Abstract
Background
Spinal muscular atrophy (SMA) is a motor neuron disease associated with progressive muscle weakness and motor disability.
Objective
This study aims to report the evaluation of nusinersen, an antisense oligonucleotide, on motor function in patients with SMA types 2 and 3.
Methods
This single-center retrospective observational study assessed nusinersen therapy outcomes, measured by HSMFSE or CHOP-INTEND scales, in patients with SMA types 2 and 3, compared to untreated patients, for at least 24 months.
Results
A total of 41 patients with SMA types 2 and 3 under nusinersen treatment were included. In 30 treated patients (mean age: 10.6 years; 14 with SMA type 2), the mean change in HFMSE scores was +1.47 points (SD = 0.4) and +1.60 points (SD = 0.6) after 12 and 24 months of treatment, respectively. In contrast, the control group (N = 37) (mean age: 10.2 years; 20 with SMA type 2) presented a mean change of −1.71 points (SD = 0.02) and −3.93 points (SD = 0.55) after 12 and 24 months of follow-up, respectively. The most severe patients under nusinersen treatment (N = 11) showed a change of +2.37 (SD = 1.13) on the CHOP-INTEND scale after 12 months of follow-up. Disease duration at the beginning of treatment was the main predictor of functional improvement. Despite functional gain and motor stabilization, treatment with nusinersen did not prevent the progression of scoliosis.
Conclusions
Our data provide evidence for the long-term safety and efficacy of nusinersen use in the treatment of later-onset SMA, and patients with shorter disease duration showed better response to treatment.
INTRODUCTION
Spinal muscular atrophy (SMA) is a neurodegenerative disease of the lower motor neurons of the spinal cord and motor nuclei of the brainstem. It leads to axial and proximal limb weakness and is associated with progressive motor function disability, ventilatory insufficiency and spinal deformity. The most common form of SMA is caused by deletions or disease-causing variants in the survival motor neuron 1 (SMN1) gene, which segregates as an autosomal recessive trait [1]. Approximately 1 in 11,000 people are affected by the disorder [2].
SMA has been classified into at least four subtypes depending on the patient’s age at the disease’s onset and the achievement of motor milestones [3, 4]. In SMA type 1, or infantile form, children never sit, even with support, and they present the disease at 0 to 6 months. In SMA type 2, clinical manifestations start between 6 and 18 months, and children are unable to walk unassisted. Children with SMA type 3 manifest the disease in the second year of life or later and are able to walk unaided. Finally, SMA type 4 is the adult form of the disease. SMA types 2, 3 and 4 are further categorized as later-onset forms [5, 6].
SMN2 is a centromeric copy of the SMN1 gene that does not provide the transcription of stable SMN protein due to the lack of exon 7 in the majority of transcripts [1]. A strong correlation between the number of SMN2 copies and SMA severity has been demonstrated [7]. Nusinersen is an antisense oligonucleotide that targets the pre-RNA of SMN2, therefore avoiding exon 7 skipping and increasing full-length SMN protein levels. Its effectiveness has been demonstrated in two pivotal trials: one for patients with SMA type 1 [8], and the other for those with SMA types 2 and 3 [9]. The US Food and Drug Administration (FDA) first approved nusinersen for the treatment of SMA in December 2016. The Brazilian National Health Surveillance Agency (ANVISA) approved its use in 2017 [10].
The CHERISH study concluded that children with later-onset SMA who received nusinersen had significant and clinically meaningful improvements in motor function compared with the control group [9]. Subsequent real-life studies confirmed the beneficial effects of nusinersen on the motor function of patients with SMA types 2 and 3 [11–17]. On the other hand, significant objective functional gain has not been demonstrated in some treated adult patients, in addition to a high rate of complications in this group [18]. In this study, we aimed to assess the safety and efficacy of nusinersen in a cohort of patients with SMA types 2 and 3 in comparison with non-treated control patients at a single Brazilian reference center.
PATIENTS AND METHODS
This was a retrospective and observational study of a single reference center in Brazil (Neuromuscular Clinic at Hospital das Clínicas, FMUSP). Patients with genetically confirmed SMA types 2 and 3 (homozygous deletion of exon 7 or with compound heterozygous mutations on the SMN1 gene) [19], and who had access to nusinersen were included. Exclusion criteria were not established. Patients with SMA types 2 and 3 followed at the same center and during the same period (at least 2 years of follow-up) who did not have access to nursinersen treatment were included in this study as a control group. The study was approved by the ethics committee of Hospital das Clínicas (FMUSP) (CAPPesq # 87773118.7.0000.0068), and written informed consent was obtained from the patients or from those responsible for the patients.
Clinical and functional evaluations
Patients who were evaluated every 6 months using the Hammersmith Motor Functional Scale Expanded (HMFSE) comprised Group 1. The scale includes 33 items, and possible scores range from 0 to 66 points; it has been validated to evaluate the progression of SMA types 2 and 3 [20]. Another group of patients (Group 2) were evaluated using the Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders (CHOP-INTEND) scale adapted to older patients and adults with severe weakness who could no longer sit. Items 5, 11, 15 and 16 of the CHOP-INTEND were modified due to the weight, size, and age of the patients in this group.
One neurologist trained in neuromuscular diseases performed all clinical evaluations (RHM), and a single physiotherapy team experienced in the use of motor function scales scored the HMFSE and CHOP-INTEND for all patients. Patients older than 18 years were also evaluated using Egen Klassification (EK) functional scale, which assesses functional daily activities in patients with SMA and Duchene muscular dystrophy [21]. For statistical analysis purposes, we used data from baseline, as well as 12 and 24 months of evaluation.
The number of hospitalizations during the follow-up due to pulmonary complications, the degree of scoliosis and history of previous spine surgery were registered at baseline in both groups. In addition, in the group of treated patients with Cobb angle assessments, scoliosis progression was analyzed at baseline and after 18 or 24 months of treatment.
Procedures
On days 1, 14, 28 and 63, all patients received 12 mg of nusinersen, administered intrathecally, with repeated maintenance injections every 4 months in accordance with the label instructions. Intrathecal injections were performed by a trained neurologist, and patients were observed for 4 to 6 hours after each procedure due to the possible occurrence of adverse events. In patients with severe scoliosis or previous spine surgery, intrathecal punctures were performed with the aid of imaging techniques such as fluoroscopy or CT scan [22].
Statistical analysis
For descriptive purposes, categorical variables were presented through relative and absolute frequencies and compared using the chi-squared test or Fisher’s exact test, as applicable. Continuous variable distributions were assessed for normality using skewness and kurtosis statistics and graphical methods. Those with normal distribution were presented as means and standard deviations and compared using an independent sample Student’s t-test. Otherwise, they were presented as medians and quartiles and compared using the Mann–Whitney nonparametric test. There were no missing data for the baseline characteristics. One patient in the control group had a missing HFMSE score at 12 months, which was imputed using the last observation carried forward method by considering the score at 6 months. Among the 24 patients from the treatment group who did not have 24 months of follow-up, scores were imputed for 11 using the same method by considering the score at 18 months.
Repeated measures general linear models were employed for the analysis of the HFMSE evolution from baseline to one and two years after the start of treatment. When the sphericity assumption was violated, the Greenhouse-Geisser or Huynd-Feldt correction was applied. Other assumptions were verified and were not violated. All tests were two-tailed, and final p-values of less than 0.05 were considered significant. The analyses were conducted using the Statistical Package for Social Sciences software (IBM SPSS Statistics for Windows, version 24.0. Armonk, NY: IBM Corp.).
RESULTS
From July 2017 to December 2019, 94 patients diagnosed with SMA types 2 and 3 were evaluated; 41 patients gained access to nusinersen treatment through health insurance (n = 18) or the Brazilian public health system (SUS) (n = 23). There was no specific patient selection for treatment based on clinical status or according to the presence of spinal deformity. Among these patients, 30 were evaluated using the HFMSE at least on baseline and at 12 and 24 months of follow-up. The other 11 patients under treatment were evaluated using the adapted CHOP-INTEND scale. Twenty-two patients (53.6%) under nusinersen treatment used noninvasive nightly ventilation. All the patients underwent annual lung function assessments as a part of standard care at the center.
Among the 53 patients who did not have access to treatment, 37 had at least three HFMSE measurements (baseline, 12 and 24 months) and were used as control group for treated patients evaluated using the HFMSE. Table 1 shows the baseline characteristics of the patients treated with nusinersen and evaluated using the HFMSE (Group 1) compared to the control group. Regarding Group 2, no control group with proper matching according to age, sex and type of SMA was identified.
Baseline characteristics of treated and control groups evaluated using the HFMSEa
y = years, HFMSE = Expanded Hammersmith Functional Motor Scale, SMA = spinal muscular atrophy, p = significance value for Pearson chi-square tests. aValues expressed as mean and SD unless otherwise indicated.
HFMSE measurements
For Group 1, mean change in HFMSE over time estimated using a repeated measures analysis of covariance model with time was +1.47 points (SD = 0.4) and +1.60 points (SD = 0.6) after 12 and 24 months of treatment, respectively. In contrast, the control group presented a mean loss of −1.71 points (SD = 0.02) and −3.93 (SD = 0.55) after 12 and 24 months of follow-up, respectively (Fig. 1).Treated patients with SMA type 2 evolved differently over time compared to treated SMA type 3 patients, but both benefited from nusinersen therapy (p < 0.001, Fig. 1).
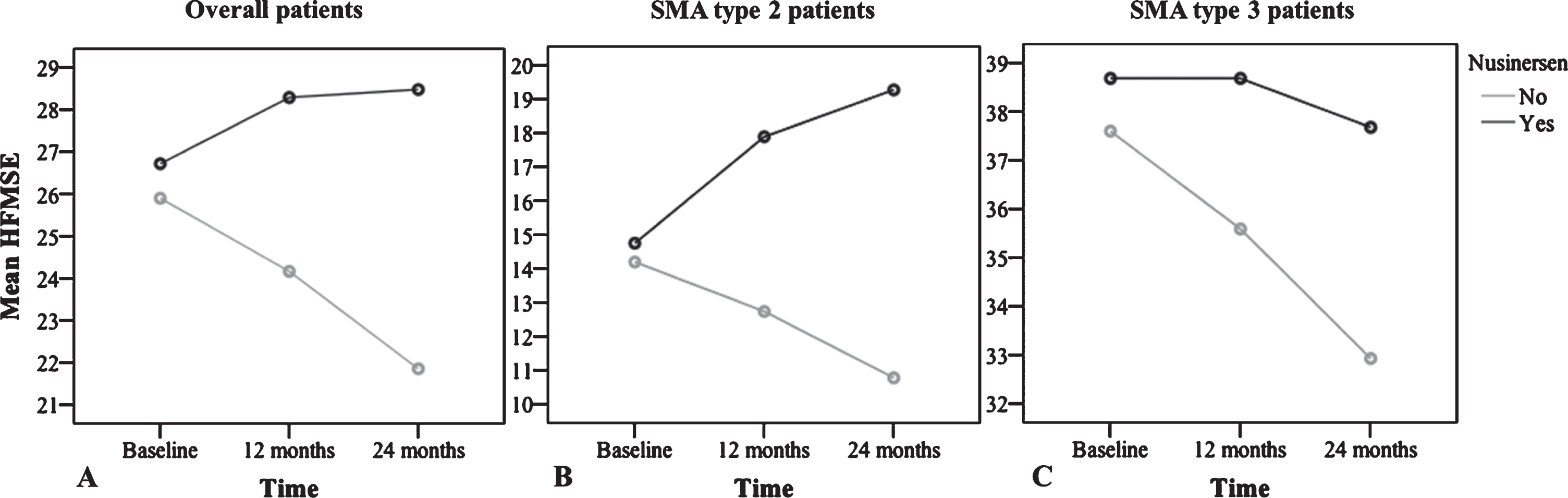
Progression in HFMSE score over time (0, 12 and 24 months) estimated using a repeated measures analysis of covariance model with time. A) Overall group of patients (SMA types 2 and 3), according to treatment; treatment effect: p < 0.001, eta-squared = 0.195. B) HFMSE progression over time in SMA type 2, according to treatment. C) HFMSE scores over time in SMA type 3, according to treatment. Mean change in SMA type 2 treated patients was +3.12 (SD = 1.26) and +4.5 (SD = 1.91) after 12 and 24 months, respectively. In the control group, mean change in SMA type 2 was −1.45 (SD = 0.9) and −3.4 (SD = 0.24) after 12 and 24 months, respectively. In SMA type 3 patients, there was no mean change (SD = 0.74) in HFMSE after 12 months of treatment, and a mean loss of −1.0 point (SD = 0.58) after 24 months of treatment was observed; the results were not significant over time (p = 0.282), but they were significant compared with the control group that presented a change of −2.00 (SD = 0.12) and −4.65 (SD = 0.19) after 12 and 24 months of follow-up, respectively.
In an exploratory analysis, in an attempt to find predictors of gain in motor function after 12 months of treatment (defined as an increase of two or more points on the HFMSE), the variables analyzed (sex, age at symptoms onset, number of SMN2 copies, absence of hip contractures and previous spinal fusion or severe scoliosis at baseline) had no significant differences. Only disease duration showed a significant difference as a predictor of increased HFMSE scores after 12 months of treatment, as shown in Fig. 2.
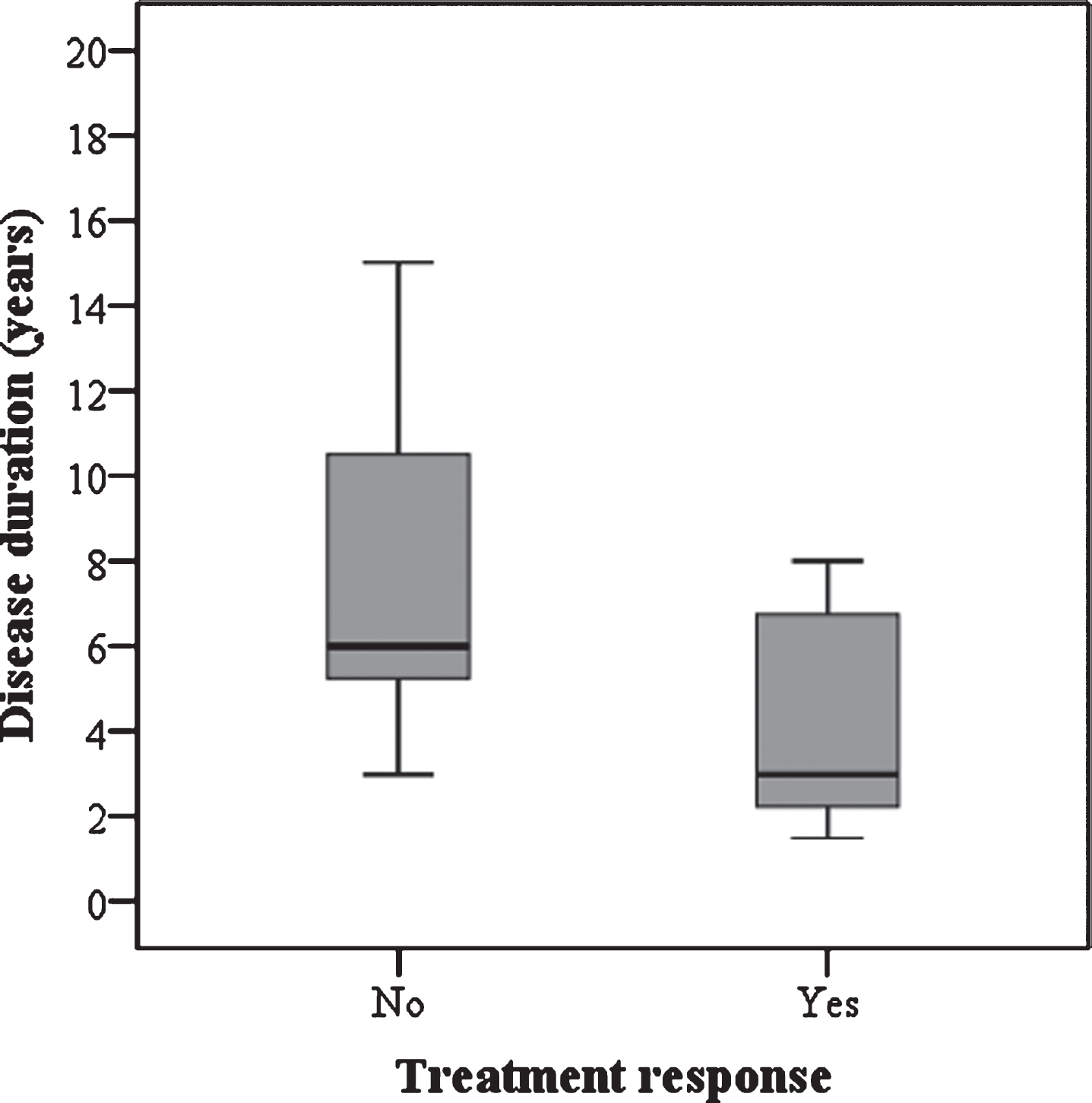
Disease duration as a predictor of gain in motor function (a change of 2 or more points in HFMSE) after 12 months of treatment. In the group that experienced motor responses the median disease duration was 3.1 years, compared to the median disease duration of 6 years in the group of patients that remained stable or worsened (p = 0.032).
CHOP-INTEND measurement
Table 2 shows baseline characteristics and results of CHOP-INTEND measurements throughout the treatment of Group 2. The mean follow-up was 16.9 months (range: 12 to 24), with a mean change in CHOP-INTEND of +2.37 points (range: −5 to 10) at 12 months of follow-up among the patients overall. In the group of patients followed for two years (n = 7), the mean change was +1.0 point (range: −5 to 8) and +3.42 (range: 0 to 14) after 12 and 24 months of follow-up, respectively. Younger patients (i.e., those between 1.5 and 4 years) achieved higher gains on the scale, although the reduced number of patients in this group made a confirmatory statistical analysis unviable.
Baseline characteristics and results of CHOP-INTEND measurement throughout the treatment (Group 2)a
y = years, mo = months, SMA = spinal muscular atrophy, CHOP-INTEND = Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders, SD = standard deviation. aValues expressed as median and (interquartile range) unless otherwise indicated.
Scoliosis progression
The analysis of a subgroup of 12 non-ambulant patients treated with nusinersen (eight SMA type 2 and four SMA type 3) who had 18 or 24 months of follow-up and whose Cobb angle was measured at baseline and at the last evaluation, showed progressive worsening of scoliosis. In these patients, the Cobb angle at baseline was 18.4° (range: 0° to 40°) and 37.1° (range: 10° to 80°) at the last assessment. This group of patients had a mean age at baseline of 7 years (range: 3 to 10 years) and a mean disease duration of 5.8 years (range: 2 to 9 years). Of these patients, three underwent spinal arthrodesis due to extensive scoliosis (Cobb angle >45°) during the follow-up period.
Adult patients and the EK functional scale
Among the 41 patients under treatment with nusinersen, nine were adults. In this adult group, six were female (66.6%), five had SMA type 3 (55.5%) and all had three copies of SMN2. Table 3 shows these patients’ clinical characteristics at the baseline and at the last treatment evaluation, as well as their motor function scale scores (HFMSE or CHOP-INTEND) and EK scale. Five patients experienced a change of at least two points on the EK scale from baseline, presenting improvement in head and trunk control, voice quality and cough strength and fatigue. Most patients (7 of 9) had previous spinal fusions, which added technical difficulty to intrathecal administration.
Clinical characteristics, motor function measurements and EK scores of adult patients at the beginning of the treatment and at the last evaluation
Pt = patient, F = female, M = male, y = years, mo = months, HFMSE = Expanded Hammersmith Functional Motor Scale, SMA = spinal muscular atrophy, CHOP-INTEND = Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders, EK = Egen Klassification functional scale, NP = not performed.
Hospitalizations during follow-up, intrathecal procedures and side effects
Group 1 had a hospitalization rate of 0.1 per patient (SD = 0.4) for causes other than the administration of nusinersen during 24 months of follow-up, whereas in the control group, the rate was 0.49 hospitalizations per patient (SD = 0.98). Although the reduction in the number of hospitalizations favored treatment with nusinersen, the difference between the two groups was not statistically significant. No patient discontinued treatment due to adverse events.
Among patients treated with nusinersen, a total of 306 lumbar punctures were performed. Fluoroscopy or computed tomography was performed on eight patients (19.5%) to guide the intrathecal administration in patients with previous spine arthrodesis [22]. Sedation was performed systematically on 25 patients (61%), either through intravenous anesthesia (propofol or midazolam) or through inhalation of anesthetic agent (sevoflurane).
The complication rate in the total number of procedures (n = 306) was low, around 4.2%, and mainly included post-puncture headaches and lower back pain. However, in the group in which sedation was used, other complications occurred: two episodes of respiratory depression and two episodes of extreme tachycardia (>180 bpm).
DISCUSSION
In the present study, we showed that treatment with nusinersen is safe and effective for patients with SMA types 2 and 3, as a statistically significant motor gain was observed after 12 and 24 months of treatment. The efficacy was confirmed by comparing the mean increase in the HFMSE scores of the treated patients with those of a group of non-treated patients. Disease duration was the main predictor for functional motor improvement.
Overall, our patients experienced a mean gain in their HFMSE scores during treatment that was similar to the results of the CHERISH study [9]. Such a gain in motor functions is not expected in the natural history of patients with later-onset SMA [20, 23]. However, when analyzed separately, patients with SMA type 2 had a greater improvement than those with type 3. Patients with SMA type 3 had a slight decline in the mean score from 12 to 24 months of treatment, but this outcome can be considered favorable based on the declines in the control group's scores. Other studies, however, have shown functional gains in patients with SMA type 3 during treatment with nusinersen [17, 24].
Our study found that disease duration at the beginning of the treatment is the main predictor of gain on the HFMSE, reinforcing findings from previous studies that showed the greatest improvements in HFMSE scores observed in children who received treatment soon after the onset of symptoms [9]. In addition, the results of the 24 months of follow-up in our patients confirm the findings of Darras et al. (2019), who provided evidence for longer-term efficacy and safety of nusinersen treatment in late onset SMA patients [24].
Interestingly, despite the favorable motor response to treatment with nusinersen, we observed worsening of scoliosis in the patients during follow-up that indicates the need for intensive preventive care for spinal deformity in this group of patients.
Real-life studies comparing treated with non-treated SMA types 2 and 3 are scarce because treatment has usually been indicated for all patients with SMA [18]. However, in Brazil, access to the drug remains restricted, as SUS does not yet have an official guideline to provide treatment for patients with SMA types 2 and 3, and a minority of patients have private health insurance that covers the treatment. At our center, treated and untreated patients were evaluated similarly for at least two years by the same team. However, although our control group was matched according to age, sex, time of disease and number of SMN2 copies, it could not be considered ideal because it was not a randomized, double-blind group selected using well-defined inclusion and exclusion criteria. Although the control group had access to standard supportive care during follow-up, there was a mean change of −1.71 and −3.93 points in the HMFSE at 12 and 24 months, respectively. This loss was greater than that described in studies of the natural history of the disease. On the other hand, Mercuri et al. (2016) showed that later-onset SMA patients between 5 to 15 years old, a range which most of our control patients fit, may have a worse outcome than patients above or below this age group [20].
Although our patients were not selected to receive treatment based on clinical condition or the presence of spinal deformities, the differences in the mean change in the HMFSE scores over time observed in our study’s treated and untreated patients might have been due to differences in the standard support of care, considering that part of the treated group had access to the drug through health insurance, and therefore may also have had favorable socioeconomic conditions. In addition, the higher prevalence of patients with severe scoliosis or arthrodesis in the untreated group may also justify a more progressive phenotype of our control group compared to the treated group [25, 26].
Very weak patients who could not sit were evaluated using the CHOP-INTEND scale adapted instead of the HFMSE. In this group, mean CHOP-INTEND scores improved by +2.37 points after the 12-month follow-up, indicating a favorable effect of the nusinersen even in very weak, later-onset SMA patients. However, we did not have a control group for these patients, and there are no available natural history data for SMA types 2 and 3 that use the CHOP-INTEND scale for comparison.
The nine adult patients evolved mostly with motor function stabilization or improvement. Despite the small number, the findings were similar to those described in other studies, which also reported improvement or motor stabilization after treatment with nusinersen [11, 16]. A recent study of a large cohort of adult patients with SMA types 1 through 4 reported a significant gain in the mean HFMSE score after 6, 10 and 14 months of treatment, in addition to an improvement ≥3 points in HFMSE scores in 28% of patients at 6 months of treatment, 35% at 10 months and 40% at 14 months [16]. On the other hand, a recent controlled and retrospective study showed no significant objective changes in adult patients treated with nusinersen [18]. Although these studies have shown great variability in responses to nusinersen, it was possible to confirm the effectiveness of the drug at least for stabilizing the disease’s progression, especially when comparing with natural history data of the adult patients [20, 27]. In addition, the EK scale used in our adult patients revealed an overall gain in several aspects, such as cervical and trunk control and improved voice quality. Another factor favored treatment with nusinersen was the lower number of hospitalizations compared to the control group, despite a lack of statistical significance.
We observed no relevant side effects causally related to the medication. A high number of intrathecal procedures was performed in our patients with a low rate of complications or adverse events (4.2%). In particular, respiratory depression and severe tachycardia occurred during anesthetic procedure. For this reason, sedation should be done with caution in these patients, since the repetitive use of anesthetic procedures may lead to complications over time, which adds risk to the treatment [28].
This study had some limitations. First, it was a single-center observational unblinded study with a small number of patients. Second, we could not exclude the influence of socioeconomic aspects for disease progression between the treated and untreated groups. Third, we did not include the six-minutes walking test (6MWT) for ambulant and scales to assess upper limb function to complement the HFMSE score in SMA types 2 and 3 [29–31]. In addition, we could not assess swallowing and pulmonary forced vital capacity in all patients due to the center’s characteristics. However, all patients were evaluated by the same team, allowing for a homogeneous medical and functional assessment compared to multicenter studies.
CONCLUSION
In conclusion, we showed that nusinersen was effective for SMA types 2 and 3 at 24 months of follow-up, but its effect on motor function was greater in patients with SMA type 2. In addition, patients with shorter disease duration showed better response to treatment. Despite the functional improvement or motor stabilization, treatment with nusinersen did not prevent the progression of scoliosis. As the later-onset forms of SMA have a slow progression, longer studies are needed to confirm the stabilization or functional gain in these patients.
FUNDING
The authors received no financial support for the research, authorship, and/or publication of this article.
CONFLICTS OF INTEREST
RH Mendonça and E Zanoteli have received financial compensation from Biogen for advisory board participation, travel expenses, and for lectures/consultant. C Matsui Jr, GJ Polido, AMS Silva, DJF Solla and UC Reed have no conflicts of interest to declare.
Footnotes
ACKNOWLEDGMENTS
We are thankful to the patients and parents who participated in this study.
