Abstract
Background:
Duchenne muscular dystrophy (DMD) is an X-linked recessive genetic muscle disorder. Respiratory muscle function is classically affected in this disease. Ultrasound recently emerged as a non-invasive tool to assess diaphragm function. However, there are only a few studies using diaphragm ultrasound (US) in DMD.
Purpose:
We aimed to assess diaphragm ultrasound patterns in DMD, their relationship with age and their association with home mechanical ventilation (HMV).
Methods:
We included DMD patients followed at Raymond Poincaré Hospital who benefited from diaphragm ultrasound and pulmonary function tests.
Results:
There were 110 DMD patients and 17 male sex-matched healthy subjects included. In all, 94% of patients were permanent wheelchair users. Median body mass index (BMI) was 18 kg/m2. DMD patients disclosed a reduced forced vital capacity (VC) (12% of predicted value), and 78% of patients were on HMV. In patients, right and left diaphragmatic motions on deep inspiration were reduced and end expiratory diaphragm thickness was borderline normal. In patients, right and left diaphragmatic thickening fractions (TF) were reduced 12.7% and 15.5%, respectively. Age and end expiratory thickness were significantly inversely associated (p = 0.005 for the right diaphragm, p = 0.018 for the left diaphragm). Diaphragm TF was significantly inversely associated with age (p = 0.001 for the right side, p < 0.0001 for the left side). Right and left inspiratory diaphragm motions were significantly inversely associated with age (p < 0.0001).
Conclusion:
This study describes the severity of diaphragm dysfunction in patients with DMD. Diaphragm US may be a non-invasive outcome measure for DMD.
INTRODUCTION
Duchenne muscular dystrophy (DMD) is an X-linked recessive genetic disorder caused by mutations in the dystrophin gene [1]. Respiratory muscles are classically involved, leading to a restrictive respiratory pattern [1, 2]. Currently, pulmonary function tests (PFT), which include forced vital capacity (VC), are used to assess lung function [3]. In childhood DMD, VC has been reported to increase with age through 10 years and then to generally stabilize from 10–18 years before declining in adulthood [4]. VC provides a global evaluation of pulmonary function, but the direct assessment of diaphragm function relies on trans-diaphragmatic pressure measurement [5]. Recently, an international workshop proposed a subset of functional respiratory tools for the evaluation of future drugs for DMD [6]. In the meantime, radiological techniques that include magnetic resonance imaging (MRI) and ultrasound emerged as biomarkers for diaphragm analysis in DMD [7–10]. Ultrasound is a non-invasive, radiation-free, accurate and reproducible tool allowing the assessment of diaphragm anatomy and function at the bedside [11–15]. In healthy patients, diaphragm ultrasound has been reported to be correlated with lung function and inspiratory muscle strength [16]. There are few studies on diaphragmatic ultrasound patterns in DMD. In the paediatric DMD population, de Bruin et al. [17] reported an increase in diaphragmatic thickness at end expiration. Laviola et al. [18] reported a decrease in end expiratory diaphragm thickness with age in young DMD. However, the authors focused their studies only on the right hemi diaphragm thickness and did not report data about diaphragm motion.
In this study, we aimed to provide the following: diaphragm ultrasound patterns in DMD and their relationship with age, and diaphragm ultrasound patterns in DMD patients with and without home mechanical ventilation (HMV).
METHODS
Subjects
For this retrospective cross-sectional study, we included DMD patients followed at the Home Mechanical Ventilation Unit of the Raymond Poincare University Hospital (Garches-France), a tertiary neuromuscular reference centre specializing in the evaluation and management of cardio-respiratory function of patients with neuromuscular disorders. We included DMD patients with diaphragm ultrasound that involved at least assessment of the right and/or left hemi-diaphragm motion and assessment of the right and/or left diaphragm thickness and thickening. From the medical records, we collected age, body mass index (BMI), peripheral skeletal muscle failure (Walton score), the presence of arthrodesis, the presence of scoliosis in patients without arthrodesis, steroid drug use, left ventricular ejection fraction (LVEF), supine VC, sitting VC, maximal inspiratory pressure (MIP), peak expiratory flow (PEF) and diaphragmatic US data available. In this unit, diaphragmatic US imaging is performed in patients consulting for echocardiography. The study was performed in compliance with the ethical principles formulated in the declaration of Helsinki and was approved by the French Regulatory Board (Commission Nationale de l’Informatique et des Libert
A group of 17 healthy male adults without any history of cardiopulmonary disease was investigated as controls by the same operator (AF) using the same diaphragmatic US protocol to obtain reference values for the investigated parameters.
Diaphragm ultrasound
Diaphragm ultrasound (Vivid E9, GE Medical Systems) was performed in patients in a supine position (45°) with standard measurements. At the Garches institution, two approaches for the diaphragmatic exam were used: analysis of the dome motion from a subcostal view and analysis of the diaphragm thickness and thickening from the apposition zone.
Diaphragm motion analysis from a subcostal view
From the anterior subcostal view, diaphragm ultrasound imaging was performed using a sector probe M5Sc-D (1.5–4.6 MHz). The right diaphragm was analysed through the liver window, and the left diaphragm was analysed through the spleen window. The transducer was placed in the anterior subcostal region with the US beam reaching the posterior part of the hemi-diaphragm [7]. From the B mode, the diaphragm was identified as a hyper-echogenic line between the interface of the lung and the liver [14]. From an applied M mode, we recorded the amplitude of excursion during quiet respiration (spontaneous breathing diaphragmatic motion, SBDM) and during deep inspiration (diaphragmatic inspiratory motion, DIM) (Fig. 1) [7, 20].
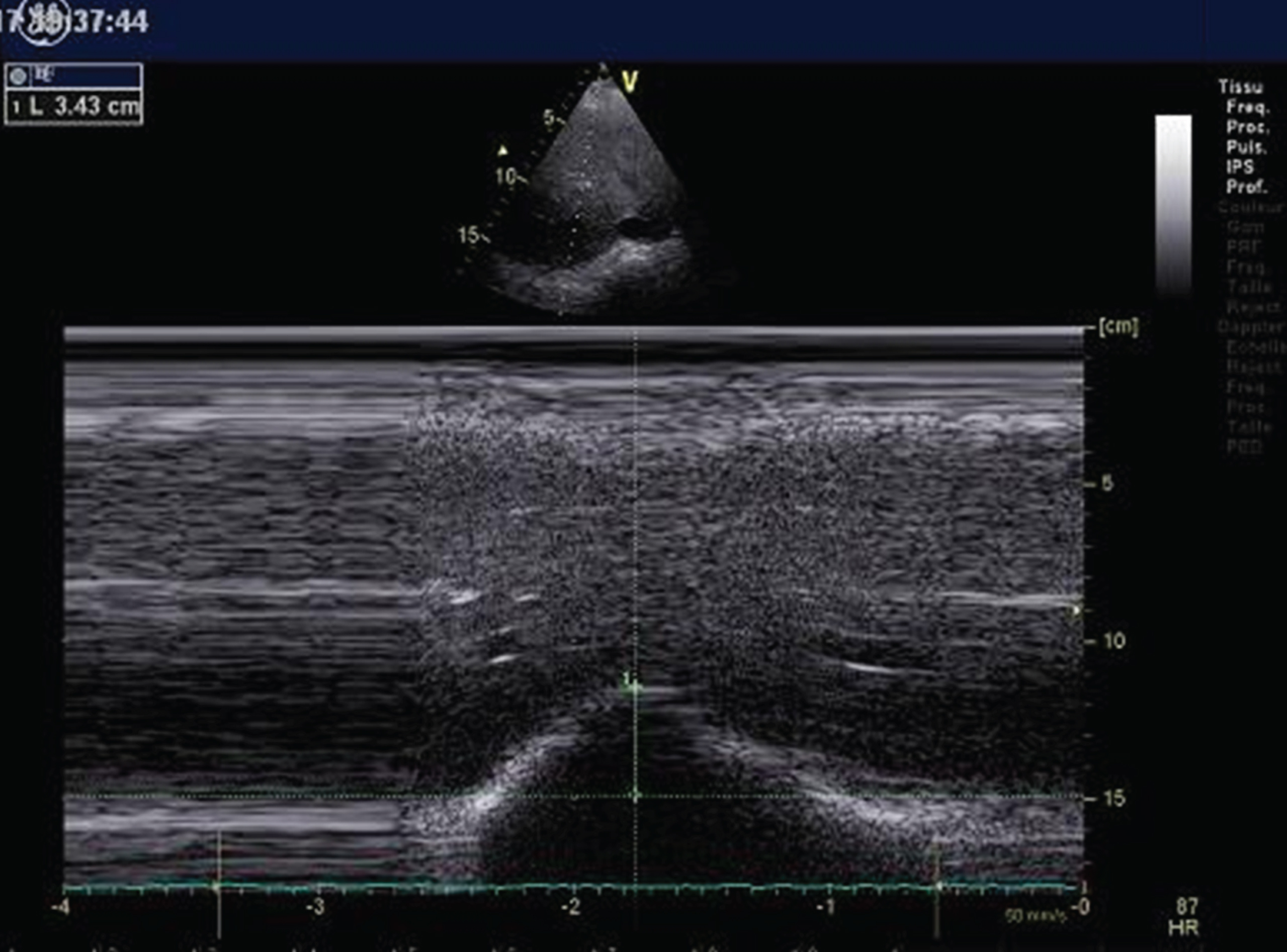
Assessment of the right hemi diaphragm motion from the subcostal view. Here is an M-mode ultrasound deep inspiration motion (34 mm) in a young Duchenne muscular dystrophy (DMD) patient.
Analysis of the apposition zone
Apposition zone (AZ) analysis with ultrasound allows the measurement of diaphragm thickness and thickening using a linear transducer 9L-D (2.4–10 MHz). The linear probe was positioned within the mid-axillary line, perpendicularly to the chest wall [12]. The diaphragm was identified beneath the intercostal muscles, as it is composed of a hypo-echogenic muscle tissue located between two hyper echogenic lines (the pleural line and the peritoneal line) (Fig. 2) [5, 15]. The diaphragmatic thickening fraction (TF) was defined as the ratio of the thickness at end inspiration - thickness at end expiration) / thickness at end expiration.
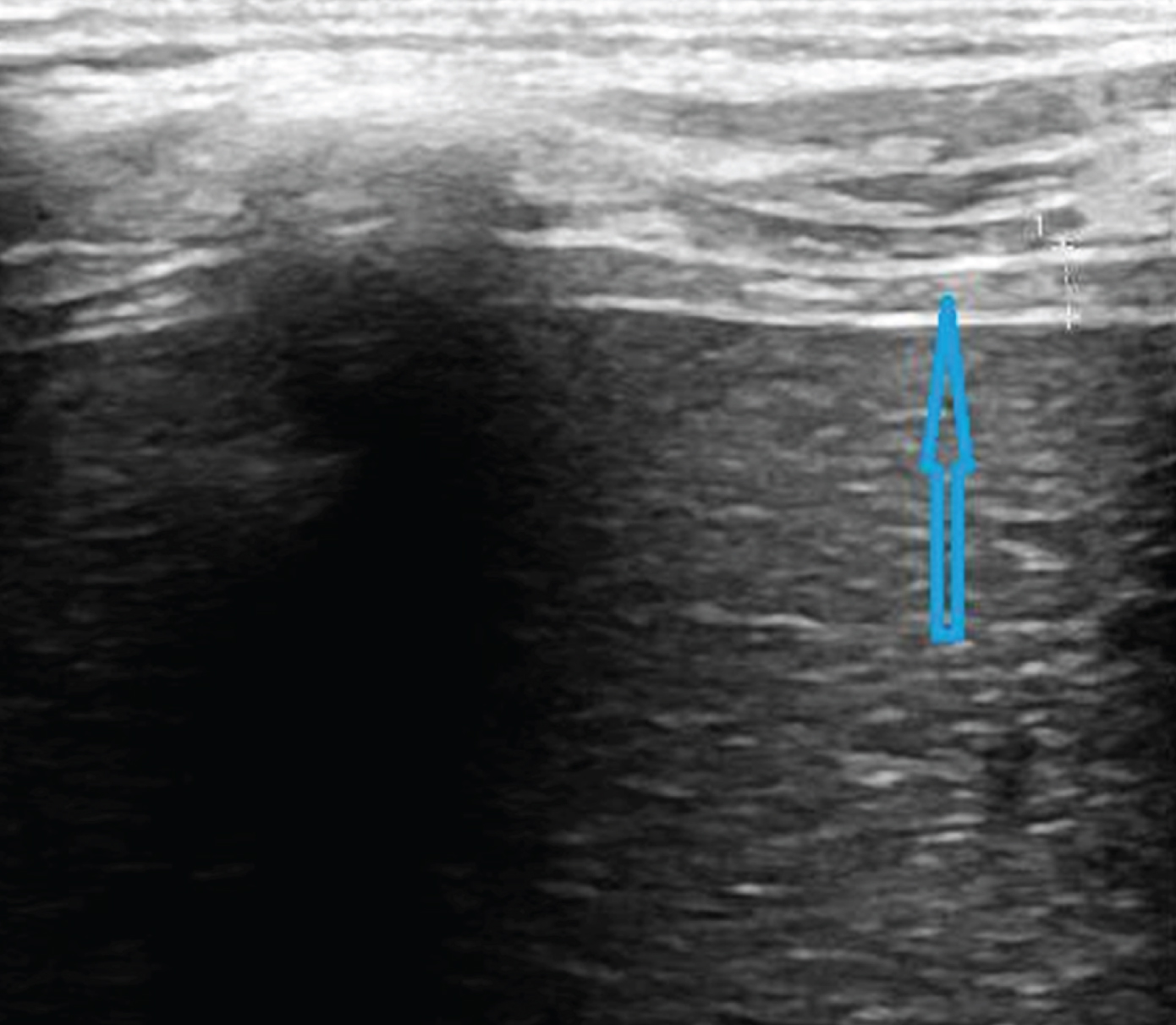
Assessment of the right hemi diaphragm from the apposition zone using ultrasound. Here is the right normal diaphragm end expiratory thickness (2.2 mm) in a young DMD patient. DMD = Duchenne muscular dystrophy.
Respiratory function and ventilation
Spirometry variables and lung volumes were routinely measured using a Vmax 229 SensorMedics System (Yorba Linda, CA, USA) using standard guidelines while the patient was comfortably seated [21]. In patients on HMV, we collected the type of ventilation (invasive or not), the duration of ventilation (nocturnal +-partial diurnal <24 h or permanent 24 h/24 h) and the tidal volume of the respiratory machine.
Statistical analysis
Continuous variables were described by the median±interquartile range (IQR) and compared by the Wilcoxon test. Dichotomous or categorical variables were described by the number of subjects and percentage and compared by Fisher’s exact test. The associations between diaphragmatic continuous parameters and respiratory data were explored by nonparametric Spearman’s correlation coefficients. Bland-Altman plots were established providing the mean bias and 95% limits of agreement for assessment of the intra-observer variability in measurement of diaphragmatic ultrasound thickness and diaphragmatic US motion. Statistical analysis was performed using R® (http://www.R-project.org/).
RESULTS
Study population
There were 110 DMD patients and 17 healthy subjects included in this study. DMD patients were mostly permanent wheelchair users (94%). Median age was 24 years [21; 31], and median BMI was 18 kg/m2 [14.4; 24.8]. A total of 14% of patients were currently on steroid therapy, and 80% of patients had a history of arthrodesis surgery. DMD patients disclosed a severe respiratory insufficiency pattern with a median VC at 12% [8–26] of predicted. There were 78% patients on home mechanical ventilation (HMV). Inspiratory muscles strength was significantly affected as indicated by the MIP value (15 cm H2O).
Diaphragm motion
Right spontaneous breathing diaphragmatic motion (SBDM) was altered with a median of 4 mm [2–6.3] compared to 15.6 mm [13.5–20] in controls. Also, right diaphragmatic motion at deep inspiration was altered in patients with a median of 9 mm [4–24] versus 84 mm [72.5–91] in controls.
Left spontaneous breathing diaphragmatic motion (SBDM) was altered with a median of 4 mm [2–5.7] compared to 12 mm [10–14.5] in controls. Also, left diaphragmatic motion at deep inspiration was altered in patients with a median of 6.8 mm [4–12] versus 75 mm [70–82.5] in controls.
Diaphragm thickness and thickening
The right median right end-expiratory thickness was 1.4 mm [1–1.7] in patients, and we found a severe alteration of the right TF with a median TF of 12.7% [10; 25] vs 67% [50.5–80.5] in controls.
For the left apposition zone, diaphragm thickness was 1.3 mm [1.1–1.7] and left hemi–diaphragm TF was severely decreased with a median TF of 15.5% [12.1; 37.9] vs 73% [59.5–113] in controls. Table 1 summarizes the clinical characteristics, respiratory function and diaphragmatic US imaging of patients and controls.
Baseline characteristics of patients and controls
Data are expressed as the median [interquartile range] or number (percentage). * = end expiratory; HMV: mechanical ventilation; LVEF: left ventricular ejection fraction (%); FVC: forced vital capacity (%); MIP: maximal inspiratory pressure; PEF: peak expiratory flow; SBDM: spontaneous breathing diaphragmatic motion; DIM: diaphragmatic inspiratory motion; TF: thickening fraction of the diaphragm; BMI: body mass index.
Diaphragm ultrasound measurement agreement
We found a higher concordance in test–retest of diaphragm ultrasound measurements for all measurements from the subcostal view and the apposition zone (Figs. 3–6).
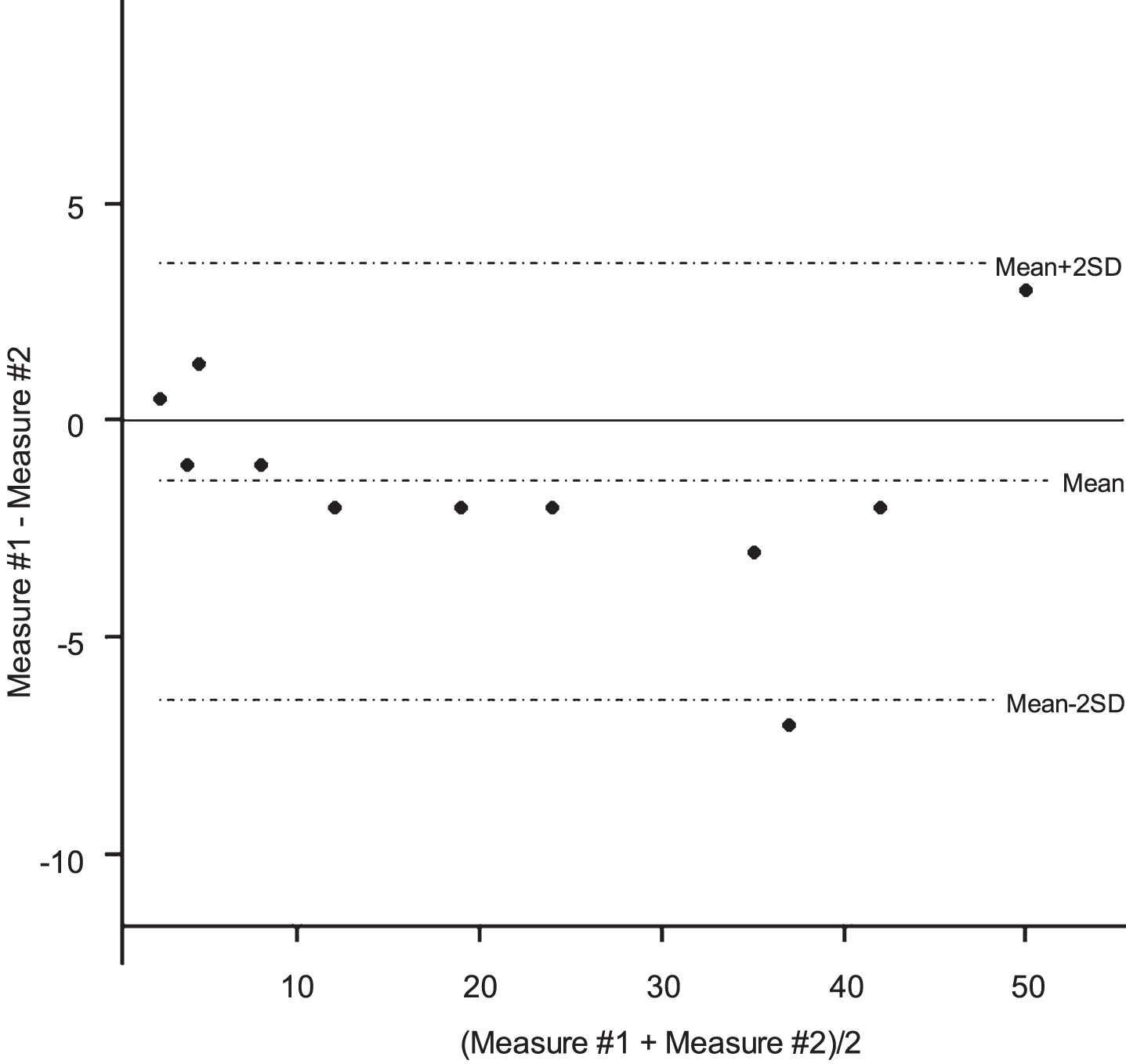
Bland-Altman plots for the right hemi diaphragm motion measurement using ultrasound.
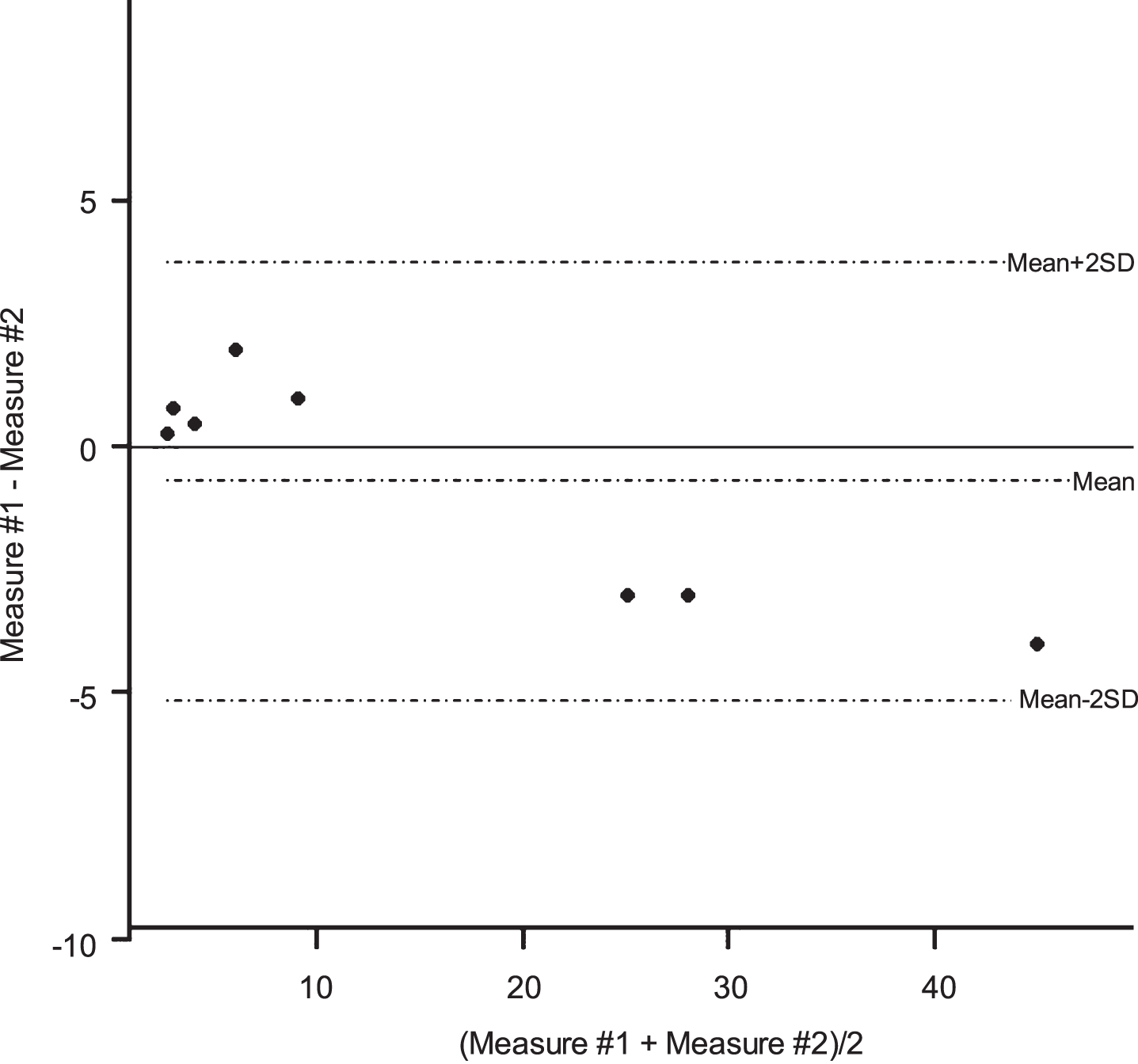
Bland-Altman plots for the left hemi diaphragm motion measurement using ultrasound.
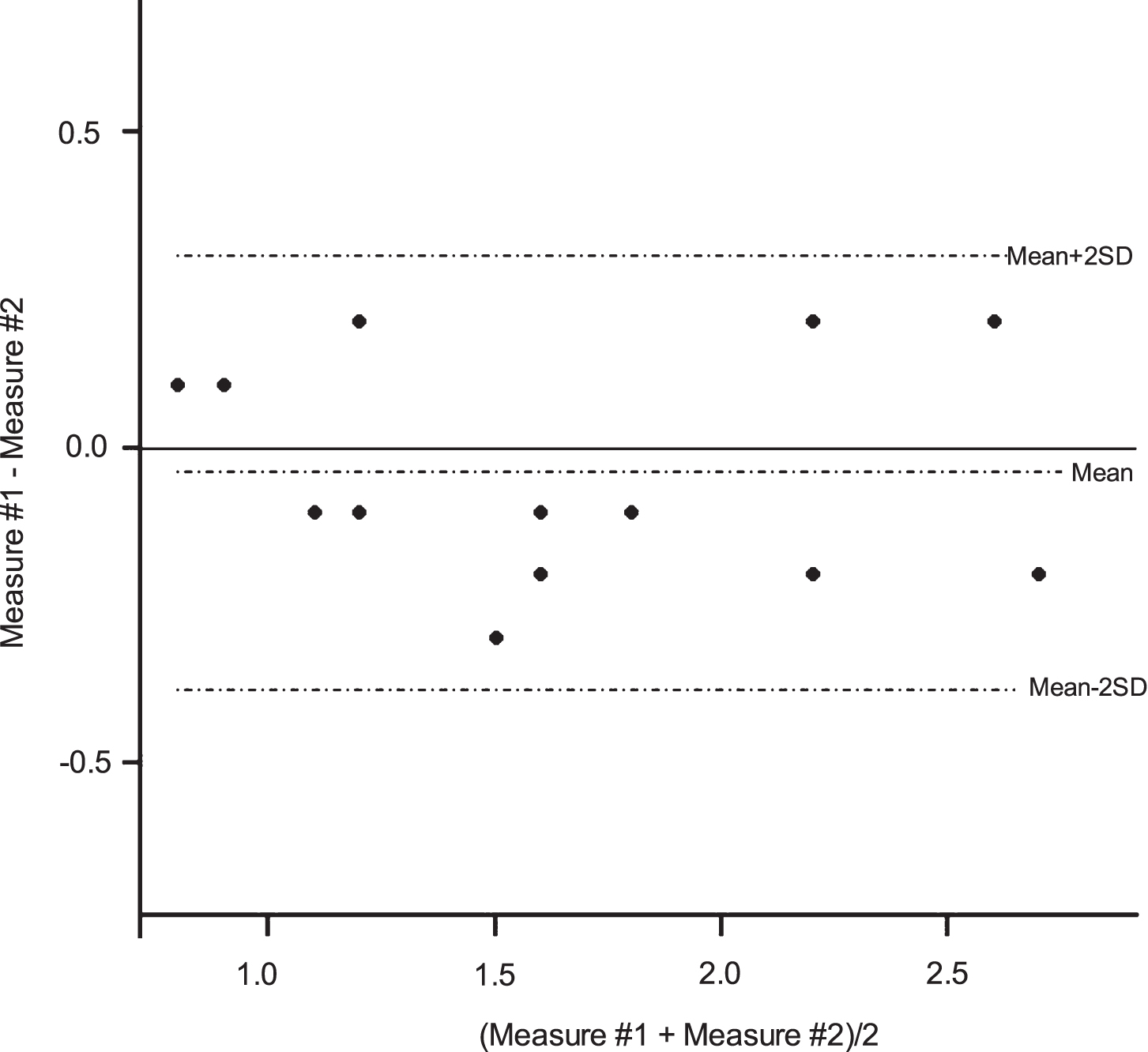
Bland-Altman plots for the right diaphragm thickness measurement using ultrasound.
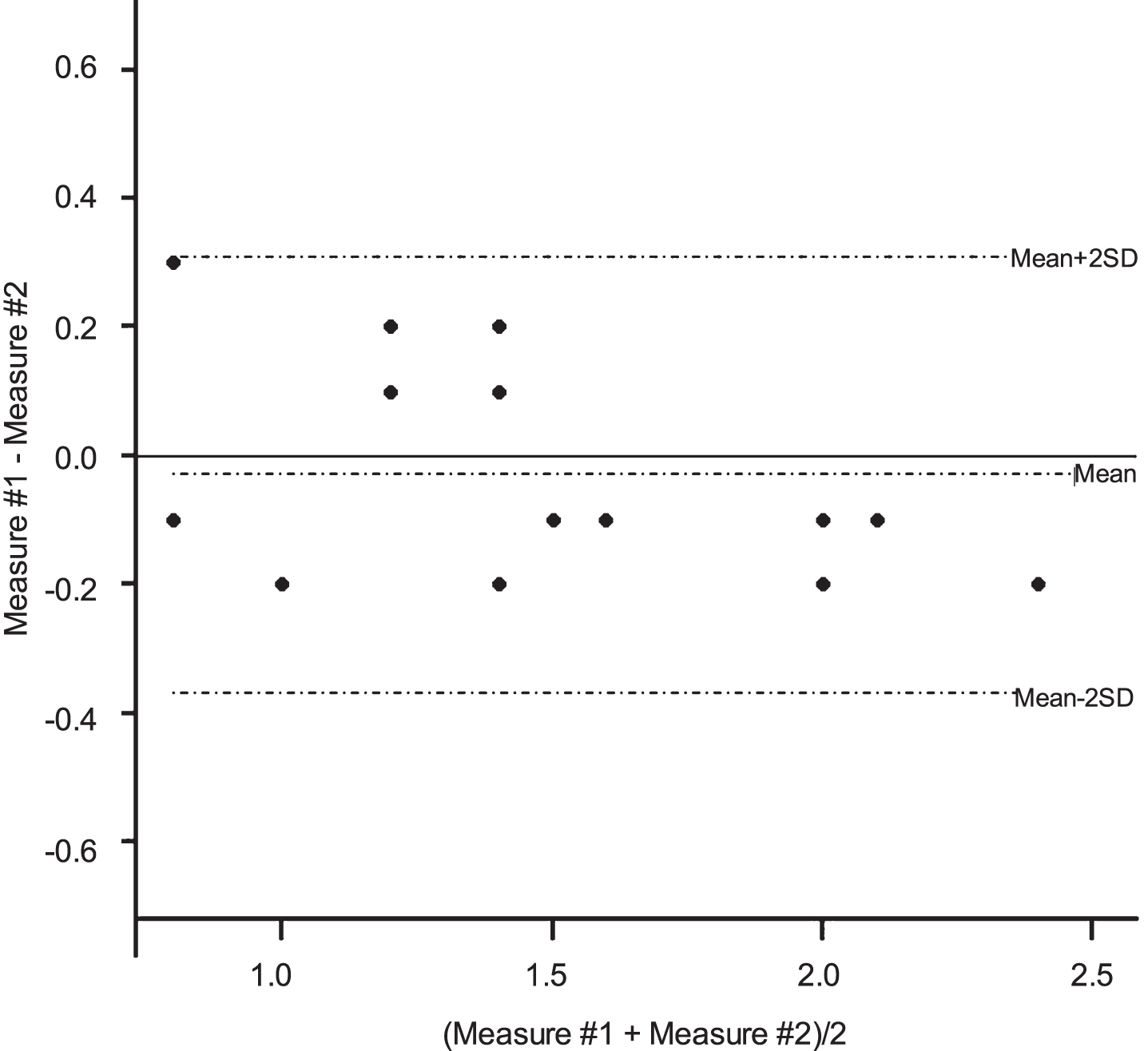
Bland-Altman plots for the left diaphragm thickness measurement using ultrasound.
Relationship between age and diaphragm ultrasound
Diaphragm TF was significantly inversely associated with age (r = –0.47 p 0.001 for the right side, r = –0.75 p < 0.0001 for the left side) (Figs. 7 and 8). Deep inspiratory diaphragm motion was also inversely significantly associated with age (r = –0.62 p < 0.0001 for the right diaphragm, r = –0.6 p < 0.0001 for the left diaphragm) (Figs. 9 and 10).
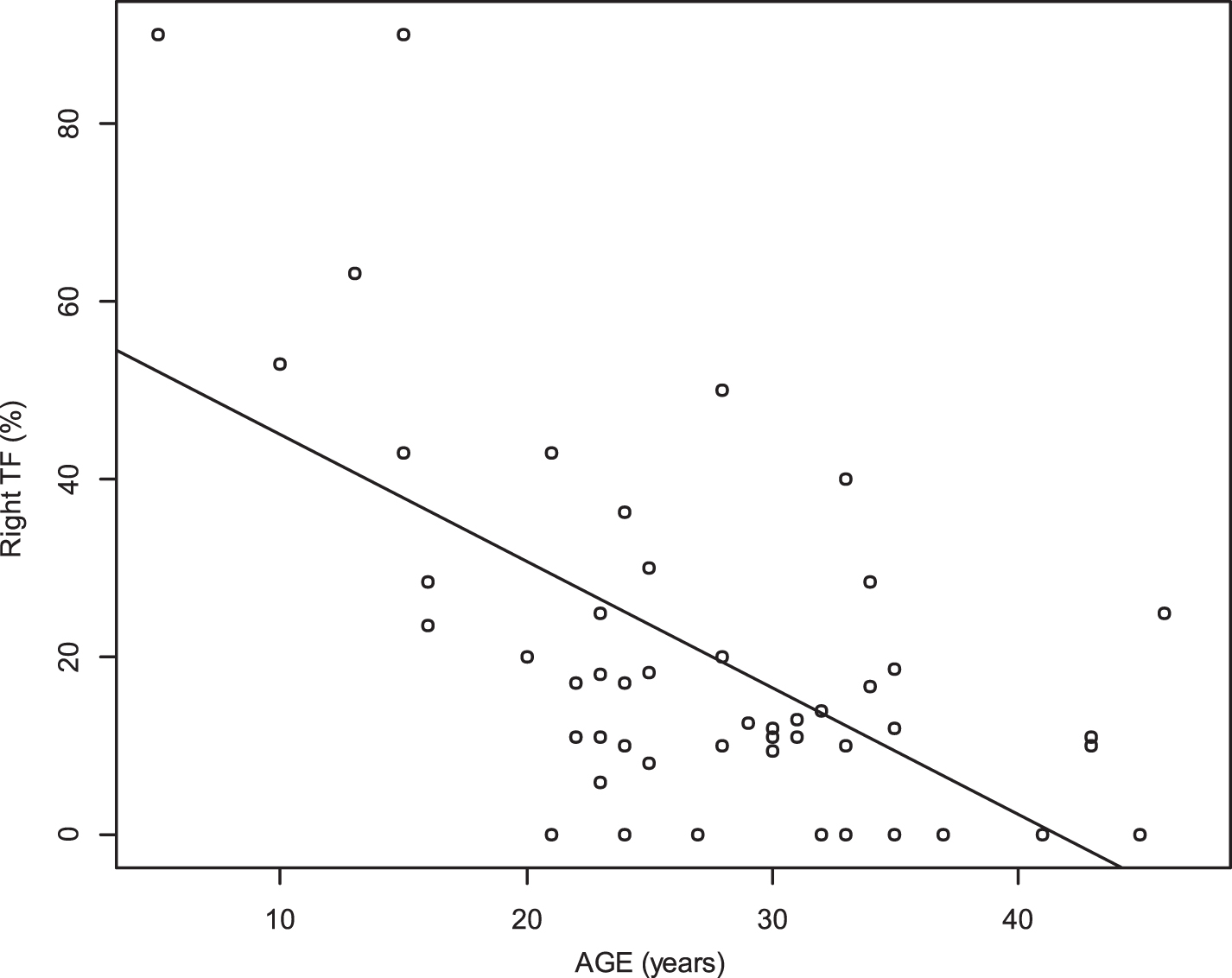
Relationship between age and thickening fraction (TF) of the right diaphragm (r = –0.47, p 0.001).
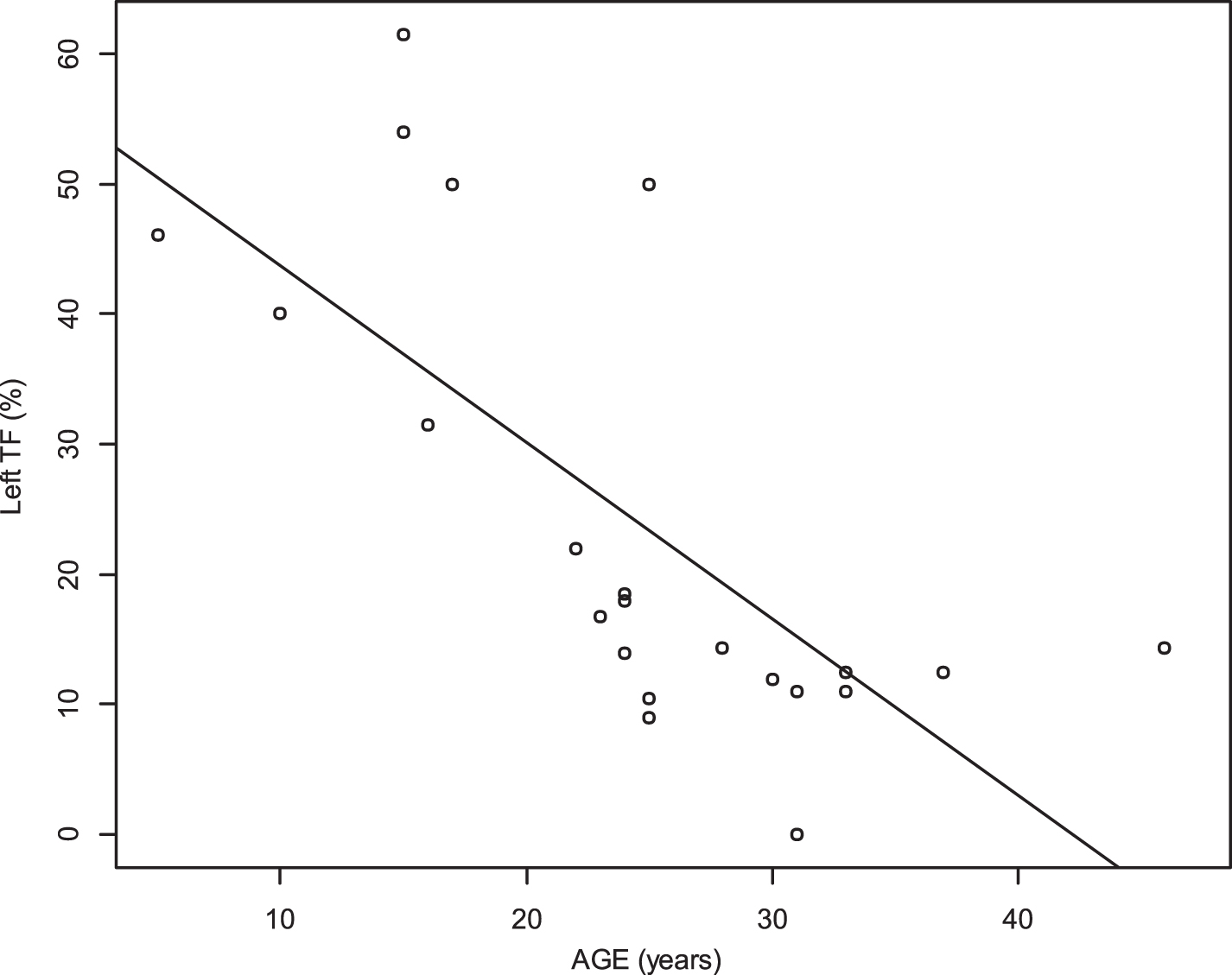
Relationship between age and thickening fraction (TF) of the left diaphragm (r = –0.75, p < 0.0001).
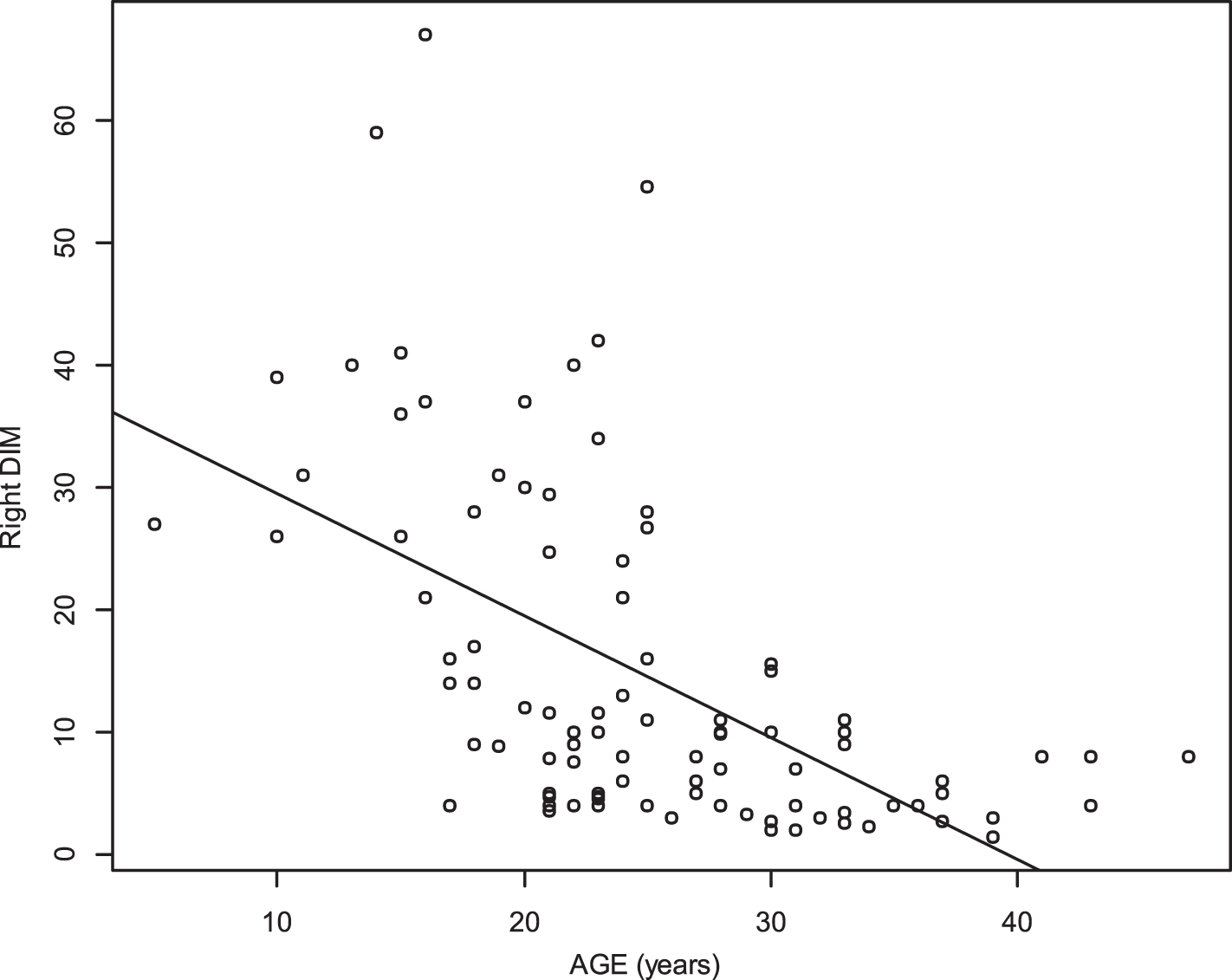
Relationship between age and deep inspiration motion (DIM) of the right diaphragm (r = –0.62 p < 0.0001).
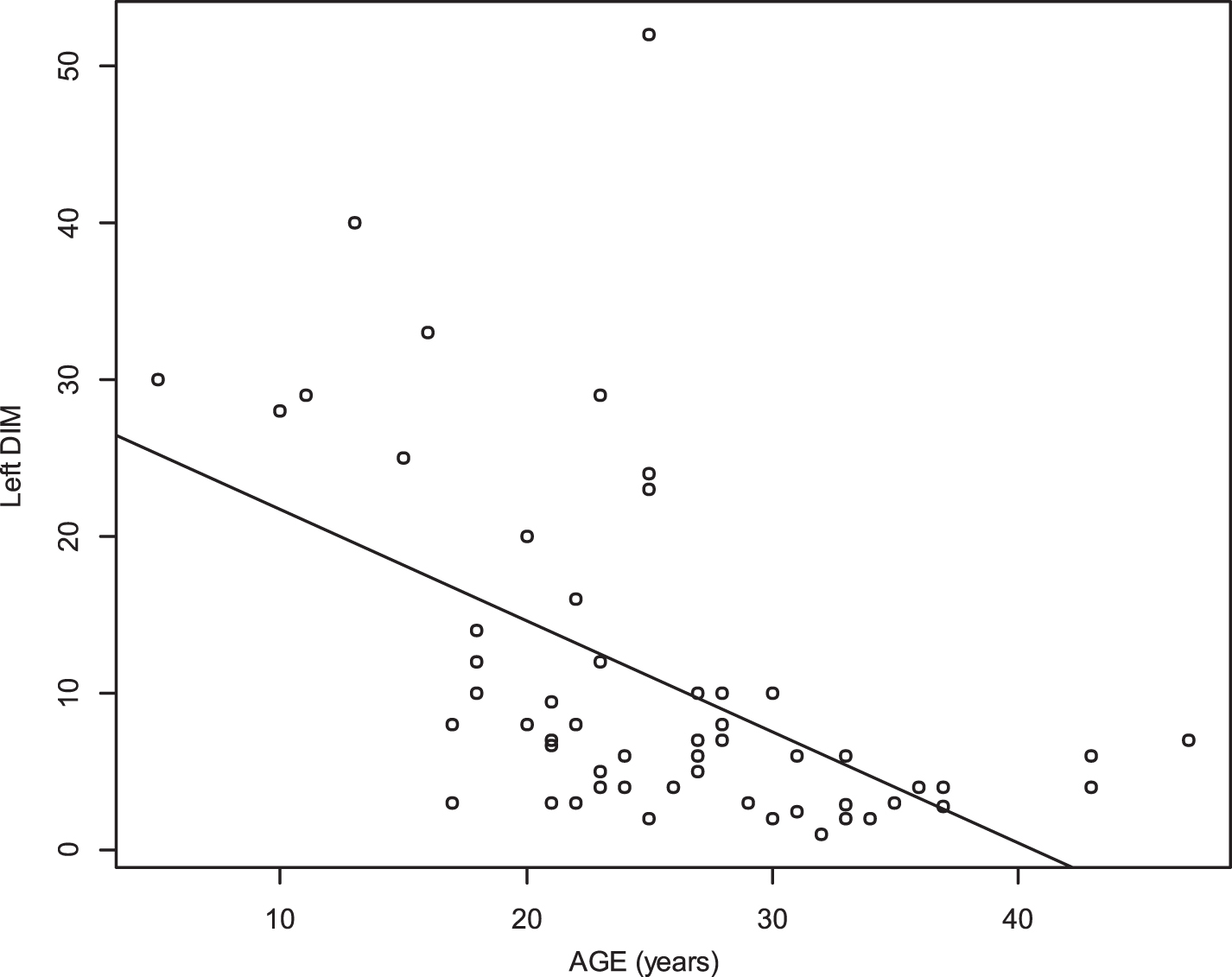
Relationship between age and deep inspiration motion (DIM) of the left diaphragm (r = –0.6 p < 0.0001).
Age and end expiratory thickness were significantly inversely associated (r = –0.33 p 0.005 for the right diaphragm, r = –0.35 p 0.018 for the left diaphragm) (Figs. 11 and 12).
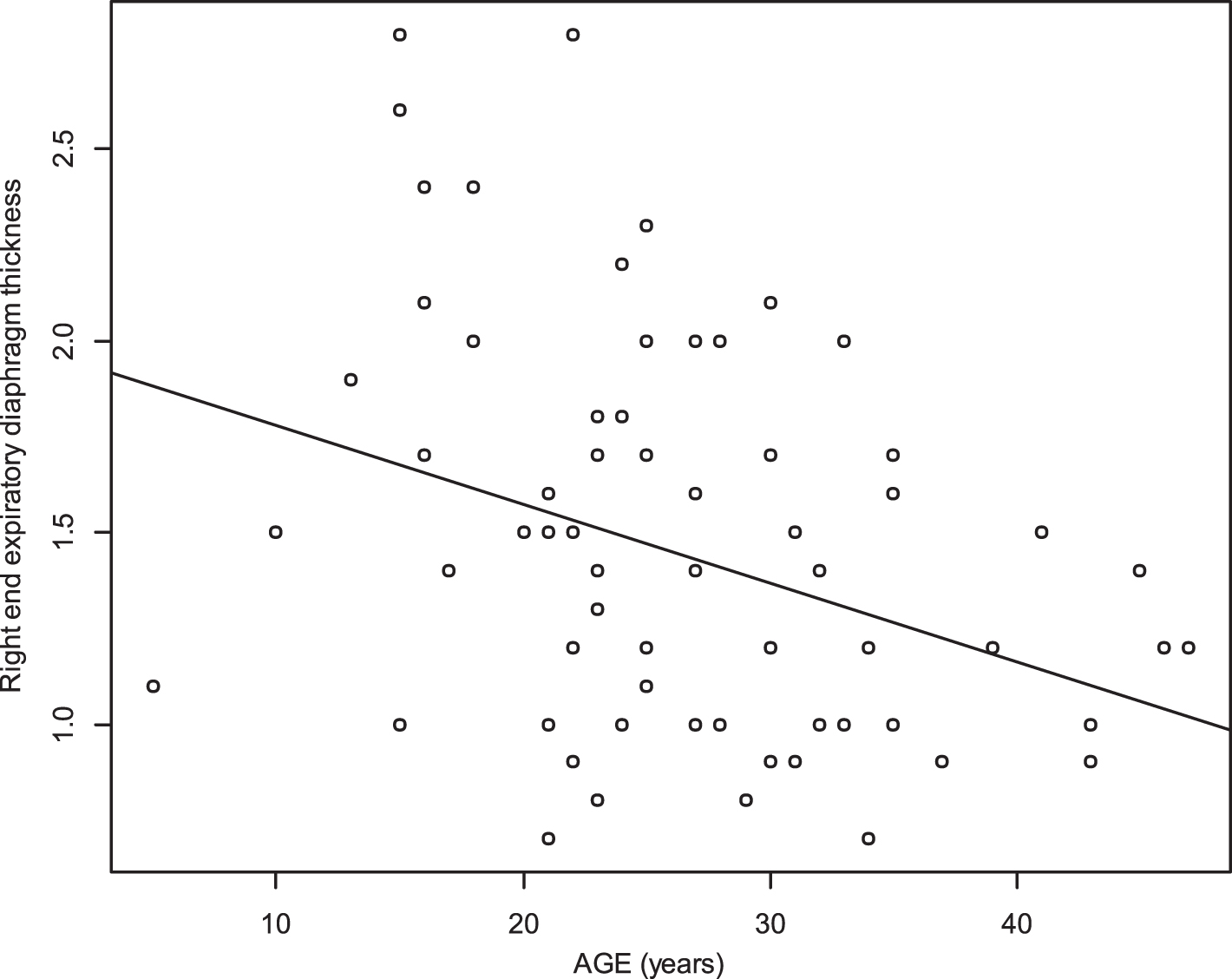
Relationship between age and right end expiratory diaphragm thickness (r = –0.33, p 0.005).
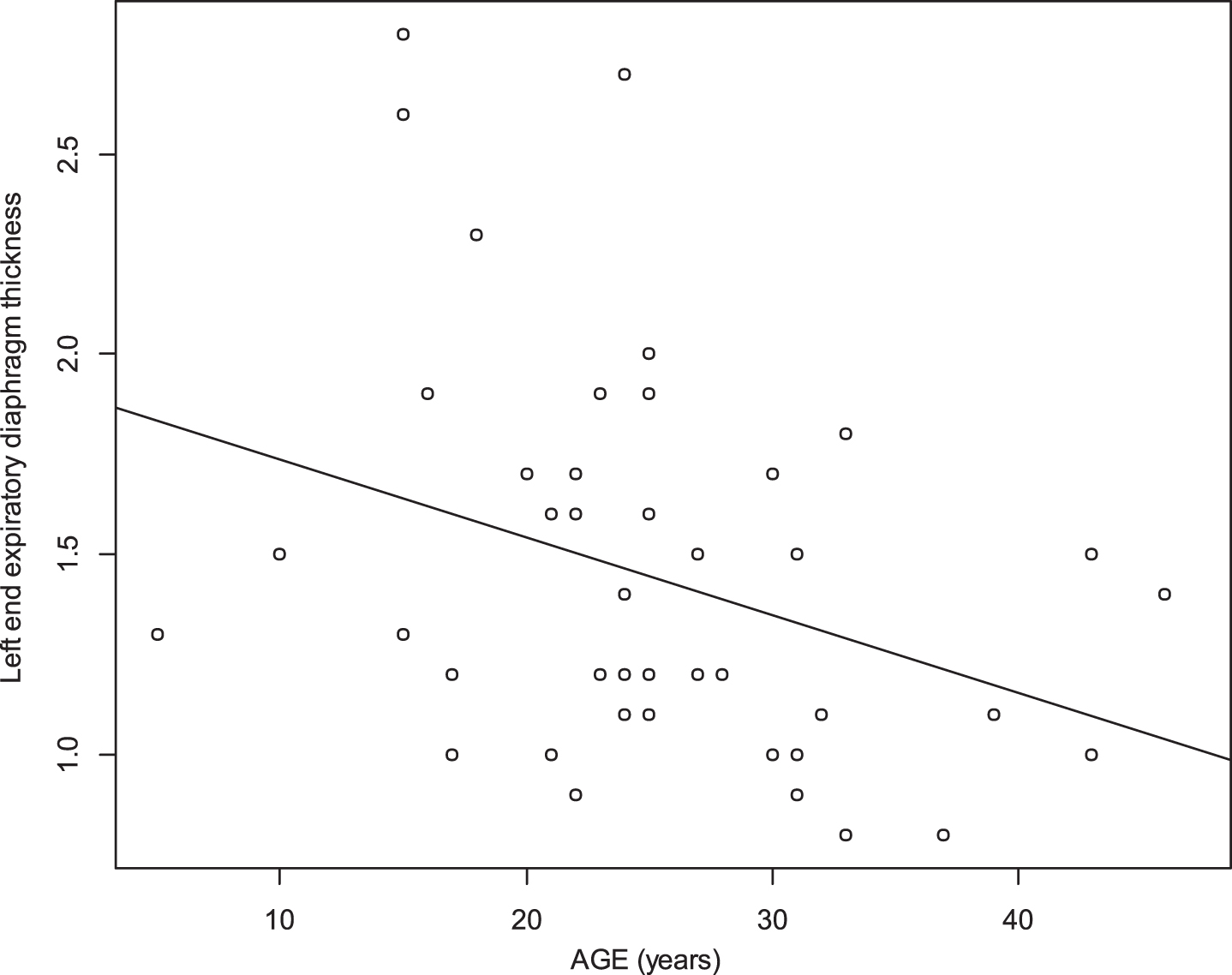
Relationship between age and left end expiratory diaphragm thickness (r = –0.35, p 0.018).
Table 2 summarizes the diaphragm thickness and thickening in patients aged < 21 years vs patients aged between 21 and 30 years vs older patients (>30 years). Inspiratory muscle strength was affected in patients with a gradient decline, as indicated by the decrease in MIP.
Distribution of diaphragm thickness and thickening in patients at three different ages
Data are expressed as the median [interquartile range]. MIP: maximal inspiratory pressure; TF: diaphragm thickening fraction; n = number of patients.
Ventilation status and diaphragm ultrasound
Patients receiving ventilation had reduced diaphragm thickness and thickening compared with patients without ventilation. Also, inspiratory strength was reduced with a gradient decline depending on ventilation status, as indicated by the decrease in MIP. Table 3 summarizes the diaphragm thickness and thickening in patients by ventilation status.
Distribution of diaphragm thickness and thickening in patients according to ventilation status
Data are expressed as the median [interquartile range]. MIP: maximal inspiratory pressure; TF: diaphragm thickening fraction; n = number of patients; Intermittent ventilation = nocturnal ventilation+-partial diurnal (<24 h); Permanent ventilation = ventilation 24 h/24 h.
Tidal volume and diaphragm US
Tidal volume was not correlated with right diaphragm TF (p = 0.8) and was not correlated with left diaphragm TF (p 0.9).
Scoliosis and diaphragm ultrasound
In our study, 15 patients did not undergo arthrodesis. Among them, 5 patients were found to have scoliosis. MIP was significantly reduced in patients with scoliosis (17 cmH20) vs patients without scoliosis (34.5 cmH20). Right diaphragm TF was available in 4 patients without scoliosis and in 1 patient with scoliosis and was 45.7% and 10%, respectively. Left diaphragm TF was available in 3/10 patients without scoliosis and 1/5 patients with scoliosis, and was 46% and 18%, respectively.
DISCUSSION
The main findings of our study were as follows: Both right and left inspiratory motions of the diaphragm were reduced in DMD patients and decreased with age. Both right and left diaphragmatic thickening fractions were reduced and decreased with age. Patients with mechanical ventilation had reduced diaphragm motion, thickness and thickening fraction. Patients with scoliosis without a history of arthrodesis had reduced inspiratory muscle strength (MIP) and reduced diaphragm TF.
Respiratory muscles had classic weakness in DMD (4, 22, 23 and 24), and the diaphragm can be assessed indirectly with the measurement of MIP. Ultrasound can provide information about diaphragm structure and function. Our study provides a global view of diaphragm motion, thickness and thickening in a large population of DMD patients. Median diaphragm end expiratory thickness was normal (1.9 mm) in patients aged < 21 years and was reduced in older patients (1.3 mm after 21 years and 1.2 mm after 30 years). Our study provides clinical values for diaphragm US for DMD patients and its association with age. From a clinical trial perspective, it may be helpful to characterize this population with ultrasound in addition to classic functional respiratory tests [25].
Undernutrition is frequent in adult patients with DMD due to swallowing disorders and muscle weakness. Also, DMD patients are typically on mechanical ventilation because of the presence of chronic respiratory muscle impairment. In our study, the median BMI of patients was reduced (18 kg/m2), and a reduction in diaphragm motion in underweight subjects was reported [14]. Also, diaphragm muscle mass was reported to be 43% lower in underweight patients [26]. Therefore, in addition to dystrophic process alteration, diaphragm thickness and thickening and motion can be affected by the presence of an undernourished state and the presence of mechanical ventilation in DMD.
Mechanical ventilation is known to affect diaphragm muscle function. In the Intensive Care Unit (ICU), diaphragmatic dysfunction is frequent and occurs rapidly after introducing mechanical ventilation [27, 28], which is the concept of ventilator-induced diaphragmatic dysfunction (VIDD) [28]. Patients with VIDD have typical diaphragmatic impairment and thinning [29]. In ICU situations, it has been reported that there is a 6% per day decrease in diaphragmatic thickness in patients on mechanical ventilation [30] and a rapid atrophy of diaphragm fibres [31]. Not surprisingly, in our study, we found that patients with HMV had reduced diaphragm thickness, thickening and motion. In mdx mice, short-term mechanical ventilation has been reported to induce diaphragmatic dysfunction with oxidative stress and the upregulation of pathways involved in muscle wasting and proteolysis [32]. However, it is difficult to address a causal relationship between diaphragm failure due to dystrophinopathy and diaphragm failure induced by the mechanical ventilation since ventilation is indicated in patients with diaphragm failure. Also, in muscular dystrophy, the fibrosis of the ribcage muscles and the presence of chest deformities may modify chest wall compliance that, in turn, may affect diaphragm motion during inspiration [10]. In our study, patients with scoliosis (who had no arthrodesis) had reduced inspiratory muscle strength compared to those with arthrodesis and reduced diaphragm TF, which was probably due to the mechanical dysfunction of the diaphragm in the context of scoliosis.
Finally, we did not find any relationship between tidal volume and diaphragm TF. Future prospective studies may be helpful for assessing the interaction between the mechanical ventilation setting and diaphragm function.
Limitations of the study
Our study had limitations due to its retrospective design and monocentric exploration. All measurements were performed by the same operator, and we therefore do not have data on inter-observer variability. These data have, however, been described previously [33]. Another limitation concerns diaphragm measurements in patients with permanent mechanical ventilation, since the diaphragm motion, in this group, results from the association of the diaphragm contraction and the passive displacement of the diaphragm induced by the pressure due to the ventilator. Finally, data are lacking about the long-term effects of steroids on diaphragms in our study.
CONCLUSION
This study describes the spectrum of diaphragm ultrasound patterns in DMD at different ages and the diaphragm spectrum of patients on home mechanical ventilation. Diaphragm US may be an augmentative, non-invasive outcome measure for DMD.
FINANCIAL/NONFINANCIAL DISCLOSURES
None.
