Abstract
Little is known about the feasibility of using long-term stored blood samples to measure neurofilament levels and about long-term changes in neurofilament levels among healthy individuals. We performed a pilot study among 26 adult men in preparation for a larger-scale study of the natural history of neurofilament levels. Median change over 14 years in pNf-H was 97.1 pg/mL (IQR: 5.0 to 242.0 pg/mL) and in Nf-L was 2.117 pg/mL (IQR: –2.691 to 3.393 pg/mL). We demonstrated the feasibility of measuring neurofilament concentrations in stored blood samples and found a trend between age and increases in Nf-L levels among adults.
Keywords
BACKGROUND
Phosphorylated neurofilament heavy (pNf-H) and neurofilament light (Nf-L) concentrations represent promising biomarkers linked with neurodegenerative and neuromuscular disorders [1–3]. However, little is known about the natural history of neurofilament levels. Studies among healthy adults have reported age-dependent increases in neurofilament levels [4–6] but drivers of these changes are not fully understood. As studies in adult populations consider changes in neurofilament levels to measure disease progression or treatment responses, benchmarks for normal rates of change will be needed. Additionally, there is a need to understand whether lifestyle factors, health conditions, and biomarkers may influence the rate of change of neurofilament levels over time.
Determining the natural history of neurofilament levels will require leveraging large, longitudinal studies with repeated blood collections and detailed neurologic disease histories. One study which can contribute to our understanding of the natural history of neurofilament changes over time is the Physicians’ Health Study (PHS), a large-scale, long-term cohort of middle-aged and older men with two blood samples collected approximately 14 years apart and more than a decade of subsequent follow-up [7, 8]. PHS represents a unique dataset for examining the natural history of pNf-H and Nf-L among men without a history of neurodegenerative or neuromuscular diseases. However, little research has evaluated the feasibility of using long-term stored blood samples to measure neurofilament levels and long-term 14-year changes in neurofilament levels among healthy individuals. Therefore, we undertook a pilot study in preparation for a larger-scale study of the natural history of pNf-H and Nf-L.
For this pilot study, we measured described changes in pNf-H and Nf-L concentrations among 50 PHS participants aged ≥40 years at baseline with no history of major neurologic or neuromuscular diseases throughout follow-up. Using blinded split samples, we examined the reliability of pNf-H and NF-L measurements from long-term stored blood samples.
METHODS
The PHS consists of two long-term trials of 29,071 men aged ≥40 years. Detailed descriptions of both trials have been published [7, 8]. PHS participants completed annual questionnaires from study enrollment in 1982 through 2012 including assessment of various neurological disorders. In addition, 22,363 PHS participants provided a blood sample in 1982 or 1996, including 9,520 PHS participants who provided blood samples at both timepoints. Samples were immediately centrifuged and separated for plasma by the participants and sent with chill packs by overnight courier. Samples were stored at –82C initially and are now stored at –130C.
To be eligible for inclusion in this pilot study, participants must have provided blood samples at both timepoints and have no self-reported history of stroke, transient ischemic attack, Parkinson’s disease, multiple sclerosis, Alzheimer’s disease, dementia, amyotrophic lateral sclerosis, or spinal muscular atrophy throughout follow-up. Due to the strong correlations between age and neurofilament concentrations in prior studies, we decided a priori to randomly select 26 men aged 40–49 years, 12 men aged 50–59 years, and 12 men aged ≥60 years at baseline in 1982 to allow us to explore changes in neurofilament levels by age group.
Blood Nf-L concentrations were determined using the ultra-sensitive single molecule array (Simoa) Quanterix platform with NF-Light® (SR-X) kit (Qua-nterix 103400). Briefly, blood samples were diluted and loaded in duplicate into wells with detector and NF-L antibody-coated magnetic beads. Average number of enzymes per bead (AEB) from fraction of positive beads (Fon) was calculated. Protein concentration was calculated based on digital determination when Fon <0.7 and by analog determination when Fon >0.7. Controls were provided by the kit. Standard curves were generated for each run and fitted concentration of samples were calculated. Samples with an out of range AEB reading were excluded.
Blood pNf-H concentrations were measured by enzyme-linked lectin assay from ProteinSimple® as previously described [9]. Briefly, cartridge wells were loaded with samples and run through a microfluidic channel containing three glass nanoreactors coated with a capture antibody against pNf-H. A detection reagent that binds to pNf-H was added to generate one relative fluorescence unit signal per glass nanoreactor. The relative fluorescence unit is used to interpolate the concentration from a calibrated standard curve (5-PL fit with 1/y2 weighting) and reported in pg/mL.
We summarized medians and interquartile ranges for pNf-H and Nf-L concentrations at baseline and 14-year follow-up as well as changes in levels be-tween the two timepoints overall and by age group. We used Spearman’s correlation to determine correlations between pNf-H and Nf-L concentrations at each timepoint, between changes in pNf-H and Nf-L, and between pNf-H or Nf-L levels and baseline age. Finally, using blinded split samples from 30 participants, we assessed assay reproducibility by calculating coefficients of variation for pNf-H and Nf-L. This study was approved by the Institutional Review Board of Partners Healthcare/Brigham and Women’s Hospital. Data supporting our findings are available from the corresponding author upon reasonable request.
RESULTS
At baseline, median age of study participants was 48 years and median body mass index was 23.8 kg/m2. As expected, given our selection criteria, most men had favorable lifestyle characteristics with 62% being never smokers and 60% reporting at least weekly exercise. Very few men reported a history of hypertension (N = 4), diabetes (N = 1), or high cholesterol (N = 5).
Table 1 shows the median and interquartile ranges of pNf-H at each timepoint and for change in pNf-H for the overall study and for each age group. Overall, the median level of pNf-H increased from 205.5 pg/mL to 301.0 pg/mL from baseline to 14-years follow-up. The median 14-year change in pNf-H was 97.1 pg/mL, with a Spearman’s correlation of 0.67 (p < 0.01) between baseline and 14 years. We did not observe a strong trend between age and changes in pNf-H (Pearson’s correlation coefficient = 0.06) (Table 1 and Fig. 1).
Summary statistics for phosphorylated neurofilament heavy and neurofilament light levels overall and by age category at study baseline for each timepoint and the change between timepoints
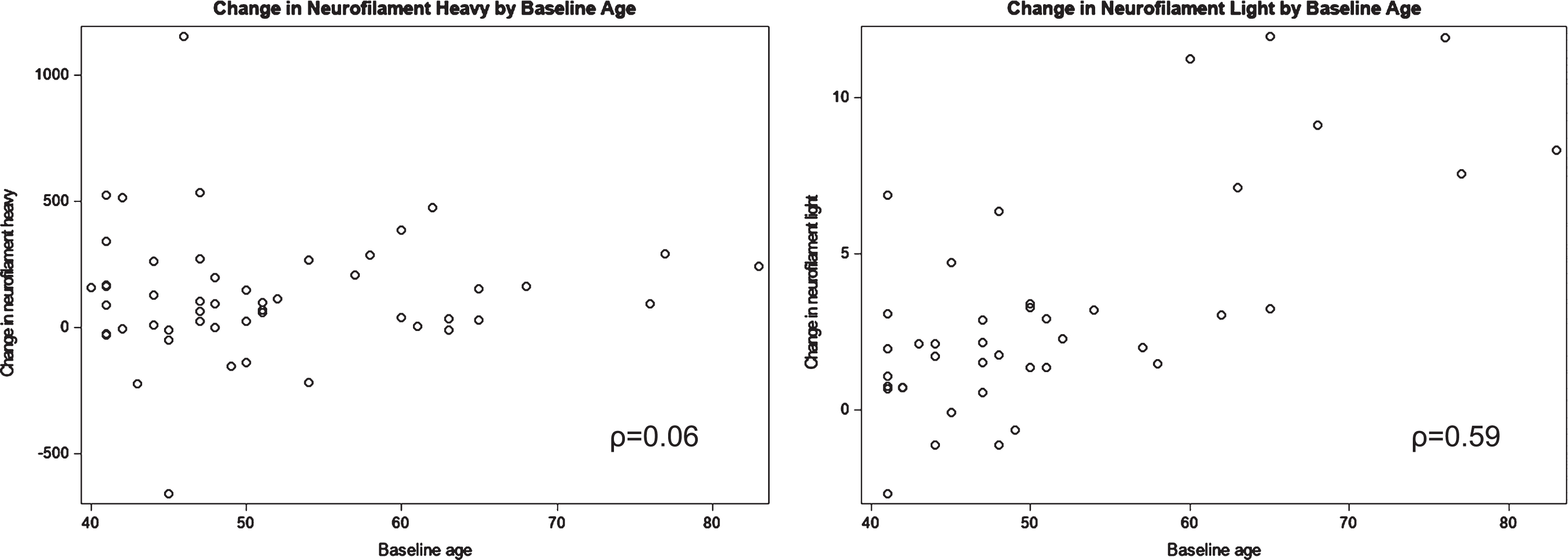
Scatterplot of changes in neurofilament heavy and light levels by age at study baseline. ρ= Spearman’s correlation coefficient.
For Nf-L, overall, the median level of Nf-L in-creased from 7.80 pg/mL to 10.83 pg/mL from baseline to 14-years follow-up (Table 1). We observed a trend of increasing Nf-L levels with increasing age category (Table 1).
The median change in Nf-L over 14 years was 2.12 pg/mL, with a Spearman’s correlation of 0.86 (p < 0.01) between baseline and 14 years. The largest 14-year changes in Nf-L concentrations were observed among men aged ≥60 years at baseline (Table 1) with a trend of larger changes in Nf-L levels with increasing age (Spearman’s correlation coefficient = 0.59) (Fig. 1).
We observed weak, non-significant correlations between pNf-H and Nf-L at both baseline (Spearman’s r = 0.28, p-value = 0.07) and 14-year follow-up (Spearman’s r = 0.21, p-value = 0.16). We observed no evidence of a correlation between changes in pNf-H and changes in Nf-L (Spearman’s r = –0.03, p-value = 0.86).
Among the 30 participants with blinded split samples, the overall coefficients of variation were 13.2 for pNf-H and 7.02 for Nf-L, indicating adequate assay reproducibility [10].
DISCUSSION
In this pilot study, we demonstrated that it was feasible to measure pNf-H and Nf-L concentrations in blood samples stored long-term with adequate assay reproducibility. Although all men included in this pilot study remained free of major neurologic and neuromuscular diseases during follow-up, most men experienced increases in both pNf-H and Nf-L levels.
As expected, we observed the largest changes in Nf-L levels among individuals aged ≥60 years. In a prior study, levels of Nf-L increased significantly over time even among individuals who were cognitively unimpaired [5]. Similarly, a nested case-control study demonstrated a linear increase in plasma Nf-L over time for dementia-free controls [6]. However, both studies enrolled older individuals and it is unknown to what extent these results generalize to younger adults. One small study among 87 healthy individuals aged 18 to 70 years without multiple sclerosis demonstrated an increase in plasma Nf-L over one year [4]. Our results expand upon these findings by examining a longer 14-year period of change.
In contrast to our finding for NF-L, we found no evidence of a trend between changes in pNf-H levels and age. A recent study observed increases in pNf-H levels over time among participants who later developed ALS, but observed relatively stable levels among 14 healthy controls (mean age 48.1 years) during a median of 1.5 years of follow-up [11].
Although pNf-H and Nf-L are both released during axonal injury [2], we did not observe correlations between pNf-H and Nf-L in either cross-sectional or longitudinal analyses. Larger studies are needed to further explore the interrelationship between pNf-H and Nf-L and whether age, long-term sample stability, and other factors may modify the association between the biomarkers.
Strengths to this study include the ability to examine long-term changes in neurofilament levels among men who remained free of major neurodegenerative or neuromuscular diseases throughout follow-up. Since the PHS only enrolled men aged ≥40 years, we were unable to explore neurofilament levels among women or younger individuals. Given the small size of this study, we were unable to explore risk factors for changes in neurofilament levels. Finally, we were unable to explore shorter-term changes in neurofilament levels to understand how neurofilament levels change across the lifespan.
Future research is needed to understand factors which predict increases in pNf-H and Nf-L concentrations, and to understand how the natural history of pNf-H and Nf-L affects future risk of neurodegenerative and neuromuscular disorders. These studies should be performed in diverse samples of men and women and assess both short- and long-term changes in neurofilament levels.
FUNDING
This study was supported by a grant from Biogen, and grants CA 097193, CA 34944, CA 40360, HL 26490, and HL 34595 from the National Institutes of Health. Dr. Rist is supported by funding from HL 128791.
COMPETING INTERESTS
VK, MP, JS, WF: Employees of and hold stock/stock options in Biogen.
PMR and HDS: Received an investigator-initiated grant from Biogen.
