Abstract
BACKGROUND:
The Andean blackberry (Rubus glaucus Benth) is one of Ecuador’s most iconic Andean berries for which a high anthocyanin content has been described.
OBJECTIVE:
The aim of the present study was to determine the chemical composition and anti-inflammatory potential of the Andean blackberry from Ecuador, with an emphasis on its effects on NLRP3 inflammasome activation and autophagy processes.
RESULTS:
Andean blackberry extracts were rich in hydroxycinnamates (coumaric acid and derivates), in addition to quercetin and kaempferol as principal flavonols. Cyanidin and its glycosides were identified as the main anthocyanins present. Andean blackberry extracts efficiently reduced oxidative stress markers in the lipopolysaccharide-stimulated RAW 264.7 cells. The extracts also caused a moderate decrease in the expression of the pro-inflammatory and antioxidant genes NFκB1, TNF, IL-1β, IL-6, and NOS2 expression, while they significantly increased the mRNA levels of both SOD1 and NFE2L2 genes. Andean blackberry extracts significantly decreased the activation of the NLRP3 inflammasome complex, as well as p62 levels, and the LC3I/LC3II ratio increased, suggesting a direct action of Andean blackberry compounds on the inflammatory response and restoration of the autophagy process.
CONCLUSIONS:
These results suggest that Andean blackberries potentially have an anti-inflammatory effect through their ability to regulate genes related to the inflammatory and antioxidant response, as well as modulate the activation of the NLRP3 inflammasome complex and autophagy processes.
Introduction
Current epidemiological evidence increasingly highlights the relationship between nutritional patterns based on the frequent consumption of fruits and vegetables with a low incidence of noncommunicable diseases, such as diabetes, cardiovascular and neurodegenerative diseases, and cancer [1]. All these pathologies have a common pathophysiological feature in their development and progression based on high production of pro-inflammatory mediators and are characterized by excessive production of reactive oxygen species (ROS) with the consequent oxidative damage on macromolecules and cellular structures [2]. Thus, the inhibition of the excessive inflammatory response and oxidative damage has been proposed as an alternative in order to minimize their damage. It is in this context that a diet rich in fruits and vegetables, with their important contribution of antioxidants and anti-inflammatory compounds, plays a fundamental role, based mainly on the ability of these compounds not only to block the excessive circulating free radicals but also on their biological effects that go beyond the simple antioxidant character [3]. Indeed, in recent decades, an increasing body of articles have described the anti-inflammatory and immunoregulatory properties that polyphenols have on different pathways related to the antioxidant response and, therefore, their contribution to protection against oxidative damage [4, 5]. Within the polyphenols described with anti-inflammatory activity, anthocyanins (ACNs) play a prominent role. Anthocyanins are natural phenolic pigments with broad and outstanding biological activity. They have potent antioxidant and anti-inflammatory activity, properties that have been closely related to their other biological effects, such as antidiabetic and anticancer activity, and their role in cardiovascular and neuroprotective prevention [6, 7]. Anthocyanins are widespread in the plant world, providing the characteristic red/blue color to various fruits, vegetables, flowers, and leaves, and their content is influenced by various factors: species, cultivars, climatic conditions, and level of maturity [8], and others [9]. Among the natural sources of anthocyanins, berries stand out (strawberries, blueberries, blackberries, blackcurrants, redcurrants, and raspberries) for being a rich source, with levels ranging from about 100 to 700 mg/100 g of fresh fruits [9].
The Andean blackberry (Rubus glaucus Benth) is one of Ecuador’s iconic berries that grows wild or cultivated, isolated, dispersed, or in groups with other plants in the Andean highlands of Ecuador. The fruits are traditionally collected and commercialized by the indigenous people of these regions [10] and they have become a popular fruit among the Ecuadorian population in general, consumed in the form of jams, wine, and boiled drinks, such as the famous “colada morada” (a traditional drink made with black corn flour and fruits) that is consumed during the Day of the Dead celebrations [11]. Although several studies have highlighted the chemical composition and biological effects of blackberries from other geographical regions [12–15], few studies have analyzed the Andean blackberry. Only a handful have described its chemical composition, evidencing its high anthocyanin content [16–18], as well as its biological properties, such as its ability to protect against oxidative damage [19]. Thus, more studies are necessary to define its chemical composition, as well as the biological effects and mechanisms that it uses to generate them, to eventually determine its true health potential and therefore justifying its consumption. With this background, in the present study we firstly determined the polyphenolic composition of the Andean blackberry fruit. In order to assess its anti-inflammatory effects, murine macrophage RAW 264.7 cells were used as an in vitro validation model both in the presence or absence of the endotoxin lipopolysaccharide (LPS). The levels of the principal biomarkers related to oxidative damage and inflammatory markers were estimated together with the activity of the main antioxidant enzymes. Finally, the expression of different genes related to antioxidant and inflammatory response was also determined with the objective of discovering the possible molecular pathways involved in the anti-inflammatory action of Andean blackberry. Finally, given the importance of the NLRP3 inflammasome complex as an important mediator of inflammatory processes, we studied the effects of Andean blackberry compounds on its activation by using an in vitro model of THP-1 human monocytes cells. At the same time, given the repairing effect of the autophagy process, and therefore its importance in cellular hemostasis, we also studied the effect of the polyphenols of this fruit on the activation of this process using this cellular model.
Material and methods
Chemicals and reagents
All chemicals and solvents were of analytical grade. Solvents, lipopolysaccharides (LPS) from Escherichia coli O55:B5, RPMI, and 1640 Medium, fetal bovine serum, penicillin-streptomycin, RIPA Buffer, hydrogen peroxide solution (H2O2), reduced glutathione (GSH), β-Nicotinamide adenine dinucleotide 2′-phosphate reduced tetrasodium salt hydrate (NADPH), glutathione peroxidase from bovine erythrocytes (GPx), L-glutathione reduced (GSH), glutathione S-transferase from equine liver (GST), 2-Thiobarbituric acid, malondialdehyde tetrabutylammonium salt (MDA), 2,4-Dinitrophenylhydrazine (DNPH), guanidine hydrochloride, streptomycin, Tris-HCl, Triton™ X-100, phenylmethylsulfonyl fluoride, leupeptin, and Bradford reagent were purchased from Sigma-Aldrich Co. (St. Louis, MO, USA), while the 2′-7′-dichlorofluorescin diacetate (DCFH) kit was purchased from Merck (Darmstadt, Germany). TRIzol™ reagent, Turbo™ DNase, and SuperScript III Platinum SYBR Green One-Step RT-qPCR kit were purchased from Invitrogen Carlsbad (CA, USA), while specific monoclonal antibodies were obtained from Santa Cruz Biotechnology (Dallas, TX, USA). A cocktail of protease inhibitors (Complete Protease Inhibitor Cocktail) was purchased from Boehringer Mannheim (Indianapolis, IN, USA). The Immun-Star horseradish peroxidase (HRP) substrate kit was obtained from Bio-Rad Laboratories Inc. (Hercules, CA, USA).
Plant materials and sample preparation
Ripe Andean blackberries (Rubus glaucus Benth) fruits were harvested during the crop season (October–December 2018) from plants growing in the highlands of the province of Tungurahua, located in the inter-Andean region of Ecuador at an altitude of between 3000 and 3200 meters above sea level. Fruits were collected at random on two different occasions from different plants based on a similar degree of ripeness and without blemishes or any signs of damage or decay. Two batches of samples (∼1 kg) were collected on each occasion and the specimens were identified using the reference vouchers for specimens deposited in the herbarium of the Jardín Botánico de Quito, Ecuador. During collection time, the fruits were kept at 4°C, protected from light, and transported to the laboratory on the same day that they were harvested. Afterward, samples were cleaned, removing remains of leaves, stems, and other residues, washed with distilled water and allowed to dry on absorbent paper at room temperature and protected from light. Finally, fruits were frozen at –20°C overnight and then freeze-dried, ground to a fine powder, and stored at –20°C until analysis.
For chemical composition determination and in vitro assays, a polyphenol-rich extract was obtained as previously reported [20]. Briefly, the fine powder of fruits (10 g) was added to 100 mL of the extraction solution consisting of methanol acidified with 0.1% HCl and Milli-Q water (80%, v/v) and stirred overnight at 3–5°C. Then, the solution was centrifuged for 15 minutes at 4000g at 4°C and filtered through a 2.5μm pore size Whatman® cellulose filter paper (Sigma-Aldrich Co., St. Louis, MO, USA). The solid residues were washed with the extraction solution as many times as necessary until no red color was observed in the extracted solution. Finally, the resulting extraction solutions were combined, water was added, and the final solution was concentrated in a vacuum rotary evaporator (∼30°C) until a dry crude extract was obtained.
HPLC-DAD/ESI-MSn characterization of phenolic acids, flavonols, and anthocyanins
The Andean blackberry crude extract (ABCext) was dissolved in 0.5 mL of 0.1% trifluoroacetic acid/acetonitrile (70:30, v/v), filtered through a 0.22μm disposable LC filter disk for analysis by HPLC, and analyzed using double online detection by diode array spectrophotometry and mass spectrometry (MS). The HPLC system consisted of a Hewlett-Packard 1200 chromatograph (Hewlett-Packard 1200, Agilent Technologies, Santa Clara, CA, USA) equipped with a binary pump and a diode array detector (DAD) coupled with an HP ChemStation (rev. A.05.04) data processing station and an MS detector API 3200 Qtrap (Applied Biosystems, Darmstadt, Germany) that was controlled by the Analyst 5.1 software. As a stationary phase, an AQUA® (Phenomenex, Torrance, CA, USA) reverse phase C18 column (5μm, 150 mm×4.6 mm) thermostated at 35°C was used. The solvents were: (A) 0.1% trifluoroacetic acid and (B) acetonitrile. The elution gradient established was: isocratic 10% B for 3 min, 10–15% B in 12 min, isocratic 15% B for 5 min, 15–18% B over 5 min, 18–30% B over 20 min, 30–35% B over 5 min, and a re-equilibration of the column to initial solvent conditions. The flow rate used was 0.5 mL/min. For detection, the DAD was set using 520, 330, and 280 nm as preferred wavelengths, while the MS operated in the positive ion mode for the analysis of anthocyanins and in the negative mode for the rest of the compounds, the spectra were recorded between m/z 100–1000. Zero-grade air served as the nebulizer gas (30 psi) and as turbo gas (400°C) for solvent drying (40 psi). Nitrogen served as the curtain (20 psi) and collision gas (medium). Both quadrupoles were set at unit resolution and EMS and EPI analyses were also performed. The EMS parameters were set as follows: ion spray voltage –4500 V, DP –50 V, EP –6 V, CE –10 V, and cell exit potential (CXP) –3 V, whereas EPI settings were: DP –50 V, EP –6 V, CE –30 V, and CES 10 V. The individual phenolic compounds were tentatively identified based on their UV and mass spectra and compared with data reported in the literature, as well as with internal standards when available.
Evaluation of anti-inflammatory properties
Cell culture and treatments
Murine RAW 264.7 macrophage cell line was purchased from ATCC (ATCC-TIB71) (Manassas, VA, USA) and maintained in RPMI 1640 medium supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin antibiotics (100IU/mL penicillin and 100 mg/mL streptomycin) in a humidified atmosphere at 37°C with 5% CO2. For all the assays, cells were used between the 4th and the 6th passage. The Andean blackberry crude extract (ABCext) was resuspended in RPMI 1640 medium to achieve the final concentration of 80μg/mL. RAW macrophages were treated with: (i) RPMI 1640 medium only (Ctrl group), (ii) lipopolysaccharides (LPS) (Escherichia coli serotype 055:B5) at 1μg/mL for 24 h (LPS group), (iii) ABCext (80μg/mL) for 24 h (ABCext group), or (iv) LPS (1μg/mL) for 24 h and then ABCext (80μg/mL) for another 24 h (LPS/ABCext group), (v) ABCext (80μg/mL) for 24 h and then LPS (1μg/mL) for another 24 h (ABCext/LPS group), and (vi) ABCext (80μg/mL) + LPS (1μg/mL) for 24 h (ABCext+LPS group). The appropriate dose/time combination of ABCext and LPS treatments was selected according to our previously reported studies using the same type of extract [19] as well as the same cell model according to the culture conditions in our laboratory [21].
Measurement of intracellular ROS level
Intracellular reactive oxygen species (ROS) levels were determined using the 2′-7′-dichlorofluorescin diacetate (DCFH) kit according to the manufacturer’s instructions. RAW cells previously treated under the same aforementioned conditions were incubated with 5μmol/L of DCFH at 37°C for 30 min in the dark at room temperature. Next, the fluorescence intensity was read using a microplate reader (Thermo Scientific Microplate Reader, Multiskan®, Winooski, VT, USA) at 485/530 nm excitation/emission wavelength. Total protein levels were determined by the Bradford method [22] and results were expressed as arbitrary units of fluorescent intensity/μg cell proteins.
Determination of antioxidant enzyme activities, biomarkers of oxidative damage in lipids and proteins, and nitrite production
After treatment, murine RAW cells were washed with PBS, incubated with RIPA buffer on ice for 5 min and the obtained lysate was stored at –80°C until analysis. For the analysis of antioxidant enzymes, superoxide dismutase (SOD) was analyzed based on the inhibition of the formation of NADH phenazine methosulphate-nitroblue tetrazolium (NBT) formazan by SOD [23]. Catalase (CAT) was analyzed following the decomposition of hydrogen peroxide [24]. Glutathione peroxidase (GPx) activity was determined according to the ability of GPx to remove H2O2 by coupling its reduction to H2O with oxidation of reduced glutathione in the presence of NADPH [25]. Glutathione reductase (GR) activity was determined based on the capacity of GR to reduce oxidized glutathione back to reduced glutathione (GSH), in the presence of NADPH [26], while glutathione transferase (GST) activity was determined by measuring the content of dinitrophenyl thioether produced by the conjugation of 1-chloro-2,4-dinitrobenzene (CDNB) with reduced glutathione [27]. SOD and CAT activity were expressed as U/mg prot/min, GPx and GR activity were expressed as nmol of NADPH oxidized/mg prot/min, while GST activity was expressed as nmol 1-chloro-2,4-dinitro benzene (CDNB)-reduced glutathione (GSH) conjugate/mg protein/min. Protein carbonyl content [28] and thiobarbituric acid-reactive substance (TBARS) [29] were used as markers of protein and lipid oxidative damage, respectively, and the results were expressed as nmol/mg protein for total protein carbonyl content, and nmol/100 mg protein for TBARS levels. Nitrite oxide (NO) accumulation in cell culture media was determined by the Griess reagent system following the manufacturer’s protocol and NO content was expressed as μM of NaNO2/105 cells.
RT-qPCR analysis of related genes to antioxidant and inflammatory responses
RNA from RAW cells was extracted using the TRIzol™ reagent followed by DNase I treatment to eliminate genomic DNA interference (Turbo™ DNase kit) according to the manufacturer’s instructions. RNA concentrations and purity were determined using a NanoDrop 2000 Spectrophotometer (Thermo Scientific, Waltham, MA, USA). Reverse transcription and subsequent quantitative real-time PCR were performed on a CFX96 Real-Time PCR System (Bio-Rad Laboratories Inc., Hercules, CA, USA) using the SuperScript III Platinum SYBR Green One-Step RT-qPCR kit and 100 ng per RNA sample. cDNA synthesis was carried out for 3 minutes at 50°C followed by an initial denaturation step at 95°C for 5 minutes. The cycling conditions comprised an initial denaturation step at 95°C for 5 min, followed by 40 cycles of: denaturation at 95°C for 30 s, primer annealing at 65°C for 30 s, and extension at 72°C for 30 s, followed by a final elongation step at 40°C for 1 min. For gene expression analysis, primers for 10 different genes associated with pro-inflammatory, anti-inflammatory, and oxidative stress responses designed online with the NCBI Primer-BLAST tool were used (Table 1). Beta-actin (ACTB) was used as a housekeeping gene. Finally, a melting curve analysis, whereby the qPCR products were run from 65°C to 95°C with an increment of 0.5°C for 0.05 s per step, was used to verify specific product formation. Relative gene expression analysis was calculated using the ΔΔCt method [30].
Primer sequences used in gene expression qPCR assays on RAW 264.7 murine macrophages treated with different combinations of Andean blackberry extract (ABCext) and/or LPS
Primer sequences used in gene expression qPCR assays on RAW 264.7 murine macrophages treated with different combinations of Andean blackberry extract (ABCext) and/or LPS
Cell culture and treatments
THP-1 cells, a monocyte isolated from peripheral blood from an acute monocytic leukemia patient, were purchased from ATCC (ATCC® TIB-202™) (Manassas, VA, USA) and cultured in RPMI media with 10% fetal serum and penicillin (100U/ml)/streptomycin (100μg/ml) at 37°C under an atmosphere of 5% CO2. To determine the effects of the Andean blackberry crude extract (ABCext) on the LPS+ATP-induced inflammasome, the cells were divided into 3 groups: (i) RPMI 1640 medium only (Ctrl group), (ii) LPS (Escherichia coli serotype 055:B5) at 500 ng/mL LPS for 4 h followed by stimulation with ATP (5 mM) for 6 h (LPS+ATP group) and LPS (500 ng/mL) for 4 h followed by ATP (5 mM) and ABCext (80μg/mL) for 6 h (LPS+ATP+ABCext group). The appropriate dose/time combination of LPS and ATP treatments for inflammasome activation was previously established by researchers at the Research Laboratory, Oral Medicine Department, University of Seville, Seville, Spain [31]. Moreover, ABCext was established according to preliminary dosage assays for protein expression induction (data not shown).
Western blotting
Whole cellular lysate from THP-1 cells was prepared by gentle shaking with a buffer containing 0.9% NaCl, 20 mM Tris-HCl, pH7.6, 0.1% triton X-100, 1 mM phenylmethylsulfonylfluoride, and 0.01% leupeptin. The protein content was determined by the Bradford method [22]. Electrophoresis was carried out in a 10–15 % acrylamide SDS/PAGE and proteins were transferred to Immobilon membranes (Amersham Pharmacia, Piscataway, NJ, USA). Next, membranes were washed with PBS, blocked overnight at 4°C, and incubated with the primary antibody solution (1:1000) specific for the detection of the following proteins or multiprotein complexes: NLRP3 inflammasome, caspase 1, caspase 1 p20 unit (p20), interleukin 1 beta (IL-1β), mature form of IL-1β (p17), the autophagy markers microtubule-associated protein light chain 3 (LC3I and LC3III), and p62. Membranes were then probed with their respective secondary antibody (1:2500). Immunolabeled proteins were detected by chemiluminescence method (Immun Star HRP substrate kit, Bio-Rad Laboratories Inc., Hercules, CA, USA). Western blot images were quantified using ImageJ software (see: http://rsb.info.nih.gov/ij/download.html).
Statistical analysis
Statistical analysis was carried out with SPSS v.24 software and Jamovi built with R statistical language [32–34]. The medians±standard deviations (SD) of three independent experiments are presented. Correlations between the different stimulation groups were processed using one-way ANOVA analysis and the post-hoc Tukey’s test with a p < 0.05 was considered statistically significant, with a p < 0.01 being considered as highly significant.
Results and discussion
Phenolic composition of Andean blackberries
In this study, a hydroalcoholic extract obtained from Andean blackberries was analyzed for its polyphenol composition by HPLC-DAD-ESI/MS n . Figure 1A shows representative HPLC chromatograms of the polyphenol profile obtained at different λ (330 and 360 nm for phenolic acid derivatives and flavonoids and 520 nm for anthocyanins), while Fig. 1B shows the specific profile for anthocyanins and Fig. 1C the specific profile of phenolic acids and flavonoids. On the other hand, Table 2 compiles the tentative identity of the peaks, assigned based on their UV-Vis spectra, mass spectral data of their (pseudo)molecular ions, and MS2 fragmentation patterns. The detected phenolic acids corresponded mainly to hydroxycinnamoyl derivatives, whereas the majority of flavonoid peaks corresponded to the group of flavonols and anthocyanins. The hydroxycinnamates here identified (coumaric acid and ferulic acid derivatives) have been previously reported in berries [4, 13]. The same is true for quercetin and kaempferol, which have been reported as the main flavonols present in this fruit [13, 35]. Among the anthocyanins reported in berries, cyanidin and its glycosides have been identified as the main components of this group of pigments [13, 35]. In fact, in our study, cyanidin was identified predominantly in its 3-form glycosidic and only two minor peaks of pelargonidin derivatives were detected. In this context, the chemical composition reported here was crucial in justifying part of the biological effects reported below, as well as those previously reported [35]. Hydroxycinnamates, for example, are among the most widely distributed phenylpropanoids present in the insoluble-bound, conjugated-soluble, and free forms in plants. They render relevant antioxidant activity by their capacity to scavenge different free radicals (FR), such as hydroxyl radical (HO•), superoxide radical anion (O2–•), peroxyl radical (ROO•), peroxynitrite (NO3–), singlet oxygen (1O2), and several organic radicals [36]. Coumaric acids have been reported as an important antioxidant component in different berries [37], while in addition to their antioxidant capacity, they have also been associated with antibacterial, anticancer, and antihistaminic effects and an enhancement of ATP production [38]. Quercetin and kaempferol were also associated with significant antioxidant, anti-inflammatory, and antitumor effects [7, 39]. This has also been evidenced for anthocyanins, which have been widely studied for their powerful antioxidant and anti-inflammatory effects [7, 40], antitumoral capacity [7, 41], and protective effects against cardiovascular diseases [5, 42].
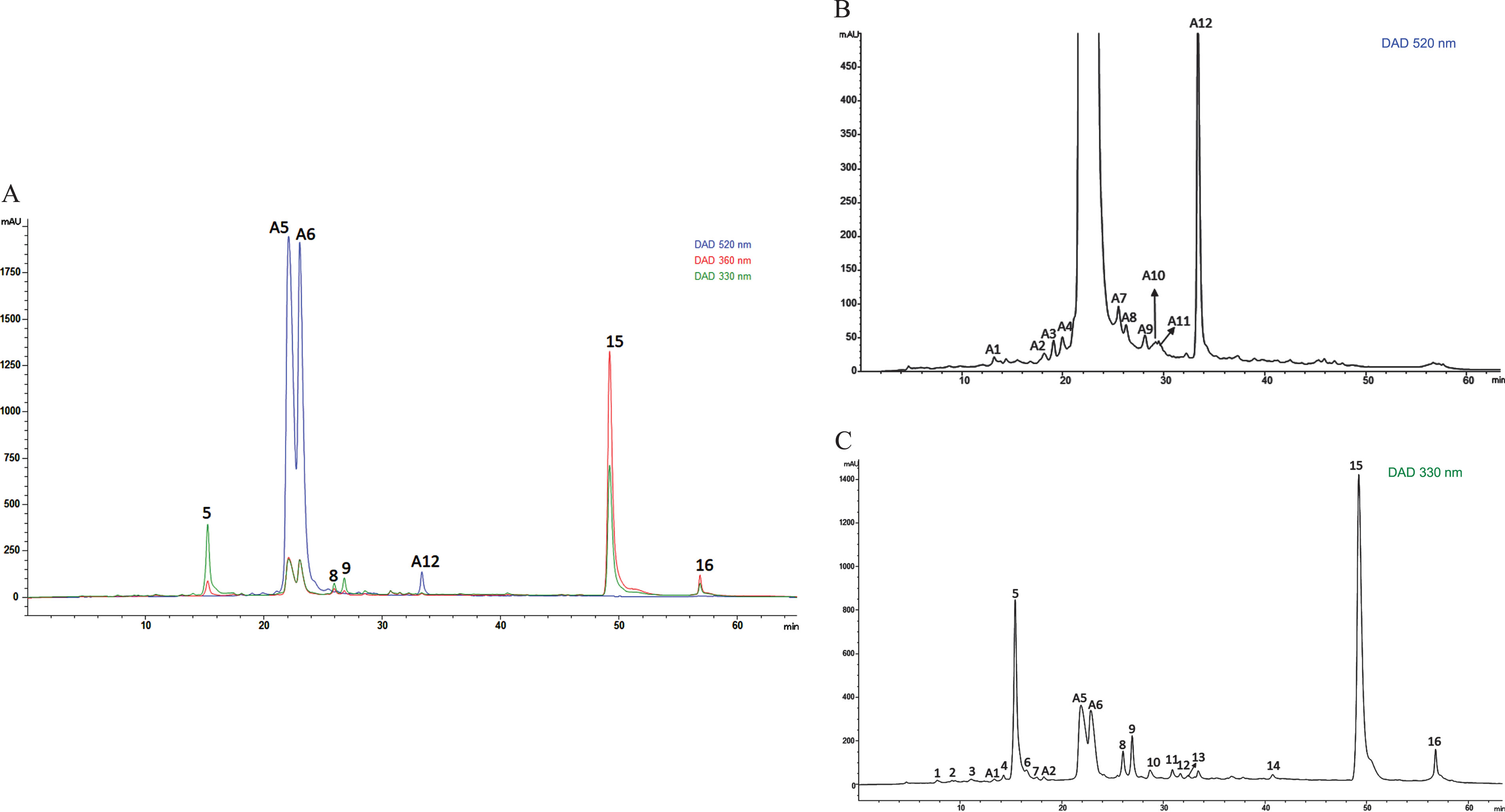
HPLC chromatograms of phenolic acid derivatives and flavonoid profile in crude Andean blackberry extract (ABCext). A) shows representative HPLC chromatograms of the polyphenol profile for phenolic acid derivatives and flavonoids recorded at 330 and 360 nm and for anthocyanins at 520 nm. B) shows the specific profile of anthocyanins (at 520 nm) and C) the specific profile of phenolic acids and flavonoids (at 330 nm).
Absorption, mass spectral data and tentative identification of the peaks detected in Andean Blackberry fruits
During inflammation, different macrophage subsets have a dual behavior. On one hand, macrophages displaying an M1 phenotype secrete pro-inflammatory cytokines and produce elevated amounts of reactive oxygen species (ROS) and nitric oxide (NO), enhancing the inflammatory status of the immune response. On the other hand, alternatively activated M2 macrophages are involved in inflammation resolution and tissue reconstruction, being responsible for apoptotic and necrotic cell phagocytosis and the secretion of anti-inflammatory, proangiogenic, and profibrotic cytokines, such as interleukin 10 (IL-10), vascular endothelial growth factors (VEGF), and transforming growth factor β (TGF-β), respectively. The excessive production of ROS and NO has been correlated with a chronic inflammation typical of some diseases, including asthma, ulcerative colitis, and rheumatoid arthritis [43]. Therefore, it is very important to investigate the role that these mediators have on these processes, together with the effects of natural extracts in improving inflammation. In line with the above, this work analyzed the effect of ABCext on the intracellular production of ROS and NO in RAW 264.7 macrophages, as well as on a group of oxidative stress markers related to oxidative stress derived from the inflammatory response (Table 3).
Values of antioxidant enzyme activities, biomarkers of oxidative damage in lipid and proteins and nitrite production
Values of antioxidant enzyme activities, biomarkers of oxidative damage in lipid and proteins and nitrite production
Values within the same column with different superscripts are significantly different (p < 0.05) by post hoc Tukey’s test.
Regarding ROS, LPS significantly (p < 0.05) increased the intracellular % of ROS up to 2.8 times compared to control cells, while the treatment with ABCext led to a slight decrease compared to the control. In the combined treatments, we found that ABCext significantly (p < 0.05) decreased the intracellular ROS levels in the LPS/ABCext group, especially in the ABCext/LPS group, compared to the LPS group, highlighting the protective effects of blackberry extract before the LPS damage (Table 3). A similar trend was found for NO production (Table 3), with the highest values (p < 0.05) found in the LPS group and the lowest (p < 0.05) in the ABCext group, with the ABCext pretreatment being the most effective (p < 0.05) compared to the other combined treatments. Our results agree with previous data, which highlighted the ability of the main blackberry compounds, such as anthocyanins, to decrease basal intracellular production of inflammatory-based ROS and NO as a predisposing factor for several noncommunicable chronic degenerative diseases [6, 44–46]. In fact, through different pathways, anthocyanins possess the ability to act as reducing agents in the electron-transfer reaction pathway, donating electrons to the free radicals with unpaired electrons [47]. However, recent studies have shown that the biological activities of anthocyanin also extend to other implicated pathways, such as the improvement of mitochondrial function [48], immunomodulatory properties [49], regulation of lipid metabolism [50], as well as mitochondrial biogenesis and functionality, modulation of intracellular ROS production, and regulation of antioxidant enzymes production involved in intracellular antioxidant defense [51].
Regarding the activities of the main antioxidant enzymes, in LPS-treated cells, a significant (p < 0.05) decrease in the activities of GPx, GR, GST, SOD, and CAT was observed compared to the control group (Table 3), while in ABCext-treated cells, similar values with the control group were measured. Interestingly, the pretreatment with ABCext (ABCext/LPS group) exerted a more protective effect against LPS damage, compared to the LPS/ABCext and ABCext+LPS. The effects of ABCext on ROS and NO levels, as well as on antioxidant enzyme activities, followed a similar behavior pattern to protein and lipid damage (Table 3). The highest values (p < 0.05) of the carbonyl group and TBARS were found in the LPS group, while the lowest (p < 0.05) were measured in the ABCext group and in the ABCext/LPS group, confirming the efficacy of the ACN-rich ABCext treatment in counteracting oxidative damage, as previously reported by our group in ACN-rich extracts from different red fruits [21, 40].
Given the direct effect of various natural compounds from, the same families as those reported here on gene regulation beyond their antioxidant power [35, 50], here we investigated the effect of the ABCext on the expression levels of genes that codify for key proinflammatory and antioxidant mediators in LPS-stimulated murine macrophages. Among the genes analyzed, we observed some interesting changes in the expression profile of 7 of these important mediators (Fig. 2). More specifically, although the nuclear factor kappa B (NFκB) expression was relatively low, not allowing for statistically significant differences between the different stimulation groups to be revealed, we observed that treating RAW cells with ABCext alone or with LPS and ABCext simultaneously shows a tendency to produce a lower NFκB expression compared with LPS-stimulated cells (Fig. 2A). In the same line, pretreating murine macrophages with ABCext before LPS stimulation appeared to confer a protective effect, though a lower NFκB expression did not reach statistical significance compared to LPS-stimulated cells.
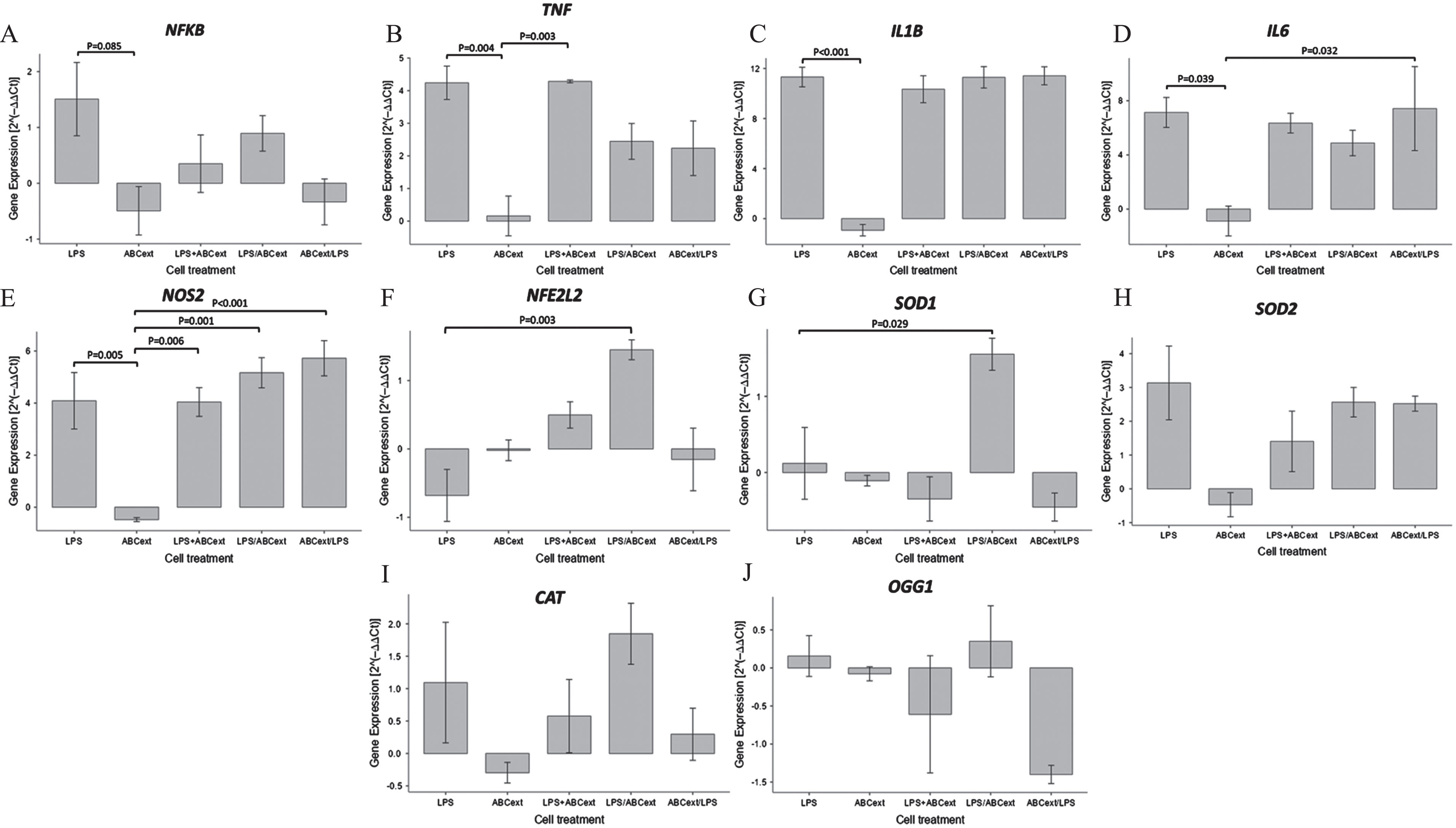
Expression of genes related to the inflammatory response and antioxidant response in RAW 264.7 murine macrophages treated with different combinations of ABCext and/or LPS. Data are expressed as mean values±SD. p < 0.05 and p < 0.01 mean significant differences between the experimental groups.
On the other hand, the analysis for tumor necrosis factor (TNF) expression showed statistically significant differences between the different stimulation groups (p < 0.05) (Fig. 2B). Additionally, we observed that mean TNF expression in both RAW cells treated first with LPS and then ABCext and cells treated first with ABCext and then LPS was almost half of the LPS-treated cells, although these values did not reach statistical significance, while stimulating RAW cells simultaneously with LPS and ABCext did not change TNF expression compared with LPS stimulation alone.
Interestingly, similar expression patterns were found between the other two proinflammatory cytokines, namely IL-1β and IL-6 (Fig. 2C and D, respectively). Indeed, for both genes codifying for these cytokines, treating cells with ABCext alone lowered expression levels compared with LPS-stimulated macrophages (p < 0.001) for each gene, respectively, as well as with constitutive expression levels. However, treating cells simultaneously with LPS and ABCext, first with LPS and then with ABCext, or first ABCext and then LPS did not manage to significantly lower the expression levels of either of these genes compared with the levels induced by LPS stimulation alone (Fig. 2).
Nitric oxide synthase 2 (NOS2) expression exhibited a similar pattern whereby ABCext-stimulated cells were found to significantly reduce NOS2 mRNA levels against LPS-treated cells (p < 0.05) and even reach lower expression levels than in unstimulated cells (Fig. 2E). However, LPS appears to be a strong promotor of NOS2 activation, as both of the treatments that involved LPS and ABCext were shown to induce similar NOS2 expression levels to the LPS alone condition.
Finally, NFE2L2 and SOD1 expression patterns were largely similar (Fig. 2F and G, respectively). Treating RAW cells with LPS was shown to lower the expression levels of both genes, although the differences between basal expression levels did not reach statistical significance. However, treating cells with ABCext after LPS stimulation incremented the mRNA levels of both genes, reaching statistical significance for both superoxide dismutase (SOD1) and nuclear factor erythroid 2-related factor 2 (NFE2L2) (p < 0.05), toll-like receptor (TLR) ligands like LPS, and cytokines like IFNG drive M1 polarization. The outcome of this stimulation is a significant increment in the expression of proinflammatory cytokines, such as IL-1β, IL-12, TNF, IL-6, chemokines like IL-8 (CXCL8), MHC-II molecules, and the production of nitrous oxide and ROS. The principal promoter of the aforementioned mediators is NFκB, a pivotal transcription factor that bridges TLR signaling and the expression of the whole armamentarium of molecules employed by classically activated macrophages [52]. Indeed, LPS, a master activator of TLR4, induced the overexpression of NFκB, TNF, IL-1β, IL-6, and NOS2, while slightly downregulating the expression of NFE2L2 that codifies for the NRF2 protein, a known regulator of anti-inflammatory, detoxifying, and antioxidant responses [53].
Treatment with ABCext, whether before, after, or concomitantly with LPS, largely reversed the LPS-induced upregulation of NFκB, suggesting an important protective role for ABCext in excessive inflammation, although generally low LPS-induced NFκB expression did not allow for statistical significance to be reached. Moreover, treating murine macrophages with ABCext before or after LPS stimulation seemed to partially mitigate TNF, but not IL-1β or IL-6 upregulation (Fig. 2C and D, respectively). This finding implies that the expression of these proinflammatory cytokines is either not exclusively NFκB-mediated in RAW cells, as LPS-induced NFκB expression levels suggest, or that these cells are so sensitive to LPS stimulation that even lower concentrations of this ligand are needed to allow the full anti-inflammatory effect of ABCext to be revealed. Additionally, the induction of NFE2L2 and SOD mRNA expression by ABCext in RAW cells, especially if added after LPS stimulation, revealed a possible antioxidant role of this extract in an endeavor to minimize oxidative stress’ adverse effects.
Overall, gene expression analysis in stimulated RAW 264.7 cells strongly suggests an important anti-inflammatory and antioxidant role of ABCext as has been previously demonstrated in ACN-rich extracts from different fruits [15, 54]. Nonetheless, the anti-inflammatory and antioxidant effects of ABCext warrant further investigation by means of in vivo models, and the exact components exerting these effects need to be identified.
Given the direct effect that molecular regulation has on the inflammatory response, and therefore on the deleterious effects that it can produce, considering the results presented here, it is imperative to know how the polyphenolic constituents of Andean blackberry can influence the regulation of the response. With this aim, we set out to determine whether ABCext administration can alleviate the inflammatory response by mediating the regulation of the NLRP3 inflammasome complex. For this, we used an in vitro model of THP-1 cells. Since NLRP3 activation requires two signals (the priming and the activation signal), we treated the cells with LPS and ATP, respectively [55]. The NLRP3 inflammasome is the best-characterized NOD-type receptor (NLR) that plays an essential role in the inflammation response. It is a multiprotein complex, also known as the caspase-1 activation platform, which once triggered by pathogens, stress, or danger signals, leads to the activation of caspase-1 (CASP1) and thus to the maturation and secretion of interleukin-1 beta (IL-1β) and interleukin 18 (IL-18), which initiate the inflammatory signaling [56, 57]. NLRP3 inflammasome plays a crucial role in the inflammation response, maintaining homeostasis by repairing tissues, eliminating pathogens, and sensing and adapting to stress levels. However, dysregulation of its activation leads to a wide range of diseases, such as metabolic, inflammatory, cardiovascular, and neurological illnesses, as well as cancer. In addition, the aging process is commonly accompanied by low-grade sterile chronic inflammation, whereby NLRP3 inflammasome is activated, thus leading to the accumulation of pro-inflammatory mediators. This process is already known as inflaming [58, 59]. For this reason, it is increasingly necessary to identify compounds, such as those present in food, that target the inflammasome to improve our general health and well-being.
The treatment with LPS+ATP in THP-1 cells (LPS+ATP group) significantly increased (p < 0.01) the expression levels of NLRP3 proteins (1.35 times), activated CASP1 (p20) (1.24 times), and activated IL-1β (p17) (0.65 times) compared to the control group (Ctrl group) (Fig. 3A), indicating that the NLRP3 inflammasome was triggered and therefore the inflammatory response was initiated. However, after treatment with ABCext (LPS+ATP+ABCext group), the levels of these proteins showed a significant decrease (p < 0.01) (0.89 times for NLRP3 protein, 0.73 times for activated caspase-1 (p20), and 0.95 times for activated IL-1β (p17) compared to the group treated with LPS and ATP (LPS+ATP group). These values are close to the basal levels, where, in the case of activated IL-1β (p17), the levels decreased below the basal values of the control group (Ctrl group) (Fig. 3A). In addition, our results showed that there is a downward trend in NFκB levels after treating the cells with ABCext (Section 3.2 of the Results and Discussion), which correlated with the observed decrease in NLRP3 inflammasome and IL-1β levels after the treatment with ABCext since NFκB mediates their expression after the priming signal, which in our case is the LPS addition. These results are promising since they suggest a possible modulatory effect of Andean blackberry polyphenols, more probably the anthocyanins, on the NLRP3 inflammasome complex, due to its ability to decrease its activation, as well as activated CASP1 and IL-1β protein levels, evidencing its potential anti-inflammatory effects. In fact, several works have already reported the modulating effect of polyphenols, e.g., anthocyanins on the NLRP3 complex [60]. It was recently reported that anthocyanin extract from purple sweet potato was able to restrict NLRP3 inflammasome activation in alveolar macrophages infected with Klebsiella pneumoniae. Furthermore, the anthocyanins extracted from the petals of Hibiscus syriacus L. were able to inhibit NLRP3 inflammasome in BV2 microglia cells by alleviating NFκB and endoplasmic reticulum stress-induced Ca2 + accumulation and mitochondrial ROS production [62]. In general, the anti-inflammatory molecular mechanisms of anthocyanins have been associated with certain mechanisms. Those that are noteworthy and are associated with the results presented here include their ability to suppress: (i) the activation of NFκB [63], and (ii) LRR, NACHT, and PYD domains containing protein 3 (NLRP3) inflammasomes by the activation of NRF2 and the thioredoxin-1/thioredoxin-interacting protein (Trx1/TXNIP) inhibitory complex [64].
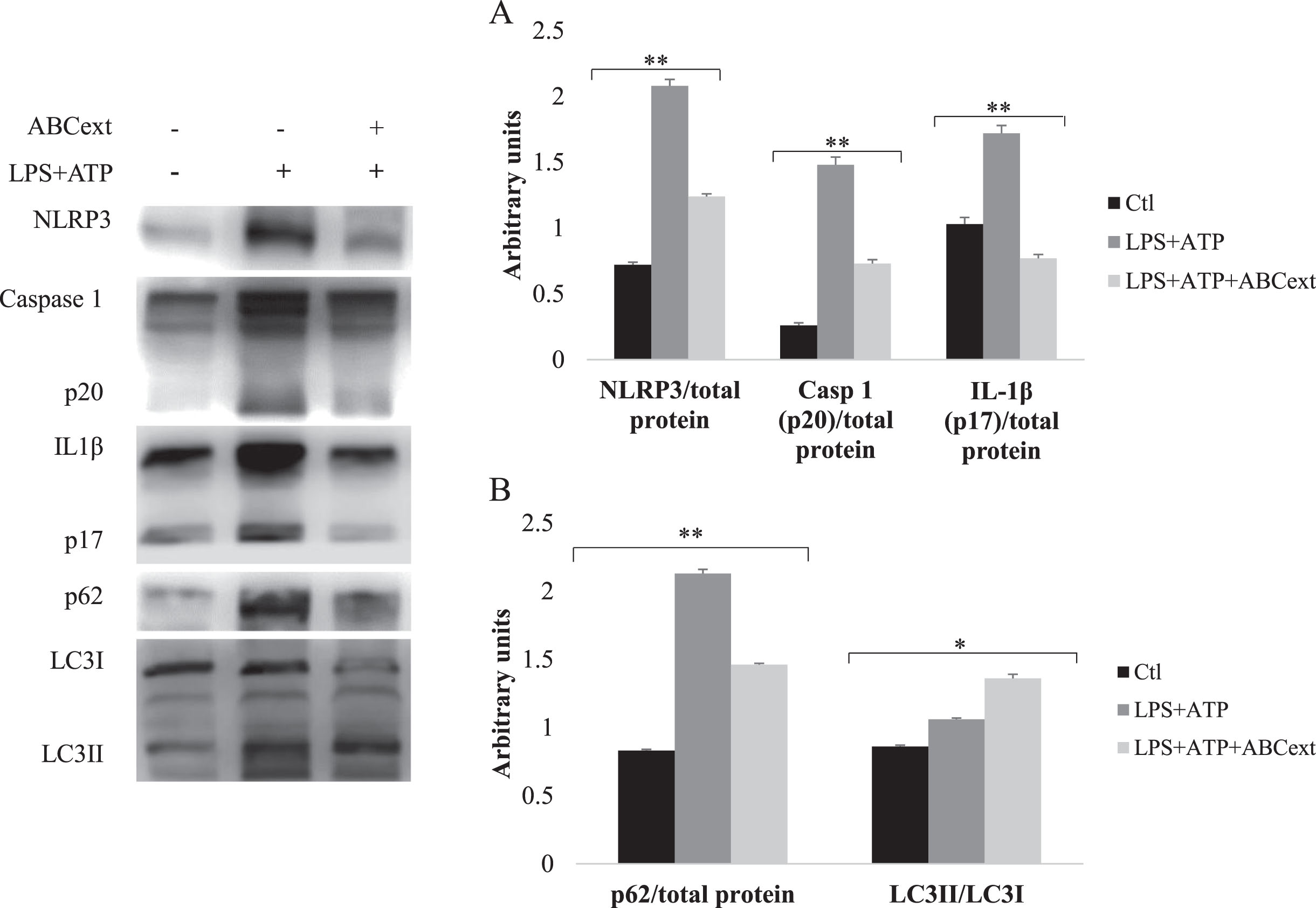
Expression levels of the proteins studied. Data are expressed as mean values±SD. *p < 0.05 and **p < 0.01 mean significant differences between the experimental groups.
After NLRP3 activation, we observed a significant increase (p < 0.01) in the levels of p62 and the LC3I/LC3II ratio in the LPS+ATP group, proteins that play a fundamental role in the autophagy process (Fig. 3B). It consists of a guided degradation of unnecessary or dysfunctional cellular components, such as misfolded proteins and dysfunctional organelles by a double membrane vesicle, or autophagosome, which are then delivered to the lysosomes for their degradation [65]. High levels of p62 together with a high LC3I/LC3II ratio suggest an impairment in the autophagy with the accumulation of non-degraded substrates. However, after treatment with ABCext (LPS+ATP+ABCext group), the p62 levels showed a significant decrease (p < 0.01) and the LC3I/LC3II ratio increased (p < 0.05), compared to the LPS+ATP group, indicating a restoration of the autophagy process (Fig. 3B). Autophagic dysfunction and NLRP3 dysregulation are associated with aging and age-related diseases. By treating the cells with ABCext after triggering the inflammatory response, a reduction in NRLP3 protein level was observed, which leads, according to other studies [66–68], to a decrease in p62 and an increase in the LC3I/LC3II ratio, restoring the autophagy flux. In light of these results, the consumption of Andean blackberry polyphenols, more probably the anthocyanins, seems certainly promising for treating inflammatory-based and age-related diseases. The results presented here agree with several studies that have demonstrated that anthocyanins from different natural sources induce autophagy [69, 70]. More recently, it was demonstrated that anthocyanins from Sambucus canadensis could significantly reduce cell senescence and lens aging by increasing autophagic and mitophagy flux and enhancing mitochondria and cell renewal through the inhibition of the PI3K/AKT/mTOR signaling pathway, which slows aging down [71].
According to the results presented here, it was possible to demonstrate a modulating effect of inflammation by the polyphenolic components of the Andean blackberry. This effect was not only due to its antioxidant capacity, evidenced in the improvement of markers of oxidative damage to cellular macromolecules, but also through its ability to regulate certain genes related to the antioxidant and inflammatory response. The components of the Andean blackberry showed a modulatory effect of the NLRP3 inflammasome complex, decreasing its activation, in addition to restoring the autophagy process, which suggests a direct action of Andean blackberry compounds on the inflammatory response and restoration of the autophagy process. Our findings require further investigation, specifically using in vivo studies and intervention models to evaluate the impact of Andean blackberry components on modulating inflammatory and antioxidant responses and the direct involvement of the NLRP3 complex in these effects.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Acknowledgments
This work was supported by Universidad de Las Américas (Grant Number: BIO.YGB.21.02) and Universidad San Francisco de Quito (Grant Number: 15748) and Proyecto PID2019-106167RB-I00 financed by MCIN/ AEI /10.13039/501100011033. Access to plant genetic resources was granted by means of the “Framework agreement for access to plant genetic resources: MAE-DNB-CM-2017-0072-M-001” celebrated between the Environment Ministry of Ecuador and the Universidad de Las Américas, Ecuador.
