was generalized fatigable weakness in contrast to the studies done by Khadilkar et al., and Singhal et al., in which the majority of the patients presented with ptosis [19, 26].
In our study, GMG (92%) was the predominant form of myasthenia. This is in commensuration with other studies from India [24, 25]. Saha et al. found diurnal variation in only 60% of the patients, whereas 93% of our patients had diurnal variation [22]. A more careful history elicitation may be the reason for this difference. The majority of our patients underwent a radioimmunoassay test to detect AChRAb, which is the gold standard because of its higher specificity and sensitivity [6]. Several studies from India have shown varying (48–60%) positivity rates for these antibodies [19, 33]. In contrast, 90% of our patients had AChRAbs, which is almost similar in both OMG (86%) and GMG (90%). This result is similar to the study on the western population by Menon et al. and on the Australian population by Blum et al which showed 91% and 86% positivity rates respectively [29, 31]. This unusually high antibody positivity rate in both OMG and GMG may be because our study population involves only individuals with thymic abnormality as well as the higher sensitivity of radioimmunoassay compared to other conventional methods of testing. This rate was significantly more common in thymoma compared to thymic hyperplasia patients suggesting its association with the pathogenesis [36]. Hence, the detection of these antibodies in a patient presenting with MG should raise a suspicion of thymoma and the need for prompt evaluation and excision of the tumor. We found one double-seropositive patient which could be due to a chance association. Thymic lesions in pure MuSKAb MG are very rare, if present, is usually not associated with clinical improvement after thymectomy [37, 38].
On HRCT of the chest, the sensitivity and specificity for detecting thymoma were 95% and 64%, respectively, whereas the sensitivity and specificity for detecting thymic hyperplasia were 44% and 96% respectively. In a clinico-radiological study by Luo et al., authors found 100% sensitivity and 88.6% specificity for thymoma whereas, 26.7% sensitivity and 98% specificity for thymic hyperplasia on contrast CT chest [39]. Their study compared the sensitivity, specificity, and accuracy of three radiological examinations, namely the CT, contrast CT, and MRI, to detect thymoma and thymic hyperplasia in patients with MG. For patients with histologically proven thymoma, they found that all three radiological methods had high sensitivity (100%) and specificity (88.6%). For patients with histological diagnosis of thymic hyperplasia, their study revealed poor sensitivity of the three radiological examinations, of which MRI had the highest sensitivity (68.4%). But the specificity of contrast CT (97.9%) for thymic hyperplasia was significantly higher than that of MRI (88.5%). The diagnostic accuracy of the three radiological methods for hyperplasia was 80%, indicating that these methods could distinguish hyperplasia and non-proliferation reasonably well.
A previous study by Klimiec et al. showed 90% sensitivity and 95% specificity for thymoma, 17.6% sensitivity and 98.6% specificity for thymic hyperplasia [14]. Their study demonstrated that both CT and MRI could effectively identify thymoma. In conclusion, diagnostic imaging plays a major role in detecting the various thymic pathologies in MG. CT is generally the first-choice technique, but distinguishing normal/hyperplastic thymus from thymoma is difficult. MRI is useful in equivocal cases to differentiate focal thymic hyperplasia from thymoma by using chemical shift sequences.
A review of thymomas in MG by Mao et al. showed a pooled estimate of the incidence of thymoma in MG as 21% [40]. In our cohort, thymoma (53%) was the most common thymic pathology in contrast to the studies done by Roy et al. and Khadilkar et al., on the north Indian population and Waitande et al., on the south Indian population in which thymic hyperplasia was more common [18, 28]. In the present study, males predominantly had thymoma, and females had thymic hyperplasia, similar to the observations of Singhal et al. and Mao et al. [26, 40]. This pathological variation in our study may be affected by the different ages of onset between both genders, as patients with thymic hyperplasia have an early peak of occurrence, whereas thymoma patients have a later peak. Patients with thymic hyperplasia had a prolonged duration of illness as compared to thymoma patients. The most probable reason could be the course of illness which is severe in thymoma patients (more ocular and bulbar symptoms) compared to the patients with thymic hyperplasia, which may have contributed to their delayed presentation.
Thymic hyperplasia is reported to occur in almost 2/3rds of MG patients and is usually reported in females and the younger age group [41]. Several Indian studies have shown varying frequencies ranging from 9–41% [18, 26–28]. In the present study, 32 patients (38%) were associated with thymic hyperplasia, which is female predominant, primarily occurred in younger patients, and all had prominent lymphoid components in the hyperplasia, with at least one-third of them displaying prominent germinal centers. Epithelial hyperplasia was not distinctly seen in any of these cases. So, this type of hyperplasia can be designated as ‘lymphoid/lymphofollicular thymic hyperplasia’ instead of true thymic hyperplasia, where increased epithelial cells are evident [41].
In our study, 53% of patients had thymoma, neoplastic proliferation of thymic epithelial cells accompanied by a variable number of lymphocytes. However, the literature reveals a varying percentage of thymoma cases in India, ranging from 17–53% in different studies on MG [18, 27]. But, none of these studies had a clinic-pathological correlation. So, a comparative analysis is difficult. Thymomas occur mainly in adults, as in the present study, wherein all the reported patients were older than 18 years of age. Sometimes, MG symptoms may develop several days or months, or even years after the excision of the thymic neoplasm [42, 43]. B2 (48%) type was the most common form of thymoma in our study, while types AB and B1 constituted 18% of the thymomas and type A was the least common similar to the study by Menon et al. [31]. This finding is in accordance with other Indian studies, but very few of them were associated with MG [44–46]. In contrast, Cesar et al. reported that type A was more common than the remaining types [47]. A previous study by Nakamura et al. showed AB predominant thymomas [48]. Type B histology was predominant in men in the current study except for B1 [45, 46].
Because of the dense lymphocytic component in type B1 thymoma and AB thymoma, these may be mistaken for thymic hyperplasia. Also, it is important not to mistake the dense lymphocytic infiltration with lymphoproliferative diseases like lymphoma and the spindle cells in type A with mesenchymal tumors. Histopathological analysis should always include the integrity of the capsule, status of the margin and the effect of treatment (if any) while subtyping the thymoma [41, 49]. Also, there may be heterogeneity within the thymoma, especially in B2 and B3 types where these subtypes may be admixture in different areas.
The pathogenesis of thymoma involvement is unclear. Some of the postulations include antigenic sharing with proteins like nicotinic AChR or other components of the NMJ. Furthermore, the density and proportion of epithelial cells and lymphocytes may not correlate with the severity and type of MG. This may suggest that lymphoid tissue in the adjacent glandular tissue may have a role as a significant number of thymomas were accompanied by lymphoid hyperplasia / prominent follicles not only within the neoplasm itself but in the adjacent region [50].
Thymolipoma constitutes up to 9% of all thymic neoplasms [10]. In a study by Huang et al., thymolipomas constituted 4.4% of all the resected thymic neoplasms in MG patients [51]. The occurrence of thymolipoma in association with MG is infrequent in India [52, 53]. Singhal et al. reported that 11% of the thymectomy specimens of MG patients showed lipomas and cysts [26]. We observed only one case of each thymolipoma and a thymic cyst. Both of these patients had elevated AChRAbs. In Indian reports, the prevalence of thymic cysts associated with MG is scarce [54–56]. The nature of the cyst was unilocular similar to a case of MG reported by Mishra et al. [54]. But, multilocular thymic cysts were the predominant forms occurring in MG according to the literature [48, 57].
The current study has certain limitations. It is a retrospective study. The study has considered patients who were subjected to surgical resection, which could be prone to selection bias. We could not perform chest MRI to characterize the thymic lesions. However, this study comprehensively evaluates MG’s clinicopathological and radiological aspects among Indians.
In conclusion, we observed earlier onset of myasthenia in females. The most common initial symptom in males and patients with thymic hyperplasia was ptosis, whereas, in females and patients with thymoma it was generalized fatigable weakness. On HPE, thymomas were more common than thymic hyperplasias and occurred predominantly in males, while thymic hyperplasia was more common in females. Cortical B2 type was the most common form of thymoma, whereas type A was the least common. Patients with thymic hyperplasia suffered from prolonged duration of illness as compared to thymoma patients. The majority of the patients underwent Radioimmunoassay for serological studies and the AChRAb positivity rate was more frequent in thymomas.
A variety of thymic lesions can occur, although many of which are benign, they can exhibit a wide spectrum of clinical, radiological and histopathological changes. Histopathology plays a significant role in distinguishing the different types of thymoma and identifying extracapsular spread. The present study emphasizes the importance and awareness of these lesions, which is essential for the diagnostic workup and approach to a patient with a thymic lesion presenting with MG.
CONFLICTS OF INTEREST
Authors do not have any conflict of interests.
DATA AVAILABILITY STATEMENT
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
