Abstract
Muscle nicotinic acetylcholine receptor (nAChR) mutations can lead to altered channel kinetics and neuromuscular junction degeneration, a neurodegenerative disorder collectively known as slow-channel syndrome (SCS). A multivariate analysis using running wheels was used to generate activity profiles for a variety of SCS models, uncovering unique locomotor patterns for the different nAChR mutants. Particularly, the αL251T and
Keywords
INTRODUCTION
The association between ionotropic receptor gating and disease has been historically studied
The nAChR is a heteropentamer ligand-gated channel composed of alpha (α), beta (β), delta (
Nevertheless, the behavioral profile of SCS models is limited and what has been described does not fully encompass the physiological presentation of SCS. Locomotor activity and its related metrics are often overlooked, ignoring the impact of conditions such as SCS. Hence, we characterized a panel of SCS mice using running wheels to examine whether these nAChR channelopathy models exhibit locomotor impairment and to what extent these alterations correlate with their pathophysiology. Here we present such behavioral characterization, including three mutations that span the acetylcholine binding subunit of the receptor (αV249F, αL251T and αC418W), two mutations in the delta subunit (
MATERIAL AND METHODS
Locomotor activity data was collected from a panel of mice expressing various muscle nAChR mutations, using running wheels. The University of Puerto Rico Institutional Animal Care and Use Committee (IACUC) approved the protocols for these experiments, which include
The experimental setup and data acquisition software has been previously described and validated by our group [18]. In brief, we utilized mouse single activity wheel chambers (Lafayette Instrument, model 80820) as a home cage, in which the animals remained for the duration of the experiment with free access to water and food. Animals were acclimated to the activity monitoring cages for 7 days with free access to running wheels, before we collected our data. Every 24 hours, food and water levels were verified and replenished as necessary. We monitored and recorded their activity using Lafayette’s Activity Wheel Monitor Software. This allowed us to record the average velocity (m/s) and duration (s) of activity events throughout a period of 24 hours, at a frequency of 1 data point per second. The data gathered from the monitoring software was sorted, mined and exported using a custom MATLAB code (see supplemental material for pseudocode), that was modified from our group’s previous work [18]. Subsequently, we organized all data points using their timestamp and their duration (seconds) and average velocity (m/min), calling an activity event when we detected consecutive non-zero velocities lasting more than 2 seconds. This allowed us to identify individual activity events throughout the 24 hours of data collected.
For single variable analysis, we filtered the data to select the activity events in the top 50% for each parameter: distance (m, meters), duration (s, seconds), and velocity (m/min, meters/minutes). We also measured: total time spent running, total distance traveled and sum of events. When a normal distribution could be determined using the Kolgomorov-Smirnov test, a parametric One-way ANOVA analysis was performed followed by a Bonferronipost-test. Statistical corrections to account for non-Gaussian distributions were used when necessary, Kruskal-Wallis Test and Dunn’s post-test. One-variable statistical analyses were performed using GraphPad PRISM 5.0®. Furthermore, three-dimensional activity profiles were generated for individual animals of each SCS strain using the 3D Histogram Macro in SigmaPlot® v. 12.0 to produce 10×10 matrices that were used to evaluate data distribution, as described previously by our group [18]. From these matrices, we selected 3×3 regions that encompassed consistent and characteristic populations of high velocity and prolonged duration events from our wild-type strains to compare to our mutant panel. We determined the region of interest (ROI) by identifying the coordinate with the highest frequency and selecting the 3×3 square surrounding it. The regions of interest were analyzed as a proportion of the total events in the heat map.
For statistical comparison, experimental controls were age- and gender-matched while being selected depending on the genetic background of the animals: FVB/NJ mice were used as a control for αV249F, αL251T,
RESULTS
The phenotype of nAChR mutation induced SCS animal models has been partially characterized focusing on grip strength and paradigms such as the hanging wire test, where an animal is place upside down while hanging on a wire between two poles, while measuring the time it takes to either fall down or cross to the one of the corner poles [6]. In brief, these models (αC418W, αL251T &
From our panel of SCS mice, the αL251T and
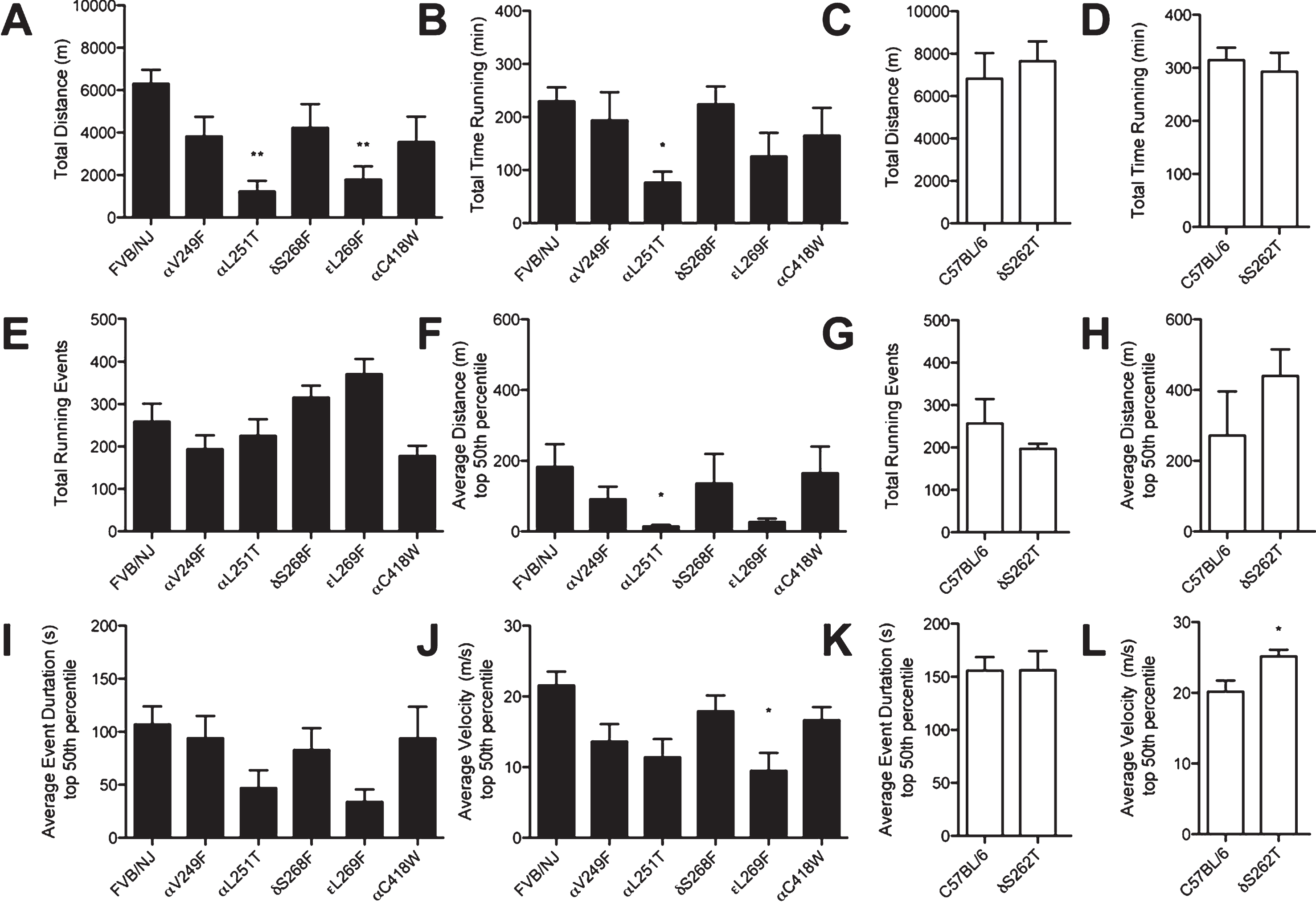
Traditional locomotor metrics reflect variability for a comparison between strains in a panel of SCS mice. Analysis of a 24 hr period of free access to running wheels in a home cage environment reveals decreases in certain facets of locomotor behavior for SCS mice when compared to controls (FVB/NJ
Multivariable analysis of our data shows qualitative differences in the distribution of events with different velocity and duration (Fig. 2A). Quantification of our three-dimensional activity profiles using pre-determined regions, based on wild-type running patterns, reveals differences in the proportion of events located in the region of interest (ROI). Of interest, the selected ROI represents high-velocity/long-duration events and is reduced across our SCS panel, revealing significant differences between the αL251T and
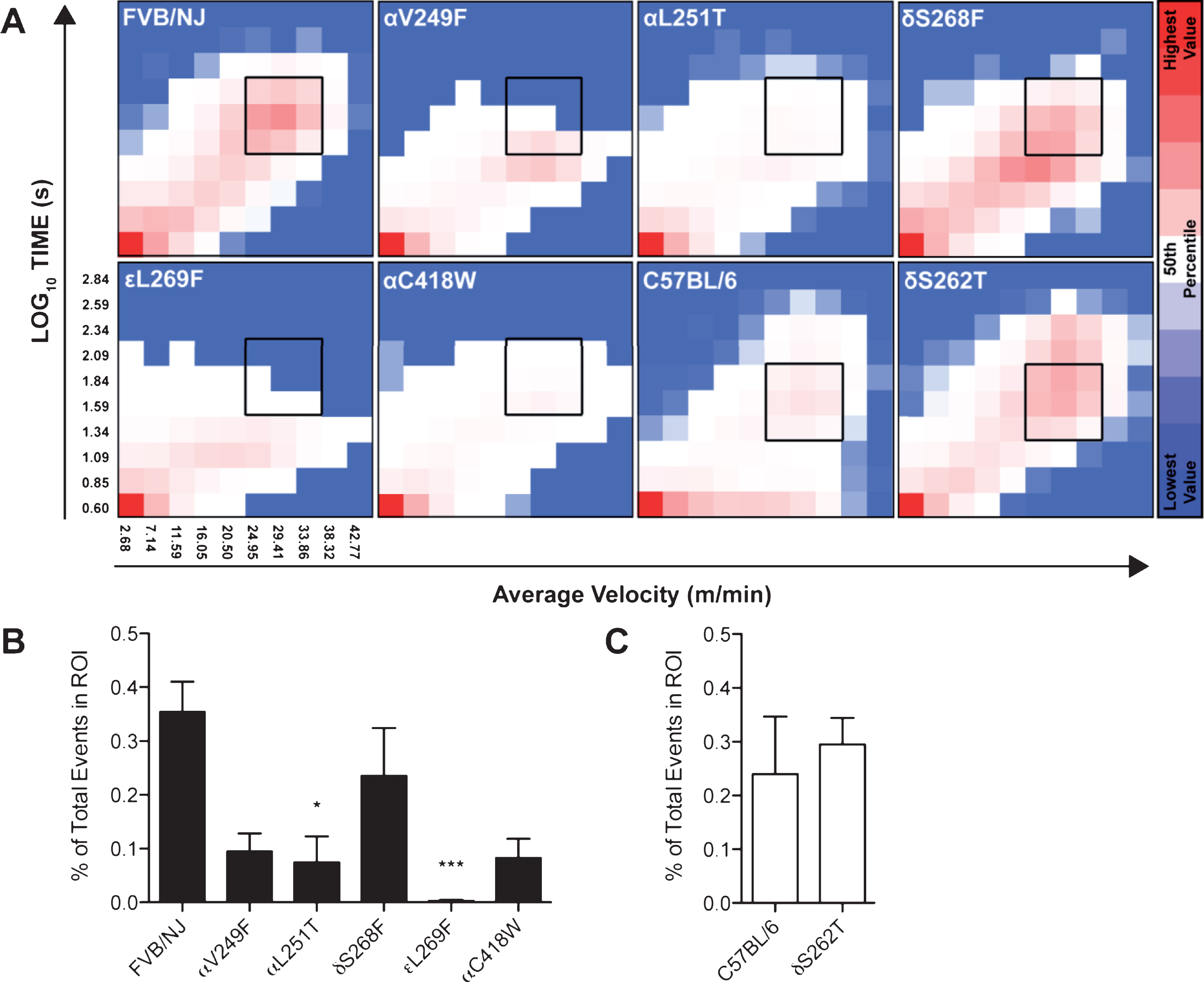
Multi-variable analysis reveals robust differences in locomotor phenotype amongst a panel of SCS mice. Generation of 10×10 matrices between the duration and average velocity of individual activity events, in combination with the frequency of such events, results in a graphical representation of locomotor activity for a period of 24 hours (FVB/NJ
DISCUSSION
Existing SCS models represent a spectrum that encompasses differences in nAChR channel gating kinetics and disease pathophysiology [6, 7]. In humans, this diversity of clinical presentations is also present, leading to a hindered understanding of the relationship between pathophysiology and phenotype [8]. Of interest to our group is the extent to which muscle weakness affects locomotor functioning in humans suffering from SCS. Various reports note that a subset of patients exhibit difficulties in locomotion, correlated with nAChR mutations [15, 16]. However, this aspect is poorly understood, and its study could provide an answer in terms of more closely relating the animal models we use to study SCS and the human presentation of such disorder.
Running-wheel analysis of SCS mice revealed impaired volunteer locomotor activity as a function of altered nAChR kinetics. Data shows a near three-fold reduction in total distance traveled, a two-fold reduction in time spent running, a non-significant but remarkable 100-fold reduction in the distance traveled during an individual running event, and a 50% reduction in the duration and velocity of individual running events for the αL251T and
Nevertheless, one-dimensional analysis of locomotor data fails to represent the complexity of our animal models. Therefore, we generated three-dimensional representations of a 24 hr activity period using the velocity and duration of individual events to come up with a new metric. The benefit of such analysis relies on its ability to segregate voluntary activity events into three-dimensional collections of points that favor detection of subtle changes. We define our analysis as a voluntary running activity profile. Three-dimensional representation of voluntary locomotor activity permits the exploration of complex motor dynamics and maximizes our capacity to detect differences between our mice models in a qualitative and quantitative manner (Fig. 2A, B and C). To further evaluate these differences we selected an ROI, based on the wild-type strain profile, which represented a discrete and consistent population of activity events. Measuring the sum of events within the ROI, we calculated the proportion of the total events compiled in the heat map and took a quantitative approach towards analyzing our three-dimensional multivariable analysis (Fig. 2B, C). Our data revealed that four of our mutant strains possess diminished voluntary motor activity within the ROI when compared to their wild-type counterpart, suggesting that these SCS models show patterns of hindered locomotor activity for high velocity and long duration running events (Fig. 2B). Although the majority of the SCS panel exhibited some degree of locomotor impairment, the
It is important to keep in mind that SCS animal models represent a wide range of disease presentation and model to a minimum the complexity and physiology of human SCS patients. Our group has previously described the clinical presentation of SCS-causing nAChR mutations in human patients, and even though SCS can sometimes possess a severe presentation, differences regarding physical exam findings and clinical score confer each one of these mutations a unique clinical scenario [3, 7]. It has been shown that SCS mice present a wide range of nAChR kinetic/gating deficits, but that only the αL251T and
However, subtle differences and variations in SCS presentation are found in the literature and attest to the need of a powerful tool to uncover phenotypical differences in a neurodegenerative model that are not appreciated when evaluated by traditional metrics. As an example, our group previously has shown that traditional metrics were not enough to detect changes in behavior using the αC418W upon membrane cholesterol depletion via statin treatment, but only when a multivariate analysis was used, then the phenotype was uncovered [18]. We believe each model is unique and in some cases, single variable data seems to come up short to describing a phenotype. Therefore, it is possible to take advantage of a multivariable approach like ours to provide a more thorough and sensitive representation. We propose this tool as one to compliment and expand a locomotor test battery in order to identify subtle changes that would be impossible otherwise. Thus, by generating profiles that accurately represent animal behavior, we will reduce the probability of failing to capture an accurate locomotor profile for these complex animal models. The applications of a model like ours should not be limited to the study of locomotor deficits in mice, but should be interpreted as a proof of concept that multivariable analysis of behavioral outputs can help resolve the subtlety and variability of behavioral experiments. Thus, such approaches will enable us to gain an alternate perspective from traditional single metrics that have long shown little or no potential for translation in clinical settings.
In brief, we have demonstrated that changes in the biophysical properties of nAChRs lead to changes in voluntary locomotor activity. Our panel of SCS mice exhibits behavioral differences that can be detected by the use of running wheels and multivariable analyses. The use of locomotion to characterize a mouse model is not new, but the ability to obtain a unique fingerprint-like signature allows us to expand our understanding of how central and peripheral nervous system disorders can manifest in rodent models [21, 22]. Our approach should not be interpreted as one necessarily confined within the variables measured in this article, the potential for further exploration of other metrics related to locomotion represent its depth and unexploited value.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by National Institutes of Health (NIH) Grants 1P20GM103642 (J.R. and J.A.L.D.), NIH Grant R01 NS033202-15A2 (C.M.G.), Research Initiative for Scientific Enhancement (RISE) Program Grant 5R25GM061151-16 (A.G.G., M.D.V. & OQ), the Minority Access to Research Careers (MARC) Program Grant 5T34GM007821-38 (J.G.G.R.; G.E. G.R. & OQ) and NIGMS P20GM103642 (J.A.L.D).
