Abstract
BACKGROUND:
Salt stress has recently emerged as one of the most significant abiotic stresses with negative impact on crop production. Photosynthesis is a vital biochemical process in plants, very sensitive to environmental constraints, including salt stress.
OBJECTIVE:
The aim of this study was to evaluate the impact of salt stress on photosystem II in the leaves of blackberry cultivars (Rubus fructicosus L.).
METHODS:
Two blackberry cultivars (Thornfree and Boysen) were exposed to salt stress (20, 40, and 60 mM NaCl) for 28 days in in vitro culture. The photosynthetic activity was evaluated by probing chlorophyll a fluorescence (JIP test) and photosynthetic pigments.
RESULTS:
The maximum quantum yield of PSII (Fv/Fm), performance indexes (PIABS and PItotal), and photosynthetic driving force (DFABS) were greatly affected by salt stress in the leaves of Boysen cultivar. Lower photosynthetic efficiency was confirmed by the increased values of absorption flux per RC (ABS/RC), trapping flux per PSII RC (TR0/RC), dissipation flux per RC (DI0/RC), decreased values of chlorophyll and carotenoid contents, disruption in the interaction between the light-harvesting complex and the core proteins of PSII, as well as disruption of the oxygen-evolving complex.
CONCLUSIONS:
The provided informations suggest that the photosynthetic apparatus of Thornfree cultivar was found to be more tolerant to salt stress compared to Boysen.
Introduction
Soil salinization is a very significant threat to ecosystems and agricultural production worldwide, leading to reduced production and quality and considerable economic losses. Beside hampered plant growth and production, numerous physiological processes in plants can be hampered by high salt concentrations, such as photosynthesis [1], stomatal conductance [2], osmotic adjustment [3], enzymatic activity, and hormone balance [4]. As one of the primary metabolic mechanisms in plants, photosynthesis is very sensitive to salinity [5]. Previous studies have revealed that salt stress decreases photosynthesis [6], damages chloroplast structure, and reduces chlorophyll content [7]. Hanachi et al. [8] noticed that growth suppression was correlated with the decline of photosynthesis. One of the consequences of salt stress is the accumulation of reactive oxygen species (ROS), which causes damage to the photosynthetic apparatus and photoinhibition of photosystem II (PSII), inhibition of plant growth, or plant death [9]. Because photosynthesis is very sensitive to various abiotic and biotic stresses, degradation of photosynthesis in plants is considered an indicator of adverse environmental impacts [10]. Chlorophyll (Chl) fluorescence is a quick, non-invasive method very often used to evaluate how environmental stresses affect PSII. Chl fluorescence measurements involve the analysis of fluorescence transients that can reveal important details about plant resistance and tolerance to stress under various adverse environmental conditions like heat [11], drought [12], and salt [13]. Although reduction of PSII activity under salt stress has already been observed in many plant species, the research on using Chl fluorescence to determine changes and behavior of PSII in berry species is limited. Blackberry is one of the most attractive and important berries, with high nutritional and pharmaceutical value and beneficial effects on human health [14]. Thus, blackberry cultivation has increased and has become an important berry species in many countries.
The aim of this research was to compare and understand the physiology of photosynthesis, with a focus on the impact of salinity on photochemistry of investigated blackberry cultivars. Through Chl fluorescence measurements, we compared the changes in PSII photochemistry of the investigated cultivars and determined their sensitivity to salinity. Since in vitro culture is carried out under controlled conditions, it is a valuble method for studying how plants will respond genetically to various environmental factors. Therefore, we used in vitro propagated plants in the experiment, as in vitro experiments have been widely suggested and utilized for the rapid evaluation of salt tolerance in tomato plants [15], apples [16], and mulberries [17]. Currently, there is no systematic evaluation to determine the photosynthetic adaptation strategies of the investigated blackberry cultivar in response to salt stress. Results from this study can provide valuble data for formulating optimized culture media for micropropagation, which can help improve the photosynthetic efficiency and effectiveness of the investigated cultivar in micropropagation. Also, this research might help create new varieties of blackberry that are better adapted to salt stress.
Materials and methods
Plant material and growth condition
The experiment was carried out in the Agricultural Institute Osijek, in Plant Tissue Culture Laboratory. Two thornless blackberry cultivars (Thornfree and Boysen) were examined in this study. Both varieties are native to the United States. They are very popular and widespread in many European countries. Aseptic cultures of blackberry cultivars were established from axillary buds. The shoots of blackberry cultivars were taken from the greenhouses of the Agricultural Institute Osijek. Blackberry shoots consisting of sterile meristems with two or three primordial leaves were inoculated in a solid medium. The medium used for culturing blackberry plants was Murashige and Skoog (MS) medium with MS macro, micro elements and MS vitamins [18]. Sucrose (30 g/L), agar (6.5 g/L), BAP (0.5 mL/L), IBA (0.1 mL/L), and GA3 (0.1 mL/L) were added to the MS medium. Plants were subjected to salt stress by placing them in 200 mL glass jars containing different concentrations of NaCl in the MS medium (0, 20, 40, or 60 mM NaCl). The pH of the culture medium was fixed to 5.8 and then autoclaved at 121° C and 1.5 atm for 20 min. Plants were subjected to salinity for 4 weeks and incubated in a growth room at 25°C with a 16-hour photoperiod (16/8 h light/dark cycle), subjected to fluorescent lamp with a light intensity of 50μmol m–2 s–. After four weeks, blackberry plants were removed from the jars, and chlorophyll fluorescence, chlorophyll, and carotenoid content were analyzed.
Polyphasic Chl fluorescence transient (OJIP)
For measuring Chl fluorescence, we used fifteen fully developed leaves of each cultivar per treatment. All measurements were conducted by Handy-PEA fluorimeter (Plant Efficiency Analyzer, Hansatech Instruments Ltd., Great Britain). The leaves of blackberry plants were illuminated with a short pulse of saturating light (3200μmol m-2 s-1, peak at 650 nm) to induce Chl fluorescence transients. The light pulse applied to the plant sample induces a rapid rise in chlorophyll a fluorescence from the initial fluorescence intensity (F0) to the maximal fluorescence (Fm). The JIP-test was used to analyze changes in Chl fluorescence from 10μs to 1 s (fluorescence intensity measured at 50μs, when all PS II RCs are open (F0, O step); fluorescence intensity measured at 2ms (FJ, J step); fluorescence intensity measured at 30 ms (FI, I step) and maximal fluorescence intensity when all PSII RCs are closed at 1 s (Fm, P step), as described in previous an reports [19, 20]. The JIP-test, provides assessment of plant photosynthetic performance with details about the efficiency of energy capture, electron transport, utilization, dissipation, and the occurrence of any stress-related distruption in the photosynthetic apparatus [21]. Table 1 shows 19 parameters that were derived based on the JIP test and were selected to monitor changes in the photosynthetic apparatus of the examined blackberry cultivars. OJIP transients data were normalized between the F0 and Fm phases and the variable fluorescence between OP was expressed as VOP (VOP = (Ft –F0)/(Fm –F0) for both cultivars. To thoroughly evaluate the O–K and O–J steps, we doubled normalized fluorescence values between O–K and, respectively, O–J steps. Normalization between O and K (300μs) steps revealed an L-band (150μs). VOK denoted the relative fluorescence between O and K (VOK = (Ft –F0)/(FK –F0)), whereas ΔVOK denoted the difference between control and salt-treated plants (ΔVOK = VOK (salt stress) –VOK (control) ). Normalization between O and J (2 ms) steps revealed the presence of the K-band (300μs). VOJ denoted the relative fluorescence between O and J, (VOJ = (Ft –F0)/(FJ –F0)], whereas ΔVOJ denoted the difference between the control plants and the salt treated plants (ΔVOJ = VOJ (salt stress) –VOJ (control) ) [22].
Description and formulae of used JIP-test parameters. All parameters are in relative units
Description and formulae of used JIP-test parameters. All parameters are in relative units
Following the measurements of Chl fluorescence, the chlorophyll a (Chl a), b (Chl b), and carotenoid (Car) contents of the same leaves were analyzed. The method of Lichtenthaler [23] was used to determine the concentrations of chlorophylls and carotenoids in leaves samples. Tissue (0.1 g) was homogenized in liquid nitrogen with the addition of magnesium hydroxide carbonate and total pigments were extracted with absolute acetone. The absorbance of extracts was determined from the sampled leaves of each blackberry cultivar and measured at wavelengths of 470 nm, 647 nm, and 663 nm, recorded on spectrophotometer (Specord 200, Analytik, Jena, Germany).
Data analysis
Statistical comparisons of differences were made by analysis of variance (ANOVA). To compare the means between different salt treatments and cultivars, the Least Significant Difference (LSD) test was conducted at the 0.05 level of probability. Values are presented as the mean±SE (n = 15 for Chl fluorescence parameters; n = 5 for Chl and Car contents. The differences between treatments and cultivars were designated by different letters.
Results
Analysis of parameters of the JIP-test
Regarding Chl fluorescence parameters, ANOVA and LSD test showed that exposure to NaCl treatments caused significant changes mostly in the leaves of Boysen cultivar. The values of F0 were significantly different between treatment and control plants only in Boysen cultivar at highest salt concentration (21% higher compared to control plants) (Fig. 1A). The Fm values were not significantly different in comparison with the control plants, in both cultivars (Fig. 1B). After being exposed to salt, the values of the initial slope of the fluorescence transient (M0) in the Thornfree cultivar did not vary significantly. In contrast, for the Boysen cultivar, values of parameter M0 significantly increased at all concentration compared to control. At 60 mM NaCl parameter M0 increased by 17% relative to control plants in the leaves of those cultivar. (Fig. 1G). In the leaves of the Thornfree cultivar, salt treatments had no noticeable impact on relative variable fluorescence at 0.15 ms (VL) and relative variable fluorescence at 0.3 ms (VK). (Fig. 1C, D). In Boysen cultivar, the VL parameter was significantly increased only at 60 mM NaCl (Fig. 1C), whereas the VK parameter was significantly increased in all salt treatments (Fig. 1D). The maximum quantum efficiency of PSII photochemistry (Fv/Fm) in salt-treated plants of both cultivars ranged from 0.75 to 0.80. Only Boysen plants that had been exposed to 60 mM NaCl showed a significant decrease in that parameter. Thornfree showed no significant difference between salt treated and control plants (Fig. 1E). Significant decline in size and number of active reaction centers of photosynthetic apparatus (Fv/F0) was found in both cultivars only at 60 mM NaCl. In comparison with Thornfree, Boysen showed higher decrease in this parameter (Fig. 1F). The quantum yields and probabilities (quantum yield of electron transport from QA–to plastoquinone (PQ) (ET0/ABS), efficiency with which an electron can move from the reduced intersystem electron acceptors to the PSI end electron acceptors (RE0/ET0), probability that a trapped exciton moves an electron into the electron transport chain beyond QA (ET0/TR0), quantum yield of electron transport from QA–to final PSI acceptors (RE0/ABS) showed no significant changes in salt treated plants in both cultivars when compared with control plants (Table 2). All stress treatments induced significant increases in parameters: absorption flux per RC (ABS/RC) (Fig. 2A), trapped energy flux per RC (TR0/RC) (Fig. 2B), electron transport flux per RC (ET0/RC) (Fig. 2C) in Boysen plants, but no significant changes in Thornfree plants (Fig. 2A–C). At 60 mM NaCl, the flux of dissipated energy per RC (DI0/RC) increased significantly for both cultivars at 60 mM NaCl. When compared to Thornfree, the dissipated energy in Boysen cultivar was higher (Fig. 2D). Parameter electron flux reducing end electron acceptors at the PSI acceptor side per RC (RE0/RC) in Boysen cultivar increased by 25% at the highest NaCl concentration. NaCl treatment did not induce significant increases in RE0/RC in Thornfree cultivar. (Fig. 2E).
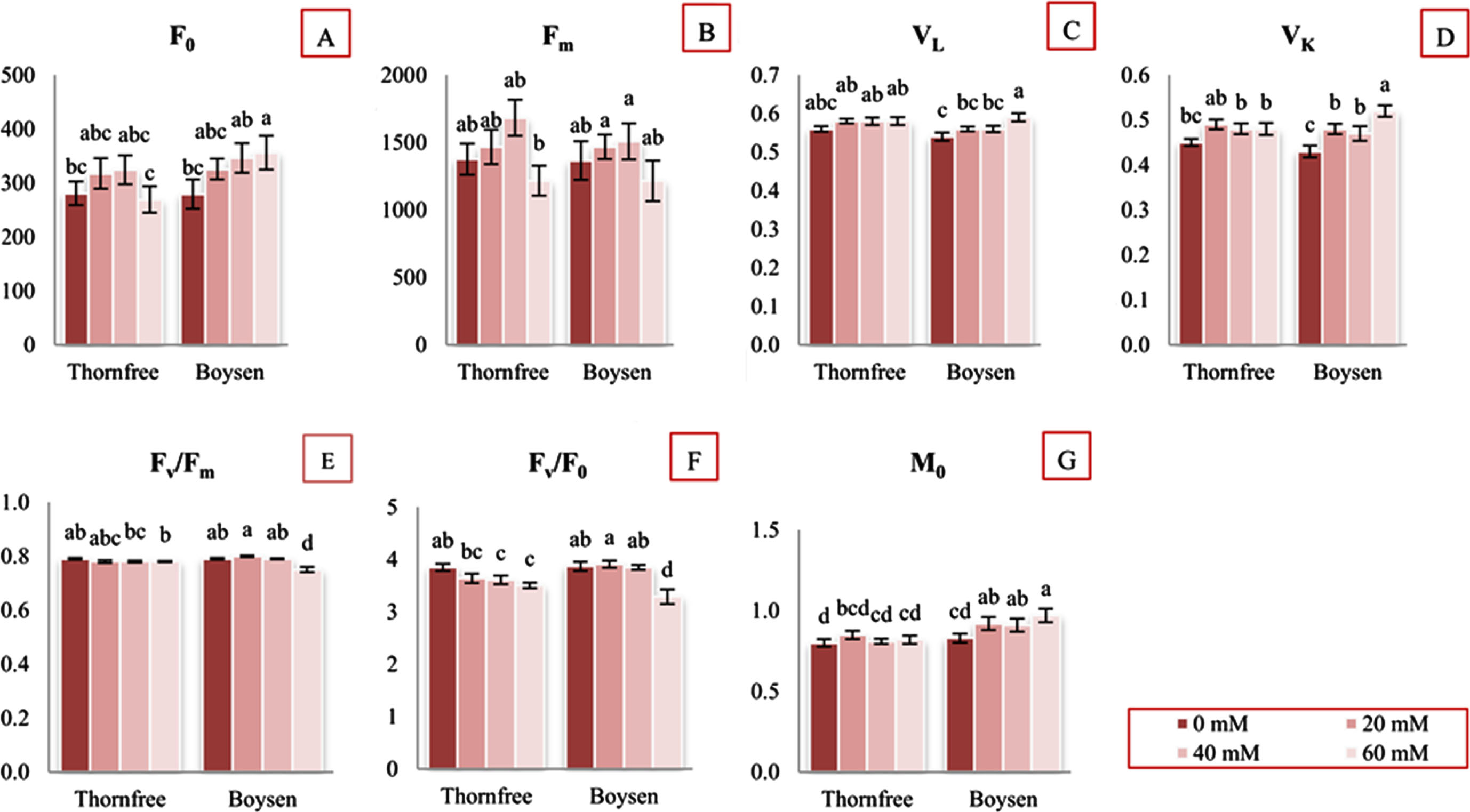
Initial fluorescence intensity, when all PSII RCs are open (F0) (A); maximal fluorescence intensity, when all PSII RCs are closed (Fm) (B); relative variable fluorescnece at 0.15 ms (VL) (C); relative variable fluorescnece at 0.3 ms (VK) (D); maximum quantum yield of primary PSII photochemistry (Fv/Fm) (E); the ratio between the trapping flux and energy dissipation flux of PSII (Fv/F0) (F); approximated initial slope of the fluorescence transient (M0) (G) of two blackberry cultivars (Thornfree and Boysen) under different NaCl salt treatment. Values are presented as relative units.
Quantum yield for electron transport from QA– to PQ (ET0/ABS); the efficiency with which an electron from PQH2 is transferred to final PSI acceptors (RE0/ET0); probability that trapped exciton moves an electron into the electron transport chain beyond QA– (ET0/TR0); quantum yield for reduction of end electron acceptors at the PSI acceptor side (RE0/ABS) of two blackberry cultivars (Thornfree and Boysen) under different NaCl salt treatment. Non significant differences at 0.05 level of probability are indicated as n.s. Values are presented as relative units
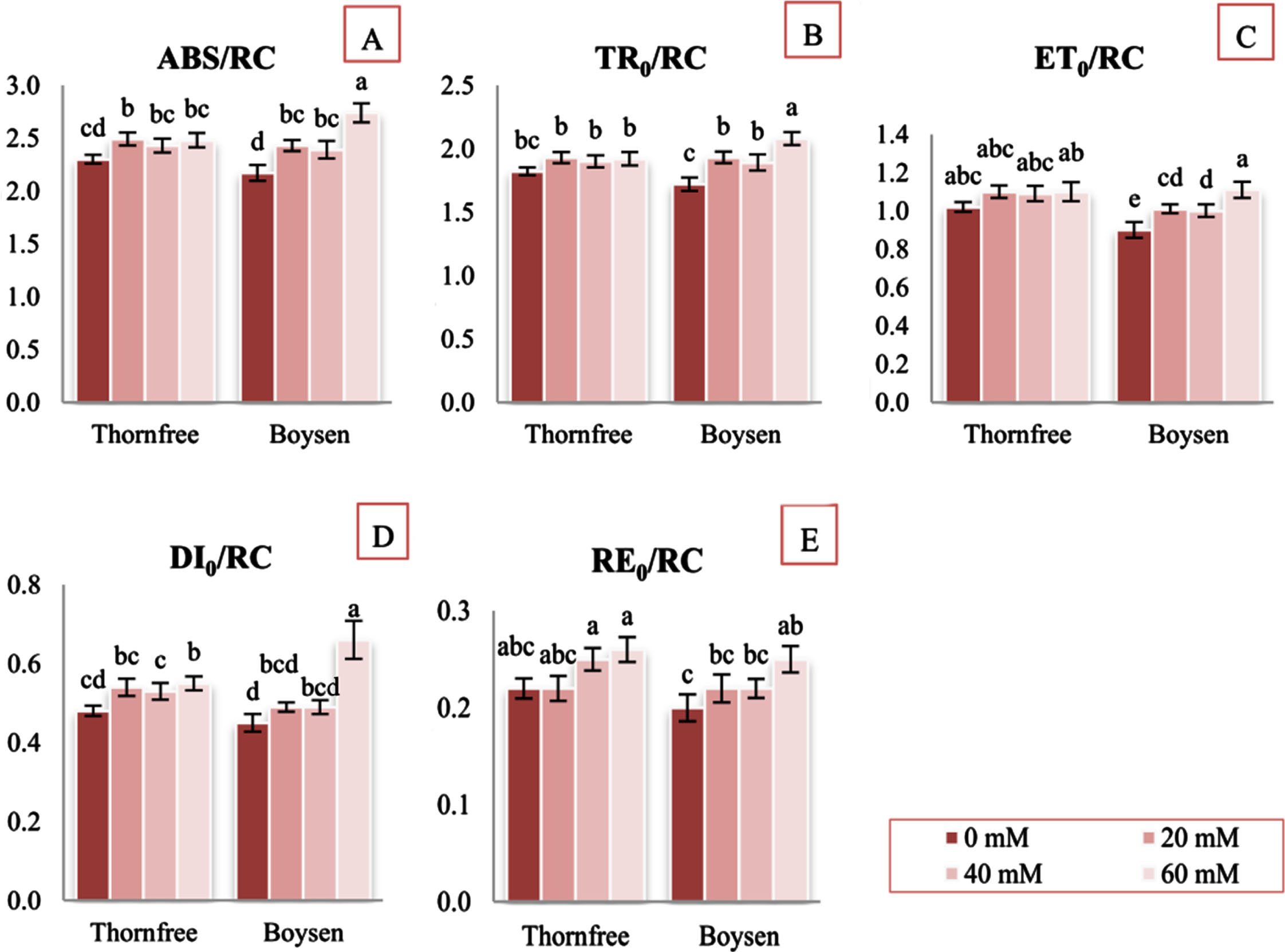
Absorption flux (of antenna Chls) per RC(ABS/RC) (A); trapped energy flux per RC (TR0/RC) (B); electron transport flux per RC (ET0/RC) (C); dissipated energy flux per RC (DI0/RC) (D); electron flux reducing end electron acceptors at the PSI acceptor side, per RC (RE0/RC) (E) of two blackberry cultivars (Thornfree and Boysen) under different NaCl salt treatment. Values are expressed as relative units.
Salinity stress significantly decreased the performance indices (performance index (PIABS) and performance index for energy conservation from exciton to the reduction of PSI end acceptors (PItotal)) during the salt treatment at 60 mM NaCl only in Boysen cultivar (Fig. 3A,B). Furthermore, the indicator of driving force in PSII based on absorption basis (DFABS) significantly decreased only in Boysen cultivar at 60 mM NaCl (52% relative to control plants) (Fig. 3C).
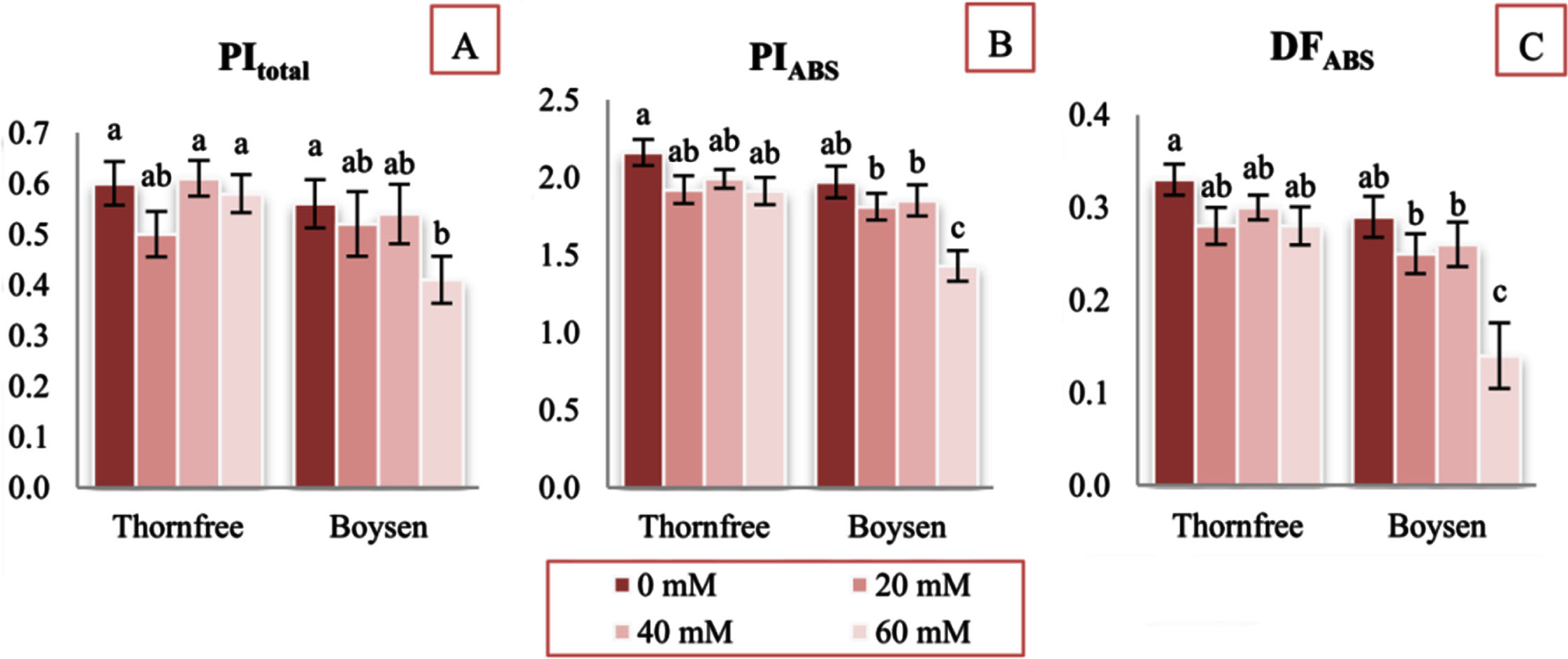
Performance index for energy conservation from exciton to the reduction of PSI end acceptors (PItotal) (A); performance index on absorption basis (PIABS) (B); driving force on absorption basis (DFABS) (C) of two blackberry cultivars (Thornfree and Boysen) under different NaCl salt treatment. Values are expressed as relative units.
Chl fluorescence transients (OJIP) in the leaves of two blackberry cultivars exposed to salt (20, 40, and 60 mM NaCl) and control conditions are presented in Fig. 4. At first sight, neither the control nor the salt-stressed plants in either cultivar displayed any visible changes in the behavior of the OJIP curves. (Fig. 4A,D). Therefore, for a more detailed examination of how salt stress affects the overall photosynthetic performance, we presented the differential curves separately for the K and L bands to analyze and compare the specific changes occurring during these steps of the fluorescence rise curve under salt stress conditions. By analyzing the K-band and L-band separately, we identified differences in the responses of the tested blackberry cultivars to salt stress. The kinetic difference between ΔVOK and ΔVOJ revealed the L- and K-bands, and both blackberry cultivars showed L-band and K-band with positive amplitudes after salt treatments. When compared to Thornfree, Boysen cultivar had a higher positive amplitude for the L-band (Fig. 4B,E). Similarly, Boysen cultivar also exhibited an increased positive amplitude for the K-band (Fig. 4C,F).
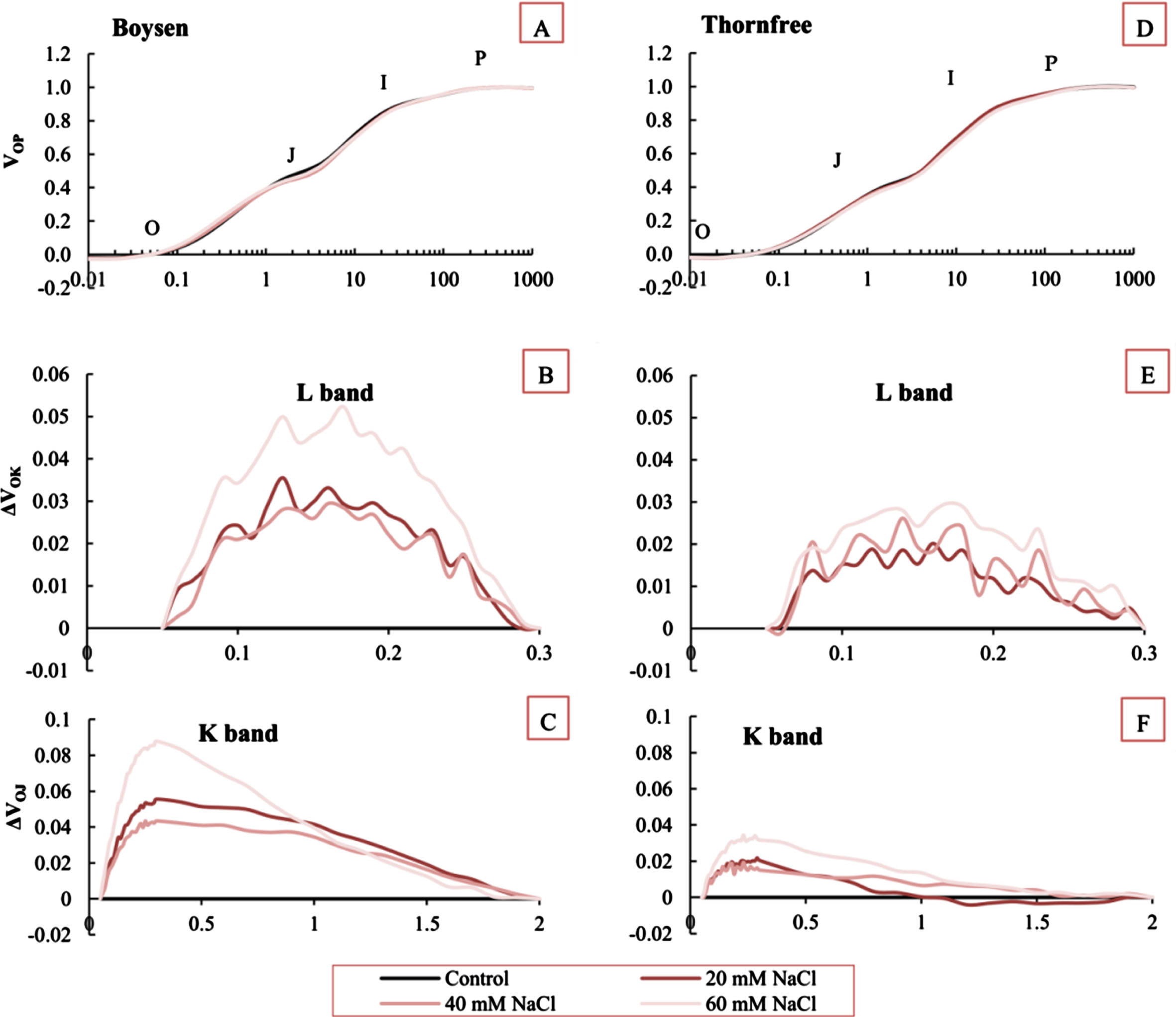
Chlorophyll a fluorescence OJIP transients curves in the leaves of Boysen (A) and Thornfree (D) blackberry cultivars submitted to different salt treatments. O, J, I and P represent the specific steps in chlorophyll a fluorescence transient in control and salt stressed plants; changes in O-K phase relative variable fluorescence intensity [ΔVOK = VOK (salt stress) –VOK (control) ] (L band) in Boysen (B) and Thornfree (E) cultivar; changes in O-J phase relative variable fluorescence intensity [ΔVOJ = VOJ (salt stress) –VOJ (control) ] (K band), in Boysen (C) and Thornfree (F). Comparisons between the salt-stressed and control plants were used to determine the differential kinetics. Data represent means of fifteen replicates.
Salinity stress affected the Chl and Car contents differently in two blackberry cultivars. Plants of the Boysen cultivar had less Chl a and Chl b levels as the concentration of NaCl increased. Chl a and Chl b contents decreased by 35% and 38%, respectively, compared to controls, in plants treated with 60 mM NaCl. In the leaves of Thornfree cultivar, treatment with the highest concentration of NaCl (60 mM) resulted in higher Chl a (by 19%) and Chl b (by 18%) contents, compared to controls. The Chl a and Chl b content, at 60 mM NaCl, was significantly higher in Thornfree cultivar compared to Boysen (Table 3). Significantly higher Car content was observed in Thornfree cultivar treated with 20 and 60 mM NaCl than in control treatment. In contrast, In Boysen cultivar, the level of Car declined as the concentration of NaCl increased. There were notable variations between the control and the treatment groups at NaCl concentrations of 40 and 60 mM, and these differences were statistically significant (Table 3).
Chlorophyll a, chlorophyll b, and carotenoid content of two blackberry cultivars (Thornfree and Boysen) under different NaCl salt treatment
Chlorophyll a, chlorophyll b, and carotenoid content of two blackberry cultivars (Thornfree and Boysen) under different NaCl salt treatment
Along with heat and drought, salt stress is also significant abiotic stressor that adversely affects photosynthesis [24, 25]. In this research, Chl a fluorescence transients in two blackberry cultivars grown in vitro were studied using the JIP-test to observe how their photosynthetic system responded to salt stress. The analyzed blackberry cultivars showed significant variations in the examined JIP-test parameters.
According to Schansker et al. [26], the OJ phase, which is light-dependent, gives data on antenna size and connection between PSII reaction centers. It reflects the state of the reduction on the acceptor side of PSII. To further study the impact of salt treatments on OJIP transient phase and examine the L- and K-bands, the OJIP transients were additionally normalized between the OK and OJ phases. In our study, salt stress induced K and L-bands in the OJIP curve. The K-band that appears at 300μs reflects the state of the oxygen-evolving complex (OEC), and a positive K-band indicates that the OEC is degrading or becoming inactive [22, 27]. The L-band appearing at 150μs is linked with the connectivity of light-harvesting complex II (LHCII) and PSII RCs [28, 29]. Increases in VK and VL values and the existence of more pronounced positive K- and L-bands in the OJIP transient curve of salt-treated Boysen cultivar leaves indicated that OEC has been destroyed and inactivated, as well as the integrity of the thylakoid membrane on the donor side of PSII [20].
The Fv/Fm represents the fraction of total energy flux trapped by PSII RCs [19] and is frequently used to describe plant photosynthetic efficiency as well as an indicator of photoinhibition or various types of stress on PSII. For many plant species, the optimum Fv/Fm ratio varies between 0.79 and 0.84 [30]. Lower values of this parameter (below 0.75), which are characteristic of stressed plants, suggest that PSII has been impaired or inactivated [6, 31]. In our study, the parameter Fv/Fm was significantly reduced only in Boysen cultivar, by the highest salt concentration, 60 mM NaCl, while no changes in Fv/Fm were observed at lower salt concentrations in either cultivar. Salinity decreased the values of Fv/Fm at 0.75 in the Boysen cultivar and caused photoinhibition in the leaves of that cultivar. Chen et al. [20] found damage on the PSII acceptor side of salt-stressed leaves and observed increases in the M0 parameter. Similarly, in our study, we noticed increased M0 levels in salt treatments, as well as PSII damage in the Boysen cultivar. Yang et al. [32] confirmed a decrease in the Fv/Fm of salt-sensitive sorghum plants, whereas Shamshiri and Fattahi [33] found no effect on Fv/Fm at lower salt concentrations. In the opinion of Krause [34] and Bussotti et al. [35] a rise in F0 is an indicator that PSII is damaged which was observed only in Boysen cultivar. The rise in F0 noticed in Boysen cultivar at 60 mM NaCl may be conected to the inactivation of the PSII RCs and the destabilization of PSII caused by photoinhibition, which led to a higher reduction in the Fv/Fm parameter. Similar results at higher salt concentration were reported by Guo et al. [36] and Dabrowski et al. [13]. A decline in the Fv/F0 was observed in Boysen cultivar. Since the ratio Fv/F0 represents the OEC’s efficiency, it can be assumed that the maximum salt concentration (60 mM NaCl) caused alterations on the donor side of PSII that were more pronounced in Boysen cultivar than Thornfree cultivar. Similarly, under salt stress conditions, the activity of the water-splitting complex or OEC on the donor side of the PSII, Fv/F0 was reduced in pea and maize [37] and sunflower cultivars [38].
According to the findings of our study, the energy flux ratios (φEo, φRo, ΨEo, δRo) in the investigated blackberry cultivars were not sensitive to salt stress. Similar to this, Dabrowski et al. [39] reported that salt stress resulted in substantial alterations only in parameters of specific energy fluxes per RCs, while the energy flux ratios were unchanged. To gain a better understanding how salt stress affects the PSII of the examined cultivars, we analyzed the absorption flux (ABS/RC), trapping flux (TR/RC) and electron transport flux (ET0/RC) per PSII reaction center (RC). Our data show that these parameters have been significantly changed only in Boysen cultivar. According to Yusuf et al. [22], a rise in ABS/RC pointed to a reduction in the amount of active PSII RC and a disruption of electron transport from QA– to QB. Increased TR0/RC typically occurs together with a rise in ABS/RC under salt stress, suggesting ineffective reoxidation of decreased QA–or electron transfer to QB and energy loss as dissipation [40]. As a result, the significant rise in both ABS/RC and TR0/RC in the leaves of Boysen cultivar may signify the inactivation of some PSII RCs by lowering the number of active RCs under the highest salt concentration, which causes active RCs to transform into a dissipative center to prevent the oxidative damage [41]. Higher increase in dissipation energy (DI0/RC) observed in Boysen cultivar supports the change in RCs functionality implayed that fractions of RCs formed dissipative centers to protect the photosynthetic apparatus from photooxidative damage. Similar increases in salt stressed maize plants have been reported by Galić et al. [42]. Previous studies noticed a connection between the decrease in Fv/Fm under salt stress and a decrease in ET0/RC [43, 44]. In our study, electron transport ET0/RC increased in Boysen cultivar despite significant decreases in parameter Fv/Fm. According to Mathur et al. [40], an increase in ET0/RC in salt- and heat-stressed wheat leaves signifies a rise in inactive centers and a decreased capacity to move electrons from QA– to QB, which results in a decline in electron efficiency.
Better photosynthetic efficiency in Thornfree cultivar was further supported by the performance indexes used to assess the PSII behavior and overall performance. They were unchanged in Thornfree but significantly decreased by salt stress in Boysen cultivar, at highest concentration. Similar to our work, Jafarinia and Shariati [1] found that the PIABS declined with increased salt stress treatments but remained stable at low salt concentrations. Furthermore, only Boysen cultivar recorded a decrease in DFABS during salt stress.
The findings demonstrated that Chl fluorescence analyses may be used for assesing the overall decline in photosynthetic apparatus and vitality of investigated cultivars. According to our study of JIP-test parameters and detection of OJIP fluorescence transients we revealed that salinity had significant effects on PSII only in the leaves of Boysen cultivar. The most evident negative consequences of salt stress on PSII functionality are most visible in the impairment of OEC functionality, disruption of the normal flow of energy from the LHCII to the RCs, and modifications in the structure and function of PSII’s RCs where some RCs converted into silent, and dissipated energy as heat.
Chlorophyll, a key photosynthetic pigment, is crucial in the process of absorbing light energy during photosynthesis. Therefore, the amount and efficiency of Chl can impact the plant photosynthetic potential [45]. Salinity can reduce Chl content by destroying Chl and inhibiting Chl biosynthesis [20]. Salt tolerance of many plant species has been evaluated using Chl content as one of the key criteria. We observed different responses in Chl content to salt treatments between investigated blackberry cultivars at different salt concentration. Our findings showed that salt treatments significantly reduced the concentration of Chl a and Chl b in Boysen cultivar, which might have related to the cultivars’ lowered photosynthetic activity. As shown in salt-stressed tobacco plants, Chl decrease is likely connected to the reduction of pigment production and the plants’ coping mechanism to prevent photoinhibition [44]. Previous studies also revealed that salt stress decrease the amount of Chl content in oat [46], radish [47], and elm [48]. However, we noticed an increase in Chl a and Chl b content of Thornfree cultivar under salt treatments. This is in line with the study of Murillo-Amadot et al. [49], who found that salt-tolerant cowpea had a higher Chl content. They considered that cultivars better adapted to salinity showed facultative halophytic behavior. The increases in Chl content have also been reported to be involved in barley [50] and rice [51] salt tolerance. Beside the fact that Car are essential light harvesting pigments in process of photosynthesis, they also protect the photosynthetic apparatus from photooxidative damage [52, 53]. According to our findings, salinity stress enhanced the Car level in Thornfree leaves, whereas Boysen cultivars exhibited a decline as a result of salt stress. Shah et al. [54] have also shown that salinity stress increases level of Car in wheat leaves in order to keep the photosynthesis system functioning efficiently. This finding supports our results regarding the lower sensitivity to salt stress observed for this cultivar. The decline of Car content in the salt stressed leaves of Boysen cultivar implies a weaker photoprotective system against salt stress and photooxidative damage, as has been described in rice plants [55]. Our findings were similar to those of previous reports, which indicated that salt tolerant plants exhibited better photosynthetic efficiency, Chl, and Car content [51, 56].
Conclusion
In conclusion, the two blackberry cultivars exhibited different reactions and adaptations to salt stress conditions. In comparison to the Thornfree cultivar, salt stress significantly reduced Fv/Fm, PIABS, Chl, and Car content, and caused more significant modifications in chlorophyll fluorescence parameters and OJIP transient curve in Boysen cultivar. Therefore, we can conclude that higher photosynthetic efficiency and Chl retention reflect a stable photosynthetic apparatus and better salt tolerance in Thornfree cultivar. Although the experiment should be repeated to examine the photosynthetic systems reaction in vivo, obtained results for Chl a fluorescence indicate that photosynthetic apparatus of Thornfree cultivar was more successful in adapting to salinity. This information can be used to select blackberry plants with better photosynthetic performance and use it in breeding programs to create salt-tolerant blackberry cultivars with enhanced productivity. Aditionally, these findings can provide valuable insights for producers who grow blackberries in areas with high NaCl content in the soil and water, and thus can help to enhance the sustainability and profitability of blackberry production in these areas.
Footnotes
Acknowledgments
The authors have no acknowledgments.
Funding
The authors report no funding.
Conflict of interest
The authors have no conflict of interest to report.
Author contributions
Conceptualization, I.M. and K.D.; software, V.T.; performance of work, D.V. and M.V.V; interpretation of data, I.M. and M.V.V.; resources, Z.Z. and K.D.; writing the article, I.M.; supervision, Z.Z. and K.D.; All authors have read and agreed to the published version of the manuscript.”
