Abstract
Background:
People suffering from spinal cord injury (SCI) undergo metabolic and physical disturbances that target the skeletal muscle, causing a progressive loss of muscle mass.
Objective:
To estimate the appendicular lean mass index (ALMI) in athletes with traumatic (T-group) and non-traumatic (NT-group) SCI, and its association with metabolic and demographic parameters.
Methods:
Wheelchair athletes with SCI aged 18 to 52 years old were included (n = 62). From DEXA assessment, we estimated the ALM index (ALMI = appendicular lean mass/height2) and classified participants according to the degree of muscle loss (ALMI < 2 SD from the reference populations). Fasting blood was assayed for glycaemia, insulin, cortisol, and IGF-1 serum levels. Data were compared by T-test and Fisher’s Exact Test; predictors of ALMI were investigated by linear regression models.
Results:
The frequency of low ALMI was 63% in overall sample, 55% T-group and 71% NT-group. Low ALMI had no significant association with the origin of injury (X2 = 1.1, p = 0.29). Linear regression analyses showed significant association, in the whole sample, between ALMI and serum levels of IGF-1 (Beta = 0.69; p < 0.001), age (Beta=–5.8; p < 0.001), percentage fat mass (Beta=–0.26; p = 0.001), and energy intake (Beta = 0.32; p = 0.02). These significances were not maintained in the NT-group sub-analyses.
Conclusions:
Low ALMI was very frequent in our SCI participants, despite exercising and independently of the type of injury. Metabolic and demographic variables associated with low ALMI were different according to the origin of injury, which possibly relies on physiopathology particularities. More studies are necessary to clarify our findings.
INTRODUCTION
Spinal cord injury (SCI) is a condition that results from a traumatic event or non-traumatic such as diseases, tumour, congenital problems or infection. This condition involves a variety of physical and psychological symptoms and disturbances, including sensory, motor, or autonomic function. Despite the classification of SCI is still under consideration, here we refer to all lesions to the spinal cord as considered by the WHO [1].
Multitudinous changes in the body composition, physiological, inflammatory and metabolic status result as consequence of this neurological disturbance depending on the level of injury, immobilisation and subsequently muscle disuse, duration and advancing age, which increase the risk of negative health outcomes [2]. Several studies have reported a significant loss of skeletal muscle adjacent to an increase in fat mass (FM) in individuals affected in a short time after the event [3, 4]. Different factors are associated with all these changes, among them the breakdown of the excitation-contraction coupling in skeletal muscles, immobilisation, physical inactivity, possible reduced protein synthesis and anabolic hormones, and increased proteolysis [4]. Therefore, these conditions expose this population to an increased risk of suffering from concomitant medical conditions such as loss of muscle mass and function, in this case referred as secondary sarcopenia, as previously proposed by an international group of specialists [5]. Sarcopenia is defined as a progressive and general loss of muscle mass and function (strength and physical performance) and recognised as an independent condition assigned to an International Classification of Disease (ICD-10-CM). This condition increases the risk of adverse outcomes, poor quality of life and premature death [5–7]. Although this condition has been extensively related to older people (primary sarcopenia), the development of sarcopenia (secondary) has been currently attributed to factors other than aging with earlier onset in the lifecycle. Some example are systemic disease, inadequate intake of energy or protein, and physical inactivity, including disease-related immobility or disability [6].
In a previous study from our research group evaluating body composition in athletes with SCI, the levels of fat-free mass (FFM) were classified in the lowest percentile of the general population [8]. In addition, a bioelectrical impedance vectorial analysis (BIVA) displayed a differentiated electrical behaviour (high resistance, low reactance and angle phase) in these participants compared with people without restrictions in physical performance. These findings were particularly more conspicuous in people with non-traumatic injury (poliomyelitis sequelae), suggesting an increased cell membrane damage in their FFM tissues [9]. Lately, Dionyssiotis et al. investigated the application of the current sarcopenia definitions of the European Working Group on Sarcopenia in Older People (EWGSOP) for SCI. A method based on the appendicular lean mass index (ALMI) was performed and estimated a high prevalence of sarcopenia in SCI-paraplegics compared with an able-bodied control group, despite unclear sensitivity and specificity of these measures [10]. Based on these statements, the first aim of our study is to explore the appendicular lean mass (ALM) in wheelchair athletes with SCI considering the origin of injury by testing the current available methods, ALM index. The second aim is to predict the association between the loss of ALM and demographic and metabolic parameters that might represent a risk factor for the development of secondary sarcopenia in this population.
MATERIALS AND METHODS
Participants
A detailed description of methods and participants of this cross-sectional study has been published elsewhere [8, 9]. Briefly, the sample consisted of seventy-four community-dwelling active wheelchair participants with paraplegia, specifically basketball players, aged from 18 to 52 years old, both genders, diagnosed with SCI from traumatic and non-traumatic origin (poliomyelitis sequelae). Participants practise physical activity from at least 1 year before participation in the study, 3 times a week, for a minimum of 1 hour per training session. Trained research staff collected anthropometric measures and 3-day 24-hour dietary recalls. Dietary intake was analysed using the NUTRI software (São Paulo Federal University). Blood samples for biochemical assessment were collected in the morning, after overnight fasting; insulin-like growth factor 1 (IGF-1), cortisol, insulin, glucose, lipid profile, blood count were assayed by radioimmunoassay. All participants signed acceptance consent. The research protocol was approved by the Committee of Ethics on Human Experiments of the University of São Paulo, Brazil.
Body composition analysis and ALMI calculation
Body composition (FM and FFM) was determined by using dual-energy x-ray absorptiometry (DEXA; Lunar Corporation, Madison, Wisconsin, USA). All individuals undertook this assessment in the same day of anthropometric measures, under the same preparation conditions. To estimate the total appendicular lean mass, we summed up the lean mass of both upper and lower extremities; this summation was used to calculate the ALMI = total appendicular lean mass/height2. Based on the population and purpose of our research, we classified the degree of ALM as recommended by the EWGSOP [6] and the International Clinical Practice Guidelines for Sarcopenia (ICFSR), who pointed that this method is especially directed to individuals who are bedridden, or with gait speed < 1 m/s, or unable to perform a chair rise [11]. The cut-off point to identify a low ALM was adopted to be lesser than 2 standard deviations below the mean for young, general reference populations. These values corresponded to 7.25 kg/height2 for males and 5.67 kg/height2 for females, according to Delmonico et al. [12].
Statistical analysis
Data are presented in means and standard deviation. Normality was explored using the Shapiro–Wilk test; since most variables had a non-normal distribution, we transformed these variables by one-parameter Box-Cox in order to make them more normal distribution-like. Comparisons of quantitative variables were made using T-test, and categorical variables were analysed using Fisher’s Exact Test. Simple linear regression analyses were performed to explore the association between ALMI (dependent variable) and demographic and metabolic variables, namely, age, IGF-1, cortisol, percentage of appendicular FM, protein and energy intake per body weight. To run these analyses, we classified participants according to the origin of their injury into traumatic (T-group) and non-traumatic (NT-group) SCI. All analyses were performed in three steps: (i) the total sample; (ii) the T-group; and (iii) the NT-group. Afterwards, we repeated these analyses for the appendicular lean mass of the upper and lower limbs separately. Statistical significance was considered at P < 0.05. Data were analysed using R Software version 3.5.1.
RESULTS
General characteristics of the sample, according to the origin of injury, are presented in Table 1. The initial sample size was 74 participants; however, twelve participants were excluded from the analysis due to the lack of information on body composition. Analyses by DEXA were obtained from a total of 62 participants, 12 women and 50 men, equally distributed in each group (T-group=31, and NT-group=31) and included in the ALMI calculation of the sample. Both groups did not present differences in age, BMI and percentage of FM, as well as serum levels of cortisol and haemoglobin. Despite no significant differences, the lipid profile and the HOMA1-IR index were slightly increased in the NT-group, contrariwise to the levels of IGF-1, which was faintly decreased in this group compared to T-group. There were no differences in energy and protein intake between groups. The NT-group had significant lower lean mass in their legs compared with the T-group, there was not found significant difference in the lean mass of their arms.
Characterization of participants
ALMI: appendicular lean mass index; BMI: body mass index; dl: decilitre; g: gram; HOMA1-IR: homeostatic model assessment index for insulin resistance; HDL: High-density lipoprotein; Ht2: squared height; IGF-1: insulin-like growth factor 1; kcal: kilocalories; kg: kilogram; kj: kilojoules; LDL: Low-density lipoprotein; μg: microgram; mg: milligram; ml: millilitre; ng: nanogram; VLDL: Very low-density lipoprotein. Data are presented as mean (SD). References values for general population: IGF-1: Male: 89.1–270.6 ng/ml, Female: 82.1–263.5 ng/ml [27]; Cortisol: 5–23 μg/dl; Haemoglobin: Male: 14–18 g/dl Female: 12–16 g/dl; Cholesterol < 200 mg/dl; HDL: Male:>40 mg/dl Female:>50 mg/dl; LDL < 100 mg/dl; VLDL: 7–32 mg/dl; Triglycerides < 150 mg/dL [28]; HOMA1-IR: Insulin resistance: 2.7, metabolic syndrome: 2.3 [29].
We found that 63% of the total sample met the current criteria for a low ALM, and therefore, higher risk for secondary sarcopenia, as shown in Table 2. When the participants were compared according to the origin of injury, we observed that the frequency of low ALM was higher in the NT-group (71%) than the T-group (55%). However, the Fisher’s Exact Test for this observation showed no significance (1.99OR, 95% CI 0.63–6.61, p = 0.29). The T-test analysis comparing the degree of ALMI in individuals classified as low ALM (based on the cut-off points) between groups, traumatic [5.7 (0.9)] vs. non-traumatic [5.5 (0.77)], exhibited no significant difference (p = 0.33). Therefore, we may infer that the low ALM has no direct relationship with the origin of injury.
Classification of the ALMI in basketball wheelchair athletes with SCI in the total sample and according to the origin of injury and gender
*Participants were classified according to Delmonico et al. [12].
In Table 3 are presented the models of linear regression analysis. We observed a significant association between the levels of IGF-1 and the ALMI (B = 0.69, p < 0.001, Fig. 1a). In the sub-analysis according to the origin of injury, we noticed that the association with IGF-1 was stronger in the traumatic group than non-traumatic (B = 0.86 and B = 0.38, respectively), and this association seemed to significantly rely on the lower limbs (B = 1.37). In the cortisol analysis, the model exhibited an inverse non-significant and weak correlation between the levels of cortisol and ALMI, possibly the levels of this hormone are not associated with the appendicular lean mass in these participants. Moreover, the regression model indicated a clear and significant inverse association between the age and the ALMI (B=–5.8, p < 0.001, Fig. 1b). Despite this significance disappeared in the sub-analysis of the NT-group, the model of separated extremities showed that this relation remained in both groups for the lower limbs (T-group B=–9.44, p < 0.05; and NT-group B=–12.48, p = 0.01). A similar tendency was found in the linear regression model for the percentage of appendicular FM; there was a significant negative association between the skeletal muscle mass and the content of adipose tissue (B=–0.26, p = 0.001, Fig. 1d). Again, this association disappeared in the sub-analysis of the NT-group, but remained in both groups for the lower limbs (T-group B=–0.46, p < 0.01; and NT-group B=–0.47, p < 0.01). There was also a positive association between the ALMI and energy intake (B = 0.32, p = 0.02, Fig. 1c), but not with protein intake, just for the lower limbs (B = 1.04, p = 0.04). Summarising, we found serum IGF-1 levels, age, energy intake, and FM as significant predictors of the ALMI in traumatic SCI, but this significance did not remain in the sub-analysis with NT-group and was pronounced for the lean mass of the lower limbs.
Associations between appendicular lean mass index (ALMI), upper (arms) and lower (legs) limbs and biochemical, dietary and body composition parameters
IGF-1 = insulin-like growth factor-1; FM = fat mass; Regression coefficients were estimated from simple regression analyses; R2 Adjusted; *Analyses were performed using box-cox-transformed values.
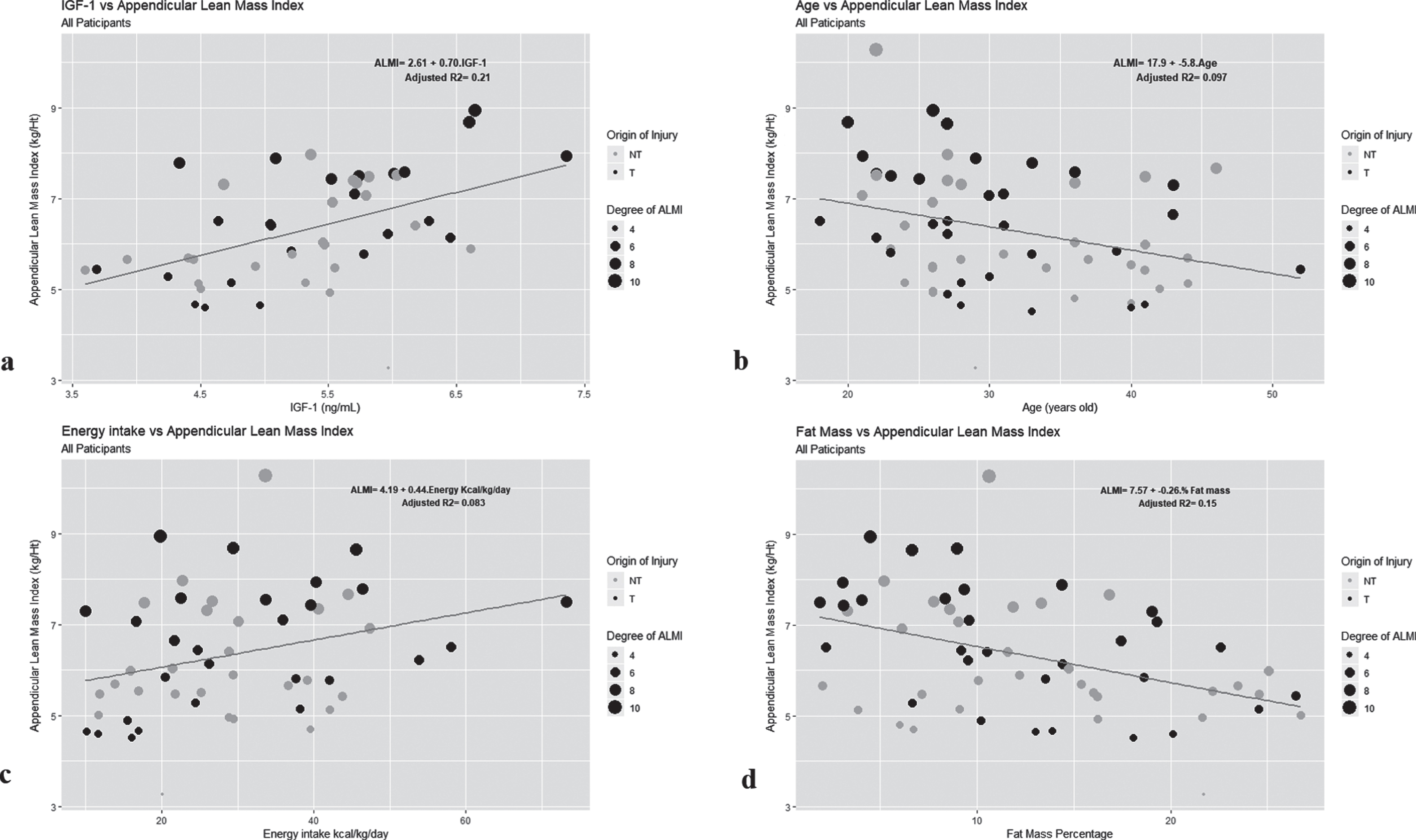
Association of the appendicular lean mass index (ALMI) scores with metabolic and demographic parameters in all participants. All associations were assessed by simple linear regression analyses Model 1 which included the whole sample, Traumatic (T) and Non-Traumatic (NT) SCI. a. Association of ALMI with IGF-1 levels (ng/ml); b. Association of ALMI with age (years old); c. Association of ALMI with daily energy intake (kj/kg/day); d. Association of ALMI with the percentage of FM (%). Light dots: NT-group; Dark dots: T-group. To facilitate the visualization of the graphs, we set the level of appendicular lean muscle mass as the size of the dots (calculated from the ALMI) and referred as the “degree of ALM”; the greater the dot, the greater the reserve of lean muscle tissue and consequently less sarcopenic.
DISCUSSION
In this work, we explored the appendicular lean mass in wheelchair basketball players with traumatic and non-traumatic SCI, and its association with metabolic and demographic factors by using the current available methods given the lack of a validated one for this population. At the same time, we tested their applicability and propose the identification of secondary sarcopenia as well as further investigation to design new techniques suited to their conditions. Remarkably, more than half of our sample presented a low appendicular lean mass index, according to the current classification in general population. The low ALM was not statistically different between groups, despite the higher percentage in NT-group. In the search for an explanation of the high frequency of low ALM in these participants we explored the association with biochemical, demographic and dietary markers. We found a significant association between the ALMI and the percentage of FM, serum levels of IGF-1, age and energy intake. Nevertheless, when the analyses were performed according to the origin of injury, this significance disappeared in the NT-group, and the extremities analyses showed that these associations apparently recline in the lower limbs.
ALMI and Associated Parameters
Consistent with our current findings, the previous publication by our research group using vectorial bioimpedance analysis (BIVA) in these participants, showed increased resistance, lower reactance, and lower angle phase, compared with the able-bodied reference population, which were heightened in the NT-group [9]. The high resistance suggests higher body fat, that might aggravate itself the loss of skeletal mass [15, 16]. To reinforce these prior findings, in the present study we found a significant negative correlation between the percentage of FM and ALMI. Similarly, in another cross-sectional study [17] exploring the frequency of obesity and sarcopenic obesity in community-dwelling non-athlete participants with chronic SCI was found a very high prevalence of sarcopenic obesity. In participants diagnosed with sarcopenic obesity the distribution of ALMI and whole-body fat was respectively lower and greater as the extent of the severity of immobility increased.
It is claimed that people who perform physical exercises are bolstered with their health benefits. If this postulate is extrapolated in the context of our hypothesis, our results would suggest that physical exercise in our participants would favour the T-group more than the NT-group. These findings were not expected, since both origins of SCI present key physiological changes related to sarcopenia. For instance, the physiologic stimulus to growth hormone (GH) release by exercise can be blunted in the SCI physiopathology; the motor neuron is impaired resulting in denervation of muscle cells, which leads to loss of mechanical loading that consequently causes muscle atrophy [2, 13]. Likewise, no significant changes in IGF-1 serum levels and body composition were reported after 6-months chronic activity-based therapy in persons with SCI; [14] therefore, physical exercise may have a lower protective effect, when compared with able-bodied individuals, at least as regards the GH-IGF-1 axis.
In addition, assumption from the BIVA might suggest an impairment in cell membrane integrity, affecting the levels of cell-surface receptor where different essential molecules, e.g. growth factors, binds to [9]. Accordingly, these facts implicate signalling and metabolic pathway alterations that are further affected by dietary changes and inflammatory status triggered by the physiopathology of SCI [2, 18]. Here, we particularly emphasise the identification of synaptic dysfunction in the neuromuscular junctions as a central feature in the pathogenesis of age-related sarcopenia [13]. Given that the pituitary GH would act on the skeletal muscle to stimulate the production of IGF-1 for protein synthesis and tissue growth, in persons with poliomyelitis, such function would be more affected due to the extensive tissue atrophy and the altered brain-muscle response. Moreover, since polio infection occurred primarily during their childhood, this group has undergone the pathological effects of sequelae for a longer time than the traumatic group, and there is a possibility that they had developed post-polio syndrome [19].
Another possible explanation for this fact may be the level of spasticity occurring in each type of injury. In SCI individuals with higher spasticity has been observed increased levels of IGF-1 [20]. Furthermore, one research exploring the correlation between skeletal muscle mass and spasticity in individuals with SCI, [21] exhibited a substantial association between spasticity and the ALMI of lower limbs, after adjusting for possible confounders. Spasticity has been recognised as a protective factor for metabolic alterations, as well as larger muscle atrophy [20, 21]. This factor probably has a different effect on the extremities affected by the injury, in the case of our participants, the lower limbs. On the other side, people with polio sequelae present a form of flaccid paralysis and loss of deep tendon reflexes as a result of muscular atrophy, secondary to the motor neuron degeneration; these factors are responsible for subsequent denervation of muscle fibres [22]. Therefore, spasticity could contribute to the relatively higher IGF-1 levels in the T-group, and to the maintenance of skeletal muscle tissue, and thus a positive strong association with ALMI, which rebounds on an improved metabolic profile, regardless of the practise of physical activity.
Regarding people with post-polio syndrome, the levels of IGF-1 and GH are inconsistent [23–25]. Moreover, the production of GH tends to decrease with age, and with it, IGF-1 levels [18]. Then, residual impairment of this infection might induce an early aging process, heightening the age-related alterations in the neuroendocrine system [24]. Also, as polio survivors age, the metabolic ravages caused by the virus were probably aggravated and consolidated over time [26]. Our findings suggest that in people suffering from SCI the age and the body fat mass may have a different influence in the upper and lower limbs when affected by the neuronal injury. Then a premature aging might be heightened in the tissues undergoing these pathological disturbances. Albeit several and complex triggers influence muscle loss in people suffering from SCI, these postulations might by some means explain the high frequency of low ALM in our participants, which was more pronounced in the NT-group.
Limitations and strengths
In this study we had some limitations. First, we could not have access to the time since injury of participants. Second, the absence of a control group was an important limiting issue to make comparisons with able-bodied population or SCI individuals who do not practise physical activity. Furthermore, the use of methods not validated for this population and the small sample size could be a limiting aspect and may induce bias. Still, this study represents a new perspective bringing about a particular issue from which many people with SCI may be suffering and may be being overlooked with no opportune intervention that might worsen pre-existing and/or associated complications. To our knowledge, there are no similar studies evaluating the ALM in athletes with SCI considering the origin of injury. This work let us recognise the necessity of more studies with improved methods, and possibly device alternative methods to assess the loss of muscle mass and the risk of developing secondary sarcopenia in this population. We also propose the investigation of muscle mass in upper and lower limbs separately adjusted by the time since injury. The identification of this condition and related mechanisms will enable the development of interdisciplinary and effective interventions targeting the underlying causes, given that the sole practise of physical activity certainly does not act as a protective factor. Consequently, it will prevent additional complications to their health status and improve the quality of life.
Conclusion
Based on our findings, people suffering from SCI undergo higher muscle loss putting them at higher risk of developing secondary sarcopenia or even primary sarcopenia as they get older, regardless of the practise of physical activity. We perceived that the potential influencing factors on the loss of muscle mass might differ according to the origin of injury, possibly associated with the metabolic disarrangements that underlie their particular physiopathology. Future studies with higher sample size and enhanced methodology are necessary for the validation of our results, and the identification of new methods to estimate the loss of muscle mass and function, and thus the risk of secondary sarcopenia in this population.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the São Paulo Research Foundation (FAPESP) under Grant number 98/02400-4, and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES) – Finance Code 001, for the scholarship (SMLR and SSMF). The authors thank to Regina Celia da Silva, nutritionist, who collaborated in the data collection.
