Abstract
Background:
Repeated neuromuscular electrical stimulation in type 1 Myotonic Dystrophy (DM1) has previously been shown to cause an increase in strength and a decrease in hyperexcitability of the tibialis anterior muscle.
Objective:
In this proof-of-principle study our objective was to test the hypothesis that noninvasive repetitive transcranial magnetic stimulation of the primary motor cortex (M1) with a new portable wearable multifocal stimulator causes improvement in muscle function in DM1 patients.
Methods:
We performed repetitive stimulation of M1, localized by magnetic resonance imaging, with a newly developed Transcranial Rotating Permanent Magnet Stimulator (TRPMS). Using a randomized within-patient placebo-controlled double-blind TRPMS protocol, we performed unilateral active stimulation along with contralateral sham stimulation every weekday for two weeks in 6 adults. Methods for evaluation of muscle function involved electromyography (EMG), hand dynamometry and clinical assessment using the Medical Research Council scale.
Results:
All participants tolerated the treatment well. While there were no significant changes clinically, EMG showed significant improvement in nerve stimulus-evoked compound muscle action potential amplitude of the first dorsal interosseous muscle and a similar but non-significant trend in the trapezius muscle, after a short exercise test, with active but not sham stimulation.
Conclusions:
We conclude that two-week repeated multifocal cortical stimulation with a new wearable transcranial magnetic stimulator can be safely conducted in DM1 patients to investigate potential improvement of muscle strength and activity. The results obtained, if confirmed and extended by future safety and efficacy trials with larger patient samples, could offer a potential supportive TRPMS treatment in DM1.
Keywords
INTRODUCTION
Myotonic Dystrophy Type 1 (DM1) is an autosomal dominant multi-systemic disorder that affects skeletal and heart muscles, endocrine system, eyes, and central nervous system [1–3]. The underlying genetic defect is an unstable CTG repeat expansion on chromosome 19 in the 3’ untranslated region of the myotonic dystrophy protein kinase (DMPK) gene. It is the most common type of muscular dystrophy occurring in adults, with a prevalence of nearly 1 in 8000 [3]. The clinical features involving skeletal muscles in this condition consist of weakness and wasting of facial, neck and distal muscles, which at a later stage progress to immobility, respiratory insufficiency, dysarthria, and dysphagia [1, 3]. DM1 is also associated with cardiovascular manifestations such as conduction defects, tachyarrhythmia, cardiomyopathy, heart failure and valvular dysfunctions [4]. Myotonia is characterized by failure of the muscle to relax after contraction and is sometimes accompanied by myalgia. Muscle dysfunction is also due to neuromuscular hyperexcitability and electromechanical deficits at rest and after a fatiguing exercise [5, 6].
Presently there is no effective treatment for this disorder. Treatment strategies for muscle impairment have focused on improving the muscle strength and reducing the hyperexcitability of the muscles. One experimental approach to treat DM1 adopted in a pilot study was to use repeated electrical stimulation of the motor point of a muscle, a method called neuromuscular electrical stimulation (NMES), which has been shown to be useful in sports medicine, stroke, spinal cord injury and cerebral palsy [7–10]. This study showed an increase in muscle strength and normalization of the curve of average rectified surface electromyographic (EMG) signal amplitude in DM1 patients [7].
Since repeated stimulation of muscles could be therapeutic, it would be worthwhile to explore the usefulness of stimulation methods that have the potential to activate multiple skeletal muscles. One approach to achieving this is using noninvasive stimulation of the cortical motor strip with repetitive Transcranial Magnetic Stimulation (rTMS). rTMS of the cerebral cortex is an important and useful technique in neuroscience research, as well as in diagnostic and therapeutic clinical investigations [11, 12]. Transcranial Magnetic Stimulation (TMS) has previously been used to study cerebral cortical excitability, sensory-motor plasticity, and central motor conduction time (CMCT) in DM1 patients [13, 14]. One limitation of conventional TMS, besides the bulkiness of the device and the large amount of current involved, is that it only allows for stimulation at one cortical site at a time. In our hospital, Santosh Helekar, in collaboration with Henning Voss of Weill-Cornell Medical College, has developed a compact portable and wearable multifocal TMS device called Transcranial Rotating Permanent Magnet Stimulator (TRPMS) that can deliver highly focal low intensity magnetic stimuli at multiple cortical sites simultaneously or sequentially [15]. It uses rapidly rotating small high strength permanent magnets to modulate currents in the brain. The TRPMS device is also ideally suited to conduct double-blind placebo-controlled studies because non-magnetized rods can be used in the same device with the same rotational parameters to provide sham stimulation that is perceptually indistinguishable from active stimulation. The subjects cannot tell the difference between active and sham stimulation because they do not feel any pain, twitch, heat or shock from the rotating magnetic field of the active stimulus [15]. The peak amplitude of the TRPMS stimulus is 7%of maximal machine output of conventional TMS [16]. It is therefore lower than the resting motor threshold and does not elicit a motor-evoked potential. However, repeated TRPMS stimulation of the cortical representation of thenar muscles in the primary motor cortex (M1) can increase the frequency of spontaneous motor potentials in healthy adults [15]. We have recently observed that repeated TRPMS stimulation for 20 min caused persistent enhancement of motor cortical excitability comparable in magnitude and duration to high frequency and theta burst forms of rTMS [17]. Repeated stimulation with the TRPMS device in preliminary studies has been found to be well tolerated [15, 18–20]. A pilot phase 1/2a randomized double-blind sham-treatment controlled TRPMS trial in chronic ischemic stroke has recently shown potentially restorative activation of stimulated cortical areas surrounding the lesion [20]. The multifocal stimulation capability provided by TRPMS allows us to activate, for the first time, nearly the entire motor strip on both sides to possibly stimulate most muscles of the body simultaneously or in quick succession. The objective of this study was to test the hypothesis that the strength and function of two affected skeletal muscles can be improved by repeatedly stimulating M1, which includes the motor representation of those muscles.
MATERIALS AND METHODS
Study design
We conducted a small pilot randomized within-patient placebo-controlled double-blind clinical study in 6 adult DM1 patients who were recruited from the Neuromuscular Disorders Clinic of the Houston Methodist Hospital. The baseline characteristics and clinical profile of these patients are given in Supplementary Table S1. The sides of active and sham stimulation in the same patient were assigned by random binary choice made by a MATLAB (Mathworks, Natick, MA) computer program using a hardware true random number generator (TrueRNG, Ubld.iT, Chase, MD). The researchers who recruited the subjects, and performed the clinical assessments, EMG recordings and data analysis were all blinded with respect to the side of active and sham stimulation. The study protocol and informed consent form signed by the subjects was approved by the Institutional Review Board (IRB) of the Houston Methodist Research Institute (HMRI). All procedures on human subjects were done in accordance with the ethical standards of the Committee on Human Experimentation and in accordance with the Helsinki Declaration of 1975.
The inclusion criteria for recruitment of study participants were: 1. Age between 20 and 60 years; 2. clinical diagnosis of DM1; and 3. medication regimen for symptomatic treatment of DM1 or comorbidities that is stable for 6 weeks prior to enrollment. The exclusion criteria were: 1. History of neurological or psychiatric disorders, other than DM1; 2. presence of metal implants or metallic or electronic devices in the head; and 3. history of drug or alcohol abuse.
Procedures
We used the portable wearable TRPMS device to noninvasively stimulate the brain (Fig. 1). This device is an investigational device not yet approved by the U.S. Food and Drug Administration (FDA), and is deemed non-significant risk by HMRI IRB in consultation with the FDA. Active or sham TRPMS stimulation treatment consisted of repeated application of 100 ms oscillatory stimuli every 5 s (i.e., at the rate of 0.2 Hz) for 40 min to the M1 using 3 microstimulators placed 3 cm apart along the precentral gyrus, the lateral two of which spanned the motor representation of the hand. One of these microstimulators was placed on the scalp projection of peak activation in a functional magnetic resonance imaging (MRI) scan during repeated voluntary gripping movements of the contralateral hand. Active and sham stimulation involved rotation of strong cylindrical magnets and non-magnetized rods, respectively. The timing, speed and duration of rotation of the motors in each case were otherwise identical [15]. We located the precentral gyrus in a brain MRI scan conducted before the start of treatment in reference to fiducial markers (vitamin E capsules) placed on a neoprene device cap (without microstimulators) worn by the patient. To map the hand motor representation, we performed a task-associated functional MRI scan while the patients performed 5 gripping movements with their left and right hands in a 14 s “on-off” block design. One side received active TRPMS and the other side received sham TRPMS in a randomized manner. One participant declined to undergo a functional MRI scan, so the hand motor representations in that case were assumed to be at the C3 and C4 loci in the 10 –20 electroencephalographic electrode montage, which corresponded to sites on the precentral gyri in the anatomical MRI scan. The correlation between C3/C4 and underlying precentral gyri has been established by several studies in the literature [21, 22] and these sites have been used to target the M1 by others [23]. We administered the 40 min stimulation every weekday for two weeks (total 10 stimulations). To monitor adverse effects and any unexpected perceptual, affective, or behavioral changes, we asked the patients to respond to a standardized questionnaire on an electronic tablet (see Supplementary Figure S1). The questionnaire results, in terms of recording of any differences in responses before and after treatment each day, and clinical assessment and EMG results were analyzed by blinded researchers.
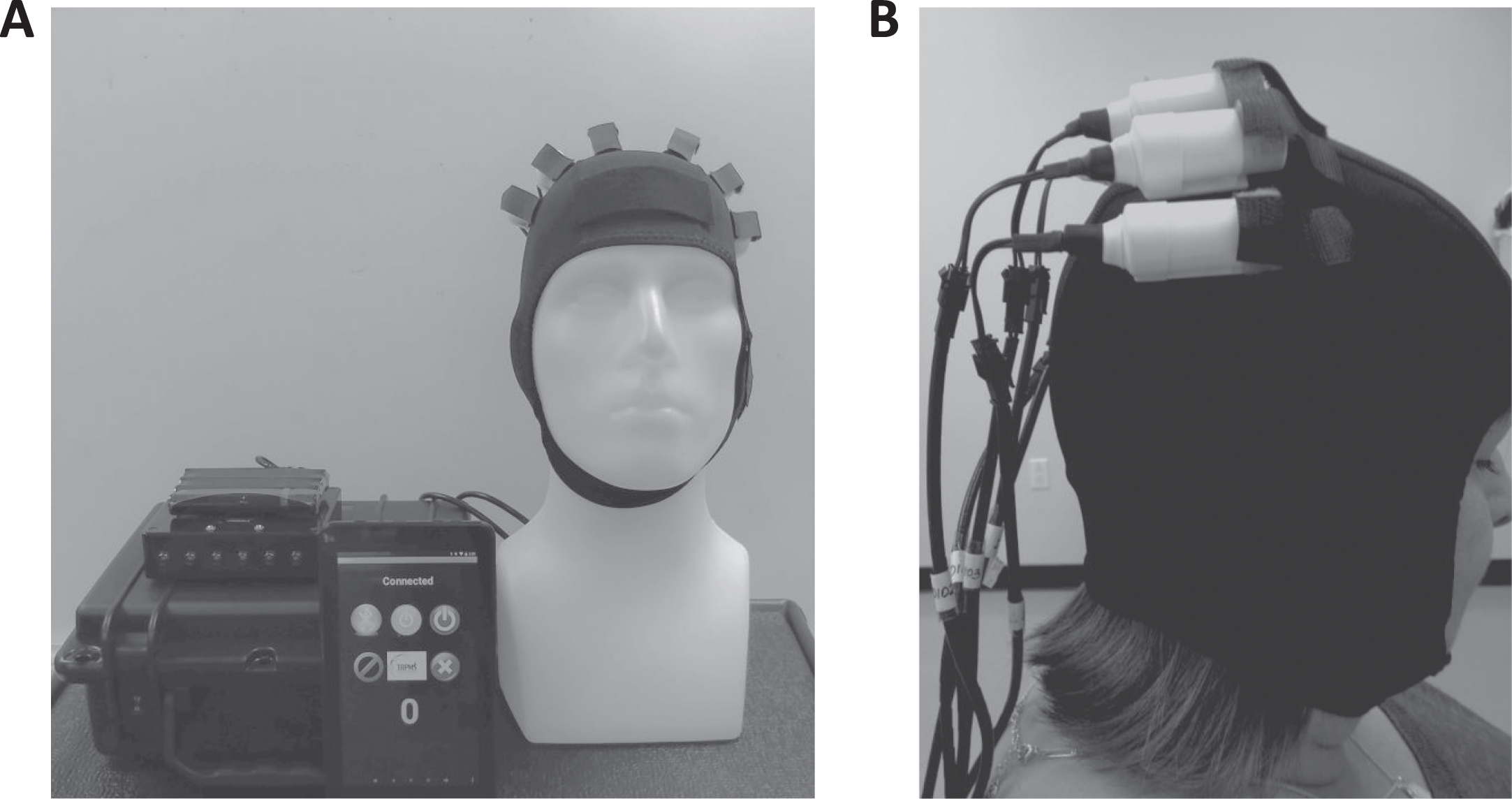
A. TRPMS device cap with attached microstimulators on M1 bilaterally and battery-powered device controller. The smartphone screen image shows the Android app used to operate the device. B. TRPMS device cap with three microstimulators attached 3 cm apart on sites along the right M1 of a DM1 patient.
Clinical assessments involved determination of muscle strength using the Medical Research Council (MRC) scale and a hand grip dynamometer. We used a standard MRC scoring protocol, which involves determining the strength of deltoid, biceps, triceps, and wrist, finger and thumb flexors and extensors for upper extremities. For lower extremities it includes measuring the strength of hip, knee, and toe flexors and extensors, and muscles causing flexion and extension of the ankles [24, 25]. The means of scores of all muscle groups in each upper or lower extremity were used as composite scores of muscle function for analysis. The time points of these assessments were immediately before treatment, and at one week, two weeks, one month and six months after the start of treatment. We also conducted EMG recordings of compound muscle action potential (CMAP) amplitudes during repetitive ulnar and accessory nerve stimulations (5 stimulus repetitions at 3 Hz, 0.1 ms stimulus duration) in the first dorsal interosseous (FDI) and trapezius (Tpz) muscles, respectively, on both sides before, and 1 min and 5 min after a short exercise test (SET), using standard procedures in published studies [26, 27]. The CMAP amplitude from baseline to the negative peak was measured manually by moving a cursor to these positions on the screen of a clinical EMG machine (XLtek Neuromax 1004). SET is a test that is often used as part of EMG recordings in myotonic dystrophy. It is well established that there is a decrease in amplitude of CMAP when the recorded muscle is subjected to a short exercise in DM1 patients compared to healthy controls [26, 27]. In this study the SET consisted of 30 s abduction and adduction of fingers for FDI and elevation of the shoulder against resistance for Tpz. Measurements of maximal CMAP amplitudes were made immediately before and after the 10-day treatment using surface electrode recordings on the FDI and Tpz [28–30].
Data analysis
We analyzed pre- and post-treatment grip strengths and MRC scores employing the Kruskal-Wallis test followed by Tukey-Kramer test for multiple comparisons, and CMAP amplitudes using the two-tailed Student’s paired t test. On CMAP amplitudes, we performed pair-wise comparison between values measured in each muscle in response to the 1st and 5th stimuli delivered to its respective innervating nerve at pre-SET, 1 min post-SET and 5 min post-SET before and after stimulation treatment, as well as between the sides affected by active and sham TRPMS. Since we performed 3 independent pair-wise comparisons (pre, 1 min post-SET, and 5 min post-SET) in each subject in each of the two muscles for the 1st and 5th CMAPs, we used the false discovery rate test to correct for multiple comparisons. We also compared the proportions of patients showing restorative changes in CMAP amplitudes by carrying out the “N-1” Chi-squared test [31].
RESULTS
All 6 adult DM1 patients (aged 45.8±11.8 years, 1 male, 5 females) who were recruited in the study completed the entire TRPMS stimulation treatment protocol. They did not report any adverse effects, or any transient or lasting changes in perception, mood, or behavior. There were no consistent differences in answers to the questions given by any of the subjects in the questionnaire presented to them before and after treatment each day. We conducted clinical assessments of motor function using MRC scoring and hand grip dynamometry at all planned time points in 5 patients. The 6th patient (female) relocated out of the state and was not available for evaluation at 6 months post-treatment. We recorded EMG bilaterally both pre- and post-treatment in 5 of the 6 patients. We did not obtain pre-treatment EMG recordings on the active TRPMS side in one patient (female).
Clinical assessments
Figure 2 shows grip strengths and composite total MRC scores at all time points plotted as boxplots. Non-parametric Kruskal-Wallis test followed by multiple comparison (Tukey-Kramer) test revealed no significant differences in these motor function values between time points due to either active TRPMS or sham TRPMS stimulation. Individual patient scores are plotted in the Supplementary Figure S2.
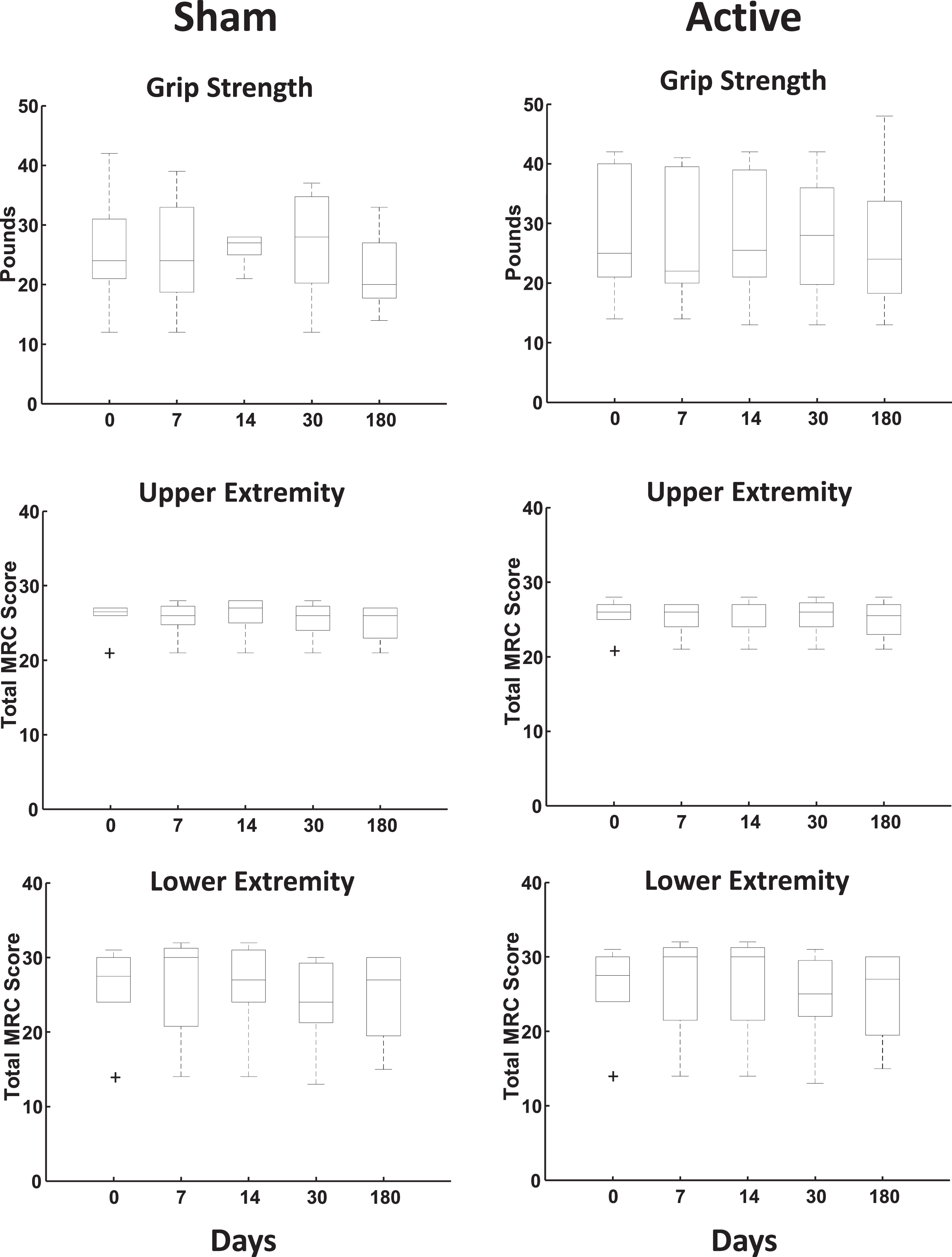
No change in grip strength and MRC scores with active TRPMS treatment. Boxplots of dynamometric grip strengths in pounds and clinically assessed total MRC scores [N = 6 in each case, except for 7 (N = 5), 30 (N = 5) and 180 (N = 4) days]. The horizontal line inside each box marks the median. The edges of the box represent the 25th and the 75th percentiles. The whiskers span extreme values not considered to be outliers by the plotting algorithm. Individual crosses mark outliers.
EMG results
In contrast, analysis of CMAP amplitude data showed a significant difference between pre- and post-treatment values on the side that was affected by active TRPMS stimulation i.e., the side contralateral to the actively stimulated cerebral hemisphere (Table 1, Fig. 3, post- minus pre-treatment changes in individual patients are plotted in Supplementary Figures S3 and S4), but not on the side affected by, and contralateral to, sham stimulation. DM1 patients show marked decrease in CMAP amplitude compared to healthy subjects after a muscle is subjected to SET [26, 27]. The increase in CMAP amplitudes of the FDI muscle measured 1 min after the SET on the side influenced by active TRPMS was more pronounced post-treatment compared to pre-treatment, if post-SET CMAP amplitudes were normalized with respect to pre-SET amplitudes (p = 0.041, T = 2.96, df = 4, Student’s paired t test, Fig. 3). Normalization was done to minimize the influence of variability across subjects. The normalized data in Tpz also showed a similar trend as that in FDI with active TRPMS, but it did not reach significance (data not shown). In terms of proportion of patients, in the normalized data all 5 patients whose data were available for analysis showed the post-treatment increase in the CMAP amplitude in FDI due to active TRPMS. On the sham-affected side, wherein data from all 6 treated patients were available, there was no increase in 5 of them (difference significant with the “N-1” Chi-squared test, p < 0.008, χ2 > 6.94, df = 1). In the Tpz muscle, the proportions of participants showing active TRPMS- and sham TRPMS-related increases in the normalized data were 5 of 6 and 2 of 6, respectively, but the difference did not reach significance. Differences between the pre- and post-treatment amplitudes of the CMAPs evoked by the 2nd –5th stimuli at both post-SET time points (1 and 5 min) and those evoked by all 5 stimuli at the 5 min time point were not significant with regard to active and sham TRPMS (data not shown).
CMAP amplitudes from EMG recordings
NS: non-significant at p = 0.05 level; *: significant with false discovery rate testing for multiple comparisons.
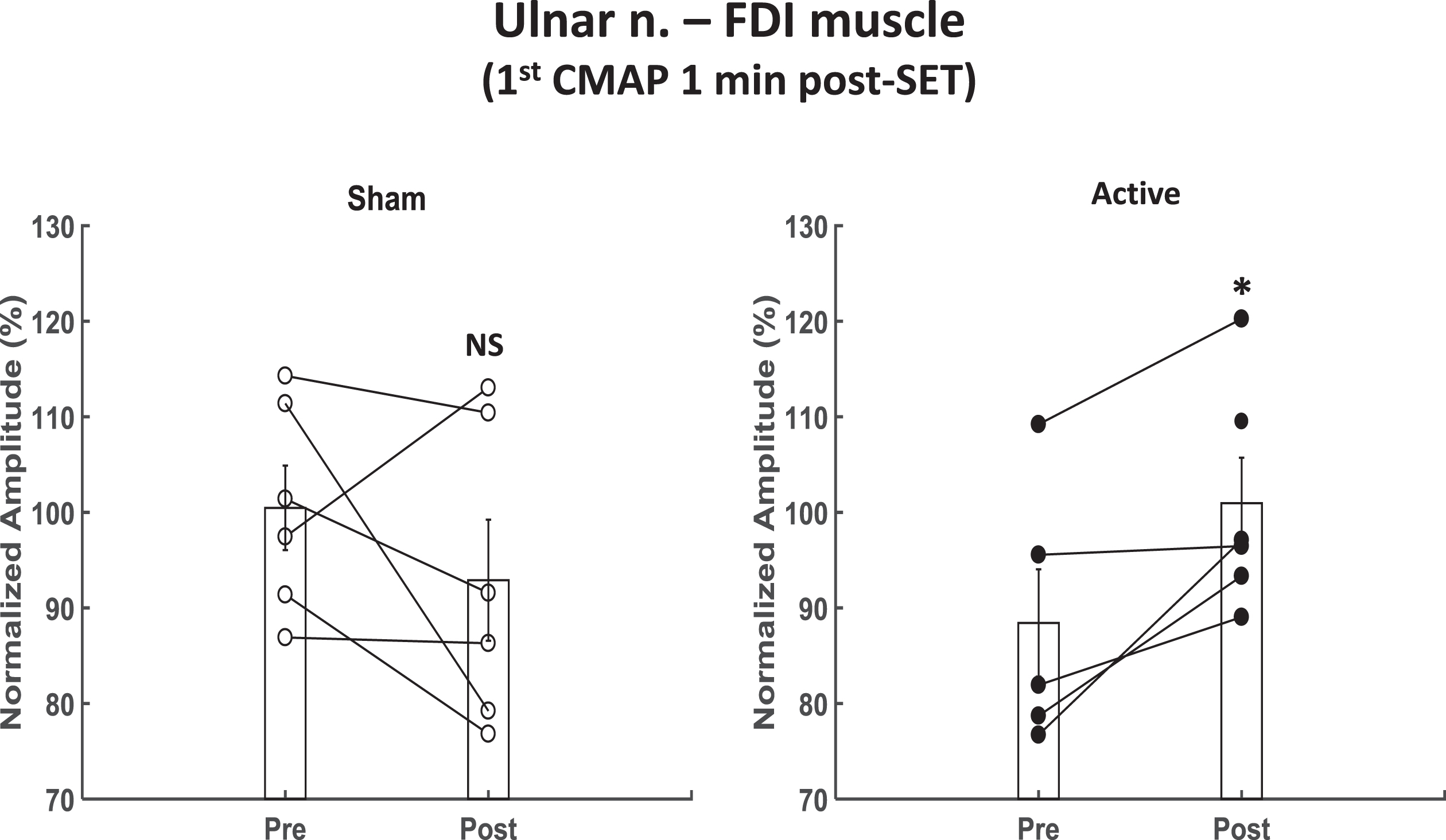
Increase in Post-Short Exercise Test (SET) CMAP amplitude with active magnetic stimulation. Line plots show change in the normalized amplitude of the 1st CMAP from pre-treatment time point to post-treatment time point immediately after a 30 s SET. Active: data from the side contralateral to the actively stimulated side. Sham: data from the side contralateral to the sham stimulated side. Bar plots represent means±standard errors of the mean across subjects at each time point. N = 5 and 6 for pre- and post-treatment, respectively. NS: non-significant. *: significant at p = 0.041 (Student’s paired t test).
In addition to the above analyses, we also compared CMAP values between the sides affected by active and sham TRPMS. As expected, there were no significant differences in amplitudes between active and sham TRPMS before treatment (Fig. 4A). However, after treatment, the active TRPMS-related amplitudes were increased in FDI (Fig. 4A and B) and Tpz (Fig. 4C). In the FDI muscle, both the 1st (Fig. 4A) and 5th (Fig. 4B) CMAP responses recorded 1 min post-SET were increased (p = 0.009, T = 4.08, df = 5; and p = 0.019, T = 3.42, df = 5, respectively, Student’s paired t test). The 1st CMAP response 1 min post-SET was also increased (p = 0.035) in Tpz (Fig. 4C). We observed increased CMAP amplitudes at this time point in both FDI and Tpz in all 6 participants. The proportion of participants showing increased CMAP amplitudes in both FDI (p < 0.034, χ2 > 4.45, df = 1) and Tpz (p < 0.002, χ2 > 9.99, df = 1, “N-1” Chi-squared test) were greater following active stimulation. Collectively, these findings suggest that repeated TRPMS stimulation might be causing an increase in the excitability of the peripheral neuromuscular pathway. These facilitatory electrophysiological changes, however, did not result in enhanced muscle strength over the test time-period.
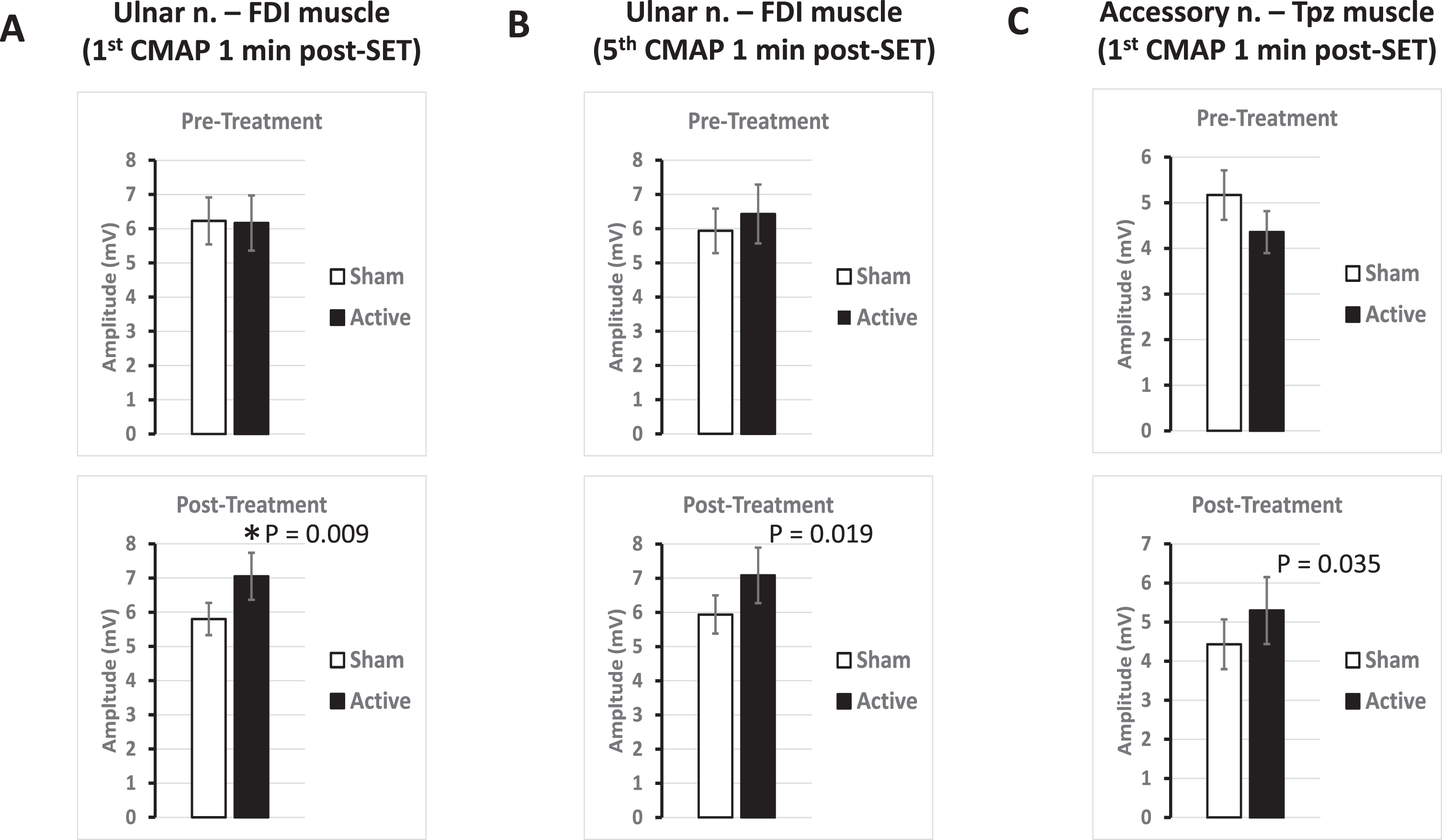
Increase in CMAP amplitudes post-treatment with active magnetic stimulation. A. Bar plots of pre- and post-treatment 1st CMAP amplitudes of the FDI on the side contralateral to active magnetic stimulation (p = 0.009, Student’s paired t test, *: significant with false discovery rate multiple comparisons test). B. Bar plots as in A for 5th CMAP amplitudes (p = 0.019, Student’s paired t test). C. Bar plots as in A in the Tpz muscle on the side contralateral to active magnetic stimulation (p = 0.035, Student’s paired t test).
DISCUSSION
We used a newly developed multifocal TRPMS cerebral cortical stimulator to conduct 40 min repetitive stimulation of the motor strip in DM1 patients on 10 weekdays to test whether this procedure enhances muscle strength and function. There were no measurable improvements in clinical assessments of muscle function post-treatment. However, comparing active stimulation of one side with sham stimulation of the other side we observed potentially restorative changes in CMAP parameters on the side contralateral to active stimulation, as opposed to sham stimulation. Because the mean normalized pre-treatment CMAP amplitude on the active side is lower than on the sham control side in Fig. 3 it is possible that the increase seen with treatment is due to regression to the mean. However, while this study is limited by the small sample size, the fact that two subjects with the lowest pre-treatment values show a decrease in the normalized CMAP amplitude on the sham-treatment side and two subjects with the highest pre-treatment values on the active treatment side show an increase, makes it unlikely that regression to the mean is the explanation for the significant increase with treatment.
The lack of clinical improvement might be because the total duration of treatment was not long enough, and treatment duration of 4–6 weeks might be required for the beneficial effects to become apparent at the gross functional level. The wide range of disease characteristics and severity may also be a reason for non-significant clinical results, and a more homogenous cohort might have shown a detectable improvement. Additionally, DM1 being a gradually progressive disease repeated courses of treatment with the TRPMS device may be needed to maintain or improve muscle strength over many years. Although to our knowledge there is no evidence for this in the literature, electrophysiological changes might also be a more sensitive indicator of the potential therapeutic benefit on sarcolemmal excitability. We recognize that possible confounding bilateral crossover effects of unilateral stimulation could have obscured the differences between the sides. Ipsilateral motor representation is well documented using TMS [32–34].
The observed TRPMS effect on CMAP could be mediated through a peripheral mechanism such as that invoked by NMES. Prior NMES study in DM1 [7] has shown that repeated stimulation of the tibialis anterior muscle for 15 days, consisting of two 60 min daily sessions, caused an increase of muscle strength, improvements in motor function tests and EMG in DM1 patients. The underlying mechanisms could be an increase in mass of the stimulated muscle [35], increased recruitment of motor units [36] and enhanced metabolism of muscle fibers [37, 38], or some combination of these processes. Another possibility raised by an experimental result in mice could be a down regulation of small conductance calcium-dependent potassium channels or SK3 channels [39], which are known to be over-expressed in the muscles of DM1 patients [40].
NMES involves direct local stimulation of the muscle. Therefore, it is impractical to use it to cause a generalized improvement in neuromuscular function in all or most muscles. Multifocal TRPMS of the M1, on the other hand, could achieve improvement in the strength and function of a significant amount of the musculature by increasing neuromuscular activity through repeated activation of the cortico-spino-motor pathway, possibly by modulation of the ongoing tonic activity of cortical neurons. If successful, this approach would allow us to produce a more global neuromuscular benefit by bilateral stimulation of the cortical motor strips. The most effective and widely used method to chronically stimulate the cortex is rTMS [41, 42]. Only two studies in the literature have reported the use of single session TMS to elicit motor evoked potentials (MEPs) in myotonic dystrophy patients [13, 43]. In both cases it was used to investigate and quantify any changes in motor cortical excitability, latency and CMCT. There are no reports on the effects of chronic rTMS stimulation of M1 in the DM1 literature. Bilateral stimulation of the motor strips is not feasible with conventional single site rTMS even if multiple devices are used because of the large physical size of their stimulating coils. TRPMS would therefore be more feasible and convenient to achieve this type of therapeutic stimulation.
Bilateral multifocal TRPMS of the M1 could potentially be useful in improving neuromuscular function in DM1. Since the TRPMS device is portable, wearable, battery operated and triggered by a user-friendly smartphone app, this magnetic neuromodulation therapy could be self-administered by DM1 patients at home. This pilot study shows that chronic TRPMS treatment is well tolerated by DM1 patients. However, its limitations are the small sample size of 6 patients that are heterogeneous in their age of disease onset, disease type, ambulatory status, and number of CTG repeats. There is therefore a need for a larger study with well-powered active and placebo arms and a longer treatment duration to evaluate the safety and efficacy of this potential novel therapy for possibly improving muscular strength in DM1 patients.
Footnotes
ACKNOWLEDGMENTS INCLUDING SOURCES OF SUPPORT
We would like to thank Mr. Eric Caballero for technical assistance and Dr. Susan Xu of the Center for Outcomes Research of the Houston Methodist Hospital for expert advice on statistical analysis. This study was funded by Shanna and Andrew Linbeck Family Charitable Trust.
CONFLICTS OF INTEREST
The corresponding author is listed as an inventor on U.S. patent numbers 9456784, 10398907, 10500408 and 10874870 covering the device used in this study. The patent is licensed to Seraya Medical, LLC. The other authors declare that they have no conflict of interest.
