Abstract
Background
Spinal muscular atrophy type 1 (SMA1) is a motor neuron disease associated with progressive muscle weakness, ventilatory failure, and reduced survival.
Objective:
To report the evaluation of the nusinersen, an antisense oligonucleotide, on the motor function of SMA1.
Methods
This was a longitudinal and observational study to assess the outcomes of nusinersen therapy in SMA1 patients using the HINE-2 and CHOP-INTEND scales.
Results
Twenty-one SMA1 patients (52.4% males) were included; the mean age at first symptoms was 2.7 months (SD =±1.5), and the mean disease duration at first dose was 34.1 (SD =±36.0) months. During posttreatment, the mean gain on the CHOP-INTEND was 4.9, 5.9, 6.6, and 14 points after 6, 12, 18, and 24 months, respectively. Starting medication with a disease duration of less than 12 months and/or without invasive ventilation were predictors of response on CHOP-INTEND. Of the patients, 28.6% acquired a motor milestone or gained at least three points on the HINE-2. The daily time for ventilatory support was reduced after treatment in most of the patients with noninvasive ventilation at baseline. No change in the daytime use of ventilation was observed in most of the patients using invasive ventilation at baseline.
Conclusions
Nusinersen produces improvements in motor and respiratory functions, even in long-term SMA1 patients. However, patients under invasive ventilation at the beginning of the treatment experience little benefit.
INTRODUCTION
Spinal muscular atrophy (SMA) is a neurodegenerative disease of the lower motor neurons from the spinal cord and motor nuclei of the brainstem, leading to axial and proximal limb weakness associated with ventilatory insufficiency and spinal deformity. The most common form of SMA is caused by deletions or disease-causing variants in the survival motor neuron 1 (SMN1) gene, which segregates as an autosomal recessive trait [1]. Around 1 in 11,000 people is affected by the disorder [2]. SMA has been classified into at least three subtypes depending on age at disease onset and the achievement of motor milestones [3]. SMA type 1 (SMA1), or Werdnig-Hoffman disease, is the severe form of the disease, and is characterized by an age at onset of 0–6 months; children are unable to sit unaided and have a median life expectancy of less than 2 years without respiratory support [4, 5].
Several studies have demonstrated a strong inverse correlation between the number of SMN2 copies and SMA severity [1, 6]. Nusinersen is an antisense oligonucleotide that targets the pre-mRNA of SMN2, therefore avoiding exon 7 skipping and then increasing full-length SMN protein levels. Its effectiveness has been demonstrated in two pivotal trials: one in patients with SMA1, [7] and the other in SMA types 2 and 3 [8]. The US Food and Drug Administration (FDA) first approved nusinersen for the treatment of SMA in December 2016 for all ages, types, and disease stages. The Brazilian National Health Surveillance Agency (ANVISA) approved it in 2017 [9]. The ENDEAR study showed that SMA1 patients under 7 months old and without ventilatory support who received nusinersen survived longer and experienced more improvements in motor function compared to those without treatment [7]. Subsequent studies, including those that use the drug in an expanded access program, confirmed its beneficial effects on motor and respiratory functions as well as survival for patients with long disease duration and variable respiratory conditions [10–17].
In this study, we aimed to assess the safety and efficacy of nusinersen in a cohort of symptomatic SMA1 patients with variable disease duration and different levels of respiratory support in a Brazilian reference center.
PATIENTS AND METHODS
This was a cohort study from a single reference center in Brazil (Neuromuscular Clinic at Hospital das Clínicas, FMUSP). Patients with SMA1 and genetic confirmation (homozygous deletion of exon 7 or with compound heterozygous mutations on the SMN1 gene), [18] with two or three copies of SMN2, and with access to nusinersen were included. No exclusion criteria were applied. Patients were included regardless of their age, disease duration, or type of ventilatory support before treatment. Patients obtained nusinersen through private health insurance or the Brazilian public health service (SUS). The study was approved by the institutional ethics committee (CAPPesq # 87773118.7.0000.0068), and written informed consent was obtained from the responsible for the patients.
Clinical and functional assessments
Patients were evaluated every 6 months through the CHOP-INTEND (Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders) functional motor scale. It included a total of 16 items, with scores from 0 to 64 points and was validated to evaluate the progression of SMA1 [19]. Patients were also assessed by the acquisition of eight motor milestones according to section 2 of the Hammersmith Infant Neurological Examination (HINE-2): voluntary grasp, kicking, head control rolling, sitting, crawling, standing, and walking. The same neurologist, who was trained in neuromuscular diseases performed all clinical evaluations. The same team of physiotherapists, all with experience in the use of motor scales, scored the CHOP-INTEND in all patients.
The type of ventilatory support (i.e. invasive mechanical ventilation [IMV] or noninvasive ventilation [NIV]) and the total daily time of ventilation use, were registered, in hours, before and after treatment. In addition, at each assessment, family members responded to a questionnaire to report the perceived benefits of treatment and the occurrence of side effects. This questionnaire included sections regarding the occurrence of infections, antibiotic use, hospitalization, daily ventilation time and the number of secretion aspirations per day in the previous six months.
Drug administration
In all patients, 12 mg of nusinersen was administered intrathecally on days 1, 14, 28, and 63, with repeated maintenance injections every 4 months in accordance with the label. Intrathecal injections were performed by a trained neurologist, and patients were observed for at least 6 hours after each procedure for the occurrence of possible adverse events.
Statistical analysis
Categorical variables were presented through relative and absolute frequencies and compared using Fisher’s exact test. The distribution of continuous variables was assessed for normality through skewness and kurtosis statistics and through graphical methods. Those with normal distribution were presented as mean and standard deviations and compared using an independent samples t test. The best disease duration cutoff points on the receiver operating characteristic (ROC) curve were 12.5 and 22 months for CHOP-INTEND response (≥4 points improvement) at 6 months and 12.5 and 26.5 months for 12 months. This finding dictated the disease duration variable categorization (<12,<24 and ≥24 months) for the purpose of results presentation.
Repeated measures general linear models were employed for the analysis of the CHOP-INTEND evolution from baseline to 6 and 12 months after start of treatment. This method allowed for the identification of variables associated with the major clinical outcome, the longitudinal evolution over time.
The predictors of motor-milestone response were identified through multivariable logistic regression. The significant variables at the 0.10 level on the univariate analysis were included in the initial models and maintained according to the Wald statistics. The Firth method was used for the logistic regression due to the phenomenon of separation observed in the variables’ treatment delay, the pretreatment type of ventilator support and the phenomenon of quasi-separation observed by sex.
All tests were two-tailed, and final p-values under 0.05 were considered significant. Analyses were conducted with the Statistical Package for Social Sciences software (IBM SPSS Statistics for Windows, version 24.0. Armonk, NY: IBM Corp.).
RESULTS
Thirty-one children diagnosed with SMA1 were evaluated from October 2017 to July 2019. A total of 21 patients (11 males, 52.4%) had access to nusinersen and were included in the study (Table 1). Eighteen patients had two copies of SMN2, and three had three copies. Baseline characteristics and functional data after treatment are presented in Table 1. The mean age at first symptoms and mean disease duration at the first dose were 2.7 (±1.5) and 34.1 (±36.0) months, respectively. No child had symptoms at birth, such as hypotonia and respiratory failure, and no child presented head control at the beginning of the therapy. Twenty-one patients were followed for at least 6 months, 15 for 12 months, 10 for 18 months, and 3 for 24 months. One patient died at 6 months of treatment due to complicated pulmonary infection.
SMA type 1 patients in treatment with nusinersen
Pt: patient, F: female, M: male, m: months, NIV: noninvasive ventilation, IMV: invasive mechanical ventilation with tracheostomy, GTT: gastrostomy, Pre-symp.: patient pre-symptomatic at first dose, d: days of life. *Patient 12 died within 6 months of follow-up.
CHOP-INTEND and motor milestones/HINE-2
At baseline, the CHOP-INTEND scores ranged between 2 and 33 (mean 13.4±9.8). Mean change in CHOP-INTEND score was 4.9 points after 6 months, 5.9 points after 12 months, 6.6 points after 18 months and 14 points after 24 months of treatment. For statistical analysis, only patients who had assessments at 6 and 12 months after treatment were included, due to the small number of patients with assessments at 18 and 24 months. Univariate analysis showed no association between CHOP-INTEND response and the variables: CHOP-INTEND score at baseline, age of symptoms onset, SMN2 copies, and sex.
The general linear model (GLM) demonstrated that change in CHOP-INTEND scores from baseline to 6 and 12 months after treatment was influenced by disease duration (p = 0.006) and baseline ventilatory support (p = 0.018) as well as an interaction between these variables (Fig. 1; Supplemental Table). Regardless of the baseline ventilatory support, the patients treated before 12 months of disease duration presented a satisfactory response, while those treated after 24 months improved less than 4 points on the CHOP-INTEND. Patients treated between 12 and 23 months of disease duration had an outcome that was largely impacted by the baseline ventilatory support (Fig. 1).
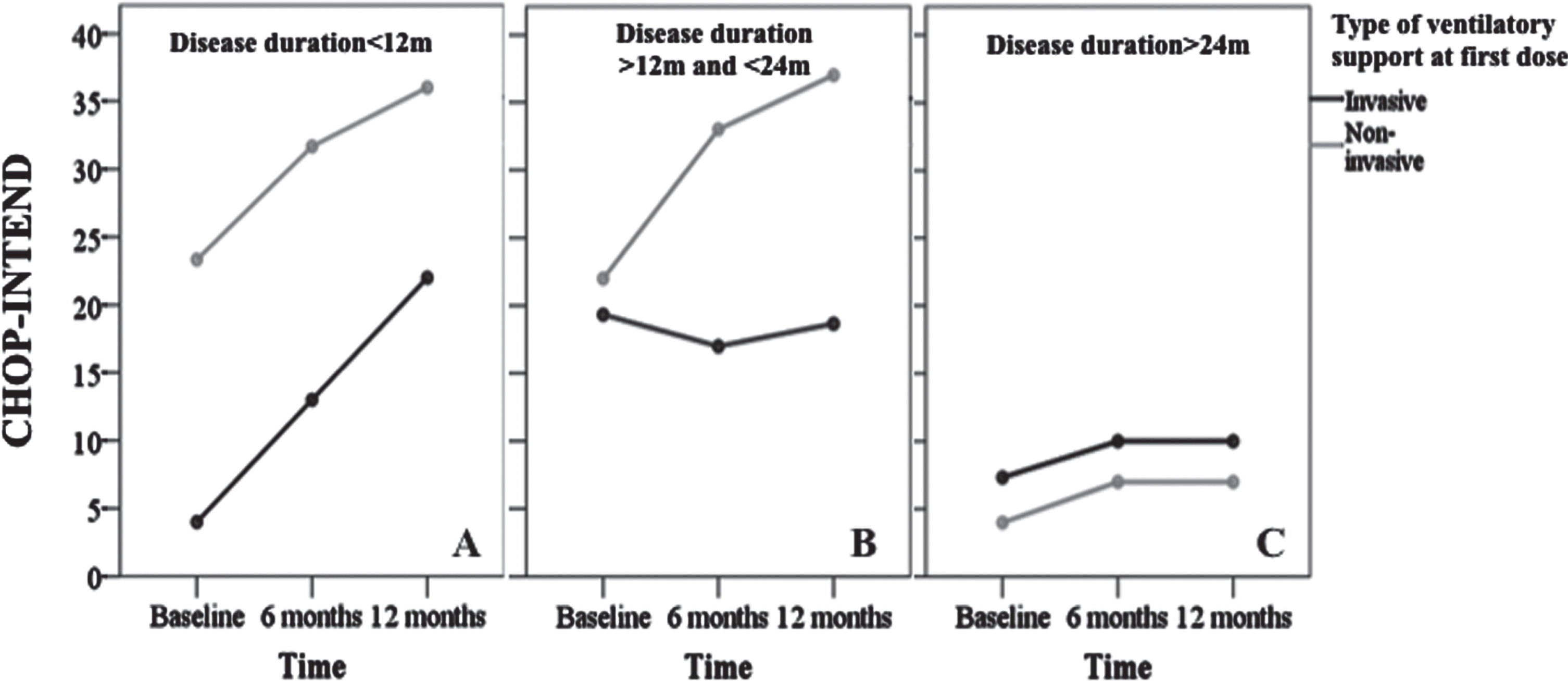
Change in CHOP-INTEND over time estimated using repeated measures, according to ventilatory support at baseline in different groups of patients with SMA1. A) Patients with disease duration of less than 12 months: NIV patients presented a mean change in CHOP-INTEND of +8.4 and +12.7 after 6 and 12 months, respectively, while IMV patients presented a mean change of +9.0 and +18.0 in the same period. B) Patients presenting a disease duration between 12 and 24 months: NIV patients presented a mean change of +11.0 and +15.0 after 6 and 12 months, respectively, and IMV patients presented a mean change of –2.3 and –0.6 in the same period. C) Patients with a disease duration longer than 24 months: NIV patients presented a mean change in CHOP-INTEND of +3.0 after 6 and 12 months while IMV patients presented a change of +2.7 after the same period of observation. Note that after 24 months of disease duration, there was no difference between patients undergoing IMV and NIV. This group of patients had an insignificant gain in CHOP-INTEND when compared to the other two groups treated under a short disease duration. Legend: m - months; NIV –non-invasive ventilation; IMV - invasive mechanical ventilation.
Among the 21 patients, six (28.6%) acquired some motor milestone or gained at least three points on the HINE-2 (Table 1); two children were able to sit independently, one sat with support, and three achieved head control. Univariate analysis showed no association between HINE-2 response and the variables: age of symptoms onset, SMN2 copies and CHOP-INTEND score at baseline. Multivariable analysis for the predictors of HINE-2 response in three different models showed that sex (female), disease duration and baseline ventilatory support were predictors of response (Table 2). There was moderate correlation between baseline ventilatory support and disease duration (correlation coefficient of 0.543, p = 0.011) and only two patients were not at IMV after 12 months.
Multivariable analysis for the predictors of HINE-2 response (n = 21) in SMA type 1 patients treated with nusinersen. Female sex, disease duration and type of ventilatory support at baseline were predictors of response
Ref.: Reference category, Coef.: Coefficient, CI: Confidence interval, SE: Standard error, OR: Odds ratio.
Respiratory support
General characteristics at baseline were similar between patients who started treatment with use of IMV and those who started with NIV (Table 3). Only disease duration at first dose differed significantly between these two groups; short disease duration correlated with NIV.
General characteristics of SMA type 1 patients under nusinersen therapy, and according to ventilatory support at baseline
IMV: invasive mechanical ventilation, NIV: noninvasive ventilation, SD: standard deviation. Data presented as n (%), except if otherwise specified.
Among the 21 patients, seven (33.3 %) were using NIV at baseline. At the last evaluation, five of these children had the daily time of ventilatory support reduced, while one child had no change and another one had a slight increase in the time of the daily use of ventilation. Fourteen patients (66.6%) were using permanent IMV (24 hours a day) at baseline; in only one, the daily time of ventilation was reduced by 4 hours after treatment (Table 1).
Nutritional support
Among patients, 20 (95.2%) used gastrostomy at the baseline or started using it during the first 2 months of treatment (loading doses). Children who were feeding orally at the first dose, whether together with gastrostomy or not, continued to use it (Table 1). Only four patients (19%) acquired speech, and all of these were under NIV.
Side effects and parent-reported questionnaires
Among the patient’s relatives, none considered interrupting the treatment at any time until the last evaluation, even among those who did not observe evident motor responses during the treatment. For 60%of the parents of patients under IMV, there was a perceived reduction in recurrent pulmonary infections and decreased need for secretion aspiration throughout the day.
Sedation was used during drug infusion on four patients (18.2%), and in one of them, the use of the anesthetic agent led to an unexpected temporary intubation. A small incidence of adverse events was reported; post-puncture headache was reported by different patients in three out 140 procedures (2.4%).
DISCUSSION
This work reinforces the results of previous studies demonstrating the beneficial effects of nusinersen regarding motor and respiratory function in patients with SMA1, even when treatment started after 7 months of age. It also confirms well-established information that the earlier the treatment begins, the better the response of motor and respiratory functions will be. However, we observed a poor effect of the drug in those cases that initiated treatment with more than 2 years of disease and that were already on IMV. Such information is relevant in the sense of planning both the indication of the use of the medication and the suspension of therapy.
In our study, it was possible to observe a gain in motor functions at different times after the onset of nusinersen therapy, as indicated by previous studies [7, 12–17]. Interestingly, the average of the CHOP-INTEND score increased progressively in patients with up to 24 months of treatment, and, despite the reduced number of patients in evaluations with more than 12 months of treatment, it was noticed in most cases that the functional gain obtained in the first year of treatment was maintained or increased after 18 and 24 months. Due to ethical reasons, our study had no control group; however, based on the natural history of SMA1, such a gain in motor functions is not observed in patients without treatment, who instead exhibit progressive loss in the CHOP-INTEND score [4, 20]. Our data showed that starting the treatment in patients with less than 12 months of disease is significantly associated with greater motor gain. Previous studies had already demonstrated that early initiation of therapy is the main determinant of therapeutic response [7, 17].
We observed that among SMA1 patients, 28.6%acquired some motor milestones or had a gain above 3 points on the HINE-2 after treatment. Such a gain in motor milestones during nusinersen therapy was also well demonstrated by others [7, 17]. In the group of patients evaluated after 12 months or more of treatment, one acquired the ability to sit unaided and another with support, and two acquired head control. In all these cases, therapy started with a disease duration of less than 12 months, and none of them was on IMV. Statistical analysis confirmed that a shorter disease duration and the absence of IMV at the beginning of treatment were significantly correlated with a higher score gain on the HINE-2. In addition to these variables, early gain in the HINE-2 score seems to correlate with the acquisition of sitting at 14 months of treatment [21]. Another factor that seems to be associated with the acquisition of the ability to sit is the presence of three copies of the SMN2 gene because in our study, two out of the three patients with three copies of SMN2, acquired the ability to sit with or without support after the treatment. Other studies have also indicated better motor response in patients with three copies of SMN2 compared to those with two copies [12, 14]. We observed in our study that female sex was also a predictor of gain in the HINE-2 score. Previous studies have shown that women have a less severe course of the disease, [22, 23] and thus can start treatment in a better functional state.
Among all patients, 66.6%started nusinersen therapy already under IMV. These patients remained on IMV at the end of the study. Only one patient had a reduction in daytime hours of ventilation. This finding is similar to that of the study of Sansone et al. (2020), [16] who demonstrated that 100%of the patients undergoing IMV at the beginning of the treatment remained stable, with no improvement for NIV. On the other hand, in the group of patients undergoing NIV, most evolved with a reduction in the daily hours of ventilation after treatment, indicating a benefit from medication on respiratory function. Such a benefit observed even in patients who started therapy after 7 months of age has been previously reported in other studies [12, 16]. In our cohort, shorter disease duration at the beginning of treatment was correlated with the use of NIV after 12 months of treatment. Such information is in line with the findings of Sansone et al. (2020) that patients who started treatment before 2 years of age had a better response regarding ventilatory function [16].
Despite the lack of evident efficacy of the drug in the pulmonary function in patients on invasive ventilation, relatives reported reduction of the number of infections and in the need of tracheal aspiration procedures. Longer follow-up of these patients may show whether such subjective beneficial effects would have a practical effect on ventilatory function. More sensitive methods, such as plethysmography and ultrasound of respiratory muscles, may be useful in monitoring these patients on IMV to assess the effects of new therapies on lung function for patients with SMA1 [24].
Among our patients already with gastrostomy at the beginning of the treatment, all of them continued to use it at the last evaluation. Similarly, Sansone et al. (2020) showed continued use of gastrostomy in most cases during treatment [16]. On the other hand, children receiving oral feeding alone or associated with gastrostomy at baseline, continued to be fed orally in the last evaluation, indicating at least a stabilization of the bulbar function. Among our patients, four acquired the ability to speak, indicating a beneficial effect of the medication on bulbar function. Application of more sensitive methods such as a swallow-video exam, in addition to the use of more specific scales to assess swallowing and speech function, would be important in deciding whether to implement oral feeding during the treatment period.
The major limitation of our study is the small number of patients, which is justified by the fact that the evaluations were carried out at the same center. On the other hand, evaluation of patients at the same center gave the advantage of a more homogeneous medical and functional assessment compared to multicenter studies.
In conclusion, the disease duration for SMA1 patients is a key predictor of the extent of motor gains during nusinersen treatment, and a longer disease duration associated with the IMV at the beginning of treatment leads to a poor therapeutic response. However, even in this group of patients, the stabilization may represent a difference compared to a clearly demonstrated decline on motor function in previous natural history studies [4]. Future studies with long follow-up on long-term patients are essential for better planning of therapy in these cases. Finally, it is important that family members are aware of these results in order to form a more realistic expectation of the effects of the medication under different conditions.
FUNDING
The authors received no financial support for the research, authorship, and/or publication of this article.
CONFLICTS OF INTEREST/COMPETING INTERESTS
RHM and EZ have received financial compensation from Biogen for advisory board participation, travel expenses, and for lectures/consultant.
Footnotes
ACKNOWLEDGMENTS
We are thankful to the patients and parents who participated in this study.
