Abstract
Objective:
To retrospectively evaluate the utility of serum and cerebrospinal fluid (CSF) levels of neurofilament light chain (NfL) and phosphorylated neurofilament heavy chain (pNfH) as biomarkers for spinal muscular atrophy (SMA) progression and response to nusinersen treatment.
Methods:
NfL and pNfH levels were quantified using single molecular array (SIMOA) in CSF of 33 adult SMA patients (SMN copy number 3–5) before and in response to nusinersen treatment. In 11 of the patients, blood serum samples were also collected. CSF NfL and pNfH from patients were compared to CSF Nfs from age-matched controls without neurological disease (n = 6). For patients, pearson correlation coefficients (r) were calculated to investigate associations between Nf levels and other functional outcome measures.
Results:
Nf levels were similar between SMA and control adults and showed no change in response to nusinersen treatment in CSF or serum. Cross-sectional analyses showed an increase in CSF NfL and pNfH with age in patients (NfL p = 0.0013; pNfH p = 0.0035) and an increase in CSF NfL in controls (p = 0.002). In non-ambulatory patients, baseline serum pNfH showed a negative correlation with multiple strength and functional assessment metrics including Revised Upper Limb Module (r = –0.822, p = 0.04), upper extremity strength (r = –0.828, p = 0.042), lower extremity strength (r = –0.860, p = 0.028), and total strength (r = –0.870, p = 0.024).
Conclusions:
Nf levels did not change in response to nusinersen in adults with SMA and were not different from controls. In patients and controls, we detected an age-related increase in baseline CSF NfL and pNfH levels. Though some associations were identified, our results suggest Nf levels are not preditive or prognostic biomarkers in this population.
INTRODUCTION
Spinal muscular atrophy (SMA) is marked by widespread loss of motor neurons in the brainstem and spinal cord resulting in varying degrees of muscle weakness [1]. Without treatment intervention, severity can range from profound congenital weakness and mortality in infancy to more mild motor neuron loss and normal life expectancy [2, 3]. Nonambulatory status is correlated with symptom prevalence and morbidity in adults with SMA [4]. SMA is a recessive disorder caused by a biallelic disruption of the SMN1 gene and retention of a paralogous gene, SMN2, leading to insuffient levels of survival motor neuron (SMN) protein. SMN2 encodes only a small amount of full-length SMN protein. SMN2 copy number correlates inversely with severity of phenotype [5].
Gene therapy approaches for SMA target SMN by either replacing absent SMN1 (via gene replacement therapy onasemnogene abeparvovec [6]) or enhancing SMN2 splicing (via antisense oligonucleotide nusinersen [7] or small molecule splicing enhancer ridsiplam [8, 9]). Until FDA approval of risdiplam in 2020, nusinersen was the only gene-targeted therapy available to adults with SMA in the US. Nusinersen is administered intrathecally via loading doses at 2-week intervals followed by perpetual maintenance doses every 4 months (12 mg per administration). Nusinersen is approved for all people with SMA, however preliminary studies were only performed in infants and children [10]. In recent years, multiple reports describing safety and efficacy of nusinersen in adults with SMA suggest that nusinersen is well-tolerated [11–17] and can lead to improvements or stabilization in some motor outcomes [11, 15]. However, objective measures (outcome measures and/or biomarkers) for monitoring treatment response remain limited, particularly for severely affected patients.
Discovery of predictive, pharmacodynamic and prognostic biomarkers in SMA are particularly important in this era of disease-modifying therapies. Several molecular biomarkers have been identified in patient tissues (blood, cerebrospinal fluid [CSF], etc) [18], including neurofilament (Nf), a marker of axonal degeneration. Nfs are space-filling structures which maximize axonal caliber and upon axonal damage are released into the CSF and bloodstream [19]. The Nf family of proteins is comprised of three proteins differentiated by weight: Nf-light (NfL, ∼68 kDa), Nf-medium (NfM, ∼160 kDa), and Nf-heavy (NfH, ∼200 kDa). Greater quantities of circulating Nfs are believed to represent active and more severe neuronal degeneration [19]. NFs have been evaluated as a disease-associated biomarker in several neurological conditions, including amyotrophic lateral sclerosis (ALS) [20, 21], Alzheimer’s disease [22], frontotemporal dementia [23], Huntington’s disease [24] and SMA [25–30].
Here we retrospectively investigated CSF NfL, CSF phosphorylated neurofilament heavy chain (pNfH), serum NfL, and serum pNfH in ambulatory and non-ambulatory adults with SMA prior to and during nusinersen treatment. Patients were enrolled in two single-center, open-label studies investigating ambulatory and non-ambulatory adults with SMA [31, 32]. CSF and blood samples were available as part of routine care. Nf levels were compared with clinical and electrophysiological measurements of disease severity and progression and motor function.
METHODS
Study design
CSF and blood serum samples were collected from patients enrolled in two prospective, open-label studies of nusinersen use in ambulatory and non-ambulatory adults with SMA, conducted at the Ohio State University Wexner Medical Center between June 2017 and January 2020 (NCT04139343 and NCT04591678). The current study is a retrospective analysis of samples collected during these trials. This study was approved by the institutional review board at The Ohio State University and written informed consent was obtained prior to enrollment.
SMA patients
Individuals aged 18 years or older with genetically confirmed 5q SMA and three or more copies of the SMN2 gene were included in the original cohorts and thus analyzed in this study. Patients had insurance approval for nusinersen or qualification for a free drug program. Individuals were excluded if they had a history of bacterial meningitis or encephalitis, spinal disease which would preclude intrathecal nusinersen delivery, use of investigational drug treatment for SMA within six months of enrollment, treatment with gene therapy, stem cells or antisense oligonucleotide, and/or investigator opinion that participant is mentally or legally unable to provide informed consent. Age at sample collection was recorded for all patients and control samples. Samples were accrued from patients enrolled in two separate studies, one in non-ambulatory adults and one in ambulatory adults. Ambulation was defined as the ability to walk 30 feet.
Motor function testing included the SMA Functional Rating Scale (SMAFRS) [33, 34], Hammersmith Functional Motor Scale Expanded scale (HFMSE) [35], Revised Upper Limb Module (RULM) [36], Manual Muscle Testing (MMT) [37], handheld dynamometry (HHD) [38] and Six-Minute Walk test (6MWT) [39]. A Maximal Voluntary Isometric Muscle Contraction (MVICT) average score was derived from 5 muscle groups including elbow flexion and extension, hand grip and knee flexion and extension tested on each side (10 total).
Upper extremity strength, lower extremity strength, and total strength score was calculated from HHD, MMT and MVICT scores at each timepoint. Upper extremity strength was calculated by adding shoulder abduction, elbow flexion, elbow extension, and hand grip (MVICT only) values from both sides. Lower extremity strength was calculated by adding hip flexion, hip extension, knee flexion, knee extension, hip abduction and hip adduction values from both sides. Total strength was calculated by adding the upper and lower extremity strength scores. MMT scores which were not standard numbers (i.e. 3+, 2–, etc.) were assigned the following numerical values: 2– = 1.67; 2+ = 2.33; 3– = 2.67; 3+ = 3.33; 4– = 3.67; 4+ = 4.33; 5– = 4.67. This approach has been applied to SMA previously [40]. Testing methodologies for strength measurement and functional assessments were consistent with prior published trials [34, 41].
Patients underwent electrophysiological testing that included assessment of maximal ulnar compound muscle action potential (CMAP) amplitude and average single motor unit potential (SMUP) amplitude determination via the Multipoint Incremental technique [42, 43]. Motor unit number estimation (MUNE) was then calculated.
Controls
The control group was comprised of six individuals who did not report neurological disease (3 males, 3 females; average age = 38.3 years, range 26–48). CSF samples were obtained in the controls. However, given the retrospective nature of this analysis, serum samples, motor outcomes, or electrophysiological assessments were not obtained.
Sample collection
We collected and analyzed 1–3 CSF samples from 33 adults with SMA at the time of intrathecal administration of nusinersen. Collection occurred prior to initial loading dose (nusinersen naïve), at maintenance dose 1 (treatment day 180) and maintenance dose 3 (treatment day 420). We studied serum in a subset of these two groups, including individuals who had at least two samples available for analysis (n = 11). CSF collection was obtained via lumbar or cervical puncture prior to each nusinersen injection depending upon patient access. Five mL of CSF was collected and centrifuged at 400 g for 5 minutes. Serum was obtained via peripheral blood draw prior to each nusinersen injection and centrifuged at 1750 g for 10 minutes. Supernatants were collected, aliquoted, and stored immediately at –80°C following processing.
Sample analysis
CSF and serum NfL and pNfH levels were measured with a single molecular array (SIMOA) platform via the SR-X Analyzer (Quanterix, Lexington, MA). Using this technique, protein molecules are captured by antibody-coated beads, labeled with a detection antibody, and isolated into wells in the presence of a fluorogenic enzyme substrate. Quantification of relative fluorescence units allow for interpolation of Nf level based on a calibrated standard curve. Results are reported in pg/mL. To analyze NfL levels in CSF and serum we used the Quanterix NfL Advantage kit (Quanterix, item #103400), and to analyze pNfH levels in CSF and serum we used a Quanterix discovery kit (Quanterix, item #102669) with an anti-pNfH monoclonal antibody. The r2 of each assay was > 0.96.
Statistical analysis
Statistical analysis was performed with SPSS and GraphPad Prism 8 (v 8.4.3). Data were assessed for normal distribution and data with non-normal distribution were assessed with non-parametric tests. Kruskal-Wallis testing was used to compare Nf levels between treatment doses in SMA patients and between SMA patients and controls. One-way ANOVA testing was used to compare Nf levels between SMA patients at different ages. Associations between CSF and serum NfL and pNfH levels and motor and electrophysiological measures were investigated via Pearson correlation coefficients. No adjustment for multiple testing was performed. A two-sided p value≤0.05 was considered statistically significant.
RESULTS
Demographics
Mean age of the participants with SMA was 38.5 years (15 males [45%], 18 females [55%], range 21–67, standard deviation [SD] = 11.8) and average age of symptom onset was 41.8 months (range 1–108 months, SD = 46.6). The majority of patients in our cohort had three copies of SMN2 (3 copies, n = 22; 4 copies, n = 9; 5 copies, n = 1). Patients were characterized as either ambulatory (n = 13) or non-ambulatory (n = 20).
NfL and pNfH in CSF and serum are unaffected by nusinersen treatment
NfL and pNfH levels before and after nusinersen treatment are shown in Fig. 1. There was no change in Nf levels in response to nusinersen treatment during the course of this study in either CSF or serum (Fig. 1A–1H). We assessed cross-sectional Nf levels at each timepoint (baseline, maintenance dose 1, maintenance dose 3) to compare all timepoints between SMA patients and controls (Fig. 1I–1L; S1 Table). This analysis also demonstrated no significant differences (cross-sectional comparison of CSF NfL p = 0.58; CSF pNfH p = 0.09; serum NfL p = 0.51; serum pNfH p = 0.84).
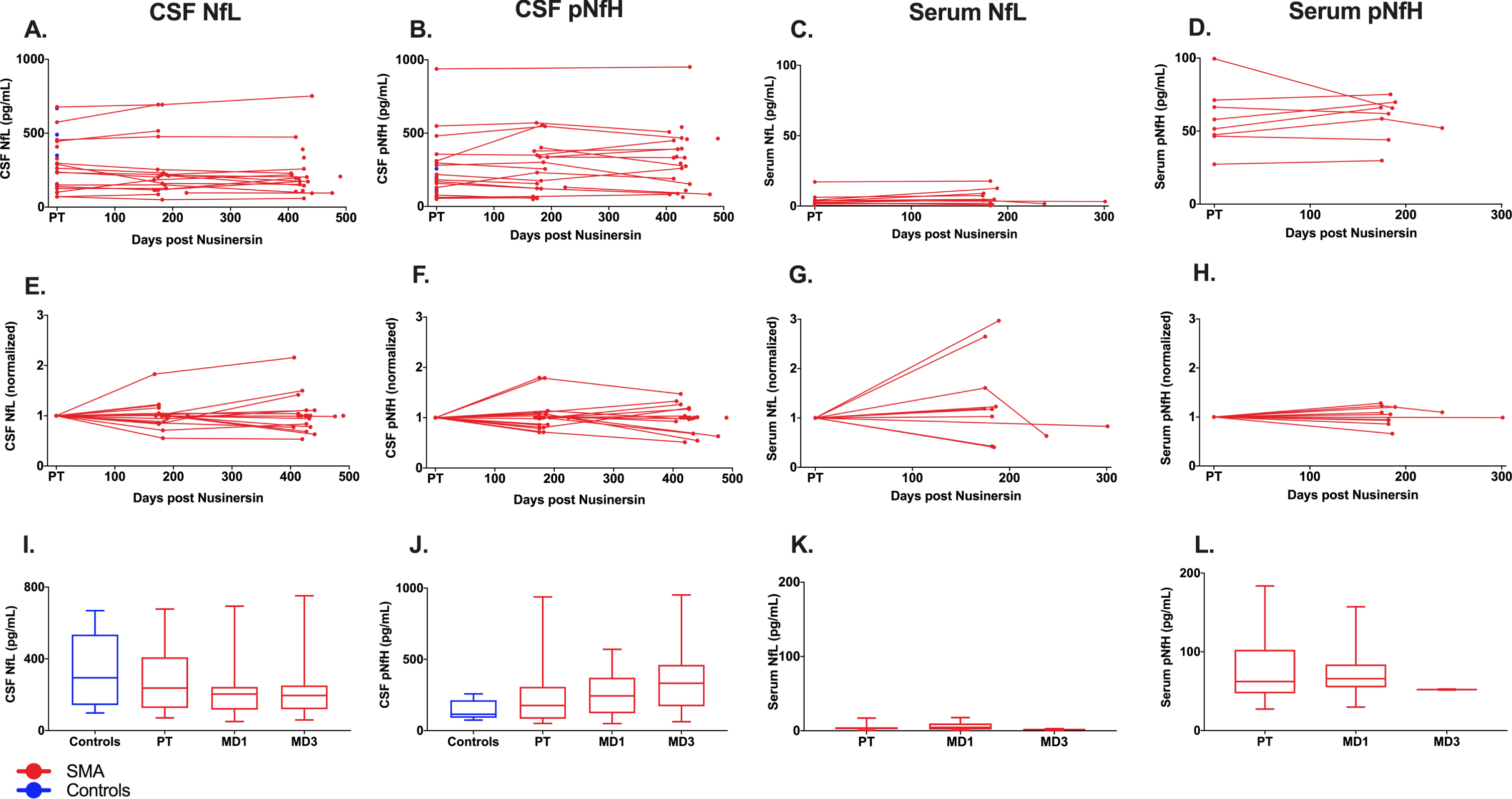
Circulating neurofilaments in SMA adults treated with nusinersen. In CSF, neurofilament-light chain (NfL) (A) and phosphorylated neurofilament-heavy chain (pNfH) (B) were quantified for all samples available. In serum, NfL (C) and pNfH (D) were quantified for patients with two or more available samples. (E-F) Normalized values for NfL and pNfH quantification in CSF and serum. (I-L) Average NfL or pNfH values at each timepoint of nusinersen treatment. Serum from neurologically healthy controls was not available for analysis. Abbreviations: PT, pre-treatment; MD1, maintenance dose 1; MD3, maintenance dose 3.
We investigated the relationship between CSF and serum Nf levels for patients with SMA who had both sample types available for analysis via correlation analysis of baseline pNfH and NfL. A positive correlation was identified between serum and CSF samples for both pNfH (r = 0.741, p = 0.01) and NfL (r = 0.666, p = 0.03). Significant positive correlation was present only in the nonambulatory cohort (serum pNfH and CSF pNfH r = 0.822, p = 0.04; serum NfL and CSF NfL r = 0.837, p = 0.03) but not in the ambulatory cohort (serum pNfH and CSF pNfH r = 0.291, p = 0.709; serum NfL and CSF NfL r = 0.596, p = 0.404).
Pre-treatment CSF Nf levels were greater in older adult SMA patients and controls
Next, we wanted to determine whether Nf levels were associated with the patient age. We performed correlation analyses across all SMA adults studied (ambulatory and non-ambulatory; n = 33; S2 Table). We identified a positive correlation between CSF NfL and patient age at baseline which remained significant at all timepoints (baseline r = 0.799 [Fig. 2A-B], p = 0.00004; 180 days r = 0.754, p = 0.00007; 420 days r = 0.779, p = 0.00005).
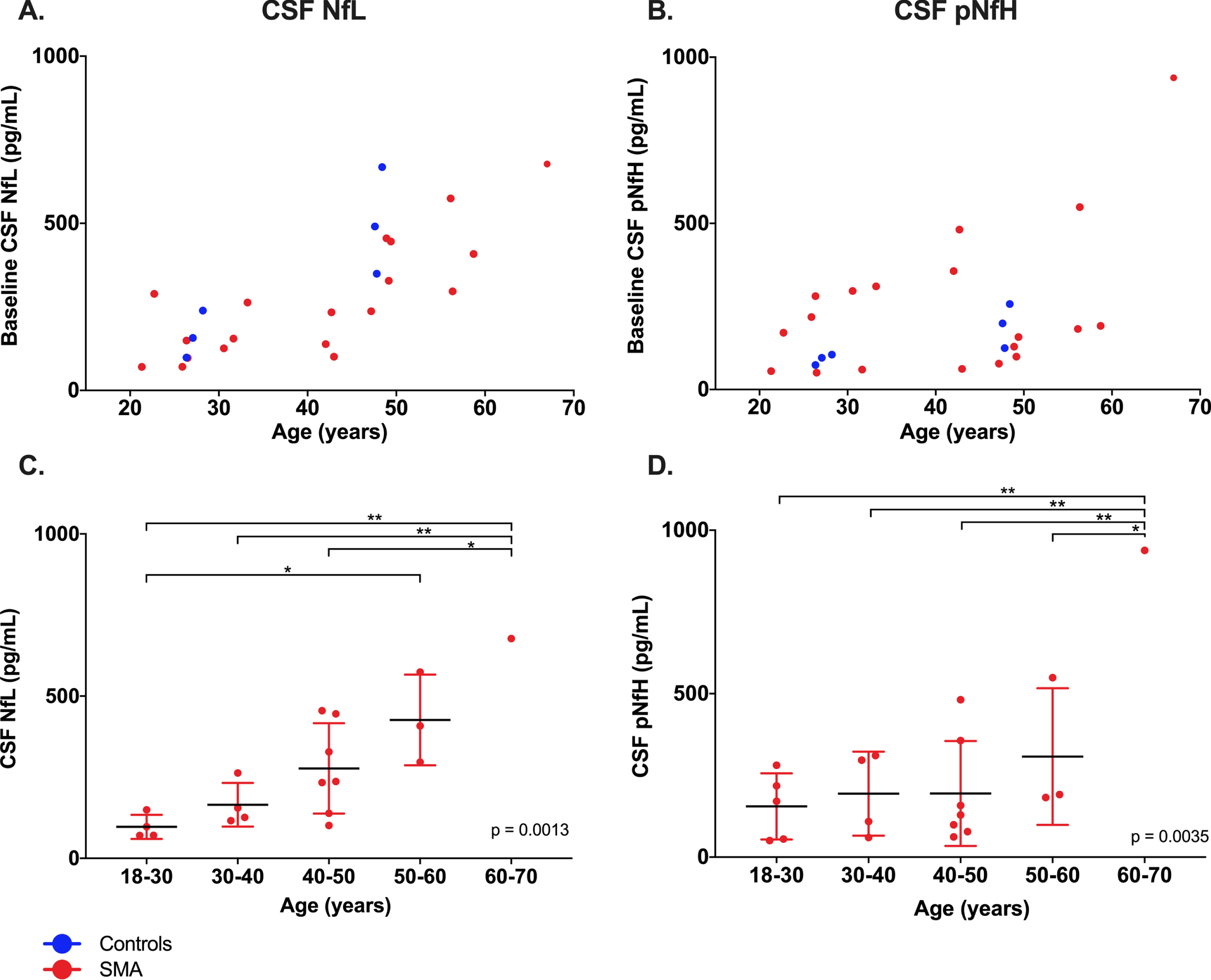
Age-related baseline increase in CSF NfL in adult SMA patients. A-B Individual data, with lines connecting samples from the same patient; C-D baseline (nusinersen naive) binned data by age (decade). One-way ANOVA was used for statistical analysis. *p < 0.05; **p < 0.01.
We then repeated the correlation analysis after stratifying for ambulatory status and found that this correlation was driven by the non-ambulatory cohort (n = 20), where a positive correlation between CSF NfL and age was also observed (baseline r = 0.867, p = 0.00005; 180 days r = 0.764, p = 0.001; 420 days r = 0.790, p = 0.004). In the ambulatory cohort, a positive correlation between CSF NfL and age was not present at baseline (r = 0.642, p = 0.243) or 6 months (r = 0.726, p = 0.165) but was present at 14 months (r = 0.795, p = 0.01). Age did not differ between the ambulatory cohort (mean age = 38.4 years, SD = 10.9) and non-ambulatory cohort (mean age = 38.5 years, SD = 12.5; p = 0.969).
When SMA patients were stratified by age (in decades), we found a significant age-related increase in CSF NfL (Fig. 2C-D; p = 0.0082) and CSF pNfH (p = 0.0035).
Positive correlation between SMN2 copy number and CSF NfL
We evaluated the relationship between SMN2 copy number and Nf level via correlation analysis. A positive correlation was identified between SMN2 copy number and CSF NfL at baseline and at maintenance dose 1 (baseline r = 0.461, p = 0.04; 180 days r = 0.490, p = 0.02). When stratified by ambulatory status, significant correlations were observed between SMN2 copy number and CSF NfL in the non-ambulatory cohort (baseline r = 0.572, p = 0.004, maintenance dose 1 r = 0.606, p = 0.01), but not in the ambulatory cohort (baseline r = 0.448, p = 0.450, maintenance dose 1 r = 0.697, p = 0.191). Ambulatory patients had significantly greater SMN2 copy number than non-ambulatory patients (ambulatory SMN2 copy number mean = 3.75, SD = 0.622; non-ambulatory SMN2 copy number mean = 3.10; SD = 0.388; p = 0.004). No relationship was identified between SMN2 copy number and age.
Nf levels correlate with functional outcomes in adult SMA patients
To assess relationships between Nf levels and functional outcomes, we performed correlation analyses after stratifying for ambulatory status. In the non-ambulatory cohort we observed multiple negative correlations between baseline serum pNfH and measures of extremity strength. Prior to nusinersen treatment, serum pNfH was negatively correlated with RULM (r = –0.822, p = 0.04). In addition, in the non-ambulatory cohort baseline serum pNfH was negatively correlated with upper extremity strength (r = –0.828, p = 0.042), lower extremity strength (r = –0.860, p = 0.028) and total strength (r = –0.870, p = 0.024). None of these correlations remained significant at subsequent timepoints. In the ambulatory cohort we observed negative correlations in three baseline functional outcomes; CMAP with serum NfL (r = –0.959, p = 0.04), SMAFRS with CSF NfL (r = –0.887, p = 0.045) and HFMSE with CSF pNfH (r = –0.919, p = 0.027). None of these correlations remained significant at subsequent timepoints.
Increased CSF Nf levels in patients with lumbar injection compared to cervical injection
Based upon each patient’s anatomy, feasibility and comfort level, some adults with SMA receive nusinersen via fluoroscopy-guided C1-2 lateral cervical injection and others via lumbar injection. This presented an opportunity to compare Nf levels between these two injection sites. Cervical injections were only performed in nonambulatory patients. We observed a significant difference in CSF NfL level at baseline (lumbar mean = 378.1 pg/mL, cervical mean = 148.3 pg/mL, p = 0.002) and CSF pNfH at baseline (lumbar mean = 346.6 pg/mL, cervical mean = 133.37 pg/mL, p = 0.031). No injection site-dependent differences were identified for NfL or pNfH levels in serum. Patients with cervical injections were younger than patients with lumbar injections (cervical mean age = 33.64, SD = 8.9; lumbar mean age = 42.05, SD = 12.58; p = 0.04)
DISCUSSION
Nf may have utility as a biomarker for treatment response in studies involving pediatric SMA individuals [25, 26]. Infants with type 1 SMA treated with nusinersen show baseline plasma pNfH levels ∼10-fold higher than age-matched healthy infants [25]. In this population, nusinersen treatment was associated with a more rapid decline in pNfH compared to those treated with sham control [25].
In contrast to pediatric populations, the utility of Nf in adults with SMA has been less convincing. Nf levels in adolescents and adults with SMA are similar to age-matched controls; this is true of baseline NfL in serum [28], NfL in CSF [27, 30], pNfH in serum [29], and NfH in CSF [27, 29]. While there has been evidence of different performance and/or monitoring potential of Nfs between SMA subtypes [28], in general, nusinersen treatment has not been shown to significantly alter Nf levels in adults with SMA [27, 28].
In our study, CSF NfL and pNfH levels in adult SMA patients were similar to levels measured in our control samples as well as those previously reported [27, 30]. Serum NfL and pNfH levels in our cohort were also unchanged throughout course of treatment with nusinersen. Prior studies have defined Nf level thresholds (45 pg/mL for serum NfL [28, 45] and 170 pg/mL for serum pNfH [29]). None of our serum samples, from either SMA patients or controls, exceeded reported thresholds.
Our observation that Nf levels were not altered by nusinersen is consistent with other reports and may be explained by the slower disease process in the adult SMA population [46–48]. In contrast, disorders with more active neuron loss such as infantile SMA and ALS do show Nf level changes [45, 49]. Adult SMA populations are known to demonstrate a greater degree of clinical heterogeneity as compared to infant SMA populations, potentially leading to greater variability in Nf level [50].
Age-related increases in NfL and pNfH have been reported previously in healthy controls [51–53] as well as in SMA type 3 patients [30]. Our results are consistent with these findings. This phenomenon may reflect natural aging independent of SMA progression and suggest normal brain atrophy throughout life [54, 55]. Age-related decreases in phosphatase activity lead to increased Nf phosphorylation in CSF, conferring resistance to degradation [56]. Increased phosphorylation may have an adaptive role, increasing the stability and alignment of Nfs in the cytoskeleton and supporting their role as cytoskeletal scaffolds.
In both ambulatory and non-ambulatory patients, Nfs demonstrated negative correlations with validated, widely-used outcome measures of motor function, including RULM, SMAFRS, CMAP, and HFSME. No correlations were identified in either group between patient age and any function outcome measure, indicating that age was not a driving factor in these signficicant findings. Each of these metrics have been associated with SMA onset, severity and progression [33, 57–59], particularly in adults with SMA [41, 60]. Previous studies have evaluated correlations between Nfs and functional outcomes [26–29]. The sample size difference (ambulatory n = 13; non-ambulatory n = 20) potentially led to underpowered analyses in ambulatory patients, especially at timepoints where data were incomplete the results of the correlations. The more uniform findings in our non-ambulatory patients may reflect a more homogenous population with similar degrees of motor neuron loss, compared to ambulatory patients who may present in various stages of disease. Finally, SMA patients with greater SMN2 copy number may more closely resemble the healthy population, accounting for the positive Nf association. We note that CSF Nf levels here are approximately the same as those of controls and it is well known that in healthy controls the serum and CSF Nf level is nonzero and rises with age [51–55].
Differences between route of administration were unexpected. Increased CSF NfL and pNfH in patients receiving nusinersen via lumbar injection compared to via cervical injection may be due to inherent differences in the clinical characteristics of each group, with the non-ambulatory cohort being more likely to have severe disease and scoliosis, prompting cervical injection. Cervical injections were only perfomed in non-ambulatory patients. Patients with lumbar injections were also older than patients with cervical injections, so the higher Nf values observed in these individuals potentially reflects the observed age-related increase in CSF Nfs in our cohort.
A limitation of this study is the relatively small number of longitudinal samples from SMA individuals and neurologically-healthy controls. In addition, the retrospective nature of this study did not allow for collecting of serum samples from controls.
Nevertheless, our retrospective analysis found that CSF and serum Nf levels do not change in response to nusinersen treatment and are largely stable throughout about a year of treatment in adults with SMA. These findings, especially in light of the observed age-related increase in CSF Nf levels in patients and controls, limit the utility of adult Nf levels to track neurodegeneration or response to treatment as a single biomarker.
Footnotes
ACKNOWLEDGMENTS
The authors would like to express their gratitude to all patients and controls who contributed to this study. This study was funded by CureSMA (Dr. Elsheikh and Dr. Arnold, award #AWD- 103358, http://www.curesma.org) and Biogen (Dr. Elshiekh, ![]() ). We are grateful for support from the William White and Family Fund. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
). We are grateful for support from the William White and Family Fund. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
CONFLICTS OF INTEREST
Dr. Kolb received compensation for consulting from Genentech, AveXis and Biogen. Dr. Elsheikh received compensation for consulting from Biogen, Genentech, Argenx and Stealth Bio-therapeutics. Dr. Arnold received compensation for consulting for La Hoffmann Roche and Genentech. All the other authors report no disclosures.
