Abstract
Background:
The necessity and optimal duration of post-thymectomy immunoregulatory medication (IM) in myasthenia gravis (MG) remain unclear.
Objectives:
This study aimed to evaluate whether IM is required after thymectomy in MG patients and to determine the appropriate duration of IM in patients without postoperative deterioration by comparing long-term clinical outcomes.
Design:
This triple-center, retrospective study included 1248 MG patients who underwent thymectomy. Among these patients, we compared outcomes between 483 patients without post-thymectomy IM and 765 patients receiving IM.
Methods:
Efficacy outcomes included post-thymectomy deterioration and minimal manifestation status (MMS) at 1 year. Safety outcomes, tumor growth, and serious complications were assessed between groups. Subgroup analyses were performed in mild cases (Myasthenia Gravis Foundation of America clinical classification (MGFA) ⩽2a at discharge), patients receiving IM for >1 year, and patients receiving IM for >2 years. Propensity score matching (PSM) was conducted for validation.
Results:
Relative to 438 MG patients underwent thymectomy with no-IM therapy, 756 thymectomized MG patients receiving concomitant IM showed significantly superior outcomes, including lower deterioration (hazard ratio (HR) = 0.44, adjusted p < 0.001), lower deteriorated MGFA class (adjusted coefficient = 0.94, p < 0.001), and higher odds of achieving MMS at 1 year (odds ratio = 2.06, adjusted p < 0.001). No significant differences were observed in tumor development (3 years: p = 0.78; 5 years: p = 0.27) or serious complications (p = 0.53). Similar trends were observed across subgroups. Among patients without postoperative deterioration, IM longer than 1 year reduced deterioration risk compared with ⩽1 year (HR = 0.67 (0.50–0.91), p = 0.01), whereas extending IM beyond 2 years offered no additional benefit (p = 0.137). PSM analysis confirmed these findings.
Conclusion:
Our real-world analysis of 1248 MG patients suggests that thymectomy alone is not sufficient to achieve satisfactory outcomes. Post-thymectomy IM is recommended for at least 1 year, while extending treatment beyond 2 years appears to offer no further advantage in patients without deterioration.
Plain language summary
Introduction
Thymectomy benefits myasthenia gravis (MG) patients in reducing the Quantitative Myasthenia Gravis score and facilitates tapering of immunoregulation drug dosage, allowing a portion of patients to achieve complete stable remission (CSR).1–4 Ever since an MGTX trial that demonstrated the effectiveness of thymectomy in nonthymomatous MG patients, thymectomy has been regarded as one of the standard treatments for MG patients. 1 Approximately 53.33% MG patients with thymectomy have taken immunoregulatory medication (IM) before surgery, but this percentage increases to 66.67% for 6 months after thymectomy. 5 Some studies of MGTX have indeed reported that thymectomy may more quickly reduce immunosuppressive drug dosage or even completely stop it, but their reported treatment goals were always gradually achieved after 2–5 years after thymectomy.1,6,7 Moreover, the benefits from thymectomy and the steroid- and immunotherapy-sparing effect typically take place nearly 1 year after thymectomy, 8 which suggests the need for additional IM during the first year after thymectomy. IM was reported to contribute little to the deterioration of MG 9 ; paradoxically, others recommended a 2-year maintenance IM after thymectomy to improve recovery. 10
The collective evidence suggests that thymectomy alone is typically not substantial enough as the only treatment for MG patients over the long term. IM should be given routinely after thymectomy to decrease post-thymectomy deterioration, even for patients with stable status during the perioperative period. However, it is important to note that the sample sizes of all these studies were very small, and outcome measures adopted were limited, that is, deterioration or post intervention status (PIS) condition.
The outstanding question arises: should we regard thymectomy as an independent treatment or as one best used in combination with IM? To answer this question, we conducted a triple-center study with a relatively large cohort to compare MG patients with post-thymectomy IM to those without post-thymectomy IM in terms of deterioration, recovery, and adverse events in an attempt to find if a period of maintenance IM is indeed needed after thymectomy.
Methods
Study design and participants
This study included 1248 adult patients from 3 medical centers, Tianjin Medical University General Hospital, Beijing Tiantan Hospital, and Shijiazhuang People’s Hospital, from January 2020 to December 2023. The inclusion criteria were as follows: (1) clinical diagnosis of MG after 2000; (2) thymectomy after MG diagnosis; (3) no other serious diseases that result in functional disability; and (4) available follow-up information. Patients with uncertain or incomplete medical histories were excluded.
Collected patient data included demographics, primary symptom, antibody type, worst Myasthenia Gravis Foundation of America clinical classification (MGFA) type before surgery, perioperative immunoregulation drug history, primary onset-thymectomy time, thymus pathology, perioperative MG crisis, postoperative immunoregulation drug history, deterioration condition during different periods, thymectomy-primary deterioration time (if deterioration was present), deterioration frequency at different time points, recovery condition, and serious complications. Enrolled patients with no-less-than 1-month of IM therapy after thymectomy were placed in the IM group, and patients without or with short-term (less than 1-month) IM were placed in the No-IM group. Patients in the No-IM group were confirmed not to have had any immunoregulation therapy until deterioration or the last follow-up time. All follow-up information was collected from the electronic medical record or telephone.
Outcomes
The primary outcome of this study was the deterioration rate, defined as the deterioration condition from the thymectomy date to the primary deterioration or last follow-up date. Other outcomes included MGFA class of primary deterioration, average time between thymectomy and primary deterioration, and minimal manifestation status (MMS) achieved in the first year after thymectomy. Tumor (thymoma relapse or other tumors) and serious complications that may cause death or disability, such as severe pneumonia, serious myelosuppression, and femoral head necrosis, were regarded as safety outcomes.
For safety outcomes, we collected IM history before events occurred or prior to the last follow-up. Patients were then reclassified into the IM or No-IM groups since some patients without postoperative IM therapy may have started IM due to deterioration before safety outcomes could be observed. Deterioration rate was calculated starting 1 month after the thymectomy finished to decrease the influence of temporary symptoms, and it included the following: (1) new symptoms or (2) worsening symptoms (worse MGFA class).
Subgroups
We further explored the differences between the two groups using the following subgroups. (1) Mild MGFA subgroup: patients with MGFA class ⩽2a upon hospital discharge; (2) 1-year subgroup: patients without any deterioration 1 year after thymectomy were selected and divided into three additional groups, the No-IM group, the under 1-year group (IM less than 1 year), and the over 1-year group (IM no-less-than 1 year). We compared patients in the over-1-year group with the patients in the No-IM group and the under-1-year group. (3) 2-Year subgroup: patients without any deterioration 2 years after thymectomy were selected and also divided into three additional groups, the No-IM group, the under 2-year group (IM less than 2 years but no-less-than 1 year), and the over 2-year group (IM no-less-than 2 years). We compared patients in the no-less-than-2-year group with the patients in the No-IM group and the under-2-year group.
Statistical analysis
Categorical variables were compared between groups using the Chi-squared test or Fisher’s exact test. Quantitative data were expressed as median (Q1, Q3) and analyzed using the Mann–Whitney test. Variables with p < 0.1 in the above tests were regarded as potential confounding factors to be considered in a multivariate model. Simple/multivariate Cox regression further identified the differences between the two groups in primary outcomes, and multivariate logistic/linear regression was used to estimate the differences between the two groups in other outcomes. A linear/box diagram was used to visualize comparative tendencies between the two groups. The same statistical analysis was used in subgroups as well. Finally, to minimize between-group differences, propensity score matching (PSM) was performed using the R package “MatchIt.” The propensity scores were derived from a multivariable logistic regression model. Patients were matched 1:1 based on the nearest-neighbor method with a caliper width of 0.1 and without replacement. We evaluated the difference between the matched groups using univariate Cox regression analysis and a nonparametric test to validate study conclusions. In addition, PSM analysis was applied across all subgroups. All statistical analysis was performed with SPSS software (version 24; SPSS Inc., Chicago, IL, USA), GraphPad Prism version 10.5 (GraphPad Software, La Jolla, CA, USA), and R (version 4.5.1; R Foundation for Statistical Computing, Vienna, Austria). A p < 0.05 was assumed to indicate significant test results for all tests.
Data availability
The datasets are available from the corresponding author upon reasonable request (contact the corresponding author).
Results
Demographics and clinical characteristics
In total, 1248 patients were selected for this study according to our inclusion and exclusion criteria. A total of 765 patients (61.3%) were included in the IM group and 483 in the No-IM group. 97.5% (1216) of patients had positive antibody results. Six patients in the IM group were Musk antibody positive, and one patient in the No-IM group was only Titin antibody positive. What’s more, 18 out of the 1248 patients were negative in any antibody type after two or more antibody tests.
Patient characteristics are summarized in Table 1. Compared to the No-IM group, patients in the IM group were younger at onset age (IM vs No-IM, 40:46 years, p < 0.001) but took more time from onset to thymectomy (IM vs No-IM, 7:2 months, p < 0.001). Patients in the IM group also showed milder onset symptoms than in the No-IM group (OMG, IM vs No-IM, 51.1%:60.5%, p = 0.001). However, the opposite is true when focusing on the worst MGFA class before thymectomy. Moreover, patients in the IM group had a higher percentage of preoperative IM therapy (IM vs No-IM, 49%:12.4%, p < 0.001), a lower percentage of thymoma (IM vs No-IM, 39%:77.6%, p < 0.001), and more perioperative MG crises (IM vs No-IM, 5.9%:2.5%, p < 0.001). There were no significant differences between the two groups in terms of gender, other accompanying immune disease, or antibody type.
Baseline characteristics of patients.

IM, immunoregulatory medication; MG, myasthenia gravis; MGFA, Myasthenia Gravis Foundation of America clinical classification; OMG, Ocular myasthenia gravis; GMG, Generalized myasthenia gravis; NA, Not applicable.
IM versus No-IM: Primary outcome
Regarding deterioration rate, patients who received IM showed less deterioration at 1, 3, and 5 years after thymectomy and had longer stable times, as well as smaller cumulative average deterioration frequency (Table 3, Figure 1). After adjusting for age, gender, primary onset symptoms, antibody type, worst MGFA class before thymectomy, immunoregulation before thymectomy, primary onset-thymectomy time, thymoma, and perioperative MG crisis, the IM group had a significantly lower hazard ratio (HR) of deterioration compared to the No-IM group (HR = 0.44 (0.37, 0.51), adjusted p < 0.001) in the multivariate Cox model, and for the simple Cox model (Table 2, Figure 2(a)).

The cumulative deterioration frequency over time.
Relative factors of post-operation deterioration.

HR, hazard ratio; MG, myasthenia gravis; MGFA, Myasthenia Gravis Foundation of America clinical classification.
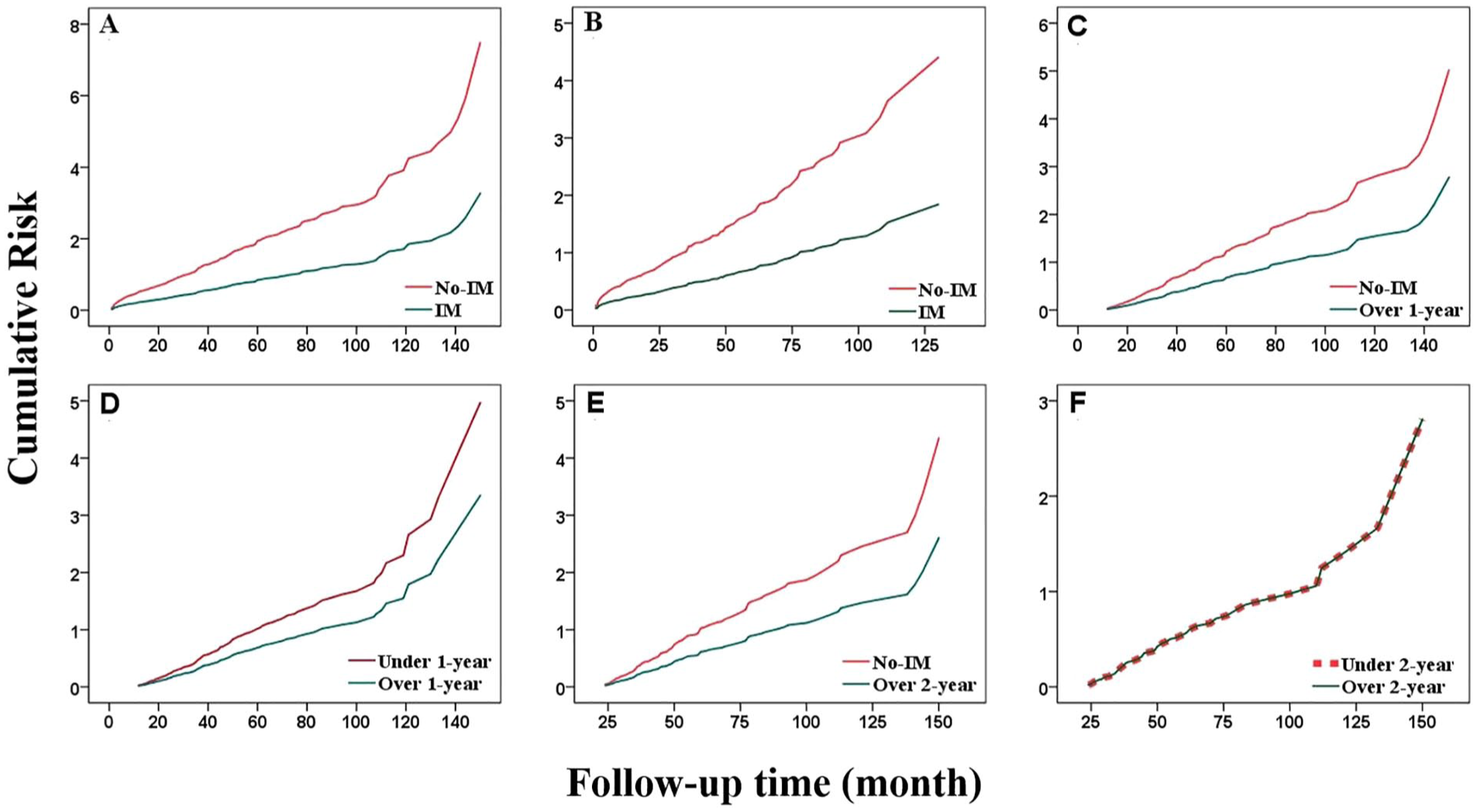
Deterioration over time between the two groups; all pictures show lower deterioration risk compared to the No-IM group. (a) Total patients. (b) Mild subgroup. (c) 1-year subgroup: No-IM versus Over 1-year. (d) 1-year subgroup: Under 1-year versus Over 1-year. (e) 2-year subgroup: No-IM versus Over 2-year. (f) 2-year subgroup: Under 2-year versus Over 2-year.
IM versus No-IM: Other outcomes
For the MGFA class after thymectomy, deteriorated patients in the IM group were more likely to get milder symptoms (coefficient = 0.94, adjusted p < 0.001) in multivariate linear regression. About 50% patients in the IM group only had new MG symptoms with no more than 2a in the MGFA class, but nearly 70% patients in the No-IM group had new MG symptoms graded above MGFA 2b (Table 3, Figure 3(b)). Conversely, the IM group had a higher percentage of mild MGFA class prior to thymectomy (Figure 3(a)).
The difference in other outcomes between the two groups.

IM, immunoregulatory medication; MGFA, Myasthenia Gravis Foundation of America clinical classification; MMS, minimal manifestation status; OR, odds ratio.

MGFA between the two groups. The IM group showed worse performance in the MGFA class prior to thymectomy (a), but had a greater percentage of mild MGFA class compared to the No-IM group (b). The IM group demonstrated a higher percentage of MMS at 1 year post-thymectomy (c). No significant differences were observed in tumor growth at the 3- (d) or 5-year (e) follow-ups, nor in the incidence of serious complications throughout the entire follow-up period (f).
A total of 68.1% patients achieved MMS 1 year after thymectomy, and the IM group had a higher percentage of such patients than the No-IM group (IM vs No-IM, 71%:63.1%, odds ratio (OR) = 2.06 (1.48, 2.87), adjusted p < 0.001). This effect also remained significant after adjusting for age, gender, primary onset symptom, antibody type, worst MGFA class before thymectomy, immunoregulation before thymectomy, primary onset-thymectomy time, thymoma, perioperative MG crisis, and immunoregulation after thymectomy (Table 3, Figure 3(c)).
IM versus No-IM: Safety outcomes
For safety outcomes, although there was a higher rate of tumor growth at 3 years after thymectomy (IM vs No-IM, 3.1%:4.1%) and a lower rate of tumor growth at 5 years after thymectomy (IM vs No-IM, 8.8%:6.6%) in the No-IM group, there was no significant difference between the two groups in terms of tumor growth at 3 or 5 years after thymectomy (IM vs No-IM, 3 years: adjusted p = 0.78; 5 years, adjusted p = 0.27), nor were there any significant differences in serious complications during follow-up between the groups (IM vs No-IM, 3.5%:4.8%, adjusted p = 0.53; Table 3, Figure 3(d)–(f)).
IM versus No-IM: Mild MGFA subgroup
In our study, 663 patients had MG symptoms under MGFA 2a at hospital discharge (IM vs No-IM: 364 vs 299). For deterioration rate, post-thymectomy, the IM group saw a decline in deterioration risk each year (HR = 0.42 (0.33, 0.53), adjusted p < 0.001) in multivariate Cox regression (Figure 2(b)). Moreover, patients in the IM group were 1.73 times more likely to achieve MMS 1 year after thymectomy compared to patients in the No-IM group (OR = 1.73 (1.1, 2.72), adjusted p = 0.02; Table 4).
The relationship between IM and outcomes in different subgroups.

HR, hazard ratio; IM, immunoregulatory medication; MGFA, Myasthenia Gravis Foundation of America clinical classification; MMS, minimal manifestation status; OR, odds ratio.
When considering safety outcomes, simple and multivariate logistic regression models both yielded no statistical differences between the two groups in tumor growth at 5 years (adjusted p = 0.1) and serious complication (adjusted p = 0.51), even after drawn by adjusting for age, gender, primary onset symptom, antibody type, worst MGFA class before thymectomy, immunoregulation before thymectomy, primary onset-thymectomy time, thymoma, perioperative MG crisis, and immunoregulation after thymectomy (Table 4).
IM versus No-IM: 1-Year subgroup
In the 1-year subgroup, 917 patients avoided any deterioration in the first year after thymectomy; 256 had no-IM therapy; 98 took IM within 1 year, and 563 continued IM for at least 1 year. IM significantly decreased deterioration rate (HR = 0.55 (0.44, 0.69), adjusted p < 0.001; Figure 2(c)). Moreover, the comparison between the under 1-year group and the over 1-year group indicates that post-thymectomy IM treatment lasting over 1 year is better than under 1 year (under 1-year group vs over 1-year group, HR = 0.67 (0.50, 0.91), adjusted p = 0.01; Table 4; Figure 2(e)).
IM versus No-IM: 2-Year subgroup
In the 2-year subgroup, 633 patients successfully maintained a stable status. A total of 184 patients had not ever taken any IM, and 67 patients received post-thymectomy IM for less than 1 year. In addition, 108 patients had a history of post-thymectomy IM for at least 1 year but less than 2 years, and 274 patients continued IM for at least 2 years. The over 2-year group had a significantly lower deterioration rate (1-year subgroup, HR = 0.60 (0.45, 0.80), adjusted p < 0.001; Figure 2(d)) compared to the No-IM group (Table 4). However, no significant difference was observed between the over 2-year group and the under 2-year group (over 2-year group vs under 2-year group, adjusted p = 0.137; Table 4; Figure 2(f)).
Propensity score matching
Multivariable logistic regression model included age, gender, primary onset symptom, antibody type, the worst MGFA class before thymectomy, preoperative IM, primary onset-thymectomy time, thymus pathology, and perioperative MG crisis was used to calculate the propensity score. After PSM, well-balanced pairs were selected, including 304 patients in the total cohort, 149 in the mild subgroup, 183 in the 1-year subgroup (No-IM vs Over 1-year), 98 in the 1-year subgroup (Under 1- vs Over 1-year), 112 in the 2-year subgroup (No-IM vs Over 2-year), and 101 in the 2-year subgroup (Under 2- vs Over 2-year). All baseline variables achieved adequate balance between the pre-IM and post-IM groups, with standardized mean differences reduced to below 0.20 and most below 0.10. Variance ratios and distributional metrics also fell within acceptable ranges, indicating that the matched cohorts were well balanced and suitable for subsequent comparative analyses. On the basis above, univariate Cox regression analysis validated above conclusion. Patients with post-thymectomy IM show lower risk of deterioration in the total cohort (HR = 0.44 (0.36, 0.54), p < 0.001) and the mild subgroup (HR = 0.51 (0.38, 0.68), p < 0.001). Furthermore, among patients without post-thymectomy deterioration, at least 1 year of immunosuppressive therapy (IM) was associated with a reduced risk of deterioration compared to no IM (HR = 0.58 (0.44, 0.78), p < 0.001) or to less than 1 year of IM (HR = 0.60 (0.39, 0.92), p = 0.018). However, no additional benefit was observed with IM durations exceeding 2 years (No IM vs >2 years: HR = 0.64 (0.45, 0.93), p = 0.016; <2 vs >2 years: HR = 0.79 (0.56, 1.11), p = 0.186; Figure 4(a)–(f)).
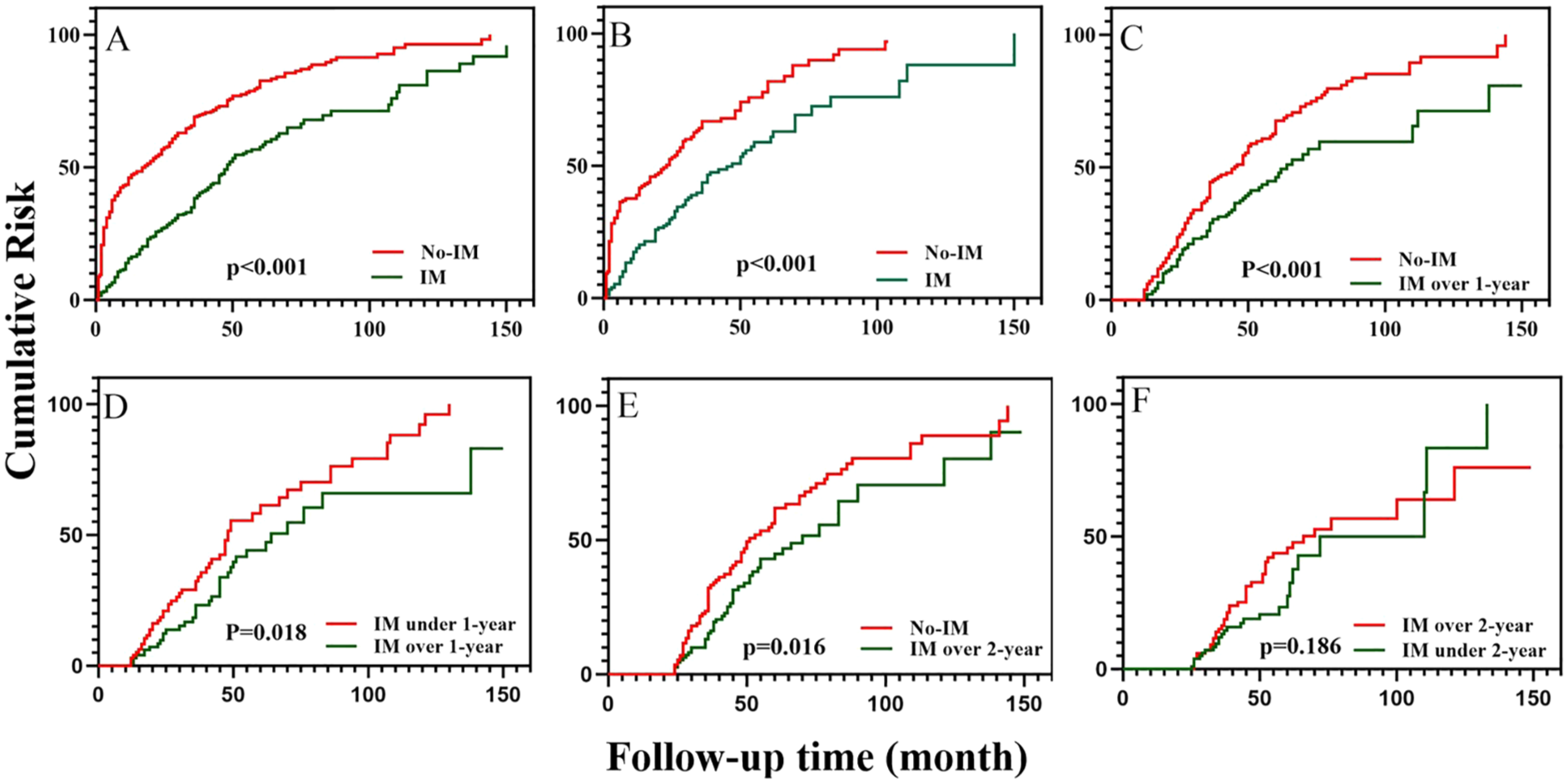
Deterioration over time between the two groups after PSM; all pictures show lower deterioration risk compared to the No-IM group. (a) Total patients. (b) Mild subgroup. (c) 1-year subgroup: No-IM versus Over 1-year. (d) 1-year subgroup: Under 1-year versus Over 1-year. (e) 2-year subgroup: No-IM versus Over 2 years. (f) 2-year subgroup: Under 2-year versus Over 2-year.
Discussion
Our study demonstrated that the addition of immunoregulation therapy post-thymectomy benefits MG patients by decreasing deterioration rate, achieving better MGFA classes, and not introducing any additional side effects in the process. Moreover, the above advantages also held true for MG patients with mild post-thymectomy symptoms. Subgroup analysis further indicated that the post-thymectomy protective effect of IM may persist for at least 1 year, but no additional benefits were observed after more than 2 years of IM treatment for those without any post-thymectomy deterioration. This conclusion is in line with the report, which indicates that 2-year post-thymectomy IM may induce better outcomes. 10 Unfortunately, one patient in the IM group and two patients in the No-IM group died due to serious pneumonia in our cohort, less likely caused by tumor growth and serious complications based on our statistical outcomes. This is supported by a previous report. 11 IM may cause other complications, such as damage to the liver and kidneys, rash, and digestive symptoms. However, we did not measure any of these mild symptoms in this study.
Our triple-center study contributes immediately to the systematic reporting of the importance of IM in post-thymectomy MG patients in terms of deterioration, recovery, and safety, as there have been few such studies to have come before. In particular, our study did not exclude MGTX outcomes. MGTX focuses more on the decline of immunoregulation drug dosage, but our study paid attention to the necessity of post-thymectomy IM. However, the MGTX followed patients for 5 years, and the observed steroid- and immunotherapy-sparing effects took place nearly 1 year after thymectomy. Thus, if no extra IM was given, those patients actually did not have enough IM during the first year after thymectomy. Our results support the idea that IM should be commonly given in the first year after thymectomy even for those with mild post-thymectomy symptoms. Post-thymectomy MG patients may benefit from maintenance IM. Nonetheless, there are still limited published reports that focus on how long IM treatment should last.
Some research indicates that about 3 years post-thymectomy IM is important for establishing CSR (CSR status) for MG patients, 12 and much research indicates that the median time for IM to help MG patients reach CSR is approximately 3 years.11,13,14 However, these studies all include a wide variety of patients, which makes it difficult to determine the maintenance time of those patients who have the potential to reach CSR. Our research preliminarily shows that for those without any deterioration after thymectomy, at least 1-year post-thymectomy IM is needed, but IM cannot decrease deterioration risk beyond 2-years.
Different deterioration rates have been observed in various studies. MGTX showed that the hospitalization rate of neuropathy was 37% after 5 years of thymectomy. The cumulative deterioration rate has also been reported to go from 16.7% at 3-years to 80% at 1-year after thymectomy.15–17 However, some studies focus on deterioration from a stable status achieved rather than calculating cumulative deterioration from the time thymectomy is finished. In addition, sample size contributes to the final deterioration rate in each study. For our study, we use deterioration to describe bad outcomes, which includes both new symptoms and aggravation of existing ones, since both deterioration and aggravation can be released by IM. Moreover, nearly 5% patients deteriorated with no absolute inducing factors 1–2 months after thymectomy and also responded well to IM. These patients were confirmed to have recovered and had been discharged to exclude perioperative deterioration. Thus, we finally chose 1 month as a time point at which to divide patients into the two groups. In our study, over 70% of patients received steroids as their initial postoperative immunotherapy. Furthermore, although tacrolimus, MMF, and cyclophosphamide (all patients with cyclophosphamide in our study had large-dose injection therapy) may require several weeks to achieve full effect, they still begin to exert a protective influence within the first month. Thus, their early protective role should not be overlooked. Fortunately, no patient was on azathioprine monotherapy. The main concern lies with azathioprine, due to its considerably slower onset of action. Therefore, while the interpretation of “time to effectiveness” is subject to some unavoidable bias, our primary outcomes and conclusions are nonetheless unaffected. Also, we found more MMS achieved in the IM group at 1-year after thymectomy, which directly supports IM benefiting patients during post-thymectomy recovery.
Additional factors linked to deterioration were also explored in our study. Interestingly, the preoperative worst MGFA class was negatively correlated with deterioration. We believe a higher MGFA class means all the more reason to start IM after thymectomy, which indirectly improves the decline of deterioration. The correlation analysis between the preoperative worst MGFA class and IM after thymectomy supports this idea, and this may also be the reason why the worst MGFA class played uncertain roles in different reports.18–20
Notably, preoperative IM in our study showed no significant difference between the two groups. Conflict exists in the literature with regard to this result,12,21–24 but more studies have shown that preoperative IM is superior to postoperative IM.21–23 Patients with preoperative therapy were able to stop IM drug due to good recovery in our study, which is different from those studies that restrict maintenance IM time. Thus, we further selected the patients with continuous IM and regrouped these patients into the pre-IM group and post-IM group. After doing so reached the same conclusion as those studies that insist pre-operative IM may benefit more for recovery.
Our study has several limitations. First, information was obtained from medical history and telephone interviews. Second, societal or economic outcomes that typically influence patients’ decisions to undergo IM were not compared. Third, we did not collect information on PIS condition after thymectomy, which may have helped us to understand the efficacy of post-thymectomy IM. Lastly, we did not determine the influence of antibody type. Currently, clinical research does not support the benefit from thymectomy for MG patients with no-AchR antibodies,25,26 and as a result, the sample of no-AchR type patients in our study was too small to depict final outcomes.
Conclusion
Our study indicates that thymectomy alone is insufficient as an immunoregulatory strategy for MG, and that the addition of IM is necessary to improve postoperative outcomes by reducing deterioration and enhancing clinical stabilization. Based on subgroup comparisons and long-term follow-up, at least 1 year of post-thymectomy IM is recommended for all patients, while extending IM beyond 2 years provides no clear additional benefit for those without postoperative deterioration. These findings support the integration of standardized IM therapy following thymectomy and offer evidence-based guidance for determining its appropriate duration in clinical practice.
