Abstract
The neurovascular unit (NVU) is involved in the pathological changes in Alzheimer’s disease (AD). The NVU is a structural and functional complex that maintains microenvironmental homeostasis and metabolic balance in the central nervous system. As one of the most important components of the NVU, microglia not only induce blood-brain barrier breakdown by promoting neuroinflammation, the infiltration of peripheral white blood cells and oxidative stress but also mediate neurovascular uncoupling by inducing mitochondrial dysfunction in neurons, abnormal contraction of cerebral vessels, and pericyte loss in AD. In addition, microglia-mediated dysfunction of cellular components in the NVU, such as astrocytes and pericytes, can destroy the integrity of the NVU and lead to NVU impairment. Therefore, we review the mechanisms of microglia-mediated NVU dysfunction in AD. Furthermore, existing therapeutic advancements aimed at restoring the function of microglia and the NVU in AD are discussed. Finally, we predict the role of pericytes in microglia-mediated NVU dysfunction in AD is the hotspot in the future.
Keywords
INTRODUCTION
Alzheimer’s disease (AD) is a progressive neurodegenerative disease characterized by decreased cognitive function and is the most common type of dementia. The classic pathological changes in AD include amyloid-β peptide (Aβ) accumulation in the brain parenchyma and around brain vessels and neurofibrillary tangles formed by hyperphosphorylated microtubule-related protein tau in neurons [1, 2]. The number of people suffering from AD is more than 50 million globally, which makes AD the fifth leading cause of death in the world [2]. With the trend in global ageing, the prevalence of AD will greatly increase in the coming years, and economic and social burdens will also increase heavily [3]. Therefore, it is urgent to reveal new pathological mechanisms and discover therapeutic targets for AD.
The neurovascular unit (NVU) is a closely connected structural and functional complex composed of microglia, astrocytes, oligodendrocytes, neurons, interneurons, vascular smooth cells (VSMCs), pericytes, brain microvascular endothelial cells (BMECs), basement membrane and extracellular matrix. In fact, the NVU is responsible for regulating microenvironmental homeostasis and metabolic balance [4]. BMECs, VSMCs, pericytes, and perivascular astrocytic end-feet make up the blood-brain barrier (BBB), which selectively controls the exchange of substances between the central nervous system (CNS) and peripheral blood. Moreover, the NVU maintains the balance of metabolism in the CNS by providing sufficient blood fluids and nutrients. The NVU regulates cerebral blood fluid (CBF) according to neuronal activity depending on neurovascular coupling [5].
NVU dysfunction has been found in the early stage of AD [4]. It includes BBB destruction, neurovascular uncoupling, and abnormal communication among NVU cells [5]. In fact, we are mainly focusing on the role of NVU dysfunction in hippocampus in AD. BBB breakdown begins and aggravates in the hippocampus and its CA1 and dentate gyrus regions in normal aging and in mild cognitive impairment (MCI) individuals [6]. Impaired neurovascular coupling has been found in the hippocampus, parahippocampal gyrus, precuneus, and posterior cingulate cortex in the MCI patients and in the hippocampus in AD patients [7]. Moreover, NVU dysfunction significantly accelerating the progression of AD. First, BBB breakdown exacerbates Aβ accumulation [8] and which destroys the microenvironmental homeostasis of the CNS [9]. Second, neurovascular uncoupling, which is characterized by a decreased response of blood vessels to neuronal activity, decreases CBF [5], increases BBB permeability [10], exacerbates Aβ deposition [11] and tau hyperphosphorylation [12], and induces neuroinflammation, oxidative stress [13], and neuronal damage [5].
As one of the most important constituents of the NVU, microglia are the main resident immune cells in the CNS, and excessive microglial activation is one of the most important changes in the early stage of AD [14]. Though early recruitment of microglia may be neuroprotective by Aβ clearance, microglial engulfment, and degradation of Aβ is suppressed and proinflammatory cytokines are increased as disease progresses [15]. In fact, Aβ activated M1 microglia depending on P2X7 receptor (P2X7R) and the poly (ADP-ribose) polymerase-1 (PARP-1) pathways while spleen tyrosine kinase pathway is critical in neuroprotective microglia in AD [16–18]. And Aβ activates NLRP3 inflammasome and induce lysosomal damage in microglia in the process of engulfment [19]. Aβ and tau induce mitochondrial dysfunction in AD brain, while mitophagy impairment in microglia upregulates neuroinflammation and suppresses phagocytosis of Aβ and tau [20, 21]. Moreover, microglia mediate NVU dysfunction in many diseases, including AD. Excessive activation of microglia destroys BBB integrity and the regulation of CBF and contributes to neurodegeneration [16, 23]. However, the exact mechanism is still unclear.
In this review, we examined microglia to clarify the mechanism of NVU dysfunction in AD, including BBB destruction, neurovascular uncoupling, and impairment in constituent cells. Furthermore, we summarized the existing AD therapies targeting microglia and NVU dysfunction to inspire researchers to go in new directions to identify medications for microglia-mediated NVU impairment.
MICROGLIA MEDIATE NVU DYSFUNCTION IN AD
Microglia mediate NVU dysfunction via BBB impairment
The influence of microglia on the BBB is largely dependent on the activation phenotypes of microglia. Aβ-activated M1 microglia have increased secretion of inflammatory factors and chemokines, including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6), interleukin-12 (IL-12), CCL-2, CXCL-10, reactive oxygen species (ROS), and nitric oxide (NO), which exacerbate damage to the BBB [24–27]. In contrast, M2 microglia are characterized by alternative activation and acquired inactivation and play an important role in the protection and repair of the BBB by suppressing neuroinflammation in the CNS [25].
Microglia affect tight junctions (TJs) and adhesion junctions (AJs) between BMECs to modulate the permeability of the BBB. Under physiological conditions, resting microglia maintain the integrity of the BBB. These cells improve the expression of TJ factors such as zonula occludens 1 (ZO-1) and occludin on BMECs
It has reported age-dependent BBB breakdown in hippocampus which is significantly aggravated in MCI patients [6]. M1 microglial activation is also found in the early stage of AD, and evidence indicates it is involved in BBB dysfunction in the early stage of AD [29]. First, proinflammatory factors derived from M1 microglia, including IL-1β, TNF-α, and CCL2, affect BMECs and directly destroy the BBB in AD. Aβ-activated microglia secrete IL-1β by upregulating the expression of P2X7 receptor (P2X7R) [16]. IL-1β increases the expression of IL-6, interleukin-8 (IL-8), and TNF-α and decreases the expression of ZO-1, enhancing BBB paracellular permeability. A study also showed that IL-1β upregulated the expression of intracellular adhesion molecule-1 and vascular cell adhesion molecule-1 to increase the transmigration of peripheral white blood cells (WBCs) into the CNS [30]. Aβ-activated M1 microglia release NO and TNF-α via PARP-1 to downregulate the expression of TJ proteins, including ZO-1 and occludin, increasing BBB paracellular permeability
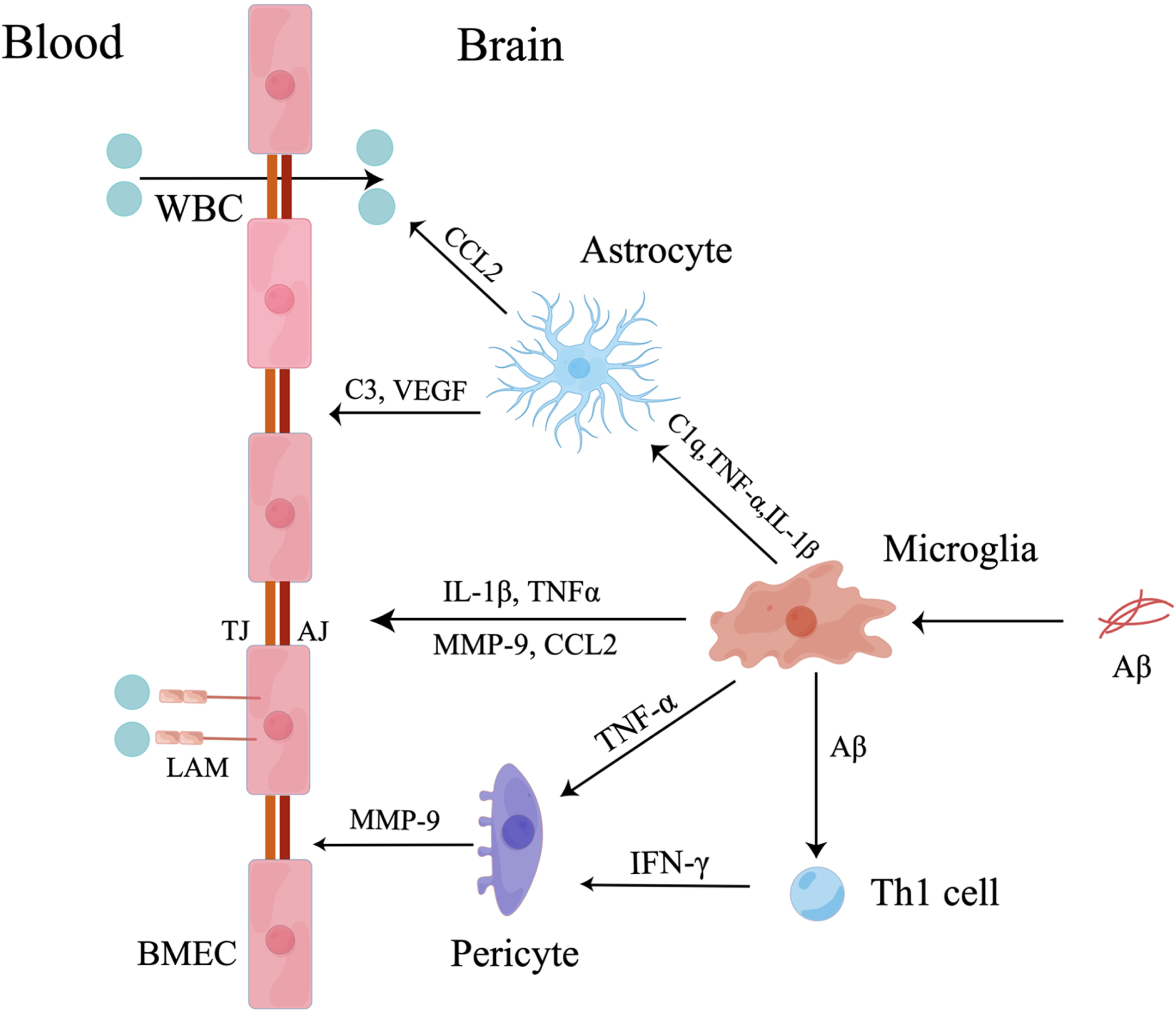
Microglia mediate BBB dysfunction. Firstly, microglia induced by Aβ secrete IL-1β, TNF-α, MMP-9, and CCL2 to destroy TJs and AJs between BMECs and increase the expression of leukocyte adhesion molecules (LAMs) on BMECs to attract peripheral WBCs. Secondly, activated microglia secrete C1q, TNF-α and IL-1β to activate astrocytes. The activated astrocytes promote transmigration of peripheral WBCs across BBB by secreting CCL2 and induce BBB breakdown by secreting C3 and vascular endothelial growth factors (VEGF). Thirdly, activated microglia mediate pericyte dysfunction by secreting TNF-α and presenting Aβ to Th1 cells. Then, activated pericytes increase the release of MMP-9 to destroy TJs leading to BBB impairment.
Increased infiltration of peripheral WBCs in the CNS exacerbates neuroinflammation and BBB dysfunction. Aβ-activated microglia and cerebral macrophages secrete chemokines and cytokines to induce peripheral monocytes to accumulate in the CNS and transform into resident macrophages to exacerbate BBB and NVU dysfunction [42]. Microglial inflammatory factors mediate peripheral WBC migration in complex ways. Proinflammatory factors (IL-1β, TNF-α, IL-6, and IL-12) and chemokines (monocyte chemoattractant protein-1, macrophage inflammatory protein-1α, macrophage inflammatory protein-1β, and IL-8) secreted by Aβ-activated M1 microglia increase the permeability of the BBB and the infiltration of monocytes into the CNS
Microglia mediate NVU dysfunction via neurovascular uncoupling
Neurovascular uncoupling is one of the most important reasons for CBF reductions in AD [48, 49]. Reduced CBF is a sign of the early stage of AD, which leads to neuronal death and neurodegeneration through metabolic dysfunction in neurons and glial cells [48, 49]. Microglia-mediated neuroinflammation has been observed in the early stages of AD [29, 50], and studies have shown a close correlation between microglia and neurovascular uncoupling in AD [51]. Multiple lines of evidence have shown that microglia modulate neurovascular coupling via the P2Y12 receptor (P2Y12R) under physiological conditions. The expression of P2Y12R on microglia is significantly decreased in AD, impairing cellular communication and the response of cerebral vessels to neuronal activity [22, 52].
Damage to neuronal mitochondria has been proven to contribute to metabolic dysfunction in neurons and neurovascular uncoupling [53]. In fact, gp91phox, a catalytic subunit of NADPH oxidase (NOX), is significantly upregulated in microglia and neurons in MCI patients [54]. A study showed that ROS derived from NOX containing gp91phox were essential for Aβ-induced neurovascular uncoupling [55]. Therefore, the interaction between microglia and neurons plays a crucial role in neurovascular uncoupling in AD. It was reported that Aβ-activated microglia promote the release of IL-1β, TNF-α, and NO and the activation of NOX by upregulating P2X7R and PARP-1 pathways in an AD mouse model [16, 17]. Increases in H2O2 and NO and proinflammatory factors led to structural and functional damage to neuronal mitochondria, contributing to increased ROS. TNF-α induced neuronal mitochondrial dysfunction via neuron-derived extracellular vesicles (EVs), leading to increased oxygen consumption and gradual ROS release from neurons [56]. As a result, increased ROS activate NOD-like receptor protein 3 (NLRP3) inflammasomes to exacerbate oxidative stress, neuroinflammation, insulin resistance and metabolic abnormalities [48, 53], leading to neurovascular uncoupling in AD.
Recent studies have shown that the primary mechanism of neurovascular uncoupling in AD is cerebrovascular component dysfunction [57]. First, activated microglia promote VSMC and pericyte dysfunction, leading to neurovascular uncoupling. On the one hand, microglia can directly mediate pericyte degeneration. Activated microglia can secrete proinflammatory factors to induce pericyte loss [26, 36–38]. LPS-activated microglia directly increased the release of ROS from pericytes by secreting TNF-α and IL-1β, leading to death in rat retinal microglia and pericytes [58]. Then, pericyte degeneration reduces the capillary response to neuronal activity, resulting in neurovascular uncoupling [59]. We think a similar mechanism by which activated microglia induce pericyte loss leading to neurovascular uncoupling is present in AD. On the other hand, activated microglia indirectly induce cerebral vessels to contract by exacerbating Aβ accumulation. Aβ-activated microglia upregulate cyclooxygenase 2 to increase the expression of IL-1β. Then, IL-1β upregulates β-site APP-cleaving enzyme (BACE-1) in neuron-like cells (SH-SY5Y cells) to exacerbate Aβ accumulation [60]. Increased Aβ deposition mediates the hypercontractive phenotype of VSMCs in small arteries in the CNS and decreases CBF via high levels of serum response factor (SRF)/myocardin (MYOCD) [61, 62]. In human and mouse models of AD, Aβ also induced pericytes around brain capillaries to contract through NADPH-oxidase-4 (NOX4)-derived ROS [63]. However, some researchers believe that VSMCs can directly contract cerebral vessels, while pericytes cannot. In contrast, pericytes influence CBF only by indirectly regulating cellular interactions [64]. Finally, the end-feet of astrocytes on cerebral vessels are very important for communication between blood vessels and neurons. The expression of aquaporin-4 (AQP-4) in astrocytic end-feet on the vessels is significantly reduced in AD and is accompanied by a prominent increase in microglial activation and proliferation [65]. Excessive levels of activated microglia may be involved in astrocyte-mediated neurovascular uncoupling, but the exact mechanism is still unknown. Overall, microglia can mediate neurovascular uncoupling in AD by downregulating the expression of P2Y12R, increasing ROS and proinflammatory factors to induce neuronal mitochondrial impairment, exacerbating Aβ accumulation to attenuate the response of cerebral vessels to neuronal activity and inducing dysfunction in pericytes and astrocytes (Fig. 2).
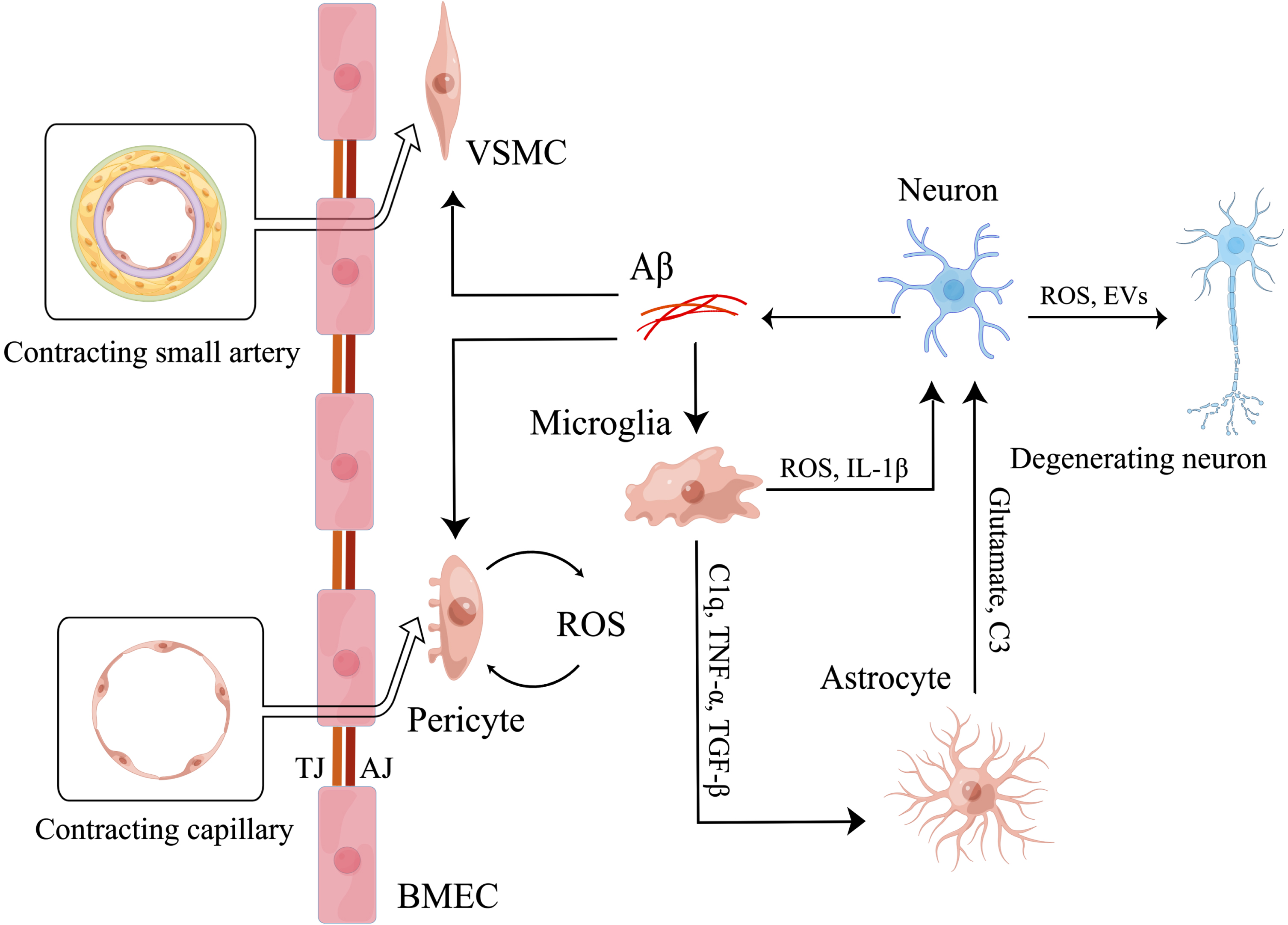
Microglia mediate neurovascular uncoupling. Firstly, microglia induced by Aβ secrete ROS and IL-1β to induce neuronal mitochondrial dysfunction directly. Then, activated microglia also secrete C1q, TNF-α, TGF-β to activate astrocytes. And activated astrocytes increase the release of glutamate and C3 to induce neuronal mitochondrial dysfunction. As a result, neuronal mitochondrial dysfunction induces degeneration of more surrounding neurons by releasing ROS and EVs, finally leading to neurovascular uncoupling. Meanwhile, microglia are involved in the regulation of neurovascular uncoupling by affecting the function of cerebral vessels. Aβ induces abnormal contraction of small arteries by promoting the conversion of VSMCs to hypercontractive phenotype. And Aβ also induces abnormal contraction of capillaries by increasing the release of ROS of pericytes. Eventually, microglia aggravate the effects of Aβ by secreting IL-1β to increase the release of Aβ of neurons.
Microglia mediate NVU dysfunction through impairment of the cellular components of the NVU
The NVU is a structural and functional complex [4]. Dysfunction of the cellular components of the NVU has been observed in AD, including changes in the distribution of capillaries, pericyte diminishment, astrocyte activation and proliferation, and the relocation of microphages [66]. Evidence support that microglia is involved in this process and potentially lead to NVU dysfunction.
Microglia mediate NVU dysfunction via astrocyte impairment
As an important constituent of the NVU, astrocytes are reactive cells in the CNS that can rapidly and simultaneously exchange information with several synapses. Astrocytes maintain the integrity of the BBB and regulate CBF with BMECs, pericytes and VSMCs [67]. Additionally, reactive astrocytes accelerate AD progression by significantly affecting Aβ and tau pathology via BACE-1 and IL-3 pathways [68, 69].
Microglia induce excitotoxicity in astrocytes and disturb the communication among astrocytes and surrounding cells to mediate astrocyte impairment, ultimately leading to NVU dysfunction in AD [70]. First, microglia play an important role in severe neuronal excitotoxicity induced by the release of calcium ion-dependent gliotransmitters such as glutamate from astrocytes [71]. TNF-α, which is mainly secreted by microglia, was shown to be essential for calcium-dependent release of glutamate by astrocytes [72, 73]. TNF-α mediates calcium ion-dependent glutamate release by activating prostaglandin E2 in astrocytes, which leads to neuronal apoptosis in an AD mouse model [74]. The expression of intermediate conductance calcium-activated potassium channels (KCa3.1 channels) on reactive astrocytes and activated microglia was enhanced in AD patients and SAMP8 mice [75, 76]. Some researchers found that microglia-derived TGF-β upregulated astrocyte KCa3.1 channels to induce astrogliosis and increased the level of calcium ions to mediate the release of glutamate during AD [71, 77]. Second, a high number of gap junctions is important for intercellular communication among astrocytes. In particular, Aβ-activated microglia suppress the expression of astrocyte gap junctions by secreting TNF-α and IL-1β, contributing to astrogliosis [78]. Third, activated microglia are involved in the regulation of astrocyte complement-dependent intercellular communication leading to NVU dysfunction. Specifically, activated microglia release C1q to activate the nuclear factor-kappa B (NF-κB) pathway in astrocytes, which release C3 [33, 79]. C3 has been reported to compromise neuronal synapses and cognitive function by acting on neuronal C3R and destroying the BBB by acting on BMECs [35, 80]. Finally, C3 attenuated microglial phagocytosis of Aβ [81].
Aβ-activated microglia induce NVU dysfunction by mediating astrocyte activation and neuroinflammation. Microglia are more sensitive to pathological changes than astrocytes [25], and the activation of astrocytes is highly dependent on microglia [82]. This finding indicates the initial role of microglia and the amplified role of astrocytes in neuroinflammation [83, 84]. In addition, microglia can mediate the phenotypic conversion of astrocytes. Similar to microglia, there are A1 and A2 astrocytes in AD. Aβ-activated M1 microglia secrete IL-1β, TNF-α, C1q, and NO to induce A1 astrocytes [27, 33]. A1 astrocytes have increased release of proinflammatory factors, chemokines and complement to amplify neuroinflammation, leading to BBB destruction and neuronal injury [33–35, 85]. Moreover, IL-1β indirectly enhances the permeability of the BBB
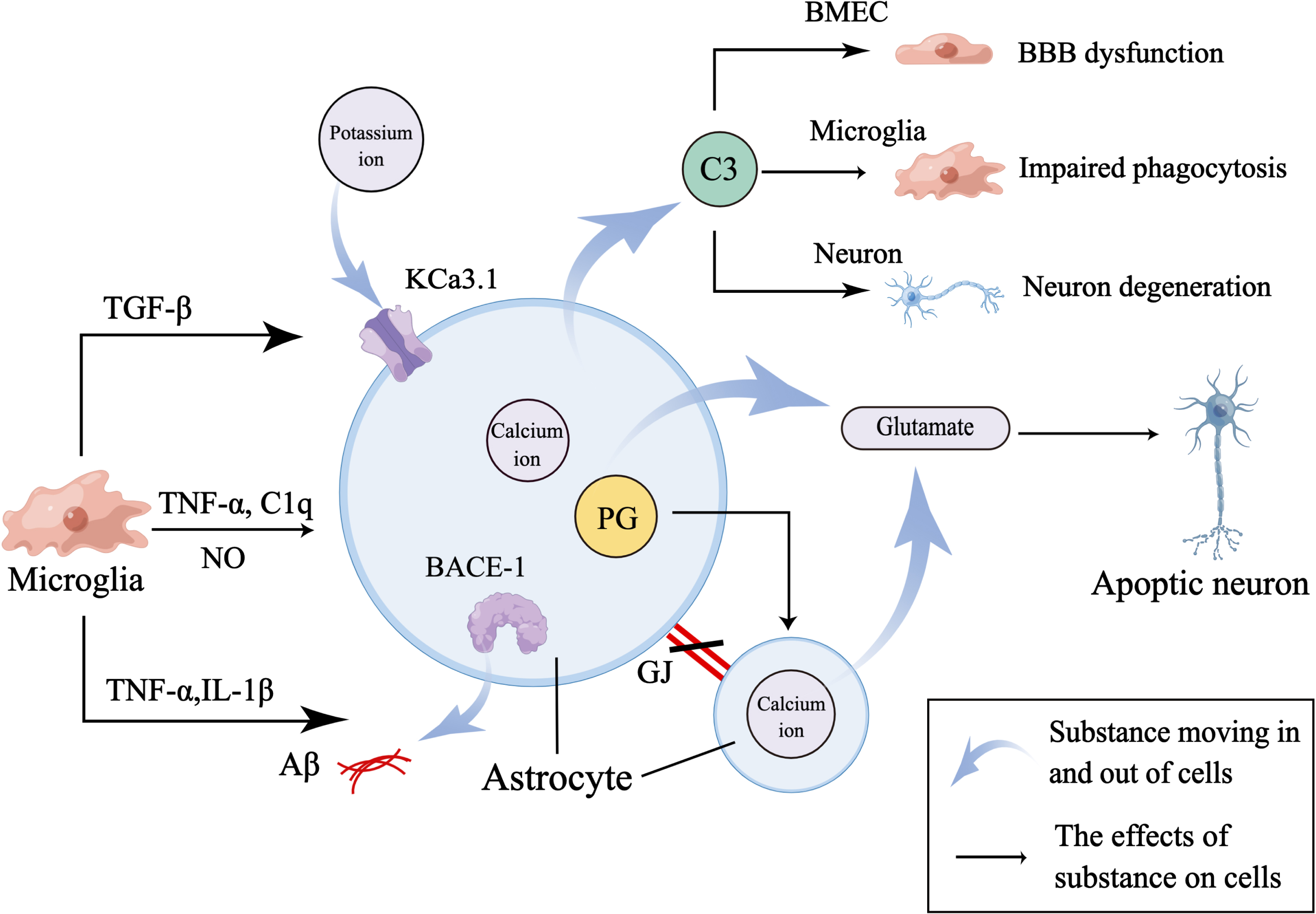
Microglia mediate astrocyte dysfunction. Firstly, activated microglia secrete TNF-α, C1q, and NO to activate astrocytes. And activated astrocytes increase the release of C3 to mediate NVU dysfunction by regulating the function of BMEC, microglia, and neuron. Secondly, microglia secrete TNF-α and IL-1β to increase the expression of BACE-1 in astrocyte to aggravate Aβ accumulation and destroy GJs between astrocytes to affect normal cellular communication between astrocytes. And TNF-α also increases the expression of PG in astrocyte to evoke calcium ion-mediated release of glutamate which induces the apoptosis of neuron. Thirdly, microglia-derived TGF-β increases the expression of KCa3.1 channel which works together with elevated calcium ion in astrocytes to promote release of glutamate.
Microglia mediate NVU dysfunction via pericyte impairment
Brain pericytes, which are important cellular components of cerebral capillaries, have features similar to those of VSMCs and form the BBB with BMECs, astrocytic end-feet and the basement membrane [88]. The coverage ratio of pericytes was proven to be associated with the permeability of the BBB [39]. Evidence has demonstrated that microglia and neuroinflammation can mediate pericyte impairment and loss in various ways, leading to NVU dysfunction in AD. First, Aβ-activated microglia induce BBB destruction via pericyte-derived MMP-9. Aβ-activated microglia secrete TNF-α in AD [26], and TNF-α promotes pericytes to release MMP-9, which leads to BBB breakdown by destroying TJs and extracellular matrix between BMECs and promoting the migration of pericytes surrounding vessels to other regions in the brain, leading to pericyte loss
Overall, it has been proven that Aβ-activated microglia induce pericyte dysfunction and loss by releasing TNF-α, IFN-γ, and TGF-β. Pericyte dysfunction and loss lead to BBB breakdown and neurovascular uncoupling in several ways. However, evidence is still needed to determine whether microglia directly mediate NVU dysfunction by regulating pericyte function in AD.
Microglia mediate NVU dysfunction via injury to neurons and interneurons
A decrease in neurons is one of the important factors in the progressively declining cognitive and memory function of AD patients, which is mediated by microglia through phagocytosis and the secretion of inflammatory factors, EVs and ROS. First, abnormal microglial phagocytosis induces neuronal damage in AD. It was reported that enhanced microglial phagocytosis damaged neuronal synapses in the early stage of AD in TREM2-variant mice, but phagocytosis was neuroprotective in the late stage of AD by engulfing Aβ [94]. Aβ-induced microglial phagocytosis of synapses was proven to be dependent on the complement pathway [79, 95]. M1 microglia secrete inflammatory factors and C1q to increase the release of C3 by A1 astrocytes, leading to the rapid death of neurons [33, 79]. Increased C3 enhances microglial phagocytosis of synapses by binding to C3R on microglia [95]. Second, microglia-mediated neuroinflammation and oxidative stress regulate neuronal function. Aβ-activated microglia have increased release of TNF-α, glutamate, and ROS, leading to excessive neuronal activation and death [16, 96–98]. In addition, Aβ-activated microglia damage neurons via Toll-like receptor-2/3 (TLR-2/3) by releasing IL-6, TNF-α, and ROS [99, 100]. Third, ATP-activated microglia produce EVs that convert aggregated Aβ to soluble Aβ, which is more neurotoxic [101]. Finally, activated microglia affect the function of interneurons and neural networks. In 5XFAD and 3x-TgAD mice, activated microglia were closely associated with damage to interneurons in AD [102]. In addition, damage to interneurons leads to dysfunction of neural networks and cognition by inducing imbalance between suppression and excitation in the CNS [103].
Microglia mediate NVU dysfunction via impairment of other cellular components in the NVU
First, microglia mediate dysfunction in BMECs and the BBB in AD by destroying TJs and AJs and increasing the entry of peripheral WBCs into the CNS. Second, microglia exacerbate Aβ accumulation [93] to affect the communication between BMECs and astrocytic end-feet [41], which contributes to neurovascular uncoupling in AD and alters calcium signaling in BMECs [104]. Third, microglia mediate the hypercontractive phenotype of VSMCs by exacerbating Aβ accumulation, leading to neurovascular uncoupling in AD. Finally, oligodendrocytes, which are the myelin-forming cells in the CNS, play a significant role in the conduction of action potentials and neuronal protection [105]. In 5XFAD mice, TREM2-variant microglia were associated with the phagocytosis of myelin and lipid metabolism in the CNS, and reactive oligodendrocytes were always accompanied by damage to axonal myelin and metabolic imbalance [106–108]. This finding indicates the potential correlation between microglia and oligodendrocytes in AD, although there is no direct evidence to support it.
POTENTIAL THERAPIES FOR RESTORING THE FUNCTION OF MICROGLIA AND THE NVU IN AD
Both AD pathologies and NVU dysfunction are present much earlier than clinical manifestations therefore the principle of medication treatment timing is as early as possible. After innumerable failure of studies of anti-Aβ, aducanumab was approved as the first disease-modifying drug of AD by FAD in 2021. However, there is still much controversy concerning the clinical benefits of aducanumab in AD [122, 123]. Because of various risk factors and complex mechanisms of AD, we believe different therapeutic approaches would be helpful to AD with different causes [124, 125]. The important roles of microglia and NVU in AD, and quickly advanced technologies make them valuable choices [4, 127]. The potential therapeutics aiming to alleviate AD progression and improve cognitive function by restoring microglial and NVU function have been described in Table 1.
Potential therapies for regulating microglia and NVU function
TREM2McAb, triggering receptor expressed in myeloid cells monoclonal antibody; A
First, inhibiting neuroinflammation is most likely helpful in improving BBB function to restore NVU function in AD. Annexin A1 (ANXA1) not only inhibits neuroinflammation by increasing IL-10 and decreasing TNF-α [128] but also alleviates Aβ-induced BBB breakdown by suppressing the RhoA-Rock pathway [129]. Second, multiple-target drugs, including dabigatran, L-norvaline and quercetin, regulate the function of several types of cells in the NVU to reduce BBB permeability in AD mouse models [116, 121]. Third, a clinical trial demonstrated that resveratrol reduced the concentration of MMP-9 in CSF to preserve BBB integrity and improved the cognitive function of AD patients [130]. Finally, rapamycin (mTOR inhibitor) was proven to upregulate TJs and inhibit the activity of MMP-9 to maintain BBB integrity in an AD mouse model [131].
Medications have also been designed to restore neurovascular coupling and increase CBF in AD. First, NAD+ intermediates have been shown to restore NAD+ concentrations to alleviate ageing-associated neurovascular uncoupling. Nicotinamide mononucleotide (NMN) rescued cerebral vessel responses to neuronal activity and cognitive function in a mouse model of aging by increasing endothelial NO-derived relaxation in cerebral microvessels and alleviating mitochondrial dysfunction [132]. NMN has also been reported to be beneficial for the cognitive function in AD mice by attenuating Aβ accumulation, synaptic loss, and neuroinflammation [133]. Second, the PARP-1 inhibitor PJ-34 was shown to improve neurovascular coupling and cognitive function by regulating endothelial-mediated vasodilation in an aging mouse model [134]. Third, twendee X has been proven to alleviate tau and α-synuclein pathology and neurovascular uncoupling in APP23+ CCH mice [135]. Furthermore, sildenafil increased CBF and improved NVU function in AD patients in clinical trials [114, 136].
Instead of restoring BBB function and neurovascular coupling alone, medications have also been designed to simultaneously regulate the function of multiple cells in the NVU to improve NVU function in AD. First, anserine can improve NVU function in AD. It was reported that anserine and carnosine improved brain perfusion and verbal episodic memory in elderly people [137, 138], although the effect of anserine on AD patients is still unclear. A recent study demonstrated the role of anserine in maintaining NVU integrity by suppressing astrocyte-mediated neuroinflammation and improving the coverage ratio of pericytes in a mouse model of AD [113]. Second, statins, including atorvastatin and pitavastatin, restored NVU function by inhibiting the activity of MMP-9 in neurons and attenuating astrogliosis, leading to improved behavioral memory and a reduction in senile plaques in an AD mouse model [119]. Third, pinocembrin was reported to suppress neuroinflammation and protect the cholinergic system to restore the function of the NVU [120]. Fourth, adipoRon, an adiponectin receptor agonist, enhanced the effects of adiponectin, whose deficiency was proven to increase Aβ-induced microglial activation and neuroinflammation. AdipoRon treatment suppressed neuroinflammation, restored microglial phagocytosis of Aβ, rescued neuronal and synaptic function, and improved spatial memory function in AD mice [111, 139]. Finally, rapamycin plays a crucial role in NVU protection. Rapamycin increases CBF, decreases BBB permeability and maintains neuronal function
Due to the close correlation between microglia and astrocytes, many medications have been designed to regulate the function of microglia and astrocytes to improve NVU function and delay AD progression. First, suppressing the activation of microglia and astrocytes and neuroinflammation may be helpful in delaying AD progression. For example, methystictin activates the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway; Nrf2 is an anti-inflammatory transcription factor that significantly reduces microgliosis, astrogliosis, proinflammatory cytokines, and oxidative stress in APP/PS1 mice [112]. Low molecular weight glucocorticoid-induced leucine zipper (GILZ) analogues, which are the peptide analogues of the p65 binding domain of GILZ, suppress the NF-κB pathway. GILZ analogues decrease Aβ accumulation, reduces activated microglia and astrocytes, and inhibits inflammatory cytokines and TLR expression in 5xFAD mice [141]. TPPU, a specific small-molecule soluble epoxide hydrolase inhibitor, improves the concentration and anti-inflammatory activity of epoxy fatty acids. TPPU significantly reversed microglia and astrocyte activation and suppressed neuroinflammation in 5xFAD mice [142]. Second, pantethine not only suppressed glial neuroinflammation in mice but also suppressed the expression of complement genes and other inflammatory genes in astrocytes [110]. Third, boldine suppresses the activity of hemichannels between microglia and astrocytes without influencing the function of gap junctions to reduce calcium ion and ATP-induced glutamate release [143]. Recently, NLY01, a GLP-1R agonist, is reported specifically to inhibit Aβ-induced microglial activation and the conversion of reactive astrocytes as well as protecting neurons in AD mice, leading to the improvement of cognitive function [144].
Medications regulate microglial function to treat AD in various ways. First, medications suppress microglia-associated inflammatory pathways and cytokines to prevent the progression of AD. For example, dapansutrile and edaravone suppress microglia-mediated neuroinflammation by inhibiting the NLRP3 inflammasome in AD [145, 146]. Artemether and tiliroside suppress microglia-mediated neuroinflammation in AD by activating the anti-inflammatory Nrf2 pathway [147, 148]. Second, some medications alleviated the accumulation of Aβ and apoptotic cells to maintain NVU homeostasis by enhancing microglial phagocytic capacity. For instance, K161 improves microglial phagocytosis of Aβ and apoptotic neurons by suppressing the Pan-SHIP1/2 pathway [149]. Magnolol improves microglial phagocytosis and the degradation of Aβ and suppresses microglia-mediated neuroinflammation [150]. TREM2 monoclonal antibodies improve cognitive function by promoting acute microglial activation and microglial phagocytosis of Aβ and suppressing chronic microglia-mediated inflammation [109]. Third, microglial phenotypic transformation from M1 to M2 is also important in AD. For example, DHCR24, TPP-MoS2QDs, naringenin, and Dendrimer-tesaglitazar were reported to induce M1 microglia to convert to M2 microglia to suppress neuroinflammation and improve microglial phagocytosis [115, 151–153]. Eventually, there are many mutant genes in microglia in AD, and some have been proven to be associated with AD, including TREM2, CD33, PILRA, and CR [154]. Interestingly, the TREM2-activated antibody AL002c has been proven to be efficient and safe for delaying the AD in mouse models and phase 1 clinical trials [155]. The TREM2 monoclonal antibody promoted the acute activation of protective microglia and suppressed microglia-associated chronic neuroinflammation to attenuate AD pathology
CONCLUSIONS: CHALLENGES AND PERSPECTIVES
In this review, we have discussed the existing mechanism of microglia-mediated NVU dysfunction and the advancements in therapeutics for restoring the function of microglia and the NVU in AD. Microglia, which are critical in neuroinflammation in AD, mediate NVU dysfunction via various pathways. Specifically, Aβ-activated microglia not only induce BBB breakdown by mediating neuroinflammation, the infiltration of peripheral WBCs and oxidative stress but also impair neurovascular coupling by mediating direct damage to neuronal mitochondria, inducing abnormal contraction of cerebral vessels and pericyte loss in AD [16, 93]. Moreover, microglia-mediated impairment of cellular components of the NVU destroys the integrity of the NVU and leads to NVU dysfunction [4]. Specifically, microglia-mediated activation and dysfunction of astrocytes regulates neuroinflammation and the destruction of the NVU. Pericyte dysfunction and loss is mediated by microglia and affect the function of the NVU by destroying the BBB and neurovascular coupling. Furthermore, microglia induce neuronal impairment and loss via abnormal phagocytosis, neuroinflammation, oxidative stress, the complement pathway and EVs, subsequently leading to NVU dysfunction. As a result, microglia mediate NVU dysfunction by destroying homeostasis in the CNS and inducing an imbalance in metabolism and neurodegeneration in the early stage of AD. Additionally, evidence support the role of microglia-mediated NVU dysfunction in AD. For example, microglia modulate neurovascular coupling via P2Y12 pathway under physiological condition while this process may be disrupted as significantly decreased expression of P2Y12 in activated microglia has been found in brain of AD patients using multispectral immunofluorescence [22, 52]. Recently, NLY01, a GLP-1R agonist, is reported specifically to inhibit Aβ-induced microglial activation and the conversion of reactive astrocytes as well as protecting neurons in AD mice, leading to the improvement of cognitive function [144].
Pericytes play an indispensable role in BBB impairment and neurovascular uncoupling in AD, which are important characteristics of NVU dysfunction [59, 157]. It was proven that microglia can increase the permeability of the BBB by inducing pericyte dysfunction and loss. Furthermore, microglia are involved in neurovascular coupling and metabolic balance by inducing pericyte impairment and loss and exacerbating Aβ accumulation. However, we know little about the mechanism of this process, and it is still unclear whether pericytes can contract cerebral capillaries and impair neurovascular coupling in AD [64]. In addition, we find that some experiments on the crucial steps in the mechanism were not performed under AD conditions. For example, a study used mouse retinal-derived microglia and pericytes to show that microglia secreted proinflammatory factors to induce pericytes to release ROS and abnormally contract capillaries, leading to decreased CBF and neurovascular uncoupling [58]. However, we do not know if this process would be the same in AD. Therefore,
Footnotes
ACKNOWLEDGMENTS
We would like to thank our supporters: National Natural Science Foundation of China (81701078), Natural Science Foundation of Heilongjiang Province of China (Outstanding Youth Foundation, YQ2022H003), China postdoctoral science foundation (2016M600261, 2018T110317), Heilongjiang Postdoctoral Financial Assistance (LBH-Z15163), Innovation and Entrepreneurship Training Program for College Students in Harbin Medical University (201910226322), Postgraduate Scientific Research and Practice Innovation Project of Harbin Medical University (YJSCX2020-03HYD).
Figures were created by Figdraw (www.figdraw.com).
FUNDING
This work was supported in part by grants from National Natural Science Foundation of China (81701078), Natural Science Foundation of Heilongjiang Province of China (Outstanding Youth Foundation, YQ2022H003), China postdoctoral science foundation (2016M600261, 2018T110317), Heilongjiang Postdoctoral Financial Assistance (LBH-Z15163), Innovation and Entrepreneurship Training Program for College Students in Harbin Medical University (201910226322), Postgraduate Scientific Research and Practice Innovation Project of Harbin Medical University (YJSCX2020-03HYD).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
