Abstract
Background:
The impact of nusinersen therapy on outcomes in adults with Spinal Muscular Atrophy (SMA) remains uncertain.
Objective:
To demonstrate whether nusinersen therapy, at currently prescribed doses, can stabilize or improve motor function in adults with SMA using existing outcome measures.
Methods:
A single-center prospective cohort study of 6 adults with SMA type 3, with inclusion/exclusion criteria intended to optimize the ability to demonstrate change using established outcome measures. Primary outcomes were the Hammersmith Functional Motor Scale-Expanded (HFMSE) and the Revised Upper Limb Measure (RULM). Secondary outcomes were the PedsQL Fatigue scale, the SMA Functional Rating Scale (SMAFRS), and the 6-minute and 10-meter walk tests (6 MWT and 10 MWT). Estimates of change in HFMSE and RULM mean scores across visits were calculated using a linear mixed effects model. Change from baseline was used for other outcome measures.
Results:
HFMSE and RULM scores over 12 months were stable or improved in all participants, with a mean increase of 2 points in each. Other measures showed high intra-individual variability. Adverse events related to the primary diagnosis, including injury and infection, significantly impacted the ability to reliably perform walk tests in the four ambulatory participants.
Conclusions:
HFMSE and RULM show potential as responsive outcome measures of motor function in ambulatory and non-ambulatory adults with SMA type 3. A time-dependent accrual of benefit of nusinersen on motor function was apparent in this cohort. More sensitive alternative measures of quality of life, fatigue, exercise tolerance, stability and ADLs are clearly needed for adults with SMA.
INTRODUCTION
Spinal Muscular Atrophy (SMA) is caused by homozygous deletions or mutations in SMN1 which reduces expression of functional survival motor neuron (SMN) protein resulting in motor neuron degeneration. Severity of clinical phenotype partially correlates with the number of copies of SMN2 [1]. SMA has been classified historically by clinical phenotype into Type 1 (never sat), Type 2 (never walked), Type 3 (walked), and Type 4 (adult onset). More recent classifications include “non-sitters”, “sitters” and “walkers”, and are further categorized as infantile or later onset [2, 3].
The anti-sense oligonucleotide (ASO) nusinersen is administered via intrathecal injection and has been demonstrated to block alternative RNA splicing of SMN2 and increase functional SMN levels within the central nervous system [4]. Clinical trials of intrathecally administrated nusinersen in infants and children with SMA types 1–3 have demonstrated significant improvement in survival, and in gross and fine motor function, with continued accrual of benefit over a period of months to years [5, 6]. The benefit of SMN augmentation in adulthood in the setting of a prolonged history of progressive denervation has not been established. However, natural history data from longitudinal studies in adults with SMA confirms that continued disease progression is expected in adulthood in the absence of disease-modifying therapies [7–10].
Nusinersen was approved by the U.S. Food and Drug Administration (FDA) in 2016 for persons of all ages with SMA. Intrathecal administration can be challenging in adult patients with a history of spinal surgery or hardware, and this group constitutes most surviving adults with SMA [11]. In addition, there is increased risk of adverse events associated with repeated intrathecal injection such as cerebrospinal fluid leak and post-lumbar puncture headache. For these reasons, it is important to document the impact and time course of any benefit of this therapy in stabilizing or improving outcomes.
Adults with SMA type 3 with long-standing muscle weakness have an incrementally slower rate of disease progression. This is not trivial as cumulative losses eventually impact daily functioning and a small decline can be life changing. However, slow progression makes establishing the benefits of a new therapy challenging. One recent well-powered cross-sectional study in adults with SMA type 2 and 3 describes relatively stable phases of muscle strength followed by more pronounced declines which differ by onset in different SMA types [10]. This suggests a need to stratify patient populations and define specific target windows for treatment. Response to nusinersen may differ by SMN2 copy number, which is heterogenous in adults with SMA.
In this single-center, prospective cohort study of nusinersen in adults with SMA type 3, we explored whether we could document improvement or stabilization of motor function using outcome measures previously validated in clinical trials of nusinersen in children and teens with SMA.
METHODS
All procedures were in accord with the ethical standards of the Committee on Human Experimentation at the Massachusetts General Hospital. This single-center prospective cohort study was intended to determine the impact of nusinersen on motor outcomes in a targeted cohort of adults with SMA type 3 followed for at least 1 year. We used the standard loading and maintenance therapy paradigm proven effective in children with SMA, with loading doses of 12 mg at baseline, 2, 4 and 8 weeks, followed by maintenance doses of 12 mg every ∼120 days thereafter. Outcome measures shown to be reliable, valid, and responsive in younger ambulatory and non-ambulatory nusinersen treated patients with SMA, including HFMSE and RULM, were used to assess gross and fine motor function [12–16].
Subjects
Inclusion criteria included: 1) age ≥18 years at the time of enrollment; 2) molecular confirmation of SMA via documented genetic testing; 3) SMA type 3a or 3b, without limitation regarding age of onset or initial diagnosis; 4) willingness to enroll in the SPOT SMA Longitudinal Population Data Repository (LPDR) at Massachusetts General Hospital; 5) ambulatory or non-ambulatory if age ≤30, or functionally ambulant if aged 31–60 years; 6) willingness to pursue clinical treatment with nusinersen; 7) willingness and ability to participate in outcome measure assessments. Exclusion criteria included: 1) limb contractures that would prohibit performance of outcomes being assessed (e.g. hip flexion contractures prohibiting prone positioning); 2) history of spinal fusion, spine surgery or significant scoliosis impacting function; 3) respiratory insufficiency requiring more than nocturnal BiPAP or CPAP. Inclusion and exclusion criteria were designed to minimize risk and target those adults considered most likely to accrue benefit and were determined in collaboration with the Partners Pharmacy and Therapeutics Committee.
Study design
Participants underwent a physical exam, physical therapy evaluation and standardized motor outcomes testing within a 2-week window prior to initiating treatment with nusinersen to establish a baseline. Participants received 4 loading doses of 12 mg intrathecal nusinersen (Day 1, 15, 30, 60) followed by maintenance dosing every ∼120 days following completion of the loading phase. Outcome measures and exams were repeated every 3–4 months, prior to each maintenance dose. All participants had follow-up data for >12 months.
Outcome measures
Standardized motor outcome measures and patient reported outcomes aimed at assessing function, fatigue and quality of life typically used to monitor progress in patients with SMA were administered by physical therapist evaluators trained for nusinersen and other clinical trials and natural history studies. In addition, participants were asked during the evaluation if they noted any change in function since the previous visit. Subjective assessment of energy levels and general fatigue was also performed. Responses were documented by the physical therapists as part of the function and motor assessments.
Motor outcome measures
Motor outcome measures were the Hammersmith Functional Motor Scale Expanded (HFMSE), Revised Upper Limb Measure for SMA (RULM), 10-meter walk/run test (10 MWT), and the 6-minute walk test (6 MWT). Each of these tests has been shown to be reliable and valid for use with persons with SMA and used as primary or secondary outcomes in nusinersen clinical trials [12, 14–17].
The HFMSE is a 33-item scale measuring functional mobility skills ranging from rolling and sitting balance to stair performance, with a maximum score of 66 points. The HFMSE was designed for persons with SMA types 2 and 3 and has demonstrated reliability and validity from age 2 through adulthood [18]. Natural history of the HFMSE performance in persons with SMA type 2 and 3 greater than 15 years of age shows a mean 12-month decline of 0.36 points and only 7.5% of demonstrate a change of more than +/–2 points, suggesting that change greater than 2 points is clinically significant [19].
The RULM for SMA is a 19-item upper limb function scale with items scored 0–2 points and a maximum score of 37 points. The RULM was designed to try to minimize the ceiling effect observed in the preceding version of the Upper Limb Module for SMA [14]. It. Inter-rater reliability and validity for the RULM has been established in persons ages 2 through adulthood [14]. A study of 12-month change in persons with SMA type 3 who were ≥15 years of age demonstrated a decline of 1.7 points per year for non-ambulatory individuals and a decline of 1.4 points per year for those who were ambulatory [20]. A Minimally Clinically Important Difference (MCID) is considered a change of 2 or more points [20], though some suggest a change of 1 point could be meaningful [21].
The 10 MWT is a timed function test measuring gait speed across 10 meters that has been validated and used in a variety of populations including SMA [22, 23]. The 10MWT provides an index of short-distance walking speed and can be used as an adjunct to measurement of walking endurance indexed by the 6 MWT. The 6 MWT provides a measure of total distance walked along a 25-meter course over 6 minutes. Comparison to available normative and reference values is not typically used due to lack of normative values specific to SMA. The 6 MWT has demonstrated test/retest and interrater reliability and has been studied extensively in the ambulatory SMA population [12]. A change of +/–24 meters has been considered clinically meaningful based on published reliability and validity data for the 6 MWT in SMA [12].
Patient reported measures
Peds QL Multidimensional Fatigue Scale (PedsQL) is a three-part patient reported questionnaire measuring negative impact of fatigue in general, sleep/rest, and cognitive domains over the month prior to assessment. The PedsQL has been validated for use in patients with neuromuscular disease including spinal muscular atrophy [24]. Scores are scaled from 0–100 with 100 indicating no fatigue/problem in each domain and 0 indicating complete or constant fatigue/problem in each domain. The PedsQL was designed for pediatric use, but has been validated for use in adults >18 years of age [25].
The modified SMA Functional Rating Scale (SMAFRS) is a 10-question scale that examines various activities and functions ranging from grooming and feeding to mobility and pulmonary health. Each question is asked on a 5-point scale (0–4) with 4 being completely independent/no modifications or increased effort compared to typical peers and 0 being completely dependent or unable. Maximum total score is 40 points. The SMAFRS was designed for teens and adults with SMA type 3 [13, 26].
At each visit, patients were asked if they noticed overall improvement, stability or deterioration since their last treatment and to describe in their own words their experience. Responses were documented as part of the physical therapy assessment. A detailed history of falls including frequency, severity, and perceived cause was also collected as a part of the physical therapy assessment.
Standard protocol approvals, registrations, and patient consents
This study was completed in compliance with and with approval of the Massachusetts General Hospital Institutional Review Board Protocol #2015P001934. Written informed consent was obtained from all participants in the study.
Analysis plan
Individual scores and group median change scores were plotted longitudinally. Statistical analysis was performed using GraphPad Prism. Shapiro-Wilk normality test was used to confirm normality of distribution in the data. The best line fit of change in HFMSE and RULM delta mean scores over time were estimated using a linear mixed effects model allowing random slope, with p < 0.05 as critical value. For the 6MWT, total distance walked over 6 minutes was measured at each visit. Raw distance data were plotted longitudinally to assess change over time compared to baseline for each individual participant. Comparison to normative values were not used in this study due to variability in available normative data sets for healthy subjects and lack of SMA specific predictive values. Factors related to SMA, including body composition, negatively impact the ability to use predictive values from normative data sets [27]. Instead, change in 6MWT distance was evaluated using published SMA clinically meaningful change (+/–24 meters) [12].
RESULTS
Six adults with SMA Type 3, with homozygous deletion of SMN1 and 3 or more copies of SMN2, were evaluated longitudinally on nusinersen treatment from August 2017 to September 2019 (Table 1, Baseline Demographics). The mean follow-up duration was 17 months with a range of 14–21 months. Of the 6 patients, 5 had SMA Type 3b and 1 had SMA Type 3a. Functional ambulation was defined as the ability to ambulate with or without walking aids and orthotics a distance greater than or equal to 10 meters. Four of the 6 participants were functional ambulators, all had SMA type 3b, and each reported intermittently using hand held assistive devices (cane or walker) for safety at home or in the community.
Baseline Demographics
*n = 4 for ambulatory assessments (6 MWT, 10 MWT); Data reported includes median (range) where indicated; Abbreviations: SMN2 survival motor neuron 2 gene, HFMSE Hammersmith Functional Motor Scale Expanded, RULM Revised Upper Limb Measure, 6 MWT 6-minute walk test, 10 MWT 10-meter walk test, SMAFRS Spinal Muscular Atrophy Functional Rating Scale.
Motor outcomes
Three of 6 participants (50%) had clinically meaningful improvement in motor function, as measured by >2-point change in individual HFMSE scores, over a follow-up period of 15–21 months on nusinersen treatment (Fig. 1a). The remaining three of 6 participants (50%) remained stable over 14 months on nusinersen, with improvements in HFMSE scores of +1 to +2. Mean HFMSE improvement was 2 points (range 1, 5) over 14 months on nusinersen treatment (Fig. 2a). Two of 6 participants (33%) showed clinically meaningful improvement in motor function as measured by >2 change in individual RULM scores over follow up of 15–18 months on nusinersen. Four of 6 participants (67%) remained stable with change in RULM score of 0 to +2 (Fig. 1b). One patient was at RULM ceiling at baseline and maintained this score over 21 months. Two patients improved by 2 points to ceiling on the RULM by 6 months on nusinersen, maintaining their scores during 13–16 months on nusinersen. One patient remained stable over 14 months on nusinersen, with change in RULM of +1. Mean RULM improvement over 14 months on nusinersen was 1.8 points (range 0, 3) (Fig. 2b). None of the patients had clinically meaningful deterioration in upper limb function and no difference in RULM score change was seen between ambulatory and non-ambulatory participants. Mean improvement and stabilization on HFMSE and RULM compares favorably to natural history data reporting a 12-month decline in HFMSE of –0.36 in SMA Type 2 and Type 3 subjects over the age of 15 [19] and a 12-month decline of 0.41 points on the RULM in subjects with SMA Type 2 and Type 3 without a relationship to age [20].
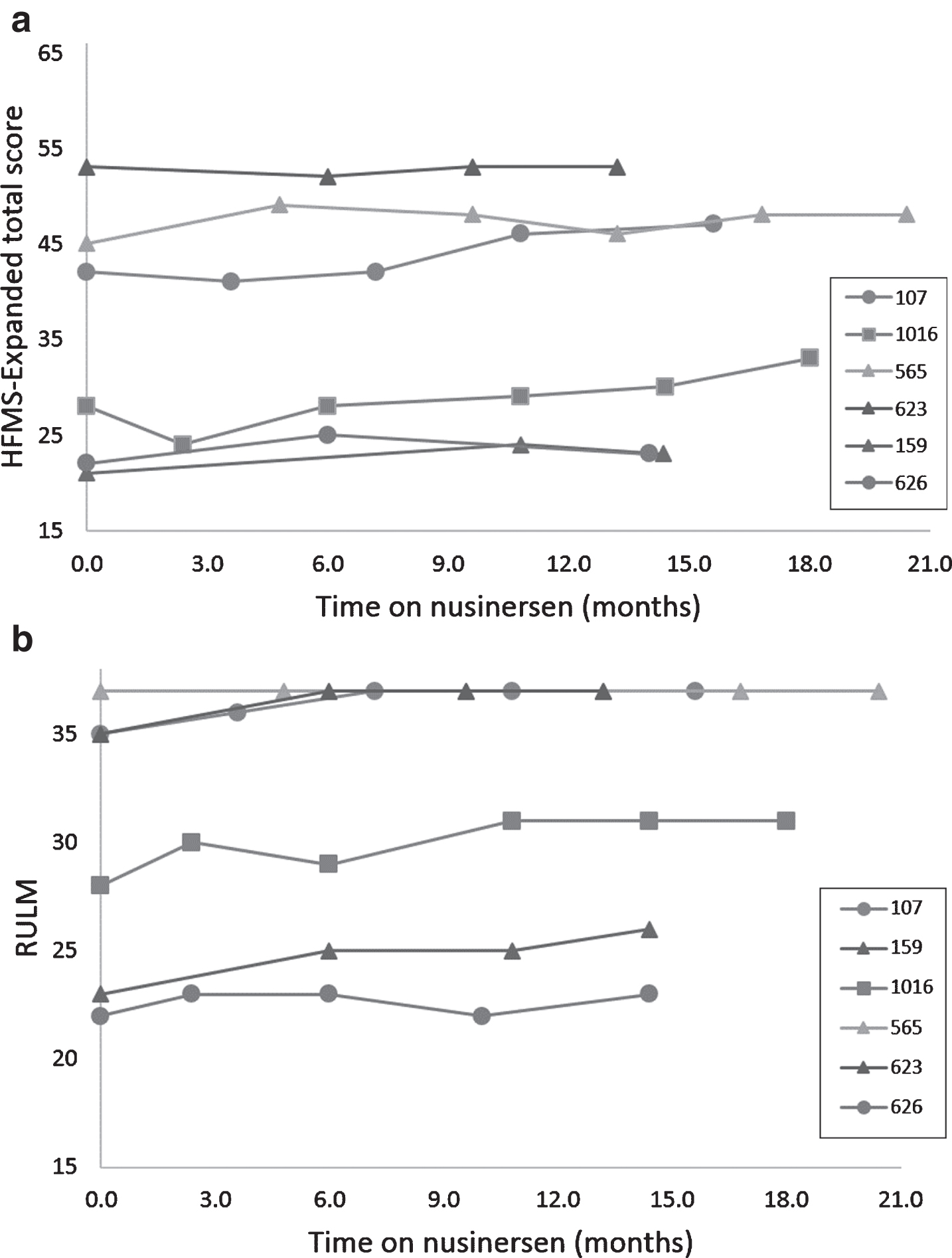
Improvements in motor function over time on nusinersen in adults with SMA Type 3. (a) Three participants (107, 1016, 565) improved by more than 2 points in their individual HMSFE scores over follow-up periods of 15–21 months and the other three participants remained stable over shorter follow-up periods of 14 months. (b) Two participants (1016, 159) improved by more than 2 points in their individual RULM scores over 15–18 months. One participant (565) was at ceiling and remained stable over follow up over 21 months. Two participants (107, 623) improved by 2 points to ceiling by 6 months and remained stable over follow up over 13–16 months. One patient (626) remained stable over 14 months.
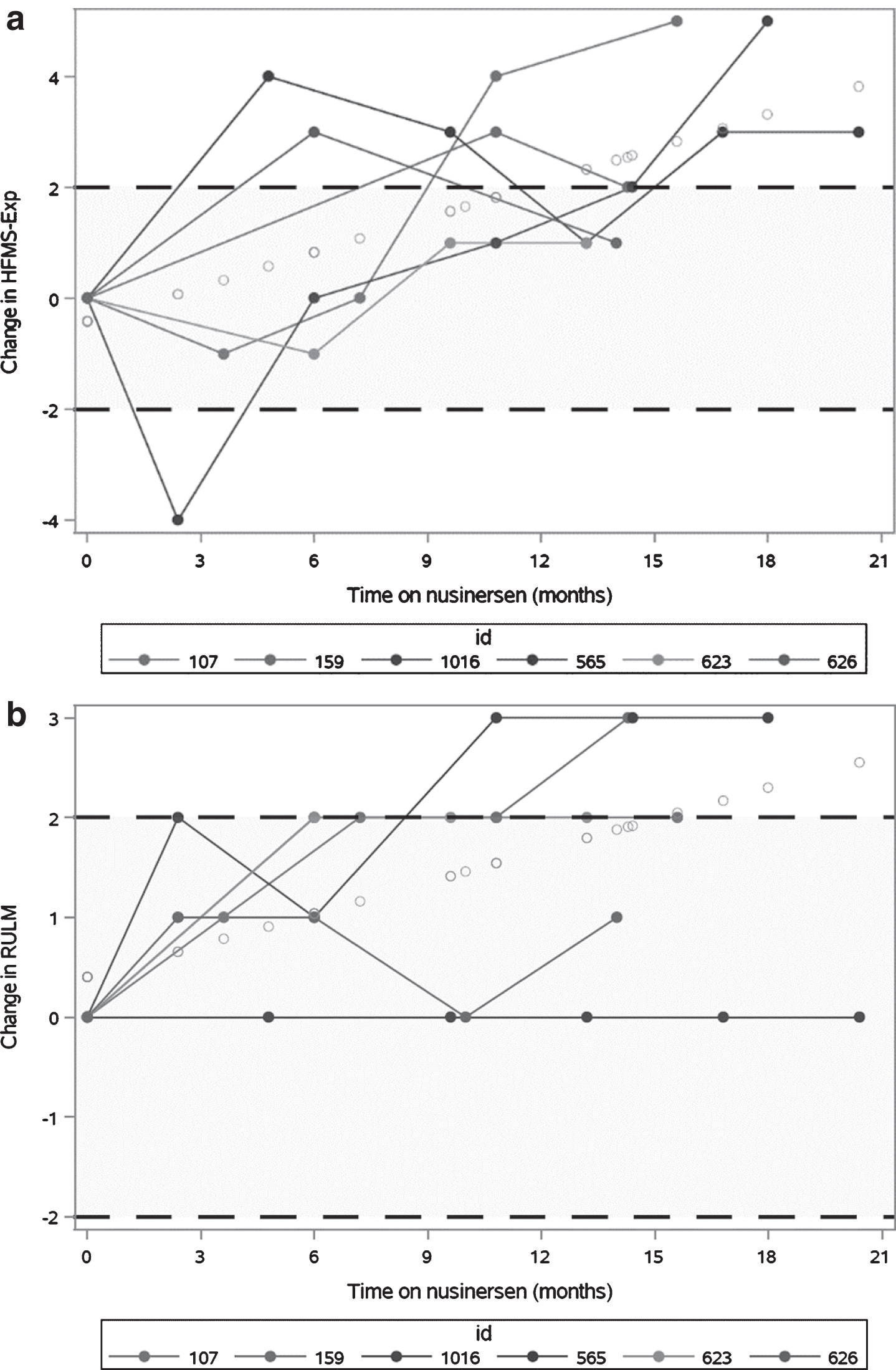
Improvement and stability in HFMSE and RULM scores over time on nusinersen treatment. Shaded areas represent scores of between +2 and –2 representing clinical stability. Unshaded areas represent scores of >2 which represent clinically meaningful change. The best line fit of change in HFMSE and RULM mean scores over time were estimated using a linear mixed effects model with random slope, p < 0.05. (a) Three participants (107, 1016, 565) had clinically meaningful improvements in HMSFE scores over follow-up periods of 15–21 months and the other three participants remained stable over shorter follow-up periods of 14 months. Estimated mean improvement in motor function was 2 points (range 1,5) over 14 months on nusinersen treatment using the best line fit of change on HFMSE mean scores, p < 0.05. (b) Two participants (1016, 159) had clinically meaningful improvement in RULM scores over 15–18 months. One participant (565) was at ceiling and remained stable over follow up over 21 months. Two participants (107, 623) improved by 2 points to ceiling by 6 months and remained stable over follow up over 13–16 months. One patient (626) remained stable over 14 months. Mean improvement in motor function over 14 months on nusinersen was 1.8 points (range 0, 3) using the best line fit of change on RULM mean scores, p < 0.05.
Across the four ambulatory participants, individual variability in direction and magnitude of 10 MWT and 6 MWT performance across visits resulted in no statistically or clinically meaningful group change when comparing baseline and final visits. One participant (626), demonstrated a clinically meaningful decline in performance on walking tests in associated with limited sleep and reduced physical activity in the setting of recurrent bouts of lower extremity cellulitis and unstable housing (Fig. 3a–b).
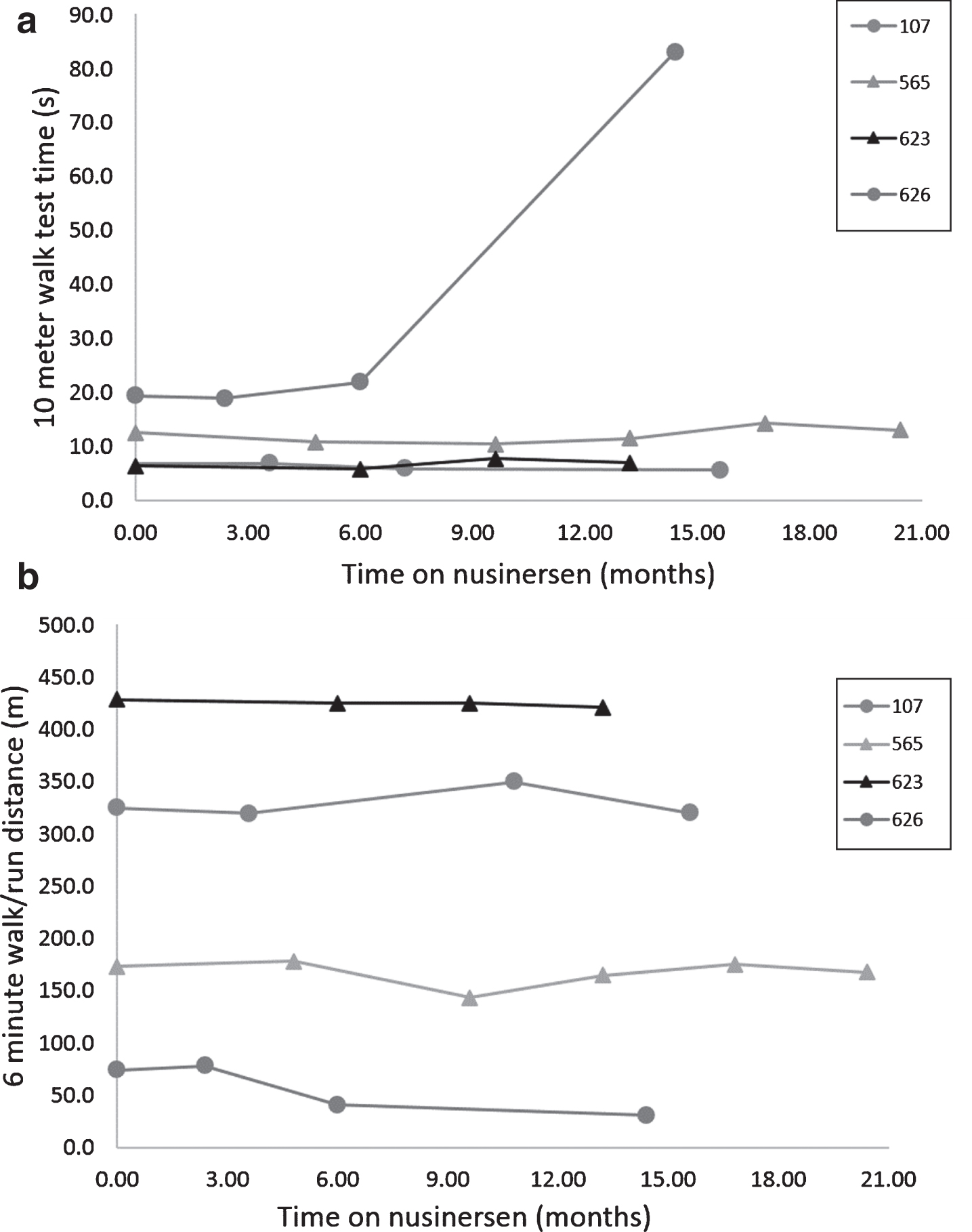
Stable 10 MWT and 6 MWT over time on nusinersen treatment. Walk test performance over time for ambulatory subjects was generally stable for the 4 ambulatory patients on the 10 MWT (a) and 6 MWT (b) on 14–21 months of nusinersen treatment, except for a precipitous decline in 10 MWT and 6 MWT performance for participant 626 due to leg cellulitis requiring hospitalization within a day of his assessment.
Patient-reported outcomes
All six participants reported a subjective improvement in energy levels and/or greater ability to tolerate work and home demands after starting nusinersen. Following completion of the loading phase, 2 of 6 participants (33%) reported that they perceived the greatest improved energy levels and/or greater ability to tolerate work and home demands during the first 2 months following each dose, but reported the perception that benefit waned in the last 1–2 months leading up to the next maintenance dose. All four functionally ambulant participants (100%) reported an overall decrease in the frequency of falls after starting nusinersen, despite two participants experiencing falls and one experiencing an episode of cellulitis requiring significant recovery time during follow-up. Additional details regarding subjectively reported perception of response is included in the case histories.
Marked individual variability in overall fatigue was seen in all measured domains (general, cognition and rest) as evaluated by the PedsQL (Fig. 4) without any obvious trends. One participant improved significantly (+11 points total PedsQL score) over 14 months but had been diagnosed with obstructive sleep apnea and underwent CPAP initiation at 3 months into follow up and weight loss therapy. There was marked individual variability in activities of daily function as measured by the modified SMAFRS with four participants reporting decline and two reporting stability or improvement (Fig. 5).
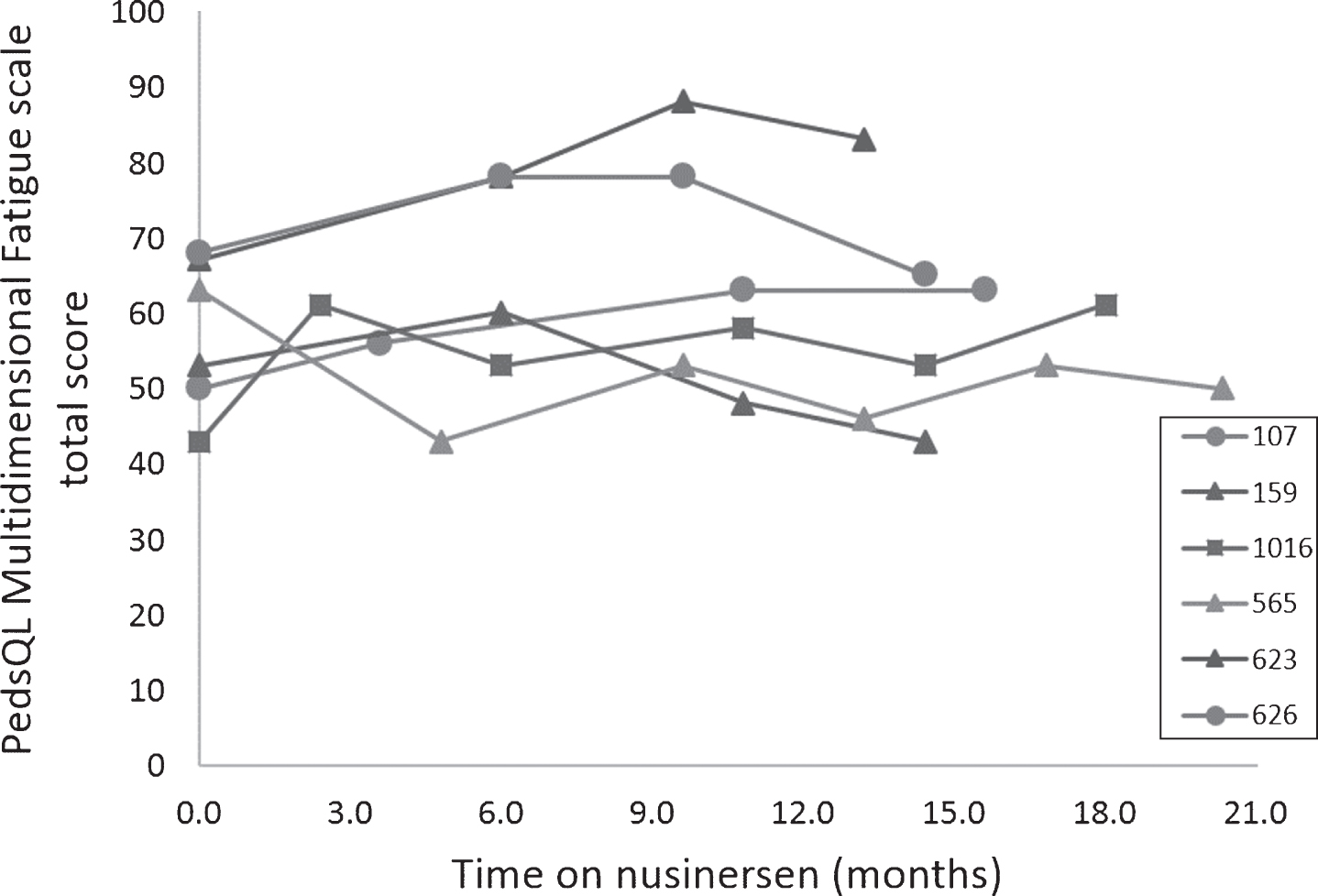
Marked individual variability on the PedsQL Multidimensional Fatigue Scale, which measures overall fatigue in all measured domains (general, cognition and rest), over time on nusinersen treatment. One participant (623) improved significantly (+11) over 14 months. However, shortly following initiation of nusinersen, he began using CPAP for obstructive sleep apnea (OSA) and began a weight loss program.
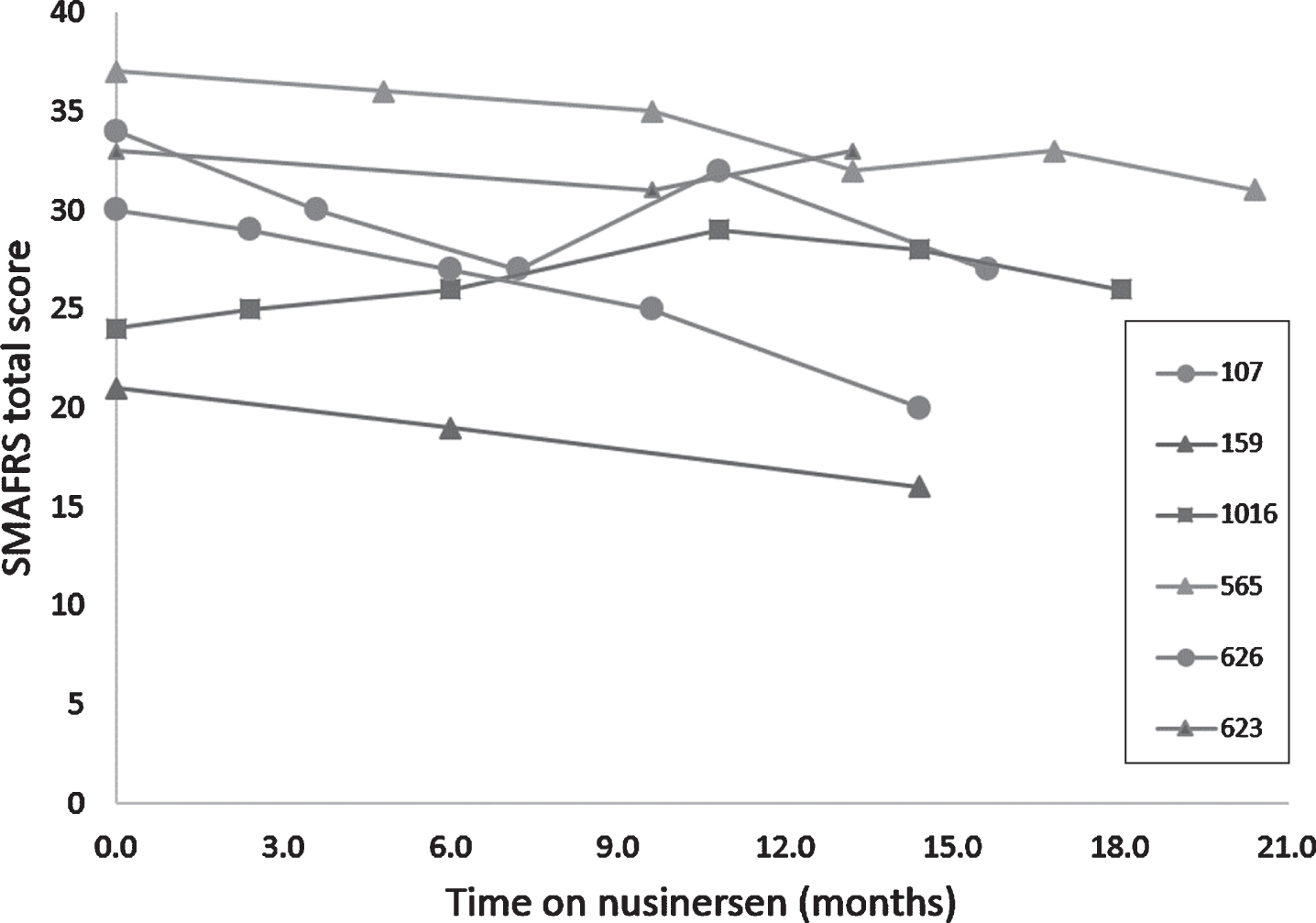
Individual variability in ability to perform activities of daily living as measured by SMAFRS over time on nusinersen treatment. There was marked individual variability in activities of daily function as measured by the modified SMAFRS with four participants reporting decline and two reporting stability or improvement
Adverse events
All participants had one or more adverse events (AEs) or injuries that precluded ideal performance or participation at one or more visits (Table 2). Two serious AEs resulting in hospitalization were reported: a fall with comminuted sacral compression fractures requiring extended hospitalization for pain control and rehabilitation, and leg cellulitis in the setting of chronic lower extremity lymphedema requiring IV antibiotic therapy. Four of 6 participants reported post lumbar puncture (LP) headaches on one or more occasions resulting in lost days from work, although none required a blood patch. Two participants reported vertigo post dosing, one acutely, resulting in an after-hours emergency room visit, and in another, vertigo persisted for a full week after dosing in association with postural headache. All post LP adverse events occurred during the loading phase.
Adverse Events
Case histories
Participant 1 (107): Adult male with SMA 3b currently in his 50s, with 5 copies of SMN2. Symptom onset in teen years included falls and lower extremity weakness. He lives independently and works full-time. Prior to initiating treatment with nusinersen he reported a gradual decline in function and increase in falls both at home and in the community. To maximize his function, he engaged in a plant-based diet and increased exercise, used the swimming pool daily and worked with a personal trainer 3–5×weekly for resistance exercises. He felt those interventions were helping to stabilize his disease progression, but still felt he was declining over time.
At enrollment, he was ambulatory at a home level using forearm crutches for beyond room distances and relied on wheelchair for community use much of the time. He had known obstructive sleep apnea (OSA) at time of enrollment and in years prior but declined treatment.
Following loading phase of nusinersen therapy, he sustained a complex sacral fracture requiring 1-month bedrest and prolonged rehabilitation leaving him non-ambulatory for 2–3 months. He was able to regain ambulation with aquatic therapy and a personal trainer progressing from use of a walker to use of forearm crutches in the months that followed allowing him to not only return to baseline ambulation as determined by the 6 MWT but exceed his baseline distance by the 10-month mark.
In addition to regaining ambulation following his injury, he reported improved endurance and energy levels, particularly in first two months following each injection.
A wearing off effect with return of fatigue and increased reliance on his manual wheelchair continued to be noted (or was noted) in the month prior to maintenance dosing.
Participant 2 (159): Adult male with SMA type 3b in his mid-20’s with 3 copies of SMN2. Symptom onset at age 4 years was characterized by inability to climb stairs like his peers. His function declined gradually over the next decade and he has been non-ambulatory since adolescence. He lives with his parents and had recently stopped working due to challenges in arranging accommodations at work for his disability, particularly for toileting issues.
At enrollment, he was able to take a few steps with maximal assist and was unable to reach overhead with his left arm. He had some difficulty with swallowing but maintained a normal diet without restrictions. He was able to stand with support for Activities of Daily Living (ADLs) and required the assist of another person for transfers. He used a power wheelchair as a primary means of mobility. He had participated in physical therapy intermittently throughout adulthood and restarted outpatient physical therapy with a focus on body weight support treadmill training and upper extremity resistance training at the start of treatment. He was diagnosed with obstructive sleep apnea during pre-treatment work up but did not utilize CPAP over the course of this study.
Following loading phase, he sustained a knee injury during a transfer, which limited his ability to tolerate standing and transfers for the next several months. He recently obtained a standing wheelchair to safely facilitate increased time in supported standing for bone density, digestion, pulmonary health, and improved pain relief which he attributes to tightness from limited mobility and prolonged sitting.
He noted improved energy and physical therapy tolerance following the loading phase of nusinersen treatment. Additional improvements noted were improved rolling and upper extremity function.
Participant 3 (1016): Adult male with SMA type 3a, in his mid-20’s at enrollment, with 3 copies of SMN2. Symptom onset at age 1 year characterized by frequent falling as soon as he started to walk. Of note, this participant has an unaffected fraternal twin and mom endorses decreased movement of participant compared to unaffected twin in utero. He lives with his spouse who helps with transfers and both work full-time. He participated in the CarniVal 2/3 trial [22] in his teenage years without apparent benefit and ultimately stopped valproic acid therapy after 1 year due to adverse gastrointestinal side effects. He remained functionally ambulatory until his later teen years, losing ambulation following a fall with resulting leg fracture.
At enrollment, he utilized a power assist wheelchair for mobility and was able to take some steps in chest deep water. He also used a standing frame daily and worked with a personal trainer weekly doing resistance exercise for extremities, core exercise, and stretching.
Following the 3rd loading dose, he experienced a cerebrospinal fluid leak and post LP headache resulting in 1-week bedrest. Weakness and fatigue were prominent for several weeks and he remained below baseline at the final loading dose. He had full recovery by 6 months post treatment initiation and reported subjectively increased energy and stamina. His course was further complicated by pressure sores and decreased time upright up till approximately 10 months post initiation of treatment. Modifications were made to work and activity to allow for increased time in standing frame to avoid complications of bedrest while he was unable to utilize wheelchair for typical lengths of time and he was able to return to pre-sore level of activity rapidly.
His time to rise from supine to sitting decreased from >3 minutes to <1 minute over the course of his first year of treatment. He endorsed a wearing off effect in the weeks preceding next maintenance dose of nusinersen but reported general improvement and improved ability to tolerate his workday.
Participant 4 (565): Adult female with SMA type 3b, in her early 40’s at enrollment, with 3 copies of SMN2. Symptom onset in elementary school, characterized by atypical gait. She is married, lives with her spouse and works full-time.
At enrollment, she used a cane for community distances some of the time and was able to climb a half flight of stairs with two hands on a railing and extreme difficulty. She was unable to rise from the floor without assist or furniture. She has a long history of anxiety/depression/chronic back pain, which impacted her function intermittently during treatment. She participated intermittently with outpatient physical therapy, primarily to address back and sciatic pain.
Following loading phase and through the early maintenance phase, she reported no apparent benefit. However, after 10 months, she was able to increase walking distances, and reported improved energy for work, ADL’s such as carrying groceries, and social events. Her spouse endorsed a decreased rate of falls.
Participant 5 (623): Adult male with SMA type 3b, in his mid-20’s at enrollment, with ≥4 copies of SMN2. Symptom onset during puberty characterized by difficulty with climbing stairs and rise from the floor. He lives independently, completed graduate education and began working full-time during this study.
At enrollment he was ambulatory with a straight cane, but able to rise from floor only with assist. He sustained regular falls, averaging 1–2 per month. He was diagnosed with obstructive sleep apnea (OSA) during pre-treatment work up and initiated CPAP use during loading phase.
Following the loading phase, he struggled with weight gain attributed to life stressors and decreased access to exercise/gym due to work schedule. Fourteen months into treatment, he noted significant improvements in mental endurance, ability to manage 60+ hour work week demands, and walking tolerance, enabling him to commute around 4 miles daily via walk and subway for work. Falls had decreased to a single fall in the four months prior attributed to environmental factors (tripped by a pet) and improved ability to get up from the floor.
Participant 6 (626): Adult male with SMA type 3b, in his early 30’s at enrollment, and ≥4 copies SMN2. Symptom onset at age 11 years was characterized by fatigue, exercise intolerance and inability to keep up with peers in sports as well as falls. He reports he was ‘clumsy’ as a child, which may have been earliest symptom. He lived alone initially, but depended on family to help with transfers, or slept in a recliner so he could get in and out of it independently. He now lives with family.
At enrollment he was minimally ambulatory and primarily used a power chair. Mobility was limited by generalized weakness, obesity and significant lymphedema resulting in recurrent cellulitis. He could walk room distances with a cane and effort (10mwt time was 80 seconds at first visit). He started a diet and exercise program with outpatient physical therapy prior to initiating treatment, resulting in significant improvement in mobility (decreased 10MWT time from 81 to 19 seconds and was able to complete 6 MWT at baseline/Day 0 testing). He had suspected chronic OSA, diagnosed at enrollment, but did not obtain a CPAP machine until 10 months post start of nusinersen therapy and did not use consistently.
Following the loading phase, he had decreased access to therapy/gym due to insurance issues but remained ambulatory at level noted during start of treatment. He sustained several falls over the course of the study and had an episode of recurrent cellulitis requiring hospitalization for intravenous antibiotic therapy, which limited ability to participate in some aspects of motor testing at the 10-month follow-up visit. Housing also became unstable, which limited his access to safe daily home level mobility between months 8 to 15 of follow up. However, he reported improved ability to tolerate longer workdays and perform ADLs. When healthy, he reported improved tolerance for walking around his home.
DISCUSSION AND CONCLUSION
This small prospective cohort study of adults with SMA type 3 was designed to maximize our chance to demonstrate clinically meaningful improvement with nusinersen therapy using existing motor outcome measures. The fluctuating performance across patient visits and the adverse events that occurred over a 14–20-month period of treatment, even in adults with relatively “mild” disease, highlights the inherent challenges in proving a given therapy is effective in stabilizing or improving motor function in the adult SMA population. A 2-point increase in scores on either scale was considered clinically meaningful improvement for our cohort [20], but fluctuating individual performance was evident and frequently associated with concomitant adverse events including falls resulting in injury that impaired performance during one or more outcomes assessments. Our data demonstrate an overall time-dependent gradual accrual of increased motor function scores on the HFMSE and RULM and add to other recently reported cohort studies showing either stability or improvement in motor function in adults treated with nusinersen [28, 29].
Walter et al. reported findings from a recent study of a high-functioning cohort of 19 adults with SMA who showed statistically significant improvement in walking endurance (6 MWT) and upper limb function (RULM), but not in motor function (HFMSE), over a period of 300 days on nusinersen [29]. However, in the context of high individual variability of responses to treatment, the statistically significant changes did not translate to clinically meaningful change as measured by effect sizes. In contrast to Walter et al., our cohort of ambulatory adults with SMA type 3 did not show consistent improvement in 6 MWT. Veerapandiyan et al. reported clinically meaningful stability or improvement in individual RULM scores in their cohort of 12 patients, all of whom had contractures and scoliosis, over 4–26 months on nusinersen treatment [28]. One of three ambulatory patients showed clinically meaningful improvement 6 MWT distance. Motor function (HFMSE) was not performed given severe contractures in this cohort. In sum, recently published reports of motor outcomes for adult with nusinersen all suggest high variability in response to treatment, yet an overall cohort response of either stability or improvement. The variability in findings across studies and within cohorts underscores the need for robust, sensitive and clinically meaningful outcome measures and biomarkers to reflect disease progression.
In our cohort, measurable improvements in motor function were modest and accrued slowly over time from initiation of nusinersen therapy, supporting a time-dependent effect. The natural history of disease progression in adults with SMA is characterized by prolonged periods of stability with cumulative declines in motor function accrued incrementally over several years, or sometimes more precipitous decline in the setting of illness or injury. Thus, it may take several months or even years to fully appreciate that stabilization of or clinically meaningful improvement in motor function has clearly occurred.
All participants in this cohort reported improvements in energy levels and/or greater ability to tolerate work and home demands, and the ambulatory participants reported fewer falls. These findings add to those reported by Veerapandiyan et al. related to subjective improvements in endurance and hand strength which translated to improved activities of daily living in their cohort [28]. Notably, patient reported outcomes used in our study, including the modified SMAFRS and PedsQL multidimensional fatigue scale, did not reflect reported benefits.
Three of the 6 participants recruited for this study had not been evaluated regularly by a center with multidisciplinary expertise in the care of neuromuscular patients for many years prior to their interest in seeking nusinersen therapy. Most were working full-time, living independently and commuting to and from appointments for treatment and assessment, and none had received the recommended standard of care monitoring. During baseline assessment, we determined that OSA was present in 4 of 6 subjects, and carnitine deficiency was noted in one subject who had been recently pursuing a restricted vegan diet. Anecdotally, many busy adults with SMA type 3 do not acknowledge OSA or respiratory compromise and are resistant to referral for diagnosis and management. Undiagnosed, untreated OSA in adults with SMA may result in adverse long-term cardiopulmonary, neurocognitive and mood outcomes, as is the case in adults without SMA [30], but untreated OSA could also confound short term responses to therapy. Worthy of note, the participant with the most improvement in fatigue on the PedsQL fatigue score had initiated CPAP concomitant with initiation of nusinersen treatment.
All participants in this study reported a subjective improvement in energy levels and/or greater ability to tolerate work and home demands with dosing, followed by a perceived reduction in benefit a few weeks before their next dose of nusinersen, suggesting a possible dose-dependent effect. Infants, children and adults currently receive the same absolute dose of nusinersen regardless of age, body weight and CSF volume of distribution. More studies are needed to confirm the ideal nusinersen dose and dosing frequency in adults, and an improved paradigm for loading that reduces the burden of 4 intrathecal injections over a 60-day period.
The main limitation of this pilot cohort study is the lack of an untreated control or comparator group, necessary to assess the possibility of a placebo effect, increased motivation with treatment or learning with repeated testing. However, even in this small cohort, we have showed clinically meaningful improvement or stabilization on individual motor function scores over time. The magnitude of the change over time and the time-dependent accrual of benefit makes it less likely that our observed changes are due solely to placebo effect, increased motivation, or learning with repeated testing. Each subject had experience with standardized outcomes used for this study from prior monitoring for at least one or two evaluations prior to enrollment, making it unlikely that their performance was influenced by learning with repeated testing. Individual variability in phenotype and motor function even in this targeted subset suggest the need for a significantly large number of subjects to perform an effectively powered, well-controlled clinical trial.
Similar to Veerapandiyan et al., we plotted individual changes in motor function scores to clearly highlight individual differences and clinically meaningful change for each motor parameter with treatment. This approach to cohort reports is particularly useful in the clinical setting to monitor longitudinal response to therapeutic intervention. Nonetheless, such observations cannot replace the need for a Class 1 randomized controlled clinical trial would provide the best evidence for definitive benefit of nusinersen efficacy in adult patients with SMA Type 3. Study design for such a trial will be critical and must incorporate a dose escalation component to help address questions regarding the ideal dosing regimen for adults with SMA. Although outside of the scope of this study, serum and CSF biomarkers that reflect neuronal survival and function should be explored. Finally, this study highlights the acute need for a broadly accepted standardized patient-reported outcomes measure that can measure what SMA adults really care about: their overall well-being.
CONFLICTS OF INTEREST
CJJY has no conflicts to disclose
SDS has no conflicts to disclose
ELT has no conflicts to disclose
RZZ has no conflicts to disclose
KJS serves on the scientific advisory board of Cure SMA. She is a consultant to Biogen, AveXis and Roche. She receives clinical trial funding from Biogen and AveXis, and grant support from Biogen for studies in SMA.
Footnotes
ACKNOWLEDGMENTS
We gratefully acknowledge the site clinical coordinators who were critical to the data collection used for this study, specifically Kendall Trautman, Maria Soledad Herrmann, and Emma Rodrigues. We gratefully acknowledge financial support from the Freeman Foundation, the Joukowsky Foundation and Cure SMA. Most importantly, we thank the participants and their families.
