Abstract
BACKGROUND:
High levels of reactive oxygen species (ROS) within the adipose tissue promote a disturbed redox balance and influence its function, impairing adipogenesis, inducing insulin resistance and stimulating adipocyte hypertrophy. Supplementation with antioxidant rich foods can reverse some of these effects. Strawberry is well known as a good source of phytochemicals; however, whether strawberry suppresses increased oxidative stress in 3T3-L1 cells remain unclear.
OBJECTIVE:
The purpose of the present work was to determine the antioxidant potential of a strawberry extract in 3T3-L1 mouse embryo fibroblast cell line.
METHODS:
3T3-L1 pre-adipocytes were induced to differentiate into adipocytes in the presence or absence of different concentrations of the strawberry extract. At the end of the differentiation period, intracellular ROS production, thiobarbituric acid reactive substances (TBARS) content, as well as superoxide dismutase (SOD) and catalase (CAT) activities and gene expressions were evaluated.
RESULTS:
In this study, we confirmed that strawberry extract markedly inhibited increased-oxidative stress in 3T3L1 cells by suppressing intracellular ROS production and decreasing TBARS content. Likewise, SOD and CAT activities and gene expressions were increased.
CONCLUSIONS:
This paper provides evidence that strawberry extract is able to scavenging free radicals and activate endogenous defense systems, highlighting its potential capacity to modulate obesity induced- inflammatory states.
Abbreviations
reactive oxygen species
Dulbecco’s Modified Eagle’s Medium
fetal bovine serum
3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyltetrazolium bromide
thiobarbituric acid reactive substances
thiobarbituric acid
superoxide dismutase
catalase
Introduction
White adipose tissue is mainly constituted by adipocytes (fat cells) and their precursor cells (pre-adipocytes) which differentiate into mature adipocytes through a complex process named adipogenesis [1, 2]. Redox environment determined by the state of the major redox pairs in the cells (e.g. GSSG/2GSH and NAD+/NADH) and reactive oxygen species (ROS) have been recognized as important modulators of this process [2]. ROS, including hydrogen peroxide (H2O2), superoxide anion (
Increased oxidative stress in adipocytes is considered an important pathogenic mechanism of obesity-associated metabolic syndrome [7]. It has been associated with insulin resistance and adipocyte hypertrophy which stimulates inflammation, altered metabolism and dysregulated adipokines secretion [2]. ROS are also required for adipocytes differentiation by regulating mitotic clonal expansion during adipogenesis [6, 7].
Conversely, supplementation with antioxidant compounds that reduce ROS levels, either by inhibiting ROS formation or by inducing antioxidant enzymes, has been demonstrated to improve insulin sensitivity, lipid metabolism and glucose homeostasis in different experimental models [2, 8]. In that sense, strawberries are recognized as a good source of phytochemicals compounds [9, 10] which present several biological activities including antioxidant [11, 12], antiatherogenic [13], anti-inflammatory [14–16] and anticarcinogenic [17–20]. However, to date the effects of strawberry in adipocytes remain unknown. Thus, the purpose of the present study was to determine the antioxidant potential of a strawberry extract in 3T3-L1 mouse embryo fibroblast cell line, one of the most well characterized and reliable models for studying adipocytes differentiation.
Materials and methods
Strawberry extract preparation
Strawberry fruits
Cell culture and differentiation
3T3-L1 pre-adipocytes were purchased from the American Type Culture Collection (ATCC ® CL-173TM) and cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) containing 10% bovine calf serum, 100 IU/mL penicillin and 100
Cell viability (MTT assay)
For cell viability assessment, 3T3-L1 pre-adipocytes were seeded at a density of 5×103 cells/well into 96-well plates and treated with increasing concentrations (from 0 to 1 mg/mL) of the strawberry extract for 24, 48 and 72 h. After treatment, 3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (2 mg/mL) were added and cells were incubated for 2 h. The formazan crystals were dissolved in 100
Assessment of intracellular ROS production by the Tali® Image-Based cytometer
Intracellular ROS levels were determined by the CellROX® Oxidative Stress Kit (Invitrogen TM, Life Technologies, Milan, Italy) according to the manufacturer’s instructions. Briefly, cells were seeded in 6-well plates at a density of 8×104 cells/well and incubated with different concentrations (10, 50 and 100
Cells’ lysates preparation
Cells were seeded in 6-well plates at a density of 8×104 cells/well and incubated with different concentrations (10, 50 and 100
Determination of lipid peroxidation
Lipid peroxidation was determined by the thiobarbituric acid reactive substances (TBARS) assay according to the method proposed by Ohkawa et al. [24]. Briefly, 300
Determination of antioxidant enzymes activity
Superoxide dismutase (SOD) activity was evaluated according to the method proposed by Kakkar et al. [25]. Briefly, 10
Catalase (CAT) activity was assayed according to the method proposed by Aebi, [26]. Briefly, 10
Western blotting analysis
Equal amounts of cellular lysates (75
Statistical analysis
Statistical analyses were performed using STATISTICA software (Statsoft Inc., Tulsa, OK, USA). Data were subjected to one-way analysis of variance for mean comparison and significant differences among treatments were calculated according to Tukey’s HSD (honest significant difference) multiple range test. Data was reported as mean±standard deviation (SD). Differences at
Results
The strawberry extract used in the present study was previously characterized by our group [27]. Pelargonidin 3-O-glucoside (29.30±0.59 mg/g) represented the main bioactive compound (about 80% of the total anthocyanins content), while ellagic acid derivatives (1.74±0.12 mg/g) and flavonols/dihydroflavonols (0.26±0.01 mg/g) were quantified in lower amounts. It presented a high antioxidant capacity as determined by the ferric-reducing antioxidant power (168.25±3.95
Cytotoxic effects of the strawberry extract
In order to evaluate the cytotoxic effects of the strawberry extract on 3T3-L1 cells a MTT assay was performed. As shown in Fig. 1, the strawberry extract did not cause significant effects (
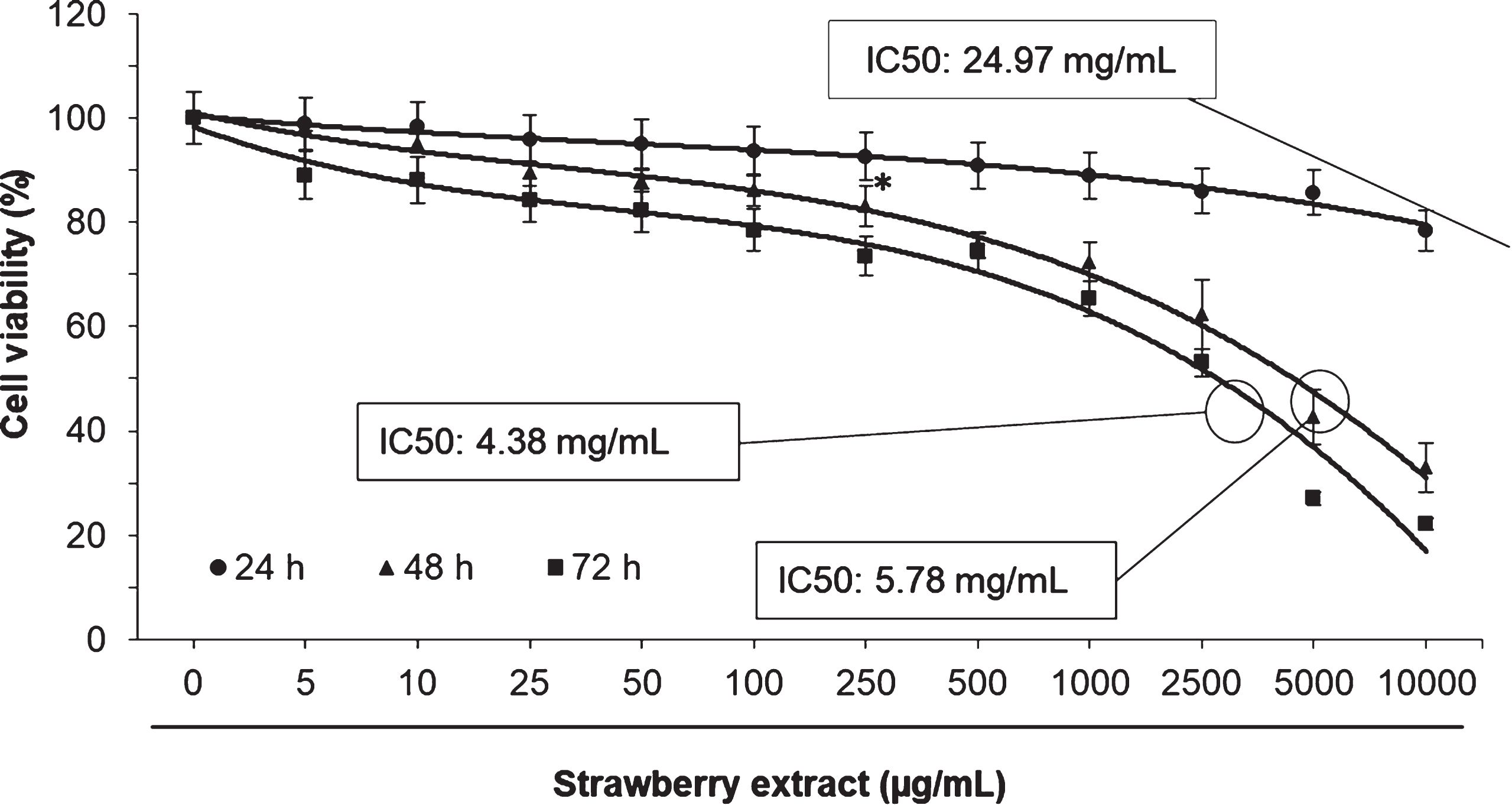
Effects of strawberry extract on 3T3-L1 cells viability as measured by the MTT assay. Cells were treated with the indicated concentration for 24, 48 or 72 h. IC50 indicates the concentration of strawberry extract which reduces the cells viability about 50%. Values are expressed as mean±SD of three independent experiments (
Once demonstrated that strawberry extract did not affect cell proliferation at concentrations ≤100
In untreated mature adipocytes, the intracellular ROS content was 0.6 times higher compared to pre-adipocytes without treatment. In both conditions, strawberry supplementation reduced the ROS levels. In pre-adipocytes (Fig. 2A), strawberry extract decreased ROS production up to 0.65, 0.61 and 0.38 fold compared to control when applied for 24 h at 10, 50, and 100
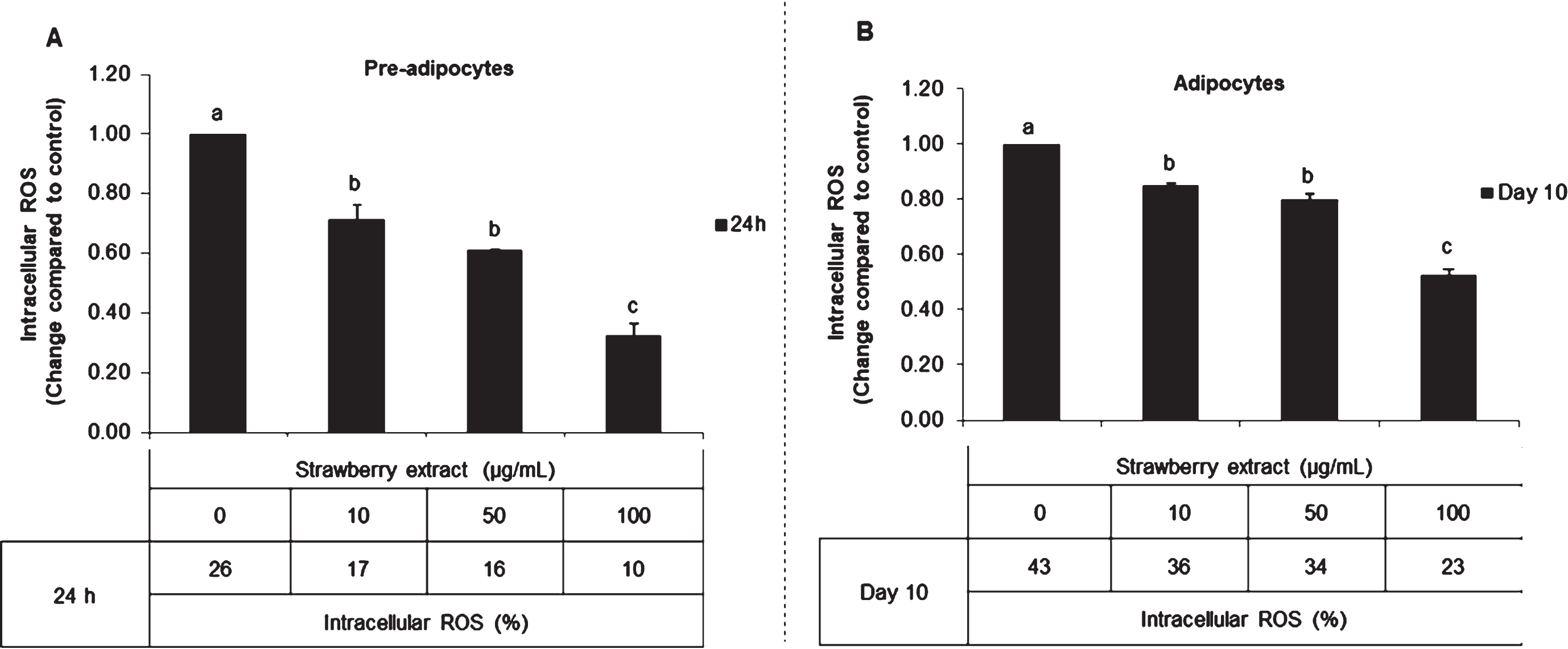
Effects of strawberry extract on intracellular ROS production in 3T3-L1 cells as quantified by the Tali® Image-Based Cytometer. 3T3-L1 pre-adipocytes were incubated with the indicated concentrations of strawberry extract for 24 h (A) or induced to differentiate into mature adipocytes in the presence or absence of the strawberry extract as described above (B). The concentration of 0
Treatment with strawberry extract also reduced lipid peroxidation as can be noticed in Fig. 3. In pre-adipocytes the highest concentration of the strawberry extract significantly (
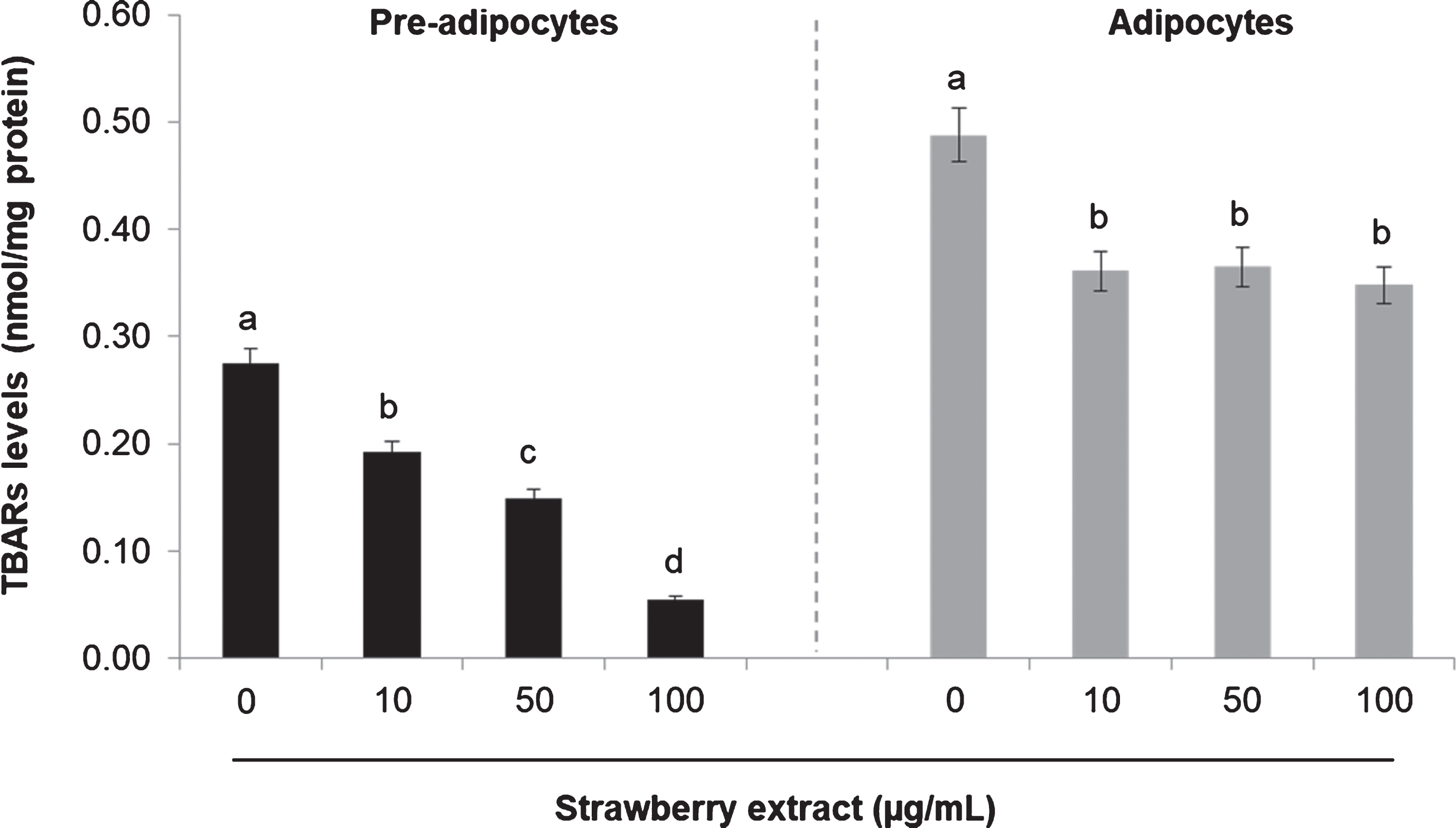
Effects of strawberry extract on lipid peroxidation in 3T3-L1 cells as measured by the TBARs Assay. 3T3-L1 pre-adipocytes were incubated with the indicated concentrations of strawberry extract for 24 h or induced to differentiate into mature adipocytes in the presence or absence of the strawberry extract as described above. The concentration of 0
The activity and the protein expression of the antioxidant enzymes SOD and CAT were also evaluated. In mature adipocytes SOD (Fig. 4A) and CAT (Fig. 5A) activities was considerably lower compared to undifferentiated cells (considering the untreated cells).
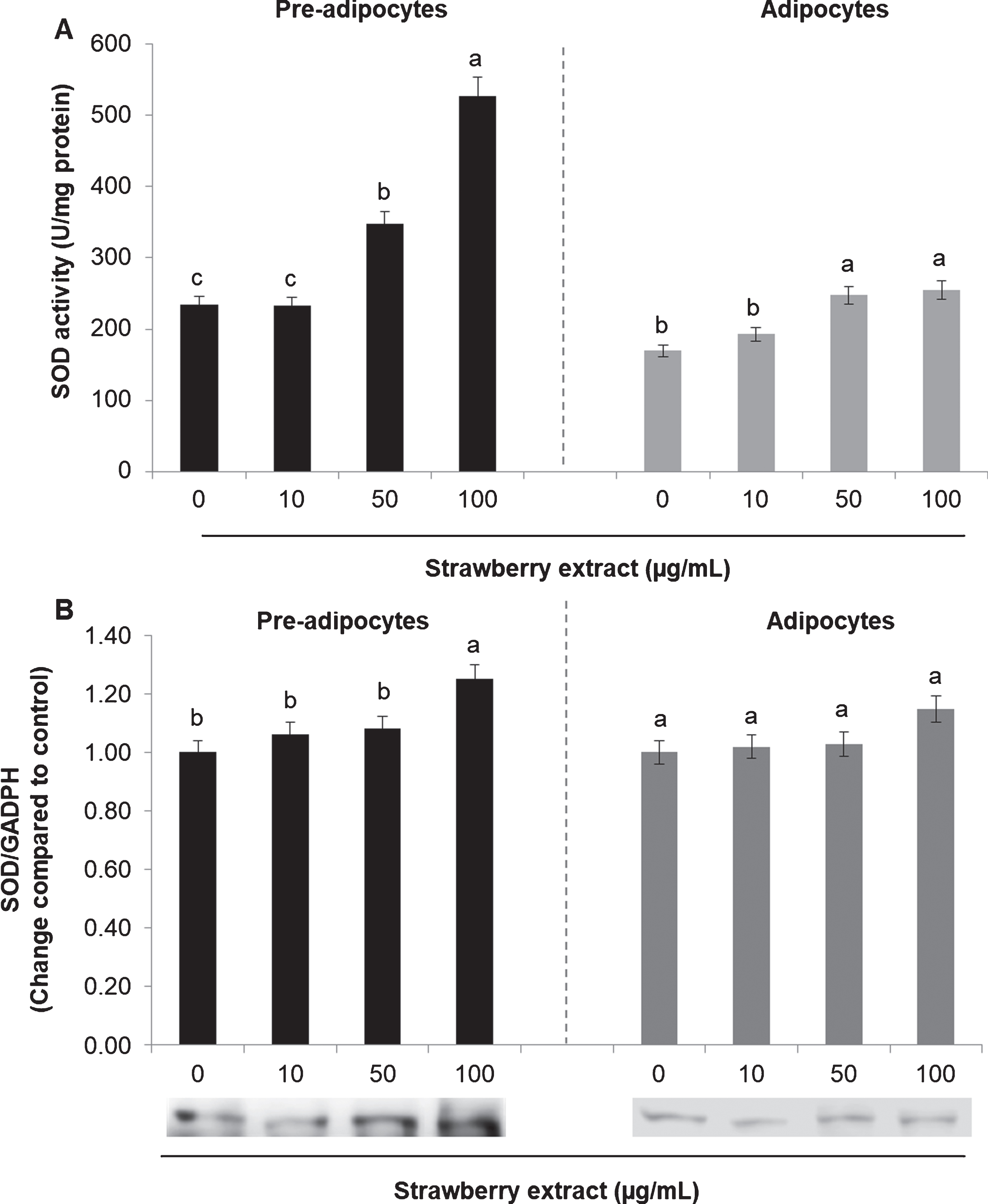
Effects of strawberry extract on superoxide dismutase (SOD) activity (A) or protein expression (B) in 3T3-L1 cells. The enzyme activity was measured by a colorimetric method while the protein expression was evaluated by western blotting. The protein signals were detected by a Lycor C-Digit Blot Scanner and quantification was made using the software Image Studio 3. 3T3-L1 pre-adipocytes were incubated with the indicated concentrations of strawberry extract for 24 h or induced to differentiate into mature adipocytes in the presence or absence of the strawberry extract as described above. The concentration of 0
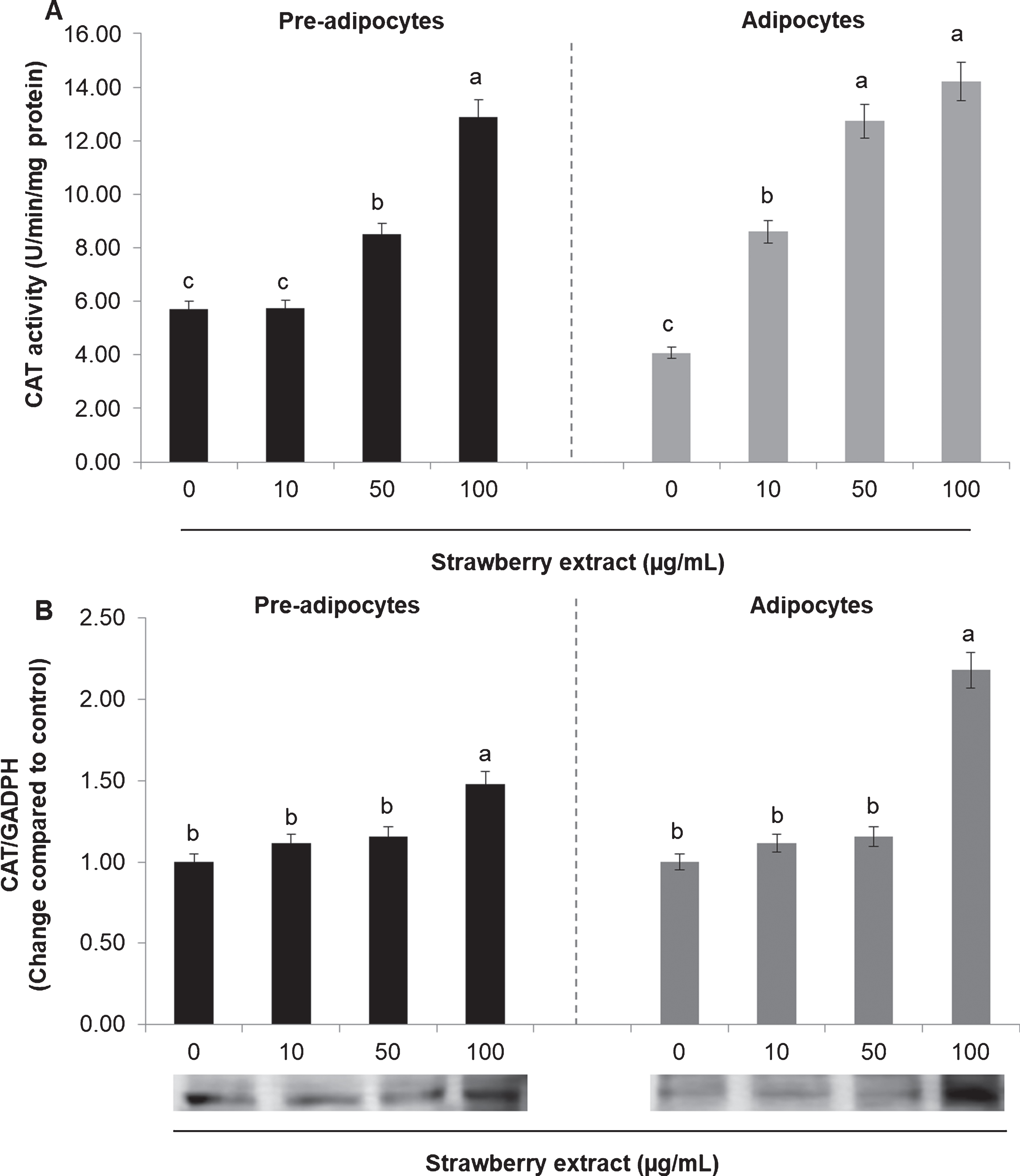
Effects of strawberry extract on catalase (CAT) activity (A) or protein expression (B) in 3T3-L1 cells. The enzyme activity was measured by a colorimetric method while the protein expression was evaluated by western blotting. The protein signals were detected by a Lycor C-Digit Blot Scanner and quantification was made using the software Image Studio 3. 3T3-L1 pre-adipocytes were incubated with the indicated concentrations of strawberry extract for 24 h or induced to differentiate into mature adipocytes in the presence or absence of the strawberry extract as described above. The concentration of 0
In both cases (either in pre-adipocytes and adipocytes) strawberry extract significantly increased (
Regarding the CAT, strawberry extract caused an augmentation in the enzyme activity in a dose-dependent manner (Fig. 5A) while it increased the protein expression only at the highest concentration (Fig. 5B) for both pre-adipocytes and mature adipocytes.
Oxidative stress has been associated to several metabolic diseases, including type 2 diabetes, insulin resistance, cardiovascular diseases and obesity. In adipocytes, ROS overproduction has been associated with multiple forms of insulin resistance [2, 7] and with some modifications in endoplasmic reticulum, mitochondrial functionality and cell signaling, that lead to inflammatory conditions [28]. The molecular mechanism involved could be based -at least in part- on ROS-induced production of the inflammatory cytokine tumor necrosis factor alpha (TNF
Likewise, increased biomarkers of lipid peroxidation such as TBARS and 4-hydroxynonenal protein adducts in adipose tissue has been significantly correlated with the body mass index in different animal and human models [2]. Also the inhibition of antioxidant enzymes has been related with increased ROS production and inflammatory states [28–31]. By contrast, mitochondrial-targeted antioxidant as well as overexpression of SOD and CAT have been associated to insulin sensitivity in mice fed a high-fat diet [2, 8]. In that sense, the observed diminution in TBARS levels as well as the stimulation of the antioxidant enzymes after strawberry treatment render this fruit a good candidate for the treatment and/or prevention of diabetes and obesity.
In conclusion, we demonstrated that strawberry supplementation significantly decreased ROS production and lipid peroxidation while increased antioxidant enzymes activities and expression in both pre-adipocytes and matures adipocytes. These effects may be attributable to strawberry polyphenols ability to scavenging free radicals and activate endogenous defense systems, highlighting its potential capacity to modulate obesity induced- inflammatory states.
Footnotes
Acknowledgments
Patricia Reboredo-Rodríguez is supported by a post-doctoral contract from Xunta de Galicia.
