Abstract
Background:
Cognitive and functional abilities in individuals with Alzheimer’s disease (AD) pathology (ADP) are highly variable. Factors contributing to this variability are not well understood. Previous research indicates that higher educational attainment (EA) correlates with reduced cognitive impairments among those with ADP. While cognitive and functional impairments are correlated, they are distinguishable in their manifestations.
Objective:
To investigate whether levels of education are associated with functional impairments among those with ADP.
Methods:
This research involved 410 African American (AA) individuals (Institutional Review Boards 20070307, 01/27/2023) to ascertain whether EA correlates with functional resilience and if this effect varies between
Results:
The results showed that EA correlated with functional difficulties in AA individuals with high levels of pTau181, such that individuals with high EA are more likely to have better functional ability compared to those with lower EA (
Conclusion:
This study extends the role of cognitive reserve and EA to functional performance showing that cognitive reserve influences the association between ADP burden and functional difficulties. Interestingly, this protective effect seems less pronounced in carriers of the strong genetic risk allele ɛ4.
INTRODUCTION
Alzheimer’s disease (AD) is the most common type of dementia. Older African ancestry individuals in US having almost two times higher prevalence and incidence compared with older non-Hispanic Whites (NHW), but substantially underrepresented in AD studies [1, 2]. Several factors influence the incidence, prevalence, and risk of AD. These include genetic, clinical, social, behavioral, and environmental determinants [3–8].
The characteristic neuropathological changes of AD, the presence of extracellular amyloid-β deposits and intracellular aggregation of neurofibrillary tangles, can now be measured using biomarkers [9, 10]. These include neuroimaging, cerebrospinal fluid (CSF), and blood-based biomarkers [9, 10]. Recent developments in the field of blood biomarkers showed that blood pTau181 levels strongly correlate with abnormal amyloid accumulation, tau deposition, and neurodegeneration [11–13]. The blood pTau181 levels have been demonstrated to differ significantly between diagnostic groups with amyloid, tau, and FDG-PET scans, further validating pTau181 as a robust biomarker for AD related pathology (ADP) [14, 15].
ADP is typically associated with the decline in cognitive, functional, and behavioral abilities seen in AD [10]. However, some studies have shown that even with advanced ADP, a subgroup of individuals can still maintain reasonable cognition and function, which reflects a better-than-expected performance in relation to the degree of ADP [16, 17]. This inconsistency between ADP and clinical symptoms underscores a possible phenotype characterized by biological pathology without clinical impairment.
‘Cognitive reserve’ (CR) may account for some of this protection from ADP [18, 19]. CR is thought to be the various thinking abilities that actively compensate for the deficits imposed by the ADP [20]. This compensation yields cognitive performance that exceeds expectations when considering life-course-related brain changes and brain injuries or disease [21].
Educational attainment (EA) is a commonly used correlate of CR. Higher EA tends to foster resilience against AD clinical manifestations, often resulting in improved cognitive performance compared to those with less education but similar levels of ADP [22–25]. Some studies also suggest an interplay between EA and the
MATERIALS AND METHODS
Study samples
We ascertained 410 AA participants through AD genetics studies from Wake Forest University (WF, North Carolina), and University of Miami (UM, Florida). These participants represented a diverse AA cohort across both Florida and North Carolina sites. All participants were classified as AA based on self-reported race-ethnicity. It is essential to acknowledge that race-ethnicity is dynamic, shaped by geographic, cultural, and sociopolitical forces and are social constructs. Therefore, while participants self-identified as AA, this classification reflects a complex interplay of social, cultural, and historical influences, rather than a distinct biological identity. The inclusion criteria for the study were having CDR scores, reported years of education,
Individual ancestral backgrounds were confirmed using genome-wide genetic data with EIGENSTRAT software [34]. Population substructure data sets were compared with those in the 1000 Genome reference panel YRI (Yoruba from Nigeria) and CEU (Utah Residents with Northern and Western European ancestry) populations. Outliers with respect to CEU population (overlapping within the cluster of CEU) were removed from the datasets [35].
Ascertainment protocols have been consistent across the sites and clinical data assessments capture sociodemographic information including years of education, medical and family history, dementia staging, AD/dementia symptoms, neuropsychological abilities, functional capabilities, and behavioral impairments. The CDR was used to evaluate functional capabilities and served as an outcome measure as described below in the Statistical Analysis section. Biomaterials were also collected at the time of study entry by trained phlebotomists. The detailed description of the study samples, recruitment and cognitive assessment are described elsewhere [27]. 253 participants were diagnosed as non-Cognitively Impaired (nCI), 12 as mild cognitively impaired (MCI), and 145 with Alzheimer disease (AD).
Genotyping
Genome-wide single-nucleotide polymorphism (SNP) genotyping was processed on a Global Screening Array and
Measurement of serum AD biomarkers pTau181
Serum concentrations of pTau181 were measured using SIMOA chemistry implemented on the Quanterix HD-X instrument (Quanterix, Billerica, MA, USA) [37] according to manufacturer’s instructions for the pTau181 Advantage V2 assay (catalog #103714). Samples were randomized according to age, sex, and diagnosis and assayed in duplicate on each plate. Means of two measures were used in analysis. Biomarker levels were log10-transformed to satisfy normality assumptions. Samples were removed if biomarker levels were greater than three standard deviations from the mean of the rest of the samples.
CDR-FUNC
The Clinical Dementia Rating scale (CDR) is a semi-structured interview that assesses cognitive and functional impairment associated with AD to allow for staging based on a global CDR score [38]. The global CDR score is derived from box scores assigned to six domains: Memory, Orientation, Judgment and Problem solving, Home and Hobbies, Community Involvement, and Personal Care. Various studies have examined the utility of these box scores for clinical and research purposes [39–41]. The application of box scores in the assessment of functional impairment has also been studied. For example, Sudo et al. developed a composite score designed to assess functional difficulties (CDR-FUNC) [42]. This score is based on the sum of CDR box scores that are aligned with functional performance (Problem solving, Home and Hobbies, Community Involvement, and Personal Care) and that may have a different trajectory than CDR based cognitive abilities. In addition, Sudo and colleagues reported that the CDR-FUNC score is strongly correlated with the functional performance as measured by the Pfeffer’s Functional Activities Questionnaire (FAQ) [42, 43].
Statistical analysis
We hypothesized that individuals with high EA and high levels of pTau181 would present less functional difficulties compared to their counterparts with low EA. We further investigated whether EA correlates with the functional resilience (abilities managing money, interacting with others, and caring for personal needs) differently between
RESULTS
The study sample consisted of 73.9% females, with a mean age of 71.08 (SD = 7.42) years and a mean log10(pTau181) level of −0.01 (SD = 0.42) (Table 1). A total of 74 individuals were classified as having high log10(pTau181) level (mean + 1SD), with 43 carrying the
Study samples characteristics
Characteristics of 74 selected individuals with high log10 (pTau181) levels
Individuals with high log10(pTau181) and low EA had significantly higher CDR-FUNC scores (median: 12.0; Q1:8.0 –Q3:12.0) compared to those with high EA (median: 3.75; Q1:0 –Q3:9.0;
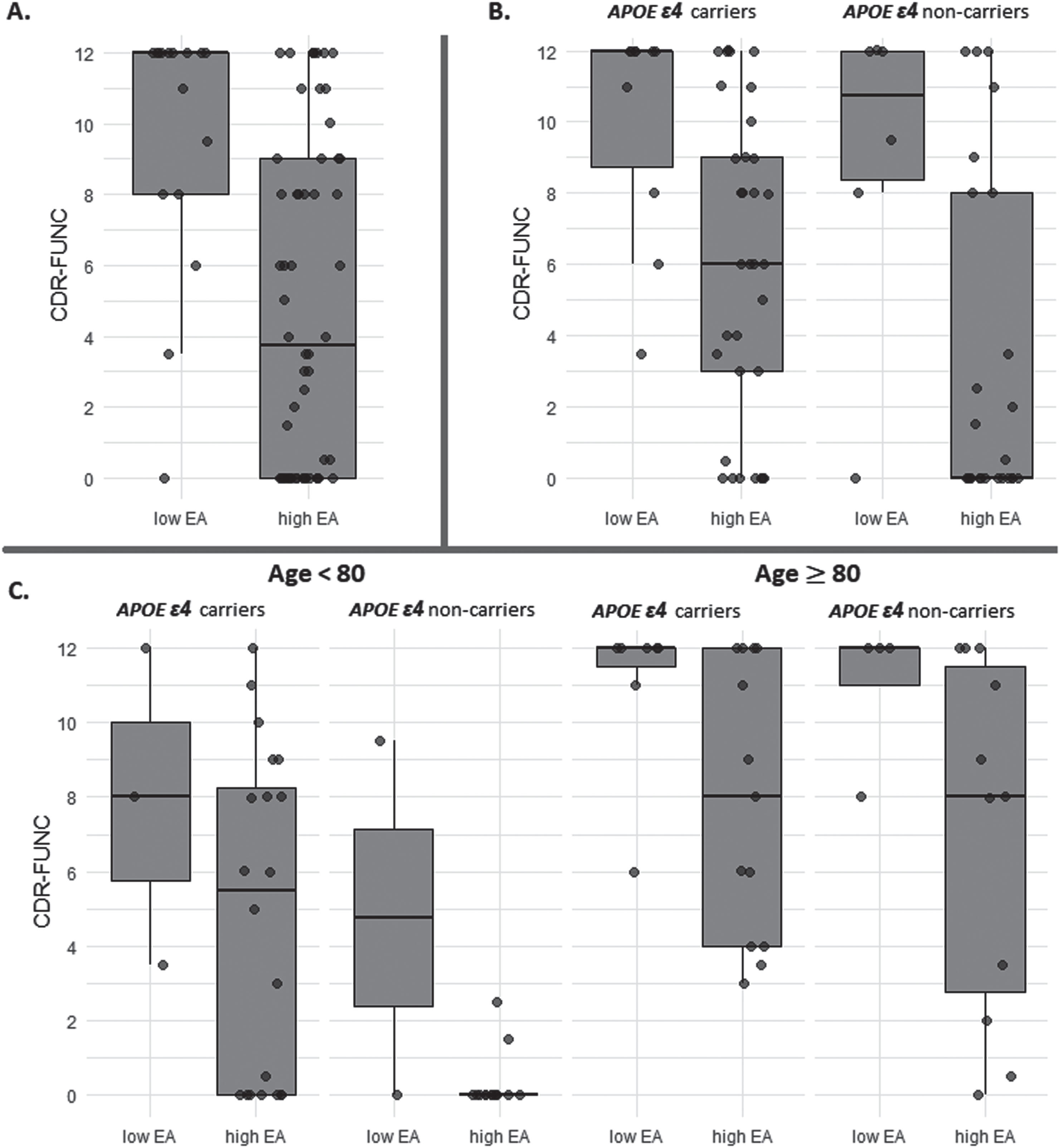
Box plot for the comparison between educational attainment (EA) levels (low and high) and CDR-FUNC score among advanced Alzheimer disease pathology individuals. (A) compare all individuals with the advanced Alzheimer disease pathology, (B) compare within subgroups stratified by the APOE ɛ4 allele, and (C) compare samples further stratified by age to investigate potential age-specific trends.
To determine the effect of the
We further stratified our samples by age to investigate potential age-specific trends. Notably, the patterns distinguishing low and high EA in these age-based subgroups were consistent with those observed in our primary dataset (Age≥80:
DISCUSSION
This study found that years of education are associated with better functional abilities in individuals with high log10(pTau181) levels. This effect was observed in both
Our results highlight a complex relationship among the genetic risk factors, ADP-associated biomarkers, and cognitive reserve and their effect on cognitive and functional outcomes of AD. The study supports the hypothesis, as suggested by Stern et al. [25], that the brain’s adaptability and efficiency in cognitive processing can mitigate the impacts of disease-related changes. This is evident in the varying functional outcomes among individuals with high log10(pTau181) levels, where those with high EA demonstrate a more robust cognitive reserve. This reserve, influenced by EA as per the model, is likely to enable the use of alternative neural networks to maintain cognitive function, aligning with the hypothesis by Stern et al. [20]. Particularly, the interaction between EA, genetic factors such as the
The trend of higher CDR-FUNC scores being associated with lower EA persists across stratified age groups. This suggests that the protective effect of high EA on functional difficulties is a consistent factor, regardless of age. However, it is noteworthy that the sample sizes decrease significantly in the stratified age groups. This reduction in sample size limits the ability to draw significant inferences about the impact of education and
Findings from this study should be interpreted with caution considering several methodological and instrumentation limitations. First, the study population is relatively small and is not representative of all African American populations. Therefore, we are unable to make universal claims that our findings will be observed in all individuals with African ancestry. Additionally, the limited sample sizes in certain subgroups, as indicated in Fig. 1, may constrain the strength of specific statistical inferences. Second, our operationalization of years in school as a proxy for educational attainment, does not capture quality of education, type of school, nature of school curriculum and location of school, all factors that could have impacted the findings. Third, the cross-sectional design of the data precluded us from making any causal claims in the associations between EA and functional status of individual with ADP. Despite these limitations, the strong community-based representation of our sample, the heterogeneity of participants across multiple sites (Florida and North Carolina) addresses concerns about not having a representative sample, and the use of gold-standard instruments to assess target variables, as well as the public health and clinical value of our findings overshadow these limitations.
In summary, this study shows the importance of using multiple variables such as genetics, education (an established social determinant of health), and AD biomarkers, to have a complete picture of the disease and its cognitive and functional outcomes. Notably, it demonstrates the need for comprehensive approaches to analyze the data, identify patterns associated with a risk of developing AD, and identify the specific combinations of factors most predictive of an individual’s risk of developing AD symptoms. Healthcare providers could use this information to identify individuals at high risk of AD and to develop personalized prevention and treatment plans for those individuals.
AUTHOR CONTRIBUTIONS
Farid Rajabli (Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Software; Validation; Visualization; Writing – original draft; Writing – review & editing); Azizi A. Seixas (Conceptualization; Methodology; Supervision; Writing – original draft; Writing – review & editing); Bilcag Akgun (Data curation; Formal analysis; Software; Writing – review & editing); Larry D. Adams (Data curation; Methodology; Writing – review & editing); Jovita Inciute (Data curation; Writing – review & editing); Kara L. Hamilton (Data curation; Writing – review & editing); Patrice G. Whithead (Data curation; Writing – review & editing); Ioanna Konidari (Data curation; Writing – review & editing); Tianjie Gu (Data curation; Writing – review & editing); Jamie Arvizu (Data curation; Writing – review & editing); Charles G. Golightly (Data curation; Writing – review & editing); Takiyah D. Starks (Data curation; Writing – review & editing); Renee Laux (Data curation; Writing – review & editing); Goldie S. Byrd (Data curation; Funding acquisition; Investigation; Writing – review & editing); Jonathan L. Haines (Methodology; Writing – review & editing); Gary W Beecham (Methodology; Writing – review & editing); Anthony J. Griswold (Data curation; Methodology; Writing – review & editing); Jeffery M. Vance (Conceptualization; Methodology; Writing – review & editing); Michael L. Cuccaro (Conceptualization; Investigation; Methodology; Project administration; Writing – original draft; Writing – review & editing); Margaret A. Pericak-Vance (Conceptualization; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Writing – original draft; Writing – review & editing).
Footnotes
ACKNOWLEDGMENTS
This investigation was supported by grants AG072547, AG074865, AG052410, AG028786 and AG032984 from the National Institutes on Aging of National Institutes of Health (NIH), HL135452 and HL152453 from the National Heart, Lung, and Blood Institute of NIH, and A2018556F grant from the BrightFocus Foundation.
FUNDING
This research was funded by the NIH, grant number AG072547, AG074865, AG052410, AG028786, AG032984, HL135452, and HL152453, and BrightFocus Foundation, grant number A2018556F.
CONFLICT OF INTEREST
The author has no conflict of interest to report.
