Abstract
Background:
Prednisone is a common treatment for myasthenia gravis (MG), and osteoporosis is a known potential risk of chronic prednisone therapy.
Objective:
Our aim was to evaluate the risk of serious fractures in a population-based cohort of MG patients.
Methods:
An inception cohort of patients with MG was identified from administrative health data in Ontario, Canada between April 1, 2002 and December 31, 2015. For each MG patient, we matched 4 general population comparators based on age, sex, and region of residence. Fractures were identified through emergency department and hospitalization data. Crude overall rates and sex-specific rates of fractures were calculated for the MG and comparator groups, as well as rates of specific fractures. Adjusted hazard ratios (HRs) and 95% confidence intervals (CIs) were estimated using Cox regression.
Results:
Among 3,823 incident MG patients (followed for a mean of 5 years), 188 (4.9%) experienced a fracture compared with 741 (4.8%) fractures amongst 15,292 matched comparators. Crude fracture rates were not different between the MG cohort and matched comparators (8.71 vs. 7.98 per 1000 patient years), overall and in men and women separately. After controlling for multiple covariates, MG patients had a significantly lower risk of fracture than comparators (HR 0.74, 95% CI 0.63–0.88).
Conclusions:
In this large, population-based cohort of incident MG patients, MG patients were at lower risk of a major fracture than comparators. The reasons for this finding are unclear but may highlight the importance osteoporosis prevention.
INTRODUCTION
Myasthenia gravis (MG) is an autoimmune disor-der of the neuromuscular junction that often requires long-term immunotherapy. Corticosteroids are a mai-nstay of treatment in MG (after symptomatic therapy with acetylcholinesterase inhibitors), however osteoporosis is a recognized adverse effect of chronic corticosteroid therapy [1–3]. A significant proportion of MG patients are elderly [4] with accumulating comorbidities, which may act synergistically with neu-romuscular weakness and fatigue to increase the risk of falling [5]. It would therefore be reasonable to hypothesize that the risk of serious fractures would be higher in MG patients than in the general population.
Despite this expectation, prior studies have shown inconsistent results [6–8]. However, the number of studies that have examined the risk of fractures in neuromuscular disease and specifically in MG is small, despite the fact that osteoporotic fractures are associated with lower quality of life in MG [9]. No specific guidelines exist for managing or preventing osteoporosis in MG, and clinicians utilize guidelines from other medical fields, such as rheumatology [10].
We have previously reported the epidemiology of myasthenia gravis in Ontario, Canada based on a large population-based cohort established through analysis of provincial administrative health databases, [11] as well as the risk of serious infections in this population [12]. In the current study we aimed to determine the risk of serious fractures in this large inception cohort, and to compare the rates of fractures relative to matched general population comparators.
MATERIALS AND METHODS
Study setting
We conducted a population-based cohort study in Ontario, Canada’s most populous province, using linked, administrative health data covering the period between April 1, 2002 and December 31, 2016. On-tario residents have universal public health insurance under the Ontario Health Insurance Plan (OHIP), the single payer for medically necessary services [13].
Data sources
Population-level health administration databases capture details of the health care of Ontarians. The-se databases are linked to allow the creation of patient cohorts and long-term follow-up. We used the Canadian Institute for Health Information (CIHI) Discharge Abstract Database to obtain hospitalization data. The CIHI National Ambulatory Care Reporting System (NACRS) was used to collect eme-rgency department (ED) visits. In both databases, coding is based on International Classification of Dis-ease-9 (ICD-9; until 2002) and ICD-10 diagnosis codes thereafter. The Ontario Health Insurance Plan (OHIP) Claims database was used to ascertain diagnoses and billing codes associated with physician services. These datasets also were used to identify comorbidities using validated case definitions. The Registered Persons Database was used to obtain inf-ormation about each patient’s health insurance status, age, sex, vital status, and location of residence. The Ontario Drug Benefit (ODB) database (which captures prescription claims among seniors and social assistance recipients) was used for MG case ascertainment (specifically pyridostigmine prescriptions). These datasets were linked using unique encoded identifiers and analyzed at ICES (www.ices.on.ca).
Use of data in this project was authorized under section 45 of Ontario’s Personal Health Information Protection Act, which does not require review by a Research Ethics Board. The datasets used in this study are held securely in coded form at ICES. While data sharing agreements prohibit ICES from making the datasets publicly available, access may be granted to those who meet pre-specified criteria for confidential access, available at www.ices.on.ca/DAS.
Study patients
Patients with MG were identified using a previ-ously-validated administrative data case definition with a sensitivity of 81.6%, specificity of 100%, and positive predictive value of 80.0% [11]. Patients were classified as having MG if they met any of the following criteria: (1) an inpatient hospitalization listing MG as primary or secondary diagnosis (ICD-9 358.0, 358.00 or 358.01; ICD-10 G70.0, G70.1 or G70.2); (2)≥5 outpatient visits associated with an MG OHIP diagnostic code (diagnosis code ICD-9 358), and 1 relevant diagnostic test (single-fibre electromyography as G458 or Tensilon testing as G419) within 1 year; or (3)≥3 pyridostigmine prescriptions within 1 year. Patients meeting the above criteria between April 1, 2002 and December 31, 2015 were included in our cohort, with the date on which the minimum criteria were met serving as the cohort entry date (index date). Patients were excluded if they had missing data on age, sex, or region of residence, were <18 or >105 years of age or resided in a long-term care residence. For each MG patient, we matched 4 gene-ral population comparators from the same administrative databases based on age (+/- 1 year), sex, and region of residence. The comparators did not have a diagnosis of myasthenia gravis, but otherwise were not selected for any other diagnoses. Comparators also required at least one instance of contact with the health care system within the last 5 years, to ensure that they were engaged users of the healthcare system. Index dates for comparators were randomly assigned, based upon the distribution of those among patients with MG.
Outcomes and follow-up
Patients were followed from cohort entry until their first serious fracture, loss of OHIP coverage, no contact with the healthcare system for at least 1 year (as a proxy for unrecorded death or outmigration), death, or the end of the study period (December 31, 2016). Serious fractures of the hip, femur, forearm, humerus, and pelvis were defined as an emergency department visit or hospitalization with an admitting diagnosis or ‘most responsible’ (primary) diagnosis of fracture, per Garg et al. (2012)’s algorithm using ICD-9 and ICD-10 diagnosis codes (CIHI-DAD and CIHI-NACRS), procedure/intervention codes (CIHI-DAD and CIHI-NACRS), and OHIP fee codes (see Supplementary Table S1 for the diagnosis and procedure codes) [14]. Fracture diagnoses accompanied by tra-uma codes were not counted as outcome events, per Garg et al. (2012)’s algorithm. In addition to a diagnosis of “any fracture”, we examined whether patients experienced fractures in the following sites: hip, femur, pelvis, forearm, and humerus.
Covariates
To describe our cohort, we captured age, sex, ru-ral status, [15] neighbourhood income quintile, and baseline comorbidities including chronic obstructive pulmonary disease (COPD), [16] asthma, [17] diabetes, [18] hypertension, [19] cancer, osteoporosis, chronic kidney disease, prior infection, history of fra-ctures (using a two-year lookback from index date, for both measures), and number of hospital admissions during the 12 months prior to cohort entry. Frailty status and comorbidity burden were assessed using the Johns Hopkins ACG® System Version 10.0. Individuals were assigned up to 32 aggregate diagnostic groups (ADGs) characterizing different medical conditions based on their healthcare use during the 2 years prior to MG diagnosis, whereby a higher number of ADGs indicates greater comorbidity burden. We adjusted for these covariates in univariate and multivariate Cox proportional hazards models.
Statistical analysis
Standardized differences were used to compare baseline characteristics of MG patients with matched comparators. Differences <0.1 were not considered to be meaningful. Crude overall and sex-specific rates of fractures were calculated for the MG patients and comparators. Time to first fracture was estimated for both groups and plotted using a Kaplan-Meier survi-val curve. Hazard ratios for serious fractures were est-imated using multivariable Cox proportional hazards regression, adjusting for the baseline character-istics listed above. All statistical analyses were performed using SAS statistical software (version 9.4).
RESULTS
A total of 3,823 MG patients were identified and matched to 15,292 general population comparators. Table 1 shows a summary of the baseline characteristics for both groups. Patients with MG had a mean (SD) age of 63.8 (16.6) years at cohort entry, 47% were female, lived in urban centers (92.4%) and fairly equally resided across neighbourhood income quintiles. MG patients had slightly higher rates of comorbidities than comparators (including asthma, hypertension, and cancer), as well as a higher ADG comorbidity index, and infection prior to index. MG patients were more likely to have either no hospitalizations or 1–2 hospitalizations in the 1 year prior to the index date.
Baseline characteristics of MG patients and general population comparator group
Abbreviations: MG = myasthenia gravis; COPD = chronic obstructive pulmonary disease; ADG = aggregate diagnosis groups. *Standardized differences less than 0.1 are not considered meaningful. #Meaningful standardized differences. **Infections occurring 2 years prior to index date. ***Fractures occurring 2 years prior to index date.
Over the study period, subjects were followed up for a mean (SD) of 6.0 (3.9) years, 5.7 (3.9) years for MG, and 6.1 (3.9) years for comparators. During follow-up, 188 (4.9%) MG patients and 741 (4.8%) comparators experienced a serious fracture, for event rates of 8.71 events (95% CI 7.51–10.04) vs 7.98 events (95% CI: 7.41–8.57) per 1,000 patient-years, respectively. Sex-specific rates did not differ between MG patients and comparators (males: 5.25 (95% CI: 4.01–6.74) vs. 4.9 (4.31–5.56), and females: 12.74 (10.62–15.16) vs. 11.46 (10.47–12.51). We observed a significantly higher fracture rate in females among both the MG and comparator groups. Table 2 breaks down frequency of first fracture by site, and illustrates that none of the specific fracture sites were more common in MG patients. Figure 1 shows the Kaplan-Meier survival curves for time to first fracture among MG patients versus their matched comparators. In this unadjusted comparison, the curves do not differ, based upon the log-rank test (p = 0.26).
Frequency of fractures according to location and study group
Abbreviations: MG = myasthenia gravis. S = Suppressed. Cells of size 1–5 are suppressed to protect patient privacy.
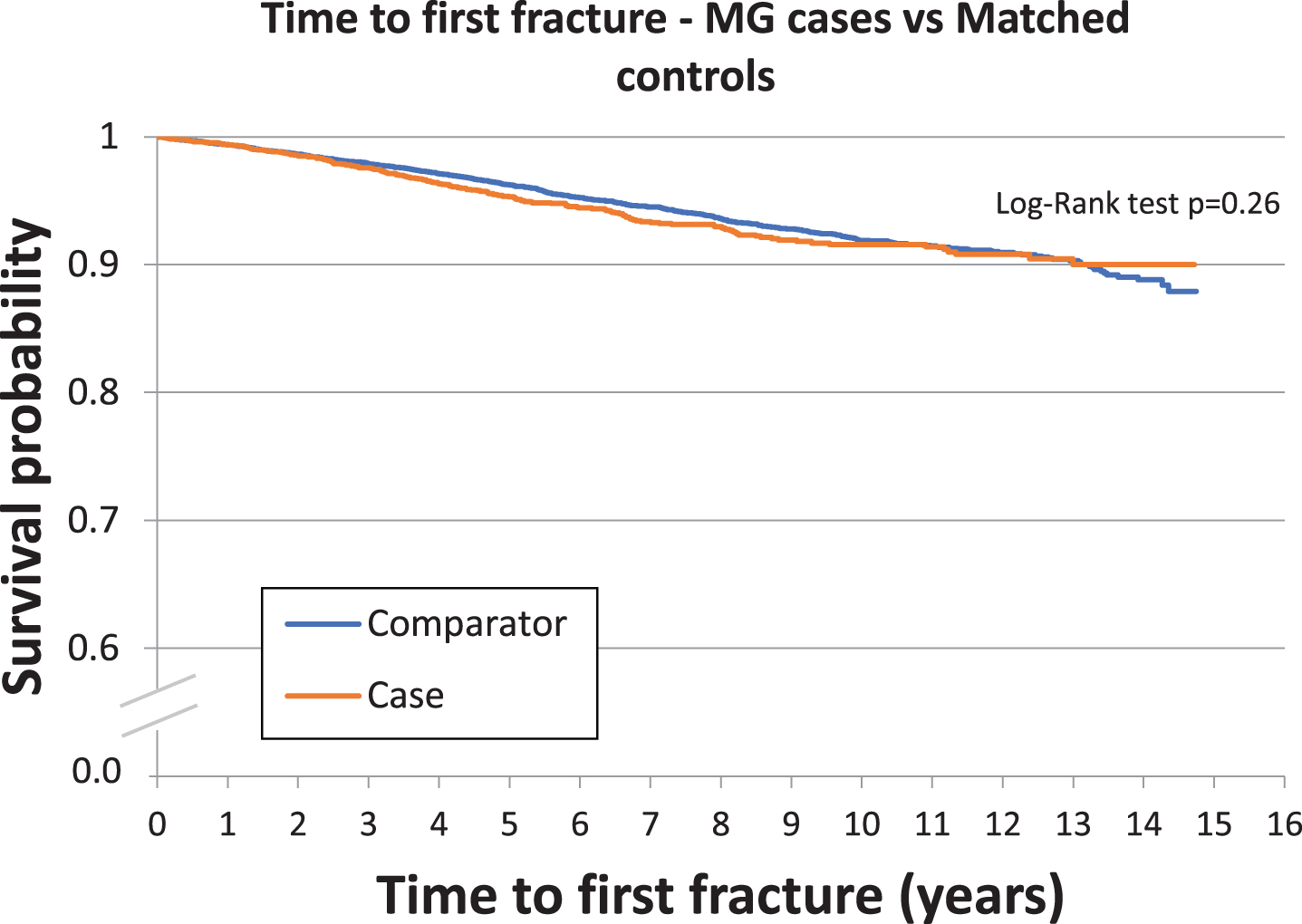
Kaplan-Meier curves showing time to first fracture for patients with MG versus comparators.
COPD, asthma, diabetes, hypertension, cancer, osteoporosis, chronic kidney disease, prior infection, prior fracture, frailty, number of comorbidities, and number of hospitalizations during the year prior to the index date were associated with fracture risk in univariate analyses. After simultaneous adjustment for all the measured baseline characteristics, MG patients showed a lower risk of serious fractures relative to comparators, with an adjusted hazard ratio of 0.74 (95% CI 0.63–0.88). Factors independently associated with greater likelihood of fracture were COPD, hypertension, osteoporosis, prior fracture, living in a rural area, presence of the ACG frailty marker, and greater ADG count (Table 3).
Association of Baseline characteristics with risk of serious fracture
Abbreviations: MG = myasthenia gravis; Q = quintile; COPD = chronic obstructive pulmonary disease; ADG = aggregate diagnosis groups. *Patients were followed from cohort entry until their first serious fracture, loss of OHIP coverage, death, or the end of the study period (December 31, 2016). **Indicates variables that were statistically significant in the multivariate analysis. ***Q5 represents the highest income quintile, Q1 represents the lowest.
DISCUSSION
This large, population-based cohort study in Onta-rio, Canada showed a significantly lower risk of se-rious fractures among 3,823 incident MG patients relative to 15,292 matched general population comparators. Our findings of a lower fracture risk are somewhat surprising (given the presence of neuromuscular weakness in MG patients, as well as pre-dnisone use) and are different than a prior study that found no increase in overall fracture rate or specific fracture types, in MG [6]. In that study, recent use of corticosteroids was not associated with higher fracture rate, but use of antidepressants, anxiolytics and anticonvulsants did show an association with fracture risk [6]. Another study reported that the rate of abnormal bone density or osteoporosis was not elevated in MG patients receiving corticosteroid therapy compa-red to what was expected in the general population [7]. By contrast, a large population-based cohort stu-dy in Taiwan found that MG patients had a higher incidence of osteoporosis versus matched comparators [8]. A potential explanation is the mean age in the Taiwanese study was 41.81 and 41.65 years for MG and comparators, respectively, considerably younger than in our study. It is possible that the higher baseline risk of osteoporosis and fractures in an older population may have diluted an increased risk in younger MG patients.
It is well-established that chronic corticosteroid therapy is associated with osteoporosis and osteopor-otic fractures,(2,3,20), including one study demonstrating an association with corticosteroid therapy duration and osteoporotic fractures in MG [9]. The seemingly contradictory finding that MG patients do not have a higher rate of fractures than matched comparators may suggest several hypotheses. First, a diagnosis of myasthenia, and subsequent prednisone use, may alert physicians to higher osteoporosis risk. This in turn may lead to more intensive counse-ling and investigations, (e.g. prophylactic bone mineral density studies). Physicians may be more likely to follow treatment guidelines and recommend supplemental calcium, vitamin D, or a bisphosphonate medication, whereas age-matched individuals may not be managed as aggressively. However, in some other autoimmune diseases (such as rheumatoid arthr-itis) there appears to be higher rates of fractures compared to the general population, [21–23] and one would expect that bone health would be managed as equally aggressively.
Another potential explanation may relate to nature of the disease; patients with neuromuscular weakness or reduced mobility may be less mobile, more cautious, or more likely to using walking aids, thus reducing the risk of falls and fractures. In addition, some of the fracture risk seen in rheumatologic disease could relate to concomitant medication use that are reported to increase fracture risk but less common in myasthenia gravis, such as opioid medications for pain [20]. MG patients are often treated with pyr-idostigmine, and some studies have demonstrated a potential anabolic effect on bone of pyridostigmine, potentially through the stimulating effect of acetylcholine on osteoblasts. In theory, this could count-eract the effects of corticosteroid on bone health [24]. Finally, our study only followed patients for a mean of 6 years, and so it is possible that beyond 6 years, there could be an increase in fracture risk.
At present, there are no specific guidelines for the management of osteoporotic risk in neuromuscular patients treated with corticosteroids, with most cli-nicians adhering to the American rheumatology guidelines [25, 26]. The most recent guidelines rec-ommend that adults on at least 2.5 mg of prednisone for ≥3 months should have vitamin D intake of 600–800 IU/day, optimized calcium intake (1000–1200 mg/day from all sources, including dietary), and consider lifestyle modifications (such as smoking cessation, and regular exercise) [26]. Additional treatment depends on the patient’s individual fracture risk and age. Patients 40 years or older and with medium or high risk of fracture are recommended to start a bisphosphonate medication, in addition to the measures above [26]. Bone mineral density should be performed within 6 months of initiation of glucocorticoid therapy in those older than 40 [26]. Since we do not know the treatment regimens for bone health or glucocorticoid use amongst the MG patients included in our study, we cannot comment on the appropriateness of these guidelines specifically for MG patients.
Our study has several limitations. The most significant limitation is availability of prescription drug data, which is limited to those covered by the ODB plan (seniors and social assistance recipients), making it difficult to properly study the effects of specific medications on the risk of fractures, or to examine whether MG patients were more likely to be on a bisphosphonate than comparators. Specifically, we do not know how many patients were treated with prednisone and at what doses. Another limitation relates to our definition of serious fractures, which we classified as those requiring emergency department visits or hospitalizations. This method may underestimate the true number of fractures (particularly milder ones). Finally, although we adjusted for multiple possible confounders, it is possible that unknown or unmeasured confounders may have influenced the association with fracture risk, such as baseline activity level or use of mobility aids. We also note that the MG cohort had a higher rate of several comorbidities versus the general population comparators; one possible explanation may relate to MG patients being investigated for comorbidities due to more frequent contact with the health care system.
In conclusion, our study draws on a very large, pro-vincial inception cohort of MG patients, to demonstrate that MG patients did not have an increased serious fracture risk when compared to matched comparators, and in fact MG patients had a lower risk of fractures. While we have proposed a few possi-ble explanations, further investigation is required. Such work will be helpful in attempting to inform bone health management guidelines, specifically for neuromuscular diseases.
CONFLICTS OF INTEREST
Drs. Widdifield, Paterson, Kopp, Barnett, Tu, and Breiner report no disclosures. Alexander Kopp and Chenthila Nagamuthu report no disclosures. Dr. Kassardjian has received speaker honoraria from Alexion and has served on advisory boards for Alexion, Akcea, Takeda, and Sanofi Genzyme.
Footnotes
ACKNOWLEDGMENTS
This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). This study also received funding from the Division of Neurology New Initiatives Fund. Parts of this material are based on data and information compiled and provided by the Canadian Institute of Health Information (CIHI) and Cancer Care Ontario (CCO). We thank IQVIA Solutions Canada Inc. for use of their Drug Information Database. The analyses, conclusions, opinions and statements expressed herein are solely those of the authors and do not necessarily reflect those of data, or the funding sources; no endorsement is intended or should be inferred. J. Widdifield is supported by a career award from the Arthritis Society. K. Tu receives a Research Scholar Award from the Department of Family and Community Medicine at the University of Toronto.
