Abstract
Background:
Infantile hereditary proximal spinal muscular atrophy (SMA) type 1 is characterized by onset in the first 6 months of life and severe and progressive muscle weakness. Dysphagia is a common complication but has not been studied in detail.
Objective:
To study feeding and swallowing problems in infants with SMA type 1, and to explore the relation between these problems and functional motor scores.
Methods:
We prospectively included 16 infants with SMA type 1 between September 2016 and October 2018. Eleven infants received palliative care and five infants best supportive care in combination with nusinersen. We compiled and used an observation list with feeding related issues and observed feeding sessions during inpatient and outpatient visits. The Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders (CHOP INTEND) was used as a measure of motor function.
Results:
All infants in the palliative care group (median onset of disease 14 days (range 1–56); median inclusion in the study 52 days (range 16–252) demonstrated symptoms of fatigue during feeding and unsafe swallowing. Symptoms were short nursing sessions (10–15 minutes), and not being able to finish the recommended feeding volumes (72%); increased frequency of feeding sessions (55%); coughing when drinking or eating (91%), and wet breathing during and after feeding (64%).
Two out of five infants in the nusinersen group (median onset of disease 38 days (range 21–90); inclusion in the study at 63 days (range 3–218) were clinically pre-symptomatic at the start of treatment. The other three infants showed symptoms of fatigue and unsafe swallowing at inclusion in the study. These symptoms initially decreased after the start of the treatment, but (re)appeared in all five infants between the ages of 8 to 12 months, requiring the start tube of feeding. In the same period motor function scores significantly improved (median increase CHOP INTEND 16 points).
Conclusion:
Impaired feeding and swallowing remain important complications in infants with SMA type 1 after the start of nusinersen. Improvement of motor function does not imply similar gains in bulbar function.
Glossary
Spinal muscular atrophy Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders Videofluoroscopic swallowing study Medical research and Ethics Committee
INTRODUCTION
Hereditary proximal spinal muscular atrophy (SMA) is a severe autosomal recessive neuromuscular disease caused by survival motor neuron (
Treatment options of children with SMA type 1 have changed dramatically due to technological advances that allow modification of the genetic machinery. Both the antisense oligonucleotide nusinersen and
Feeding and swallowing problems are one of the most important complications of SMA type 1 [5–7]. Dysphagia in SMA type 1 is described as disturbed and weak sucking, problems with handling oral secretions, weak swallowing with dysfunctional airway protection, and gastroesophageal reflux (GER) [7–10]. Dysphagia in SMA type 1 may also lead to other problems such as poor weight gain, discomfort and risk of aspiration pneumonia [9–14]. Early and regular monitoring of feeding difficulties at an early stage is recommended in the standards of care [5], but a systematic approach to identify feeding and swallowing problems has not been reported in the literature.
The aim of this study was to describe characteristics of dysphagia in a cohort of infants with SMA type 1 who either received palliative care or best supportive care in combination with nusinersen. The second aim of the study was to explore the relation between functional motor scores and bulbar problems.
PATIENTS AND METHODS
Patients
We prospectively included 16 consecutive patients with SMA type 1, who visited the outpatient clinic of the Netherlands SMA Center at the University Medical Center Utrecht from September 2016 to October 2018. All patients had a homozygous deletion of the survival motor neuron (
The study protocol was approved by the local medical ethical committee (No. 09-307). Parents of eight infants gave informed consent. Eight other infants (all receiving palliative care) visited the hospital once and then received palliative care at home. We could not obtain informed consent because parents were overwhelmed by emotions after hearing the diagnosis. The clinical data of these infants were used anonymously (in accordance with Article 458 of the Dutch Law of Medical Treatment Agreement).
Treatment with nusinersen versus palliative care
Treatment with nusinersen for infants with SMA type 1 became available in the Netherlands in May 2017 as part of the expanded access program (EAP) and was reimbursed from August 2018. Parents of children with disease duration of <26 weeks, and in relatively good clinical condition (i.e. without apparent signs of (impending) respiratory insufficiency) were offered the choice between palliative care and treatment with nusinersen in combination with best supportive care [5]. We arranged palliative care if children were in an advanced stage of the disease, i.e. with signs of respiratory insufficiency.
Feeding and swallowing assessment
Two of the authors (AMBH and LEH), who are both experienced speech and language therapists specialized in pediatric feeding and swallowing problems in neuromuscular diseases, compiled an observation list based on clinical best practice and the relevant literature [7, 15–23]. The presence of feeding abnormalities in all infants was systematically assessed, using the predefined items during inpatient (e.g. for nusinersen treatment) and outpatient visits as part of clinical care.
The items of the observation list covered four main topics:
1. Fatigue related to oral feeding:
We documented the length and frequency of a nursing session and the presence of sweating when eating or drinking.
2. Unsafe swallowing:
We documented symptoms of unsafe swallowing (coughing, clearing the throat, and wet breathing) during oral feeding. We observed coughing, due to posterior drooling (i.e. saliva spilled over the tongue into the hypopharynx). We also asked parents if they recognized these symptoms and had noticed them prior to the hospital visit.
3. Regurgitation of food:
We asked whether infants frequently spit up milk or food or continued to swallow after finishing eating or drinking.
4. Respiratory system:
We observed the presence of an abdominal breathing pattern and documented respiratory rate, weakness of voice and cough, and frequency of respiratory infections.
Registration of swallowing by videofluoroscopic swallowing study (VFSS)
We performed VFFS in infants from the nusinersen group when we suspected unsafe swallowing. A Philips MultiDiagnost Eleva C-arm was used at a pulse rate of 15 per second. The images were stored in the picture archiving and communication system (PACS). We used an iodine-containing non-ionic radiocontrast agent. We mixed the agent with the milk or pureed food. We visualized the first suck-swallow-breathe sequence, and/or sips of pureed food with the bottle and/or spoon the child was used to. We used the 8-point Penetration-Aspiration Scale, ranging from 1 (‘material does not enter the airway’) to 8 (‘material enters the airway, passes below the vocal folds, and no effort is made to eject’) [24].
Motor function assessment (CHOP INTEND score)
Two of the authors (MAGCS and DRW), both experienced pediatric physiotherapists, evaluated the motor function using the CHOP INTEND score at the start of the treatment and at 2, 6 and 10 months after the first gift of nusinersen. This instrument consists of 16 items and assesses the strength during reflexive, spontaneous, or goal-directed movement. The maximum score is 64 points. Lower scores indicate poorer motor function [25, 26].
Relationship between functional motor scores and bulbar problems
We used wet breathing as an outcome measure of unsafe swallowing. To evaluate the effect of nusinersen treatment we used the following categories: ‘no wet breathing’, ‘inconsistent wet breathing’ (during feeding), ‘consistent wet breathing’ (during and after feeding). These scores were compared with the CHOP INTEND values at the start of the treatment, and at 2, 6 and 10 months after the first gift of nusinersen.
Statistical analysis
We used descriptive statistics for patient characteristics, results of the feeding observation list, and CHOP INTEND scores.
RESULTS
Characteristics of patient group
We included 16 out of 19 (84%) consecutive infants with SMA type 1 from May 2016 until October 2018. One child passed away immediately after the diagnosis. Parents of the other two infants could or did not want to participate. Eleven out of 16 (69%) enrolled infants received palliative care and five (31%) received treatment with nusinersen in combination with best supportive care. The parents of three infants (27%) declined treatment and opted for palliative care. Patient characteristics of the palliative care and nusinersen treatment groups are displayed in Table 1. The median onset of disease symptoms, start of tube feeding and CHOP INTEND was higher in the nusinersen group. The age at inclusion and weight did not differ between groups.
Patient characteristics
Data are n or median (range). *Not available from 1 patient. **Growth curves based on the fifth national growth study in the Netherlands (www.tno.nl/groei). SD: standard deviation. ***Infant 7 was prenatally diagnosed. ****Not available from 4 patients.
Feeding problems in the palliative care group
All infants demonstrated weak sucking. Most infants did not manage to suck with the required vacuum with their tongues, resulting in clicking sounds and/or spilling of milk. The nursing sessions were frequently short and infants failed to drink the recommended feeding volumes. We observed the following symptoms of unsafe swallowing: 1. coughing; 2. clearing the throat during drinking and 3. wet breathing during and after feeding. Three infants (27%) coughed when drinking, but showed no wet breathing, while seven infants (64%) demonstrated a combination of coughing and wet breathing.
Parents of six infants (55%) reported that they increased the frequency of feedings in order to compensate for feedings that were not finished. Six infants (55%) demonstrated sweating when feeding. Respiratory rate was >60 per minute in nine infants (82%) (Table 2).
Feeding characteristics of infants with SMA type 1 in the palliative care group, at inclusion in the study
*Percentage of ‘yes’. – = No, + = Yes.
Four infants (36%) showed symptoms of regurgitation, as they swallowed frequently after tube feeding. All infants had an abdominal breathing pattern. Their cough and cry was weak. Three infants (27%) had respiratory infections.
All infants started tube feeding directly or within a few days after visiting the hospital.
Feeding problems in the nusinersen group
Two infants (7 and 9) who were clinically pre-symptomatic at enrollment did not show symptoms of feeding problems (Table 3). The parents of both infants had previously lost children with SMA type 1 (the sibling of infant 7 died at the age of 2 months, the sibling of infant 9, at the age of 7 months). Nusinersen was started seven days (infant 7) and 56 days (infant 9) after birth respectively. The other three clinically symptomatic infants had a weak suck and cough and showed wet breathing when eating or drinking. After the start of treatment with nusinersen, sucking and swallowing improved. For example, infant 10 was able to breast-feed for a few minutes without symptoms of unsafe swallowing. She also received partial tube feeding. Two months after the start of treatment, breast feeding sessions had normalized (i.e. without coughing or wet breathing).
Feeding characteristics of infants with SMA type 1 in the treatment group, at inclusion in the study
*Percentage of ‘yes’. – = No, + = Yes.
This initial improvement of bulbar function was temporary in all five treated infants. Between the ages of 8 to 12 months respiratory infections and coughing or wet breathing reappeared. VFSS showed silent aspiration in four out of five (80%) infants. The first VFSS examination of the fifth infant (infant 8) at the age of 8 months was not successful. At the age of 21 months a second VFSS demonstrated laryngeal penetration. All infants treated with nusinersen eventually needed tube feeding (median start at 382 days (range 63–445).
Relationship between functional motor scores and bulbar problems
The median CHOP INTEND score at baseline was 19 (range 4–35) in the palliative group, and 34 (range 17–46) in the nusinersen group. The infants with CHOP-INTEND scores in the range of 4–35 points all coughed or showed wet breathing during or after drinking. The feeding observations of the two clinically pre-symptomatic infants with CHOP INTEND scores of 43 and 46 at inclusion in the study showed no symptoms of fatigue or unsafe swallowing.
After 6 nusinersen gifts the median CHOP INTEND was 48 (range 42–59 points). In this first year of treatment, the median increase in the CHOP INTEND score was 16 points (range 7–26). Despite this improvement, all five treated infants showed a deteriorating swallowing function between the ages of 8 to 12 months (Fig. 1). Unsafe swallowing was confirmed with VFSS.
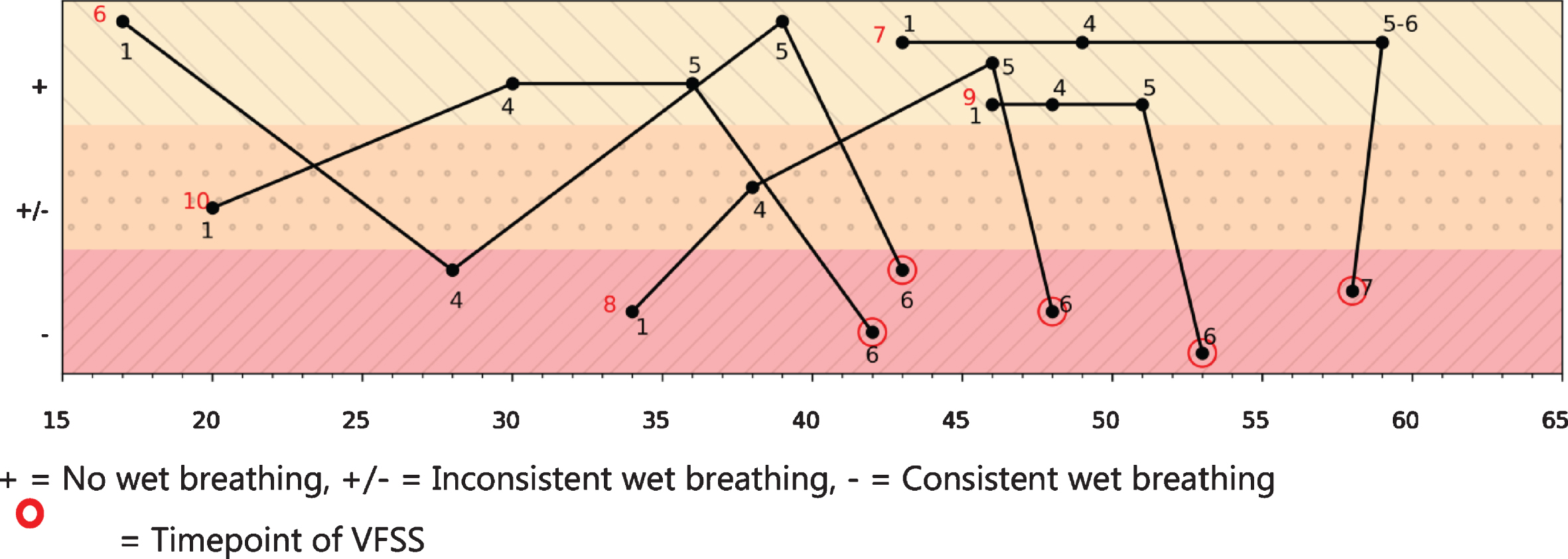
The CHOP INTEND scores (x-axis) in relation to bulbar function (y-axis), during the course of treatment with nusinersen (gift 1,4,5,6) of infant 6–10. + = No wet breathing, +/– = Inconsistent wet breathing, – = Consistent wet breathing = Timepoint of VFSS.
DISCUSSION AND CONCLUSION
In this study we systematically assessed feeding of infants with SMA type 1. As expected, all clinically symptomatic infants showed varying symptoms of insufficient drinking and fatigue during feeding, which eventually resulted in unsafe swallowing. These symptoms appeared gradually and parents could not specify when problems started. Treatment with nusinersen improved swallowing quality in the first half year of life but deteriorated in all, including 2 infants in whom treatment had been started when they were clinically pre-symptomatic. The gains in motor function as shown by increasing CHOP INTEND scores were not matched by improvement of bulbar function. After initial improvement of the swallowing and feeding abnormalities, the swallowing function deteriorated between the ages of 8 to 12 months. Symptoms and severity of dysphagia in the nusinersen group were comparable to the palliative care group. Our data suggest that continuous monitoring of feeding problems in treated infants with SMA type 1 is important.
SMA affects motor neurons in the spinal cord and the brainstem [27] and abnormal bulbar function is a well-known complication of SMA type 1 and 2 [7–10, 28]. Feeding is an important topic in the standards of care [5] but there are few studies that have systematically addressed the nature and prevalence of abnormalities in infants with SMA type 1. We observed feeding abnormalities in all infants, although symptoms varied. The most frequent abnormalities were coughing and shortened nursing sessions. This latter symptom easily goes unnoticed, as parents often increase the frequency of feeding with the aim of achieving sufficient intake and probably not always inform caregivers of these adaptations. Choking was easily overlooked because all infants demonstrated weak, ineffective coughing. We also found that ‘wet breathing’ occurred after a period of (soft) coughing when drinking and that in these cases VFSS showed, without exception, silent aspiration or silent laryngeal penetration. In addition, many children showed signs of fatigue, including sweating and an increased respiratory rate. High respiratory rate further complicated the coordination of swallowing and breathing when drinking, which may be associated with feelings of distress and aspiration [22, 23]. Limited endurance of muscle activities is a feature of SMA [29] that probably also affects bulbar muscles during swallowing and chewing. Our findings indicate that signs of relevant feeding abnormalities may be subtle and that feeding observations should be performed systematically and at short intervals.
This is to the best of our knowledge the first study that has systematically investigated the impact of nusinersen on bulbar functions. Recent findings suggest that nusinersen upon intrathecal administration disseminates throughout the central nervous system [30], but that delivery to motor neurons in the brainstem may be less efficient than to the lumbar and thoracic segments of the spinal cord [31]. Our data suggest that this has functional consequences, since even children in whom we started treatment with nusinersen before the onset of muscle weakness, demonstrated symptoms of abnormal feeding before their first birthday. Feeding abnormalities may not only be seen in infants treated with intrathecally administered nusinersen. Data from the phase 1 trial with AAV9-
It is important to note that motor function scores, which are used to evaluate response to treatment, do not necessarily completely correlate with bulbar function and do not predict the absence of bulbar dysfunction.
CONTRIBUTORS
AMBH contributed to study design, analyzed and interpreted data, and drafted the manuscript. IC, critically revised the manuscript. RIW critically revised the manuscript. FA critically revised the manuscript. MAGCS critically revised the manuscript. DRVDW critically revised the manuscript. EG, critically revised the manuscript. WLP contributed to revising the manuscript for content, including writing for content, study concept or design, analysis or interpretation of data, statistical analysis, study supervision or coordination, obtaining funding. LEH contributed to revising the manuscript for content, including writing for content, study concept, analysis or interpretation of data, statistical analysis, study supervision or coordination.
FUNDING SOURCE
This study was supported by research grants from the Prinses Beatrix Spierfonds, WOB 14–25, and by Stichting Spieren voor Spieren.
ROLE OF THE FUNDING SOURCE
The funding source which provided support for the conduct of the research had no role in study design; in the collection, design, and interpretation of the data; in the writing of the report; and in the decision to submit the article for publication.
DECLARATION OF INTERESTS
AMBH,RIW, IC, FA, MAGCS, DRVDW, EG, LEH declare no financial or other conflicts of interest. WLP receives research support from the Prinses Beatrix Spierfonds, Stichting Spieren voor Spieren and Vriendenloterij. His employer receives fees for ad hoc consultancy services to Biogen, Avexis (member scientific advisory board), and Novartis (member data-monitoring committee).
Footnotes
ACKNOWLEDGMENTS
The authors thank all parents of the infants who participated in this study.
