Abstract
Alzheimer’s disease (AD) accounts for most dementia cases, but we lack a complete understanding of the mechanisms responsible for the core pathology associated with the disease (e.g., amyloid plaque and neurofibrillary tangles). Inflammation has been identified as a key contributor of AD pathology, with recent evidence pointing towards Reelin dysregulation as being associated with inflammation. Here we describe Reelin signaling and outline existing research involving Reelin signaling in AD and inflammation. Research is described pertaining to the inflammatory and immunological functions of Reelin before we propose a mechanism through which inflammation renders Reelin susceptible to dysregulation resulting in the induction and exacerbation of AD pathology. Based on this hypothesis, it is predicted that disorders of both inflammation (including peripheral inflammation and neuroinflammation) and Reelin dysregulation (including disorders associated with upregulated Reelin expression and disorders of Reelin downregulation) have elevated risk of developing AD. We conclude with a description of AD risk in various disorders involving Reelin dysregulation and inflammation.
INTRODUCTION
Dementia is a symptom of neurodegenerative diseases associated with significant quality of life impairment for patients and families [1–3]. Alzheimer’s disease (AD) typically results in progressive memory decline preceding dysfunction to numerous cognitive domains including spatial navigation, executive functioning (e.g., attention, decision making, and planning) and language [1]. Although treatments for AD provide temporary symptom relief at various stages of the disease and evidence consistent with slower disease progression has been shown with currently approved treatments, these treatments do not cease disease progression in terms of clinical presentation or neuropathology [4–6]. This is partly related to our incomplete understanding of the mechanisms responsible for the development of AD. Reelin, an extracellular matrix protein, could be a valuable therapeutic target for AD as a variety of studies show dysregulation of Reelin cleavage and Reelin signaling occurs in AD [7–9]. Promoting Reelin homeostasis could be a valuable therapeutic avenue for preventing disease onset and/or reversing AD pathology, although additional work is needed to support Reelin-based therapeutics in the context of AD.
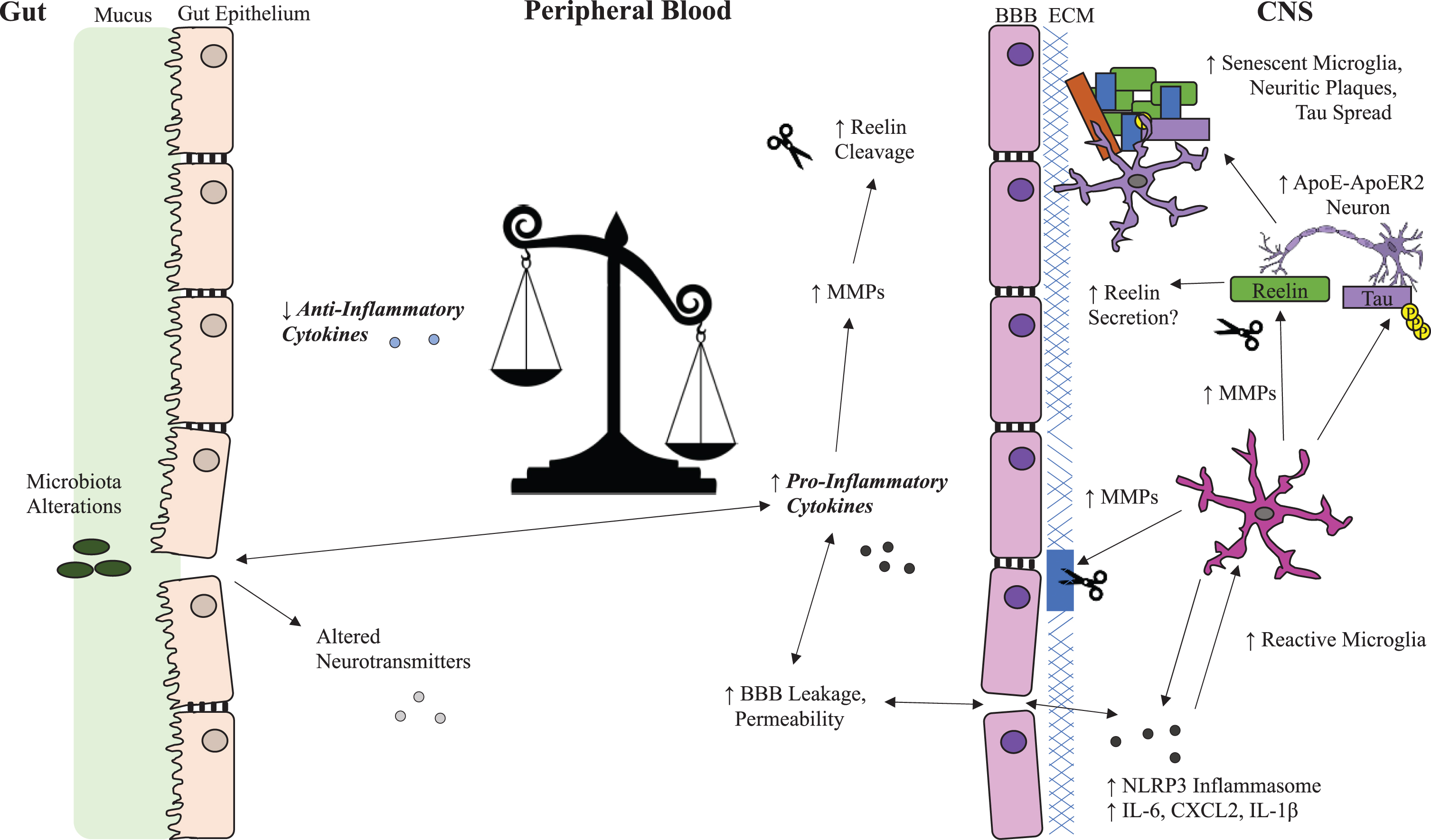
Key pathological features of AD include senile plaques composed of amyloid-β (Aβ) peptides and neurofibrillary tangles (NFTs) of hyperphosphorylated tau protein [10, 11]. Aβ is a by-product of cleavage of amyloid-β precursor protein (AβPP) which is a protein necessary for healthy neuronal development and function [12]. Tau is a microtubule-protein that confers microtubule stability and phosphorylation of tau allows microtubule dissociation, which is critical for healthy neuronal vesicular transport [13]. Hyperphosphorylation of tau destabilizes microtubules and leads to the formation of NFTs [14]. Reelin is a stress-sensitive and inflammation-sensitive protein that inhibits cleavage of AβPP into Aβ, reduces the toxicity of Aβ and inhibits the phosphorylation of tau [15–21]. Within AD and experimental models of AD pathology, research indicates inflammatory processes play a key role in the development of the disease [22]. A role of Reelin in inflammatory conditions has recently been identified, suggesting enhancement of Reelin cleavage may serve an important anti-inflammatory function that, over time, promotes core pathologies of AD due to a reduction of effective Reelin signaling [23, 24]. We propose a mechanism whereby peripheral inflammation and/or neuroinflammation compromise Reelin homeostasis and leads to the development of AD pathology, as outlined in Fig. 1. In this review, connections between Reelin, inflammation and AD are described and the basis for the loss of Reelin homeostasis hypothesis of AD is outlined.
Alzheimer’s disease and inflammation
Inflammation is a tightly regulated protective process involving activation of immune and non-immune cells in response to infection, toxic compounds, damaged cells, and irradiation with the purpose of restoring homeostasis [25]. When dysregulated, inflammation can persist beyond the days or weeks required to resolve an inflammatory stimulus. Inflammation is presently considered a key promoter of AD pathology as reviewed elsewhere [26]. Inflammation is observed in the early stages of AD and exacerbates AD pathology experimentally, suggesting inflammation is a key pathological feature of AD rather than a by-product of the disease [27, 28]. Phosphatidylinositol 3-kinase (PI3K) and glycogen synthase kinase-3β (GSK3β) play important roles in both promoting and resolving inflammation [29, 30]. GSK3β also plays a central role in AD as a regulator of tau phosphorylation [28, 31–33]. GSK3β activity is regulated by Reelin signaling and is further described in the Reelin overview section. One of the most well-established genetic risk factors for late onset AD is the
Microglia, the resident immune cells of the central nervous system (CNS), are considered the major drivers of neuroinflammation and are responsible for a variety of important regulatory functions in both healthy and disease states [41]. Microglia produce reactive oxygen species and secrete pro-inflammatory cytokines and matrix metalloproteinases (MMP) that contribute to blood-brain barrier (BBB) breakdown [41–46]. Functions of MMPs include cleaving extracellular matrix proteins which increases permeability of the BBB and allows infiltration of peripheral immune cells and inflammatory cytokines into the CNS, which have been reported in AD [47–52]. Microglial proliferation, morphology, and survival are regulated by cytokine signaling and are susceptible to inflammation [53, 54]. Hyperinflammation promotes microglial priming, increasing susceptibility to evoke an exaggerated inflammatory response to secondary inflammatory stimuli and also makes microglia prone to cellular senescence, an arrest of cell cycle that impairs organelle function and halts proliferation [22, 55–57]. Microglia senescence prevents microglia from effectively serving their numerous functions required for healthy neural activity [58]. The impact of microglial senescence and microglial dysfunction (involving impaired phagocytosis, spreading of amyloid beta and tau aggregates, and maladaptive synaptic element pruning) on AD progression was recently reviewed by Lau et al. [55]. Tau pathology causes neurons to externalize phosphatidylserine which triggers microglial phagocytosis, promotes microglial senescence, and causes a positive feedback loop of seeding tau aggregates and spreading of tau-based pathology [59].
Experimental evidence shows inflammation is a key regulator of AD pathology. In AβPP/PS1 transgenic mice (which express mutant human AβPP with the Swedish mutation (K6790N, M671L) and human Presenilin 1 with the M146V mutation) and 5xFAD mice (which express mutant human AβPP with the Swedish, Florida (I716V) and London (V717I) familial AD mutations and human PS1 harboring two familial AD mutations, M146L and L286V), interferon (IFN)-
The viral mimetic polyinosinic-polycytidylic acid (Poly(I:C)) has also been applied experimentally to elevate inflammatory cytokines. Prenatal exposure to Poly(I:C) predisposes wild-type mice to develop increased hippocampal AβPP and proteolytic fragments of amyloid, increased phosphorylated tau and impairment of working memory in adulthood [27]. A subsequent immune challenge in adult mice exposed prenatally to Poly(I:C) exacerbated neuropathological features of AD including AβPP proteolytic fragment deposition, tau aggregation, reactive astrogliosis, and microglial reactivity suggesting inflammatory challenges, particularly in early life, predispose animals to develop AD-like pathology upon future inflammatory events [27]. Peripheral exposure of 4–6 month old C57/BL/6J mice to Poly (I:C) for 7 days also impaired contextual fear conditioning and increased hippocampal Aβ levels 72 h after the final Poly(I:C) exposure, suggesting Poly(I:C) exposure does not just increase AD-like pathology when delivered during the gestational period, as adult-mice also developed greater AD-like pathology and induced cognitive impairment after Poly(I:C) exposure [65]. Splenectomy exacerbates AD-like pathology in AβPP/PS1 mice (which express APP695 with the Swedish mutation and human Presenilin1 lacking exon 9) further implicating the peripheral immune system to AD [66, 67]. In contrast to the various studies showing inflammation promotes or exacerbates AD pathology, Th-2 biased Aβ-specific T-cell infusions reduced plaque-associated microglial reactivity, vascular amyloidosis, and cognitive impairment without infiltrating into the CNS, suggesting modulation of peripheral inflammation can be a valuable therapeutic target for AD [68].
There exists a complex interaction between peripheral and central inflammation in the context of neurodegenerative diseases including AD. Chronic peripheral inflammation, indicated by elevated C-reactive protein (CRP) levels, is associated with age-related cognitive decline and all-cause dementia [69]. The gut microbiome modifies peripheral inflammation and mice transplanted with gut microbiota from human patients with AD show activation of the NLRP3 inflammasome, heightened hippocampal microglial reactivity and cognitive deficits [70]. Cognitively impaired adult humans with brain amyloidosis have higher levels of cytokines (IL-6, CXCL2, NLRP3, IL-1β) compared to controls and cognitively impaired individuals with no amyloidosis, and these alterations were associated with higher abundance of proinflammatory gut microbiota, specifically Escherichia and Shigella taxa, and lower levels of Eubacterium rectale, suggesting diet is a modifiable risk factor for AD [71]. Supporting a role of diet in modifying peripheral inflammation and reducing AD risk, adherence to the Mediterranean diet, which has antioxidant and anti-inflammatory properties, is associated with reduced risk of developing AD [72–76]. Future treatments might involve regulation of peripheral inflammation and/or BBB permeability given the various lines of evidence showing peripheral inflammation contributes to AD pathology.
Reelin overview
Reelin is a pleiotropic extracellular matrix protein associated with regulating neuronal migration in embryonic development and in adulthood, promotes synaptic plasticity, cell adhesion and cell proliferation [77–79]. Reelin dysregulation is seen in various neuropsychiatric conditions including schizophrenia, bipolar disorder, major depression, and AD [80–83]. Reelin is a large multi-domain protein consisting of a C-terminal of predominantly basic amino acids, eight Reelin repeat domains of 350–390 amino acids, and a F-spondin-like N-terminal domain [84]. Outside the CNS, Reelin is expressed in the liver, kidney, blood (erythrocytes, platelets), bone marrow, lymphatic tissues, enteric nervous system, and the yolk sac during development, serving a plethora of functions including regulating cell growth, cell cycle, cell motility, cellular adhesivity, hemostasis and platelet spreading [85]. Although the full-length Reelin molecule is large, at approximately 410 kDa, evidence of Reelin in caveolar vesicles of brain endothelial cells suggests Reelin could be actively trafficked into the brain from the periphery [86]. Reelin was first recognized for its important roles in embryonic neurodevelopment, and therefore, most of our understanding of Reelin relates to the central nervous system wherein Reelin has been considered a homeostatic regulator of synaptic plasticity [87]. In the brain parenchyma Reelin promotes cortical synaptogenesis, dendritic spine development and maturation, memory formation, hippocampal plasticity, neurogenesis, neuronal migration, and maturation and integration of dentate granule neurons, as reviewed elsewhere [88].
Reelin signaling is transduced through binding of Reelin to the apolipoprotein E receptor ApoER2, very-low density lipoprotein receptors (VLDLR), and interactions with
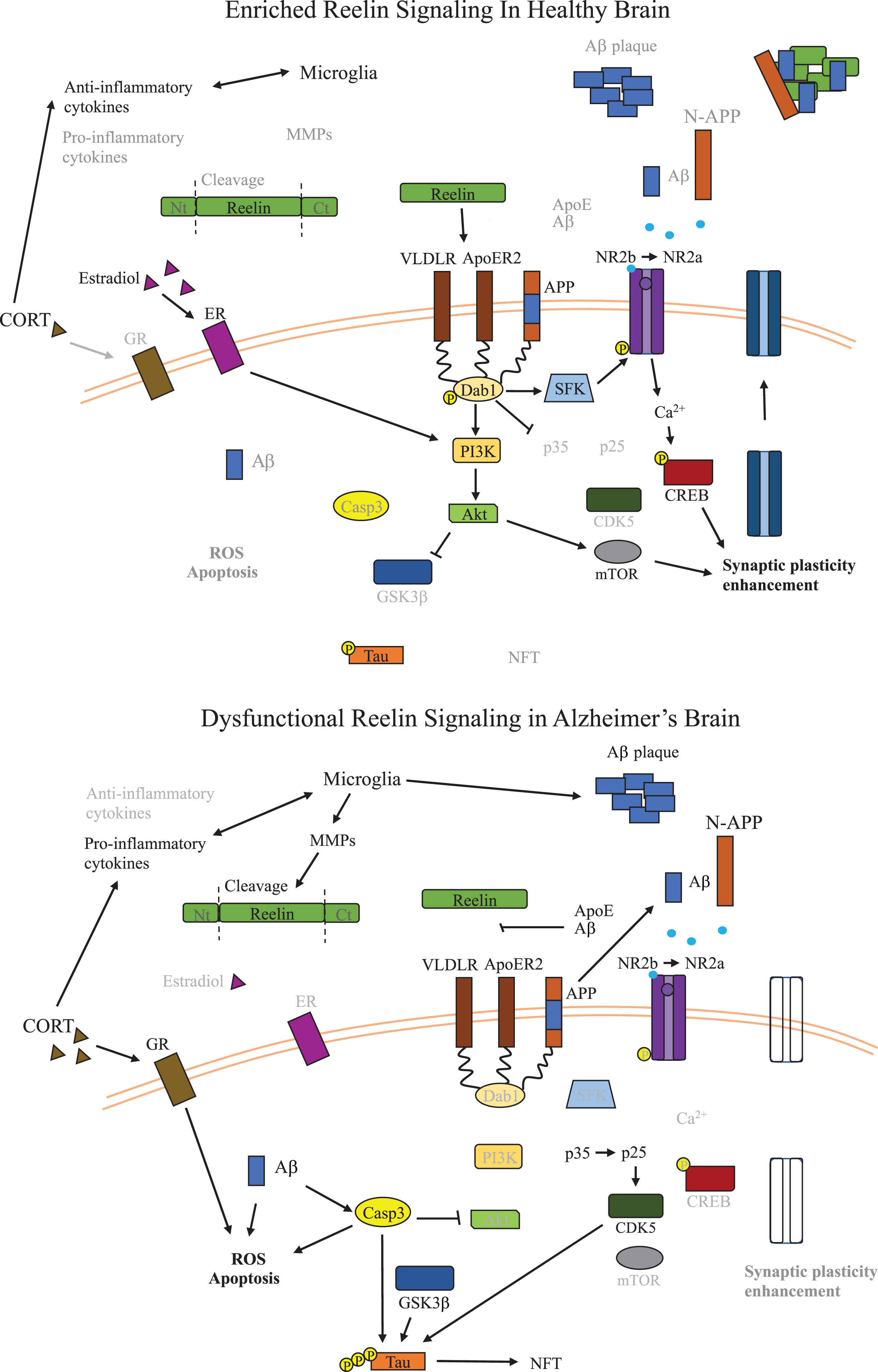
Reelin signaling also leads to phosphorylation of PI3K which inactivates GSK3β through phosphorylation of GSK3β at serine 9 [105]. As mentioned, GSK3β signaling is a key source of tau phosphorylation that is dysregulated in AD and is modified by inflammation [28, 106]. Reelin signaling also regulates mechanistic target of rapamycin (mTOR), which regulates metabolism, immune response, and autophagy, as well as extracellular signal-regulated protein kinases 1 and 2 (ERK 1 and 2), which control cell proliferation and cell death [107–110]. Both mTOR and ERK1/2 are dysregulated in AD and expression of mTOR has been correlated to both total and phosphorylated tau in AD [107, 110–113]. Figure 2 outlines Reelin signaling pathways in the context of AD. An extensive review of Reelin signaling pathways beyond the scope of AD is described elsewhere [114].
Reelin in cognition
Although many disorders involving Reelin dysregulation, including but not limited to depression, schizophrenia, AD, epilepsy, and multiple sclerosis, are widely recognized to present cognitive alterations, whether Reelin dysregulation is responsible for these alterations is not yet clear [7, 115–117]. For example, although AD and major depression involve reduced plasticity, lower spine density and disruption of associative learning and memory, it is unclear whether Reelin dysregulation is a major driving force for such dysfunction in these contexts. In addition, the impact of Reelin therapeutics have not been evaluated in humans and likewise, direct roles of Reelin treatment in normalizing cognition in humans have yet to be shown. Therefore, rather than speculate on the roles of Reelin in known neuropsychiatric disorders in which Reelin is dysregulated and cognition is affected, we focus here on the impact of Reelin signaling on cognition in rodent models.
Two relevant animal models for studying the roles of Reelin in cognition are the heterozygous reeler mouse (HRM) and models of chronic stress. HRM have a mutation leading to deletion of roughly 126 kb in one Reelin allele resulting in approximately 50% reduced Reelin expression [118]. Although there exists the Reeler mouse line, which are homozygous Reelin mutants devoid of Reelin expression, these mice have major brain malformations [119, 120]. While the Reeler mouse has been pivotal to identifying the roles of Reelin in brain development, it is difficult to conclude whether any behavioral deficits observed are a result of insufficient Reelin signaling and not resulting from the major brain malformations. Therefore, the HRM model, which has normal neuronal migration and brain development, is a more applicable model for assessing the role of Reelin signaling in cognition across the lifespan. Deficits observed in the HRM model include reduced dendritic spine density, altered synaptic plasticity and behaviorally deficits have been observed in associative learning, memory, and pre-pulse inhibition [121–124]. Reversal learning, the process of learning to inhibit previously rewarded actions, is also impaired in HRM [123, 124]. Impairment of HRM in reversal learning, particularly with increased perseverative responses, implicates Reelin in inhibitory processing and impulsivity, consistent with the roles of Reelin in regulating synaptic plasticity [96, 126].
Reelin supplementation through intraventricular injection can also influence behavior and normalize various deficits associated with Reelin depletion in rodent models such as the HRM, including enhancement of hippocampal synaptic function and normalization of LTP, fear conditioning, associative learning, and spatial memory [98, 128]. Supplementation of wild-type mice with Reelin through bilateral intraventricular injection has also been shown to improve context-dependent learning and memory and to enhance LTP [98]. In a mouse model of Angelman syndrome (a condition characterized by synaptic dysfunction and reduced Reelin expression) caused by a UBE3A null mutation, bilateral intraventricular injection of Reelin rescued dendritic spine density, synaptic function, learning and memory [125]. Reelin levels are also reduced in the dentate gyrus sub-granular zone in rodent models of chronic stress which induce behavioral changes including despair-like behavior and spatial memory impairment [129–131]. Either intravenous or intraventricular injection of recombinant Reelin resolves stress-induced depression-like behavior in rodents, stress-induced spatial memory deficits and Reelin cell counts in the dentate gyrus [129–131]. Additionally, a single peripheral Reelin injection reverses alterations to LTP induced by chronic stress [132]. Reversal of behavioral and neurobiological alterations with an intravenous injection of Reelin suggests that either Reelin crosses the BBB and/or peripheral Reelin indirectly normalizes Reelin expression in the brain to restore neural functions of Reelin following chronic stress. That Reelin supplementation resolves behavioral and neurobiological impairments of low Reelin signaling suggests Reelin therapeutics could be valuable for treatment of AD and other disorders where Reelin signaling is disrupted.
Reelin in AD
As mentioned above, Reelin signaling regulates a variety of pathways important for AD. In support of a key role of Reelin signaling in the context of AD, recently a gain of function mutation to the Reelin gene, Reelin-COLBOS, was identified as providing resistance in familial AD, and when inserted into the human tau transgenic mouse model (which express human tau with a P301L substitution), the Reelin-COLBOS mutation reduced tau hyperphosphorylation and intensity of neurofibrillary tangles [133]. In addition, a large single nucleus RNA sequencing study identified the loss of Reelin inhibitory neurons in the prefrontal cortex as a key factor for predicting level of cognitive impairment in AD, as individuals with maintained cognitive functioning had higher levels of Reelin expressing inhibitory neurons than those with lower scores in cognitive testing [134]. Reelin has been shown to be reduced in the entorhinal cortex at all stages of AD and was undetectable in severe AD entorhinal cortex samples [135]. Another study showed fewer Reelin positive cells in post-mortem frontal cortex, hippocampus, and entorhinal cortex in AD, as well as lower levels of full length Reelin [7]. Co-staining for Reelin, tau and Aβ in AD brain samples showed dense aggregations of Reelin positive cells within and surrounding amyloid plaques [7]. Reelin also binds to AβPP and reduces β cleavage, which may relate to accumulation of Reelin in amyloid plaques [7, 15]. Dab1 phosphorylation reduces β-cleavage of AβPP to Aβ
Understanding Reelin cleavage and fragmentation is important for interpreting the various findings of Reelin expression in AD as a variety of studies show altered proteolytic processing of Reelin occurs in AD [8, 83]. The full-length Reelin molecule is composed of 8 epidermal growth factor repeats that can be cleaved at Reelin repeat 3, or the N-terminal cleavage site, and C-terminal cleavage between repeats 6 and 7 [88, 137]. The N-terminal cleavage at repeat 3 leaves an approximately 180 kDa and 250 kDa Reelin fragment whereas the C-terminal cleavage between repeats 6 and 7 leaves an approximately 310 kDa fragment and 100 kDa Reelin fragment [9, 137]. The Reelin central fragment composed of repeats 3–6 is required for effectively inducing downstream signaling and N-terminal cleavage reduces the duration and range of Reelin-mediated Dab1 phosphorylation [138–141]. A variety of molecules are responsible for Reelin cleavage, many of which also serve anti-inflammatory functions [142]. Increasing the activity of these enzymes may serve to restore inflammatory homeostasis, but in chronic inflammatory conditions such as AD, increased enzymatic activity could promote development of AD through Reelin cleavage. Identified Reelin proteases include tissue plasminogen activator, meprin
Multiple studies support Reelin cleavage being dysregulated in AD. Increased levels of the inactive 180 kDa N-terminal Reelin fragment were found in the cerebrospinal fluid (CSF) of individuals diagnosed with frontotemporal dementia and AD, where the expression of the 180 kDa Reelin fragment correlated to changes in MMSE score [8]. Another study also reported increased levels of the 180 kDa Reelin fragment in the CSF in AD, which was also seen in frontotemporal dementia, progressive supranuclear palsy, and Parkinson’s dementia [154]. Elevation of Reelin mRNA and the 180 kDa Reelin fragment were also observed in frontal cortex samples of patients with AD, again suggesting proteolytic processing of Reelin is dysregulated in AD [83]. In contrast, a recent study evaluating Reelin in the CSF of individuals with AD showed a reduction of full-length Reelin, elevated levels of the 310 kDa fragment and the 100 kDa fragment, lower levels of the 250 kDa fragment, but no change to the 180 kDa fragment [9]. Although these results also show dysregulated Reelin cleavage in AD, they conflict with the previously mentioned studies observing elevated N-terminal cleavage products in AD, instead showing evidence of elevated C-terminal cleavage products. Reports of Reelin aggregates within and surrounding amyloid plaques and various studies showing dysregulation of Reelin cleavage, particularly increased expression of the N-terminal Reelin fragments that have reduced signaling capacity, indicate a reduction of Reelin bioavailability and signaling capacity occurs in AD [7–9, 83]. Altered levels of Reelin fragments strongly suggests Reelin cleavage is dysregulated in the context of AD, which has been reviewed elsewhere [137, 154]. Table 1 provides an overview of human studies linking AD to Reelindysregulation.
Overview of human studies of Alzheimer’s disease evaluating relationships between Reelin and AD pathology
CSF, cerebrospinal fluid; FTD, frontotemporal dementia; HC, healthy controls; WB, western blotting; EC, entorhinal cortex; IHC, immunohistochemistry; CR, Cajal Retzius cells; FC, frontal cortex; MCI, mild cognitive impairment; HPC, hippocampus
Evidence derived from experimental rodent models of AD also suggests Reelin signaling regulates the two hallmark pathological features of AD, amyloid and tau pathology. As mentioned above, N-terminal cleavage by ADAMTS-3, which is secreted by excitatory neurons, dysregulates Reelin-mediated Dab1 signaling [143, 144]. Relatedly, although Reelin is predominantly expressed in inhibitory neurons, Reelin is uniquely expressed in excitatory neurons in the entorhinal cortex, precisely where AD pathology is most severe, and atrophy first presents [20, 135]. The mechanisms regulating ADAMTS-3 secretion in the context of AD are not yet clear, but experimentally reducing ADAMTS-3 reduced Aβ levels in the cerebral cortex of 3-month-old APPNL - F mice (which express APP with the Swedish and Iberian (I716F) mutations) which otherwise display Aβ accumulation, increased synaptic loss, microgliosis, and astrogliosis around 6 months of age [150, 155]. Alike human samples, Reelin immunostaining has been shown in amyloid plaques in the hippocampus and cortex of wild-type mice and transgenic mice expressing mutant human AβPP (with the Swedish, Dutch (E693Q) and London mutations) and mutant human presenilin-1 (with mutation M146L) [156, 157]. Conditional Reelin knockout causing loss of Reelin beyond 2 months of age, when crossed with Tg2576 mice (which express APP with the Swedish mutation), was associated with deficits in the Morris water maze and in contextual and cued fear conditioning at 7 months of age that were not seen in Tg2576 mice or in mice with conditional Reelin knockout not crossed with Tg2576 mice [16].
Loss of Reelin signaling has also been shown to exacerbate tau hyperphosphorylation in rodent models of AD [15, 158]. For example, relative to double human AβPP transgenic mice (which express the human APP695 gene containing the Swedish and Arctic (E693G) mutations) not crossed with HRM, double human AβPP transgenic crossed with HRM had elevated levels of amyloid plaque distribution, accumulation of hyperphosphorylated tau and elevated microglia reactivity [18]. In contrast to studies showing the impact of Reelin depletion, Reelin overexpression in transgenic mice expressing mutant human AβPP (with Swedish and Indiana (V717F) mutations) delayed Aβ plaque formation and normalized deficits in recognition memory [17]. Similarly, in the VLW transgenic mouse model of tauopathy (which overexpress a 4-repeat isoform of human tau bearing three frontotemporal dementia linked with parkinsonism-17 mutations (G272V, P301L and R406W)), overexpressing Reelin normalized tau phosphorylation, resolved deficits in latent inhibition, and enhanced LTP [19]. Evidence of Reelin overexpression and supplementation reversing AD pathologies experimentally again suggests that Reelin-based therapeutics could have value in treatment of human AD, although this requires empirical validation. Table 2 shows key
Overview of key animal studies evaluating relationships between Reelin and AD-like pathology
Tg, transgenic; WB, western blotting; KO, knock-out; IHC, immunohistochemistry; HRM, heterozygous Reeler mice; HPC hippocampus; WT, wild-type; EC, entorhinal cortex; LTP, long-term potentiation.
Reelin and inflammation
Although the roles of Reelin in inflammation are not yet completely clear, several reviews describe roles of Reelin in inflammation [159, 160]. A potential role of Reelin in immunity was identified in the HRM model as altered splenic T lymphocyte proliferation and down-regulation of numerous immunity-related genes including IRF8, which is required for development and maturation of myeloid cells, and NFIL3, which controls regulatory T-cell function and stability, were observed [161–164]. Reeler-, Dab1- and Apoer- mutants were also shown to lack microglial immunoglobulin G Fc receptors in the cerebellum, suggesting healthy microglia might require Reelin signaling [165]. In the periphery, hepatic stellate cells, which respond to and release various inflammatory molecules, are thought to the be major source of peripheral Reelin [166]. A variety of studies show dysregulation of Reelin signaling in inflammatory conditions. Evidence from inflammatory disorders of elevated peripheral Reelin expression suggest elevated Reelin levels increase NF-
In the dextran sulphate sodium (DSS) model of ulcerative colitis, colonic Reelin expression was upregulated in wild-type mice due to decreased methylation of the Reelin promoter following DSS exposure, and Reeler mice exposed to DSS presented with elevated disease index scores and higher mortality rates [168]. Elevated peripheral Reelin levels have also been shown in COVID-19 and experimental autoimmune encephalitis (EAE) [23, 24]. Reelin upregulation correlated with elevation of pro-inflammatory cytokines in humans with mild and severe COVID-19 symptoms, whereas asymptomatic COVID-19 infection was associated with normal Reelin expression [23]. Experimentally, treatment with CR50, an antibody that reduces peripheral Reelin levels without affecting Reelin in the CNS, in transgenic mice expressing human angiotensin-converting enzyme 2, protected against COVID infection [23]. Similarly, depleting peripheral Reelin levels in EAE with the CR50 antibody normalized monocyte infiltration into the CNS through reducing cell adhesion molecule expression and prevented development of EAE symptoms [24]. In contrast to these disorders, where inflammation is linked to elevated Reelin signaling, systemic inflammation through exposure to LPS or maternal administration of Poly(I:C) has been shown to reduce CNS Reelin levels and induce learning and memory impairment, suggesting peripheral inflammation can, over time, lead to a depletion of Reelin in the CNS [21, 170]. As described above, chronic stress also depletes Reelin in the brain, and we found atrophied spleen white pulp induced by chronic stress could be reversed by a peripheral Reelin injection at the acute dosage associated with antidepressant-like effects [131, 171]. In this model, promoting Reelin homeostasis through a single peripheral injection of Reelin recovered both stress-induced atrophy of lymphatic germinal zones as well as hippocampal Reelin, suggesting returning Reelin to homeostatic levels promotes healthy inflammatory responses in disorders of low Reelin expression [131, 171]. As Reelin is a pleiotropic molecule, evidence suggests the anti-inflammatory mechanisms responsible for promoting Reelin homeostasis in models of low Reelin expression through Reelin supplementation are very different from the anti-inflammatory mechanisms of Reelin depletion in inflammatory disorders of elevated Reelin levels. Evidence suggests restoring Reelin homeostasis, either through supplementing Reelin in disorders of low Reelin levels or depleting Reelin in disorders of high Reelin levels, both normalize inflammatory changes.
MICROGLIA AND REELIN DYSREGULATION HYPOTHESIS OF AD
As described above, there is an intricate relationship between immune dysregulation, inflammation and Reelin homeostasis in the context of AD. As mentioned above, microglia are involved in a variety of functions in both healthy and disease states and are implicated in a variety of pathological features associated with AD [41, 172]. Microglia are key regulators of inflammation and BBB integrity that produce various MMPs, particularly at sites of inflammation, although understudied in the context of Reelin dysfunction [45, 46]. MMPs increase BBB permeability through regulating cleavage of extracellular matrix proteins, including Reelin, which increases infiltration of peripheral immune cells and inflammatory molecules into the CNS [47]. Treatments such as resveratrol, an antioxidant commonly associated with red wine, and microRNA miR126a-5p reduce production of microglial MMP-9 and protect against inflammation and inflammation-induced BBB permeability [173, 174]. As the tight regulation of extracellular matrix proteins is disrupted and the permeability of the BBB is increased via increased MMP activity, peripheral inflammatory signaling molecules can elevate neuroinflammation by promoting inflammatory secretions from microglia. Additionally, as mentioned above, various molecules sensitive to inflammation cleave both Reelin and AβPP. This suggests that over time, initially protective anti-inflammatory mechanisms become destructive in the context of AD [148, 149]. Additionally, CXCL12/CXCR4 signaling regulates microglia reactivity, and plasma CXCL-12 is depleted in both early AD and the Tg2576 mouse model of AD [175, 176]. In cultured SH-SY5Y neurons, exposure to CXCL-12 dose-dependently increased Reelin secretion and depleted intracellular Reelin, which was blocked by a CXCR4 antagonist [177]. Disruption of CXCL12/CXCR4 signaling in AD could also alter Reelin secretion and intracellular Reelin expression in the absence of enhancement of pro-inflammatory microglia secretions.
The Reelin receptor VLDLR is expressed on microglia in brain tissue derived from individuals diagnosed with AD and in healthy controls, and VLDLR is upregulated in microglia at sites of plaques in AD [178]. Although direct interactions between Reelin and microglia have not been reported, to the authors’ knowledge, dysregulation of Reelin signaling could impact microglia motility and/or kinase activity through VLDLR. VLDLR is integral for regulating the radial distribution and proliferation of oligodendrocyte precursor cells and may have similar functions for microglia [77]. Microglia may selectively migrate to vulnerable neurons presenting increased Reelin secretion, ApoER2 upregulation, and containing initial tau aggregates. Microglial phagocytosis of selectively vulnerable neurons and their surrounding environments likely contributes to enriched Reelin deposits around neuritic plaques, as microglia engage in partial exocytosis of digested contents when exhausted and/or senescent [55].
In AD, inflammation and ongoing oxidative stress likely build into positive feedback loops to cause lipid peroxidation, a process by which reactive oxygen species react with lipids [55]. Enriched lipid peroxidation permanently cross-links ApoE into ApoER2 in the medial temporal lobe of individuals diagnosed with AD or mild cognitive impairment [179]. Although this requires empirical confirmation, if Reelin is unable to bind to cross-linked ApoE-ApoER2, lipid peroxidation of selectively vulnerable neurons could lead to compensation of reduced Reelin signaling through increasing secretion of Reelin and ApoER2 upregulation. These mechanisms account for enriched ApoER2 levels, inhomogeneous Reelin distribution via enriched Reelin deposits found in dystrophic neurites, most likely around vulnerable neurons involved in developing tau pathology, and Reelin homeostasis conferring protection against tau pathology and cognitive decline associated with AD. Increased Reelin production and cleavage in selectively vulnerable neurons may initially protect against inflammation but also inadvertently sustain progression of AD.
This hypothesis predicts disorders of both inflammation (both peripheral inflammation and neuroinflammation) and Reelin dysregulation (including disorders associated with upregulated Reelin expression and disorders of Reelin downregulation) would be associated with elevated risk of developing AD. Some research indicates inflammatory diseases altogether are not associated with increased AD risk, suggesting an additional factor is involved in increased risk of AD in specific inflammatory diseases [180]. Disruption of Reelin homeostasis could be an important secondary factor as a variety of disorders of both Reelin dysregulation and inflammation are associated with elevated AD risk including depression, epilepsy, autism, and multiple sclerosis [81, 181–191]. Menopause, a midlife period of neuroendocrine alterations manifesting in reproductive senescence, is also associated with both significant AD risk and inflammatory alterations [192–195]. Reelin expression is coregulated with estrogen in ovarian follicles, with highest levels of Reelin co-occurring with the highest expression of estradiol, suggesting loss of estrus cycles could alter Reelin production, impacting the elevated risk for development of AD [196]. Although in healthy aging plasma Reelin levels are not affected by sex or age on the group level, longitudinal data and association with future disease risk should be evaluated to address whether individuals with transient dysregulation of Reelin during menopause are at greater risk of developing AD [24].
Temporal lobe epilepsy, which is associated with decreased RELN RNA, has also been linked to both heightened inflammation and elevated AD risk [116, 197–199]. Pathogenic variants of the Reelin gene are seen in roughly 1/5th of families with autosomal dominant lateral temporal epilepsy, and depletion of Reelin has been shown experimentally in rodents following long-term kindling, which induces seizures and an epilepsy-like state through repeated electrical stimulation of epileptogenic brain regions [185, 200]. Autism spectrum disorder (ASD) is marked by both inflammation and RELN methylation, is frequently associated with RELN polymorphisms and is a condition associated with elevated risk of developing AD [181, 201]. Relapsing remitting multiple sclerosis (MS) is another inflammatory disorder associated with downregulated RELN expression and a recent epidemiological study showed a 2-fold increased risk of AD in MS [117, 202].
As mentioned above, COVID-19 has been linked to Reelin dysregulation and COVID-19 infection is associated with worsened AD pathology and elevated risk of developing AD, although further epidemiological evidence is still required to validate this association [23, 203–206]. The role of Reelin dysregulation in COVID-19 and AD risk also requires further evaluation. Reelin dysregulation was also described above in the context of experimental colitis, and some experimental and epidemiological evidence shows colitis is linked to inflammation and AD risk, although connections between colitis and elevated AD risk are not consistent across epidemiological studies and may be modifiable with treatment [207–209]. Although Reelin dysregulation in these disorders is not clearly linked to altered risk of AD, the numerous conditions linked to both inflammation and Reelin dysregulation associated with increased risk for developing AD warrants investigation into the potential causal role(s) of Reelin dysregulation in the development of AD, particularly in the context of these disorders and other disorders where both inflammation and Reelin dysregulation are observed. Given the numerous lines of evidence showing dysregulation of Reelin cleavage in AD and the direct connection between Reelin signaling in the hallmark pathological features of AD, evaluation of Reelin therapeutics in the context of these disorders is warranted to evaluate whether treatments that restore Reelin homeostasis influence risk of developingAD.
CONCLUSIONS
As we learn of the causal factors responsible for AD pathology, multiple lines of evidence suggest dysregulation of Reelin homeostasis and inflammation are key to development of the pathological features of AD. Recent evidence suggests inflammation, which has widely been recognized for contributing to AD, disrupts Reelin homeostasis. Improving our understanding of how Reelin homeostasis is affected by inflammation, and whether Reelin therapeutics that restore Reelin homeostasis can regulate inflammation and restore microglia function, could lead to the development of novel treatments for AD. Further research is warranted into the mechanisms responsible for altered Reelin signaling in AD and the evaluation of Reelin-based therapeutics should continue in the context of preclinical models of AD.
AUTHOR CONTRIBUTIONS
Brady S. Reive (Conceptualization; Writing – original draft; Writing – review & editing); Victor Lau (Visualization; Writing – review & editing); Carla Sánchez-Lafuente (Visualization; Writing – review & editing); Alex Henri-Bhargava (Writing – review & editing); Lisa E. Kalynchuk (Funding acquisition; Supervision; Writing – review & editing); Marie-Eve Tremblay (Conceptualization; Funding acquisition; Supervision; Writing – review & editing); Hector J. Caruncho (Conceptualization; Funding acquisition; Supervision; Writing – review & editing).
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgements to report.
FUNDING
We would like to thank the financial support from NSERC, CIHR, CGS-M and Dr. Tremblay and Dr. Caruncho’s Canada Research Chairs which helps to make this research possible.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
