Abstract
Background:
Oxaliplatin-induced peripheral neuropathy negatively affects the quality of life for patients with gastrointestinal cancers and may cause neuropathic pain. Measures of peripheral nerve structure or function, such as intraepidermal nerve fiber density (IENFD) during treatment could reduce neuropathy severity through individualized dose reduction.
Objective:
The aim was to evaluate the predictive values of IENFD, quantitative sensory testing (QST), and nerve conduction studies (NCS) for significant neuropathy and neuropathic pain.
Methods:
Fifty-five patients were examined prospectively before, during, and six months following treatment using skin biopsies, QST and NCS. Clinically significant neuropathy six months after treatment was defined as reduced Total Neuropathy Score of more than five and neuropathic pain was assessed according to International Association for the Study of Pain criteria.
Results:
Thirty patients had a clinically significant neuropathy, and 14 had neuropathic pain. Vibration detection threshold (VDT) before treatment was correlated with clinically significant neuropathy six months after treatment (OR 0.54,
Conclusions:
Vibration detection thresholds and thermal detection thresholds may be useful for prediction of clinically significant and painful neuropathy, respectively. However, low to moderate sensitivity and specificity may limit the predictive value in clinical practice.
Keywords
INTRODUCTION
Oxaliplatin reduces mortality following gastrointestinal tract cancers, but leads to chronic chemotherapy induced peripheral neuropathy (CIPN) in approximately 75% and chronic neuropathic pain in 49% of patients [1]. CIPN following oxaliplatin treatment is generally irreversible [2]. No effective prevention has been developed, [3] and worsening of CIPN after cessation of treatment complicates clinical decisions regarding the duration and total dose of oxaliplatin treatment in individual patients based on symptoms of peripheral neuropathy alone.
Objective markers could identify patients at increased risk of CIPN before or during the early phases of treatment and may aid in adjusting oxaliplatin dose more precisely to balance the risk of CIPN against the desired antineoplastic effect.
Quantification of small nerve fibres in skin biopsies from the distal leg may provide a measure of early, possibly subclinical, peripheral nerve damage, measured as a reduction in intraepidermal nerve fibre density (IENFD), as suggested in a study of patients with diabetes without neuropathy [4].
Nerve conduction studies (NCS) assessing distal nerve segments have been shown to be predictive of severe chronic oxaliplatin-induced neuropathy [5]. The predictive value regarding neuropathic pain was not examined.
Assessment of peripheral nerve function is also possible through quantitative sensory testing (QST) of both small and large fibre modalities. In oxaliplatin treatment, cold hyperalgesia has been correlated with severe neuropathy, [6] but the predictive value of this finding was not examined.
We previously reported the results of a prospective study on early changes in these peripheral nerve measures during oxaliplatin treatment, [7] and found that sural nerve amplitude and conduction velocity, vibration detection thresholds, mechanical detection threshold and cold detection threshold were significantly reduced during treatment.
Based on the same study population, the aim of this study was to determine whether early changes in IENFD, QST or NCS during the first three months of oxaliplatin treatment could predict clinically significant or painful peripheral neuropathy assessed by neuropathy scores six months after end of treatment.
MATERIALS AND METHODS
The methods employed in this study have previously been described in detail [7]. Patients scheduled for treatment with oxaliplatin (adjuvant treatment or metastatic cancer) against colorectal or oesophago-gastric cancers at the Department of Oncology at Odense University Hospital were screened for inclusion. Patients between 18 and 80 years without pre-existing peripheral neuropathy or predisposition for peripheral neuropathy (e.g. diabetes mellitus, hypothyroidism, rheumatic disease, malnutrition, MGUS or treatment with medication associated with neuropathy) were eligible for inclusion. Patients were examined five times at the Department of Neurology: Before treatment (week -1), at least 10 days after the patient had received 25% (week 6) and 50% (week 12) of the total planned dose, after completion of treatment (week 24), and six months after completion of treatment (week 52). There was an interval of at least 10 days between treatment and examination to reduce the risk of interference from acute oxaliplatin-induced peripheral neuropathy.
Study procedures comprised skin biopsies from the distal leg, QST, and NCS. Neuropathy symptoms and signs were assessed using Neurological Symptom Score (NSS), [8] Neuropathy Impairment Score (NIS), [9] INCAT disability sum score (INCAT DS) [10] and INCAT sensory sum score (INCAT SS) [11].
Skin biopsies were processed and IENFD determined according to EFNS and PNS guidelines [12].
QST was performed according to DFNS (Deutscher Forschungsverbund Neuropathischer Schmerz) standards [13] and included evaluation of cold detection threshold (CDT), warmth detection threshold (WDT), heat pain threshold (HPT), mechanical detection threshold (MDT), mechanical pain threshold (MPT), and vibration detections thresholds (VDT) at the dorsum of the right foot. Furthermore, vibration sense was assessed using a biothesiometer (BIO-MEDICAL INSTRUMENTS CO., Newbury, Ohio) balanced on the tip of the first toe with a pressure corresponding to the weight of the instrument. The applied voltage (range 0–50) was gradually increased until vibration was perceived. The mean of three measurements was used.
Conventional NCS were performed on the sural (calf), peroneal, and tibial nerves bilaterally using standard surface electrodes.
The overall severity of the neuropathy was assessed using the reduced total Neuropathy Score (rTNS) (range 0–28), [14] which incorporates symptoms, signs, and some results of the sural and peroneal NCS. At follow up examination, patients completed the Neuropathic Pain Symptom Inventory questionnaire (NPSI) [15] to assess the severity of neuropathic pain.
Clinically significant neuropathy was defined as rTNS > 5. The results of nerve conduction studies were not included in rTNS for this classification to avoid circular reasoning regarding correlations between changes in nerve conduction variables and significant neuropathy. Neuropathic pain was defined according to the International Association for the Study of Pain criteria as probable in the presence of relevant symptoms and sensory signs without abnormal NCS or skin biopsy and definite with symptoms, signs and abnormal NCS or skin biopsy [16]. Severity of neuropathic pain was assessed using the NPSI.
Study procedures were approved by The Regional Ethical Committee for Southern Denmark (reference number S-20120077), and written informed consent was obtained from all patients.
Data analysis
Analyses were carried out in separate subsequent steps. First, all variables were analyzed at group levels (selected data shown in Figs. 1 and 2) for selection of variables to be included in subsequent logistic regression analysis. The reason for this pre-analyses was to avoid inclusion of to many highly correlated variables (multicollinearity) in the logistic regression analysis. The three variables with the most significant differences regarding baseline values and changes during the early phases of treatment (weeks 6 and 12) were then analyzed in a logistic regression analysis. For each time point, we calculated the odds ratios (95% confidence interval) of the association of these values with risk of significant neuropathy / neuropathic pain six months after treatment, compared with patients without this disorder / symptom. Analyses were carried out unadjusted, and adjusted for age, sex and total oxaliplatin dose. For VDT, CDT and sural SNAP changes from baseline to 25% and 50% of planned treatment (weeks 6 and 12) decreases in values were used for analysis to reflect deterioration of peripheral nerve function. Finally, we performed ROC analysis for variables with a significant correlation in the logistic regression analysis (VDT at baseline for clinically significant neuropathy and changes in CTD and HPT for neuropathic pain) to determine predictive cut-off values, sensitivity and specificity. Cut off values were determined as the value with the largest product of sensitivity and specificity in the ROC analysis. Analyses were performed using Stata/IC v. 15 (StataCorp LLC, College Station, TX, USA).
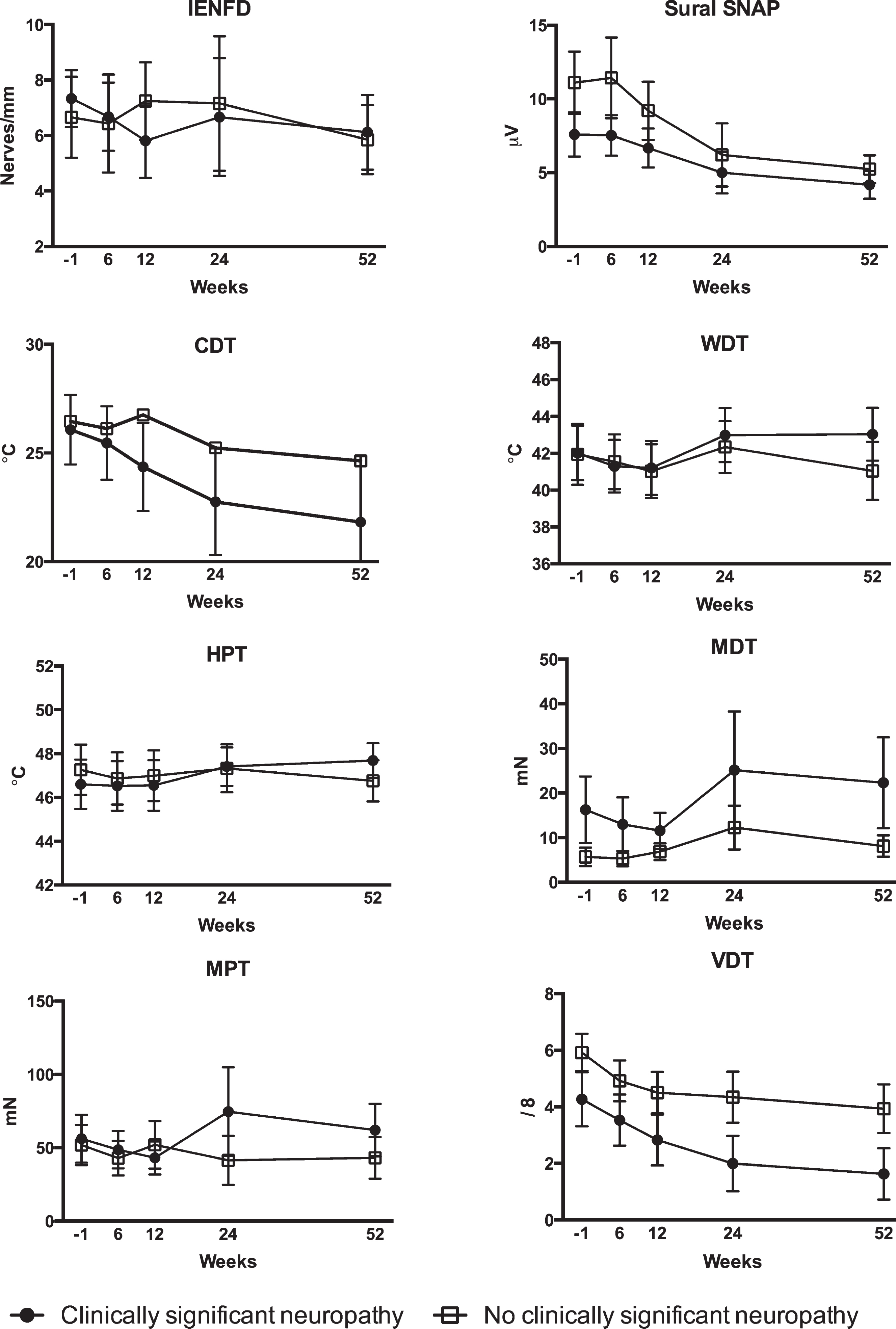
Results of skin biopsy analysis, sural nerve conduction studies and quantitative sensory testing before (week -1), during (weeks 6 and 12), after (week 24), and at examination six months after (week 52) treatment in patients with and without clinically significant neuropathy six months after treatment. IENFD: Intraepidermal nerve fiber density; SNAP: Amplitude of the sensory nerve action potential; CDT: Cold detection threshold: WDT: Warmth detection threshold; HPT: Heat pain threshold; MDT: Mechanical detection threshold; MPT: Mechanical pain threshold; VDT: Vibration detection threshold.
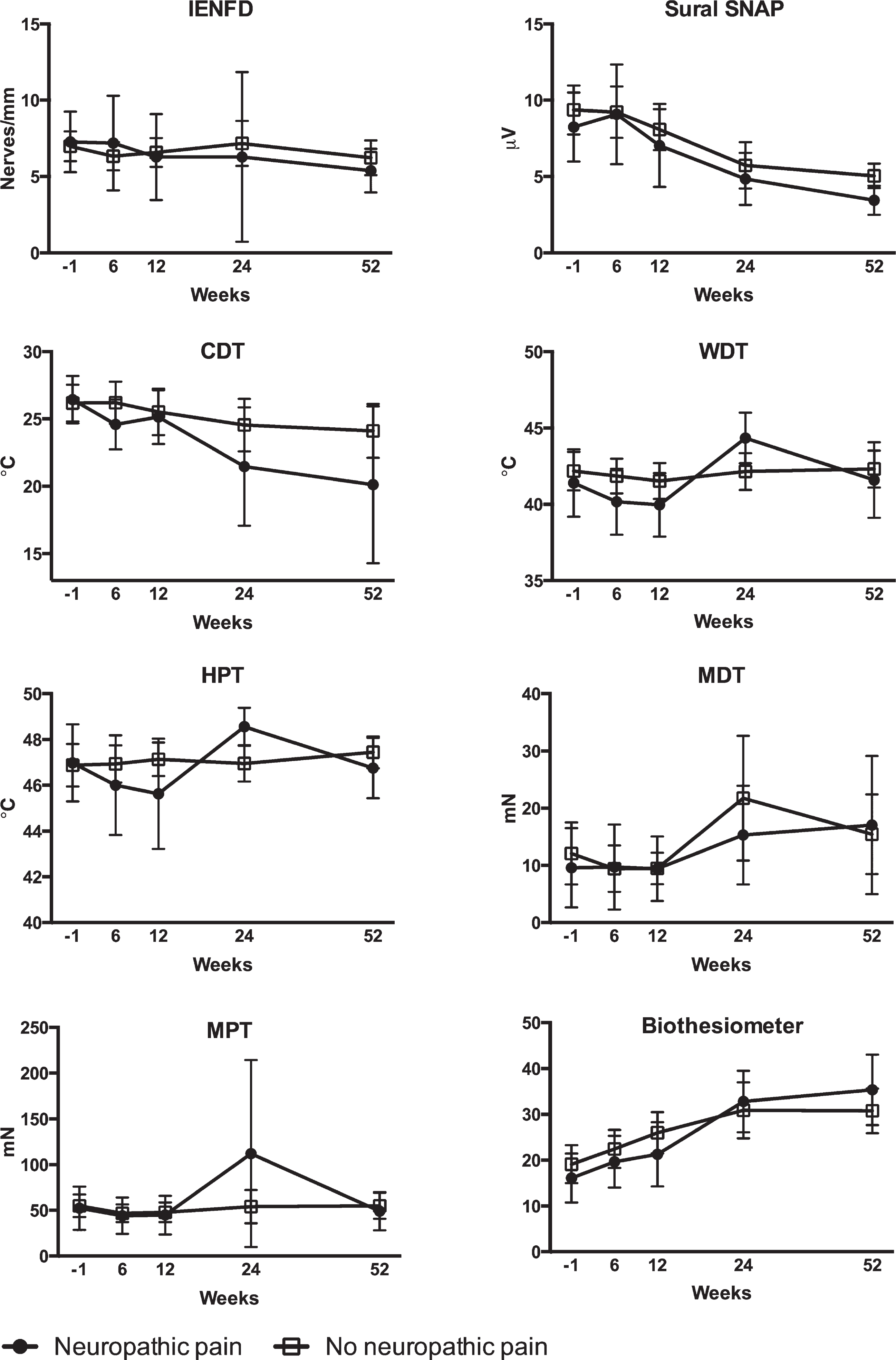
Results of skin biopsy analysis, sural nerve conduction studies and quantitative sensory testing before (week -1), during (weeks 6 and 12), after (week 24), and at examination six months after (week 52) treatment in patients with and without neuropathic pain six months after treatment. IENFD: Intraepidermal nerve fiber density; SNAP: Amplitude of the sensory nerve action potential; CDT: Cold detection threshold: WDT: Warmth detection threshold; HPT: Heat pain threshold; MDT: Mechanical detection threshold; MPT: Mechanical pain threshold.
RESULTS
One hundred ninety-six patients were screened for inclusion, and 92 refused to participate. Twenty-four patients were not included due to logistic reasons (unable to participate in baseline examination prior to initiation of chemotherapy). Eleven patients were excluded due to previous chemotherapeutic treatment (
Thirty patients had a clinically significant neuropathy and 14 patients had neuropathic pain six months after treatment. Eleven of the 14 patients with neuropathic also had clinically significant neuropathy. Patient characteristics are presented in Table 1.
Patient characteristics
NSS: Neurological Symptom Score; NIS: Neuropathy Impairment Score; INCAT DS: Inflammatory Neuropathy Cause and Treatment disability sum score; INCAT SS: Inflammatory Neuropathy Cause and Treatment sensory sum score; NCS: Nerve conduction studies; NPSI: Neuropathic Pain Symptom Inventory; rTNS: reduced Total Neuropathy Score.
Based on preliminary analysis, including considerations to avoid multicollinearity, VDT, CDT and sural SNAP were included in the logistic regression analysis of clinically significant neuropathy and CDT, HPT and biothesiometer were included in analysis of neuropathic pain.
Clinically significant neuropathy
A significant correlation was found between lower VDT at baseline and clinically significant neuropathy six months after treatment (OR 0.54) (Table 2). There was no correlation with baseline values or changes between baseline and 25% or 50% of planned dose for any of the other variables. Baseline VDT measurements in patients with and without significant neuropathy are presented in Table 3.
Logistic regression analysis for selected measures of nerve function and clinically significant neuropathy and neuropahic pain six months after treatment
Biothesiometer: Vibration detection assessed by biothesiometry; CDT: Cold detection threshold; HPT: Heat pain threshold; OR: Odds ratio (reference: No clinically significant neuropathy or no neuropathic pain); SNAP: Sensory nerve action potential; VDT: Vibration detection threshold.
Measures of nerve function with significant correlation with significant neuropathy or neuropathic pain six months after treatment
CDT: Cold detection threshold; HPT: Heat pain threshold; VDT: Vibration detection threshold.
ROC analysis showed that the maximal product of sensitivity and specificity was obtained at a cut off of 5 (arbitrary scale). Sensitivity of a baseline VDT value of less than 5 for the prediction of clinically significant neuropathy six months after treatment was 76.0% (95% CI 54.9% to 90.6%) and specificity was 53.3% (95% CI 34.3% to 71.7%).
Consistently, baseline VDT in individual patients had a very significant overlap between patients with and without significant neuropathy six months after treatment (Fig. 3).
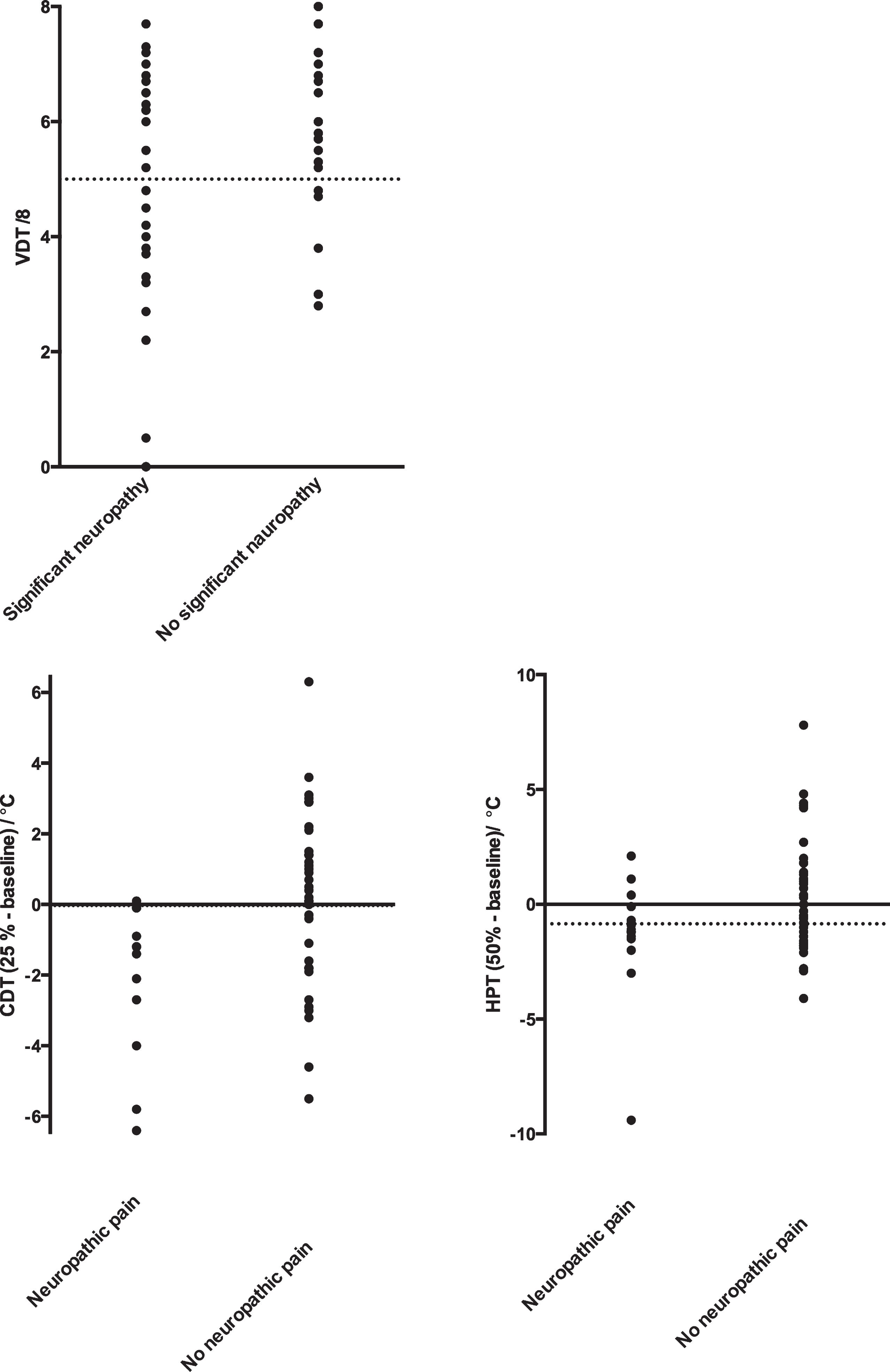
Measures in individual patients of vibration detection threshold (VDT) at baseline and changes in cold detection threshold (CDT) and heat pain threshold (HPT) with and without clinically significant neuropathy and neuropathic pain six months after treatment, respectively. Dotted lines represent cut off values based on ROC analysis.
Neuropathic pain
Ten patients had definite neuropathic pain and four patients had probable neuropathic pain.
Logistic regression analysis (Table 2) showed significant correlations between reduced CDT after 25% of the planned oxaliplatin dose (OR 1.38) and reduced HPT after 50% of the planned dose (OR 1.91) and neuropathic pain. Changes in CDT and HPT in patients with and without neuropathic pain are presented in Table 3.
For change in CDT and the prediction of neuropathic pain, ROC analysis resulted in cut off value of –0.05 °C. Using this cut off value a sensitivity of 92.3% (95% CI 64.0% to 99.8%) and a specificity of 64.9% (95% CI 47.5% to 79.8%) was found.
For change in HPT and the prediction of neuropathic pain, ROC analysis resulted in cut off value of –0.85 °C with a sensitivity of 64.3% (95% CI 35.1% to 87.2%) and a specificity of 70.0% (95% CI 53.5% to 83.4%).
Consistently, changes in CDT and HPT in individual patients revealed a very significant overlap between patients with and without neuropathic pain six months after treatment for both CDT and HPT (Fig. 3).
DISCUSSION
The proportions of patients with clinically significant neuropathy and neuropathic pain of 55% and 25%, respectively, in this study were lower than previously reported [1]. This discrepancy is likely due to CIPN diagnosis in previous studies frequently being based on neuropathy symptoms, as opposed to our study, which also incorporated objective measures of peripheral nerve function in the diagnosis.
The major strength of this study is that multiple tests of peripheral nerve function were performed simultaneously before and during the early phases of treatment resulting in a direct comparison of their predictive values. Previous studies have typically focused on specific tests.
We identified vibration detection threshold before treatment and early changes in cold and heat detection thresholds as predictors of clinically significant neuropathy and neuropathic pain six, moths after treatment, respectively. However, sensitivity of the estimated cut off values was only moderately good except for CDT, and specificity was low. Further, the precision of the estimated sensitivity and specificity was low, and larger studies are needed to confirm the results. One explanation for the moderate prognostic values of these QST measures could be that the method is subjective (psychophysical) and relies on patient cooperation.
In a small study, possibly affected by selection bias due to low participation rate at follow-up, cold allodynia after three cycles of oxaliplatin, which is otherwise considered a sign of acute neurotoxicity, was correlated with severe chronic PN [17]. In this study, we did not examine cold allodynia, but our observation of a reduction in HPT after 25% and 50% of planned dose probably reflects a similar hypersensitivity during the early phases of treatment.
In a study of nineteen patients examined prospectively during one year, including comprehensive QST, higher heat detection threshold at baseline and increased heat and cold detection thresholds before treatment cycle two were correlated with chronic grade 2 or 3 neuropathy [18].
Regarding NCS, studies examining distal sensory nerve segments such as the dorsal sural nerve [5, 19] and the median nerve [20] found correlations between changes from baseline to mid-treatment and clinically severe neuropathy at end of treatment. It is possible that distal sensory segments are better suited for predictive purposes than the sural nerve at the calf, which we examined. In the latter study, however, there was also a correlation between changes in proximal sural nerve studies and severe neuropathy, which we were not able to demonstrate. An increase in superexcitability of more than 15%, which is a measure derived from nerve excitability testing, prior to 5 of 12 chemotherapy cycles was able to identify 80% of patients with moderate or severe chronic neurotoxicity based on neuropathy symptoms after treatment [21]. This change was not found in patients with mild neurotoxicity and preceded development of symptoms. However, nerve excitability testing is a method still restricted to research laboratories and it is not immediately applicable in clinical practice.
We expected to find a correlation between reduced IENFD reflecting small fibre loss and neuropathic pain. IENFD was reduced during oxaliplatin treatment in a prospective study of eight patients [22]. It is possible that regeneration of small nerve fibres during treatment could explain the lack of correlation. Regeneration of peripheral nerves has been assessed through quantification of growth-associated protein 43 [23] in diabetic neuropathy, but not in oxaliplatin induced neuropathy, and this could be the subject of further studies.
A limitation in this study was the arbitrary definition of clinically significant neuropathy based on the rTNS which combines the severity of neuropathy symptoms and signs. This approach has previously been used, [24] but the correlation to patient related outcomes such as quality of life scores, which may be more clinically relevant cannot be determined. Another limitation is the relatively low number of patients. The relatively small fraction of patients with neuropathic pain to some degree limits the interpretation of the results.
In conclusion, we found correlations between measurement of vibration detection thresholds before treatment and significant polyneuropathy and changes thermal detection thresholds during the early phases of treatment and neuropathic pain six months after treatment. There were no correlations with IENFD or NCS variables. However, our data indicate that the potential predictive value of QST measures in clinical practice is limited due to overlap between groups of patients with and without neuropathy at follow-up examination resulting in low to moderate sensitivity and specificity.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
FUNDING
The study was supported financially by the Danish Cancer Society (grant no. R56-A3139) and The Region of Southern Denmark.
