Abstract
Background
Education promotes cognitive reserve (CR), potentially buffering Alzheimer's disease pathology (ADP). However, the education-CR relationship may differ by population and genetic background.
Objective
To examine education, APOE ε4, and functional scores in a Puerto Rican (PR) cohort with varying plasma pTau181, an ADP biomarker.
Methods
A subset of 514 PR older adults with “high” (>mean+1SD) or “low” pTau181 (<mean-1SD) was analyzed. Functional scores were derived from the Clinical Dementia Rating scale (CDR-FUNC). The primary analysis examined educational attainment (EA; high ≥12 years versus low <12 years) as a categorical proxy for CR. Nonparametric tests compared CDR-FUNC across EA and APOE ε4 strata. A secondary analysis applied a hurdle model, examining years of education as a continuous predictor on (1) the odds of any impairment (CDR-FUNC > 0) and (2) severity among impaired participants, adjusting for age and sex.
Results
High EA was associated with better CDR-FUNC than low EA within the high pTau181 group (n = 80; MedianLow_EA = 7, MedianHigh_EA = 0; p = 0.011). The hurdle model similarly showed that each additional year of education reduced the odds of any functional impairment in the high-pTau181 group (OR = 0.89, 95% CI [0.79–0.99]). No significant education × APOE ε4 interaction was observed, though a negative trend suggested that increasing education attenuated ε4-related impairment.
Conclusions
Greater education is linked to functional preservation in PR older adults, particularly in those with elevated ADP, suggesting potential CR-mediated resilience. APOE ε4 may worsen outcomes with decreasing education, suggesting both educational and genetic factors should inform strategies to mitigate cognitive decline globally.
Introduction
Alzheimer's disease (AD) accounts for 60–80% of dementia cases, making it the most common cause. 1 Historically, the diagnosis of AD required confirmation of specific patterns of its canonical neuropathological features, namely amyloid-β (Aβ) and tau, during autopsy. 2 Advances in neuroimaging and fluid-based biomarker technologies have enabled increasingly accurate quantification of these features in vivo. 3 Consequently, diagnostic frameworks for research have evolved such that AD is now viewed as a neuropathological disorder characterized by abnormal levels of Aβ, tau, and neurodegeneration in living individuals, distinct from its downstream clinical outcomes.4,5 This shift in perspective underscores an important observation: while elevated AD pathology (ADP) increases dementia risk, some individuals remain cognitively unimpaired despite having a high neuropathological burden.6–9
Cognitive reserve (CR) is a theoretical construct that explains variability in cognitive trajectories in the presence of ADP. 10 Broadly, CR encompasses the adaptive mechanisms that enable the brain to compensate for injury and maintain function despite accumulating pathology. It is considered a dynamic entity shaped by multiple factors including genetics, education, and lifestyle. As a latent construct—unable to be measured directly—CR is typically inferred from proxy variables such as education, occupational complexity, or measures of aptitude (e.g., IQ). 11 A related concept is resilience, which refers to the maintenance of cognitive function despite pathology or other adverse factors. 12 While CR is thought to contribute to resilience, the two are not interchangeable; CR represents one mechanism through which resilience may be developed. Given the limited treatments for dementia, identifying modifiable factors that enhance CR—and potentially mitigate the deleterious effects of ADP—is a key focus in cognitive aging research.
For pragmatic and empirical reasons, education is perhaps the most frequently used proxy variable for CR. 11 Education is (1) associated with cognitive outcomes in older adults, (2) straightforward to collect, and (3) intuitively interpretable in the context of CR (e.g., higher education equates to increased CR). Moreover, research suggests that operationalizing education categorically as educational attainment (EA)—measured by the highest level of education completed (e.g., high school or college degree)—may better capture milestone-related effects of education that contribute to CR, beyond total years of schooling. 13 A meta-analysis investigating the effect of education on incident dementia found that comparing individuals who had at least completed high school to those who had not provided the EA threshold most strongly associated with differences in dementia-free survival time, suggesting a possible plateau in the contribution of education towards CR at higher levels of attainment. 14 Nevertheless, the extent to which education contributes to CR may vary depending on specific mediating factors, including demographic properties. Studies have reported conflicting outcomes regarding the moderating effect of education on cognitive decline across different populations. 15 Additionally, research on MRI biomarkers of brain integrity has found population-specific differences in their impact on cognition, suggesting that factors contributing to CR, including education, may play a role in these differences. 16 Collectively, these findings emphasize the need to examine the interplay between education, CR, and cognitive outcomes across populations to inform the development of global strategies for AD prevention.
The effect of education on cognitive function may also depend on the presence of specific genetic risk factors for AD. The strongest genetic risk factor for late-onset AD is the APOE gene, which encodes three primary alleles found at the population level: ε3 (reference), ε4 (risk-increasing), ε2 (risk-decreasing). 17 Prior studies suggest a potential synergism between education and APOE status, where the magnitude of effect of education/CR on cognitive function varies based on whether an individual carries the ε4 allele.18,19 Nevertheless, there are discrepancies in the relative magnitude of these effects across APOE ε4 carrier status; some studies have reported a more pronounced effect of education on cognitive function among ε4 carriers compared to non-carriers, while others report a weaker effect among ε4 carriers.20–22 Additionally, the effect of APOE status on changes in the “functional” aspects of cognition—those involved in activities of daily living that promote independence (e.g., personal care, managing finances, social interaction)—is less understood. Previous research on the relationship between APOE ε4 and functional performance measures, such as the Instrumental Activities of Daily Living (IADL), have reported either no effect or an association with increased functional decline among ε4 carriers.23,24 Similarly, the relationship between education and the preservation of functional abilities is understudied.
Taken together, these observations highlight the importance of further investigating how education, alongside genetic risk factors like APOE, influence various aspects of cognition, including those related to functional abilities. Previously, our group examined the relationship between EA, APOE ε4, and functional performance in an African American cohort with elevated plasma pTau181. 25 Building on that work, the present study investigates this relationship in a Puerto Rican (PR) cohort, a population that represents the second largest Hispanic subgroup in the United States. PRs—part of the broader Caribbean Hispanic population—have been reported to experience a more severe AD symptomology and greater AD-related mortality compared to other ethnic groups, which may reflect differences in healthcare access, education, economic conditions, and other lifestyle or environmental factors.26,27 Given their substantial representation in the U.S. population, identifying factors that promote CR and protect against cognitive decline in this population is of critical importance. To accomplish this, we first examined whether high versus low EA was associated with functional performance in older PR adults using non-parametric tests. Additionally, we assessed whether APOE ε4 carrier status was associated with differences in functional abilities among individuals with elevated pTau181, across low and high EA strata. As a secondary analysis, we modeled years of education continuously using a hurdle framework to evaluate its association with CDR-FUNC and APOE ε4 while accounting for covariates and non-normal outcome distribution.
Methods
Sample selection
This is a primary study conducted by the authors as part of ongoing genetic studies of AD. Study participants were enrolled primarily from the seven principal health regions defined by the Puerto Rico Department of Health. A small subset of participants residing in the continental United States, who were related to participants living in PR, were also enrolled. The general ascertainment and clinical evaluation procedures are thoroughly described in Feliciano-Astacio et al., 2019. 28 Participants were screened using the Modified Mini-Mental State Examination (3MS) and CDR.29,30 Those exhibiting cognitive impairment also underwent a comprehensive neuropsychological assessment to provide a consensus diagnosis of definite, probable, or possible AD or mild cognitive impairment (MCI) by a clinical adjudication board.31,32 Study procedures were approved by the Institutional Review Boards (IRB) of the University of Miami and the Universidad Central del Caribe. Written informed consent was obtained from all participants following a detailed explanation of study procedures. In cases where participants were severely cognitively impaired, proxy consent was obtained from a family member.
Participants were considered for the present study if they: (1) were screened using the CDR, (2) had plasma measures pTau181 available, and (3) had APOE genotype information available. A total of 514 individuals met these criteria.
Outcome variable: CDR-FUNC
The outcome measure for this study was derived from the Clinical Dementia Rating (CDR) scale. 33 The CDR is a widely used and well-validated staging instrument for dementia, administered through a structured interview that applies a 5-point scale (ranging from 0 to 3: 0, 0.5, 1, 2, 3) to assess cognitive and functional performance across six primary domains: memory, orientation, judgment and problem solving, community affairs, home and hobbies, and personal care. The CDR has demonstrated strong reliability and validity across different cultural and linguistic contexts, including Spanish-language and Hispanic samples. 34 Multiple approaches have been developed to generate an aggregate score from the domain ratings (e.g., by weighted algorithm or unweighted domain sum).35,36 To focus on functional capabilities, this study used the functional CDR score (CDR-FUNC). 37 The CDR-FUNC is calculated by summing the ratings from four domains—judgment/problem solving, community affairs, home/hobbies, and personal care—producing an overall score between 0 to 12, with higher scores indicating increased functional impairment.
Plasma pTau181 measurement and APOE genotyping
The SimoaTM pTau181 Advantage V2 assay (Quanterix) was used to measure blood plasma pTau181 concentrations (pg/mL) as previously described. 38 Samples were randomized across assay plates based on age, sex, and cognitive status, and each was measured in duplicate. The mean of the duplicate readings per sample were used for the analysis. Additionally, pTau181 concentrations were log10-transformed to provide a closer approximation to a normal distribution. APOE genotyping was performed using polymerase chain reaction (PCR) and restriction fragment length polymorphism (RFLP), as detailed in Saunders et al. 39
Analysis
The primary analysis assessed EA —defined categorically as “high” (≥ 12 years) versus “low” (< 12 years)—and its association with CDR-FUNC. The 12-year cutoff was selected based on prior evidence that high school completion represents an EA threshold with especially pronounced differences in cognitive outcomes compared to other cutoffs. 14 This approach was complemented by a continuous-years framework in the secondary hurdle analysis to examine linear effects of education on CDR-FUNC.
Plasma pTau181 measurements were obtained from 514 participants across three primary assay batches over two years. This study focused primarily on individuals with elevated pTau181, defined as a log10(pTau181) concentration exceeding one standard deviation above the mean. To account for batch effects in the pTau181 distributions, the “high” log10(pTau181) designation was assigned within each batch. Additionally, because higher EA may independently contribute to better functional performance, even in the absence of ADP, we also assessed its effect on the CDR-FUNC among individuals with “low” pTau181—based on a log10(pTau181) measurement less than one standard deviation below the mean.
Given the overrepresentation of CDR-FUNC scores equal to 0 relative to scores greater than 0, the primary analyses employed non-parametric statistical methods. Differences in CDR-FUNC between EA strata were assessed using the Mann-Whitney U test within both the low and high pTau181 groups. Because individuals in the low pTau181 group were younger on average, an age-stratified analysis was conducted by selecting participants aged ≥ 75. Additionally, to examine whether APOE influenced the relationship between EA and functional performance, individuals with high pTau181 were analyzed by their ε4 carrier status. Specifically, the Mann-Whitney U test was used to compare CDR-FUNC scores between ε4 carriers and non-carriers in both low and high EA groups.
The secondary analysis used a hurdle model framework to account for the zero-skewed CDR-FUNC distribution while allowing covariate adjustment. The hurdle models used in this study comprised two components: (1) a binary component estimating the probability of a non-zero outcome (e.g., CDR-FUNC > 0) using logistic regression, and (2) a positive-outcome component modeling the distribution of non-zero values (e.g., among those with CDR-FUNC > 0) using a generalized linear model assuming a gamma distribution with a logarithmic link function. These models were applied separately within the low and high pTau181 groups, with years of education (continuous) as the main predictor, adjusting for sex and age at exam. To assess whether APOE ε4 modified the relationship between education and CDR-FUNC within the hurdle framework, an additional model including an interaction term between ε4 carrier status and years of education was fitted in the high pTau181 group.
Results
A detailed summary of the demographic features and APOE genotype frequencies for individuals in the low and high pTau181 subgroups is provided in Table 1. The average number of years of formal education did not differ significantly between the two groups (p = 0.12). Additionally, individuals with high pTau181 had significantly worse functional performance, as measured by the CDR-FUNC, than those with low pTau181 (p < 0.001).
Demographic characteristics and APOE genotype frequencies for individuals with “low” and “high” pTau181.
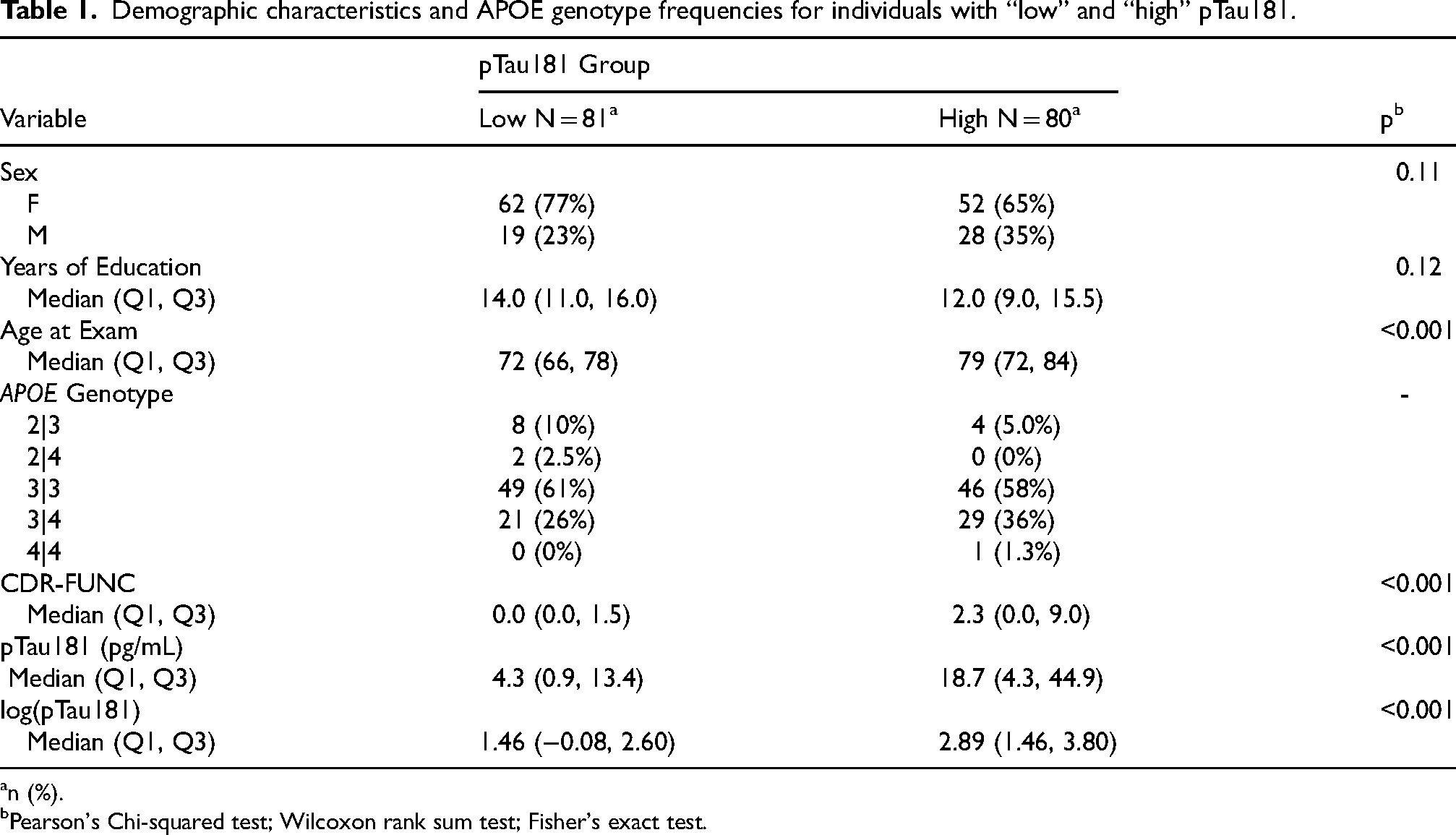
n (%).
Pearson's Chi-squared test; Wilcoxon rank sum test; Fisher's exact test.
CDR-FUNC by EA level in high and low pTau181 groups
Table 2 summarizes age, sex, APOE genotype, and CDR-FUNC scores across dichotomized EA levels (low/high), for the low and high pTau181 groups. Among individuals with high pTau181, high EA was associated with better functional performance (lower CDR-FUNC scores) compared to low EA (n = 80; MedianLow_EA = 7, MedianHigh_EA = 0; p = 0.011). A similar trend was observed in the low pTau181 group, though the difference in functional performance between EA strata was more modest (n = 81; MedianLow_EA = 0, MedianHigh_EA = 0; p = 0.032). Figure 1 illustrates the distribution of CDR-FUNC scores across EA strata for both pTau181 groups.
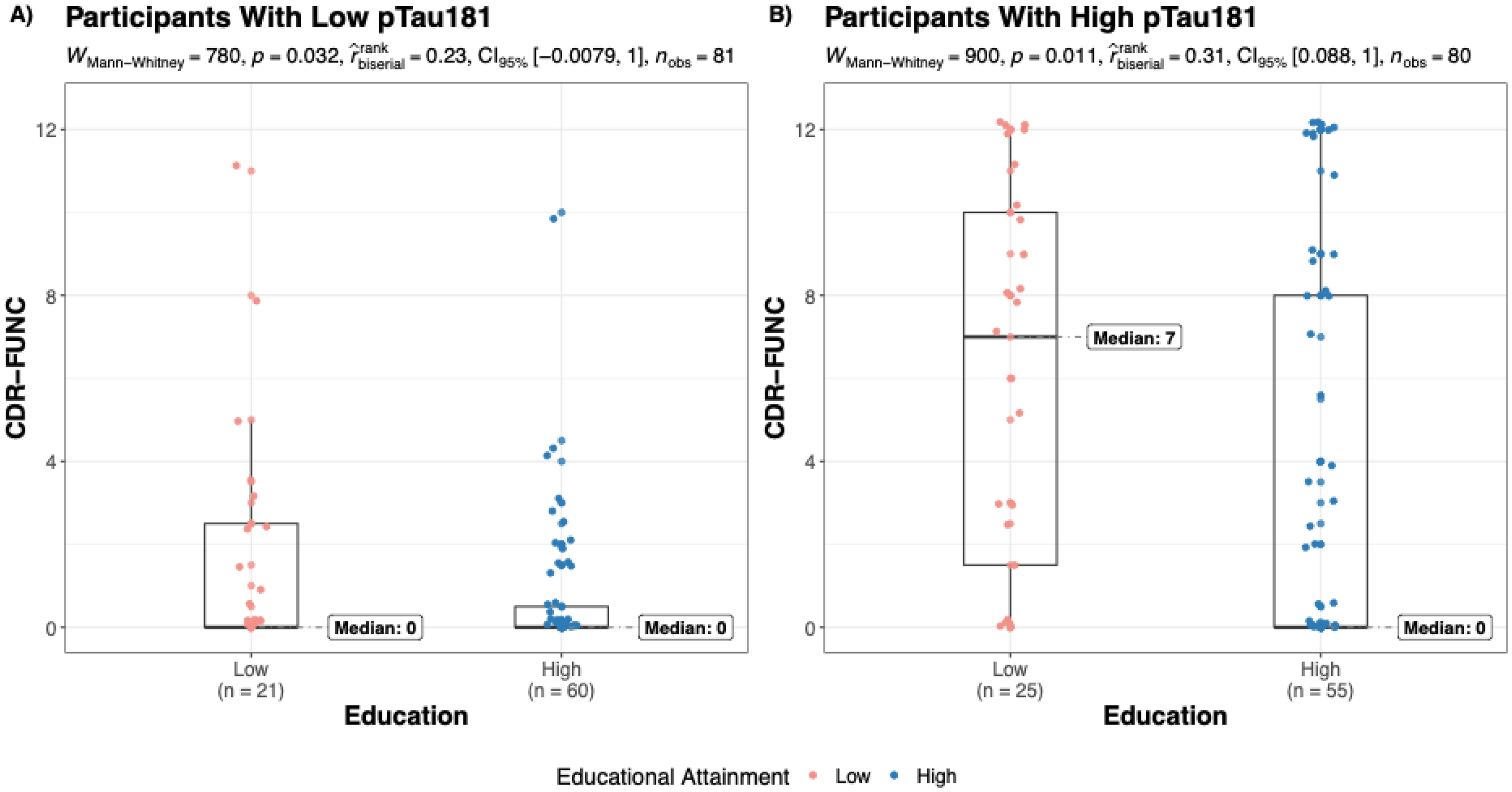
Effect of educational attainment on CDR-FUNC among participants with low and high pTau181. (A) Among individuals with low pTau181, those with high EA have modestly better functional performance than those with low EA. B) In the high pTau181 group, a more pronounced difference in functional performance is observed between low and high EA individuals.
Demographic characteristics, APOE genotype frequencies, and CDR-FUNC scores across dichotomized EA levels, grouped by pTau181 status.
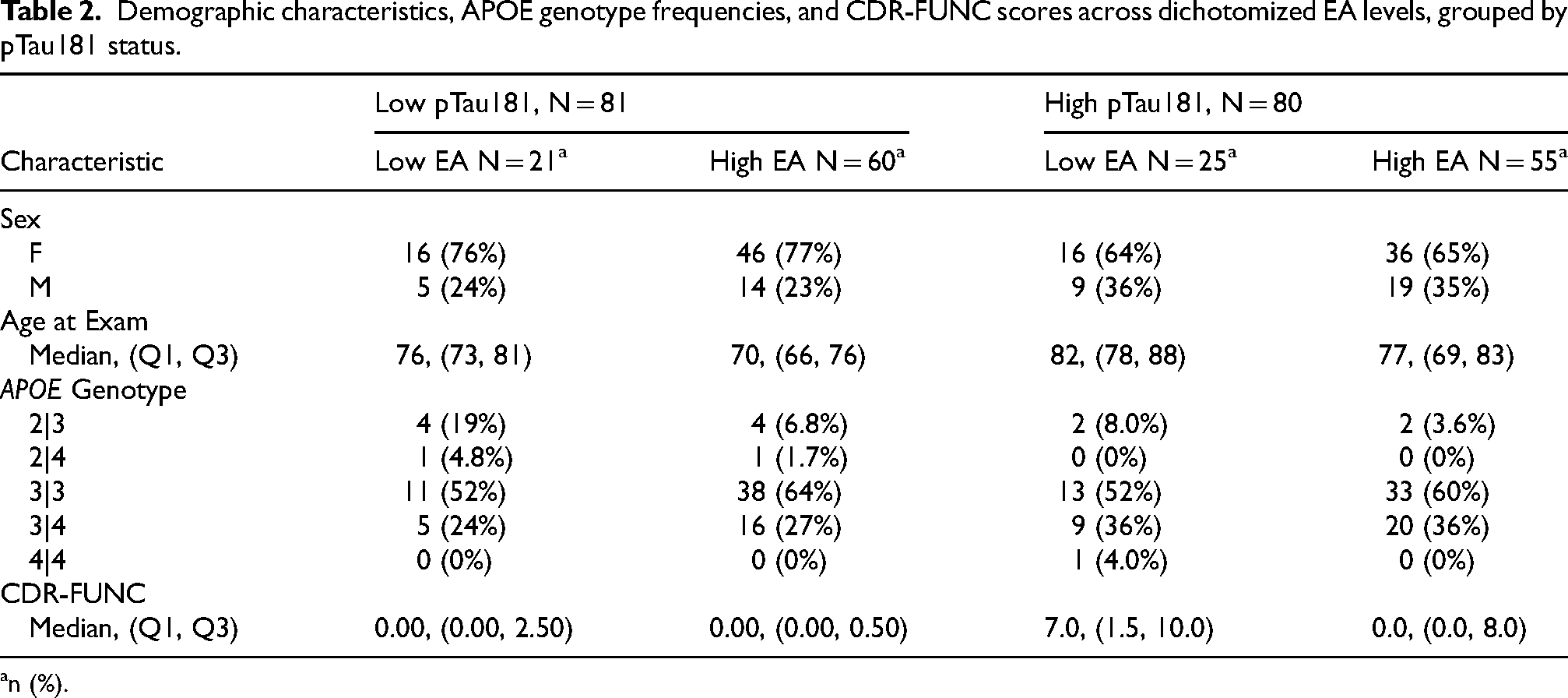
n (%).
In the age-stratified analysis (≥75 years), high EA was associated with better functional performance compared to low EA in the high pTau181 group (MedianLow_EA = 7.5, MedianHigh_EA = 0.5; p = 0.047). In contrast, among individuals aged ≥ 75 with low pTau181, functional performance was only nominally different between low and high EA strata (MedianLow_EA = 0.75, MedianHigh_EA = 0; p = 0.27). These results are presented in Supplemental Figure 1.
CDR-FUNC by APOE ε4 carrier status in high and low EA groups
Among those with high pTau181, APOE ε4 carrier status showed a trend towards worse functional performance differences in the low EA subgroup, where ε4 carriers had greater CDR-FUNC scores than non-carriers (Medianε4 non−carrier = 2, Medianε4 carrier = 8; p = 0.06). However, this association was weaker within the high EA subgroup (Medianε4 non−carrier = 0, Medianε4 carrier = 0.25; p = 0.41). These results are illustrated in Figure 2.
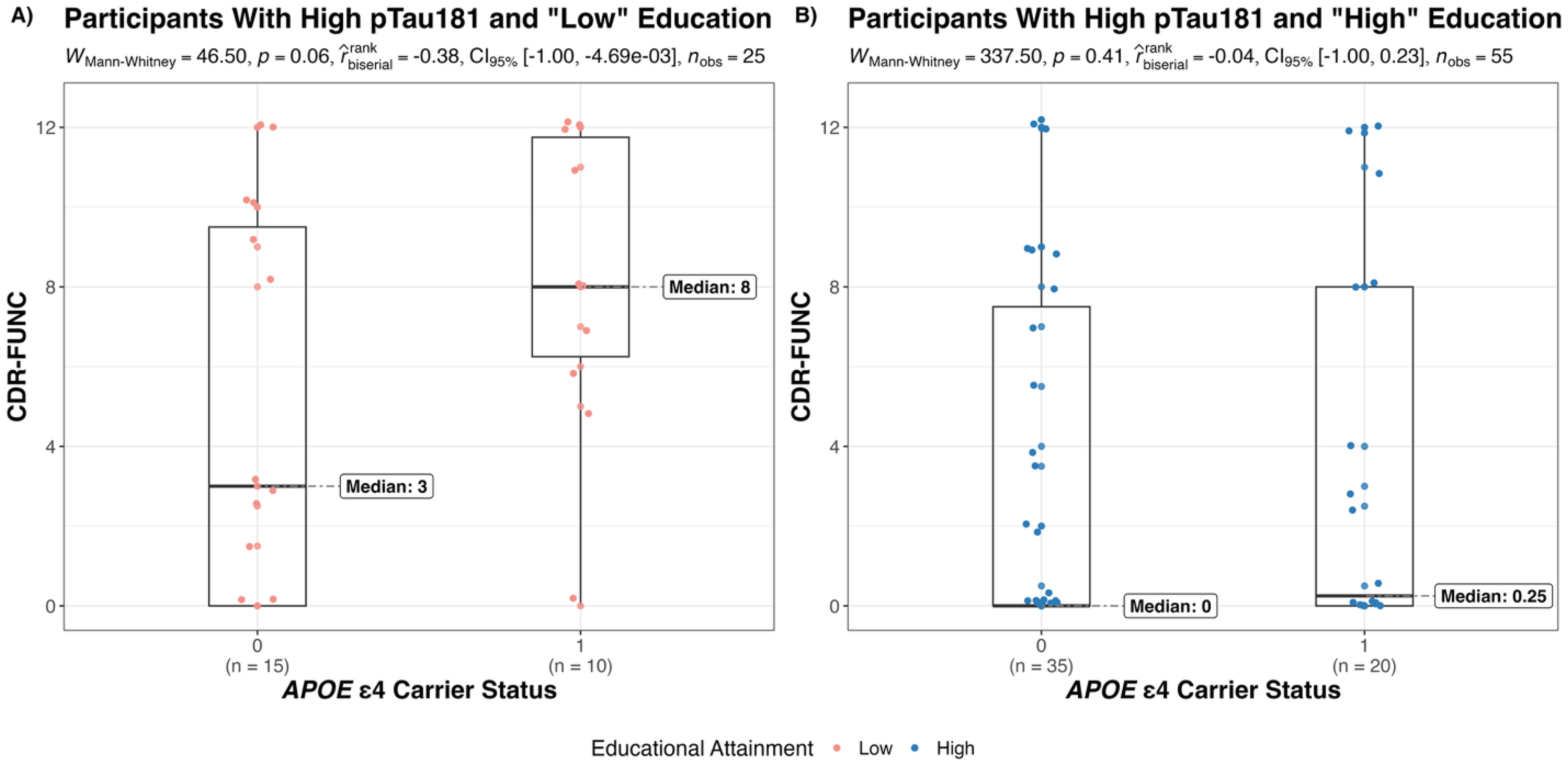
Effect of APOE ε4 carrier status on CDR-FUNC among participants with high pTau181, grouped by educational attainment. (A) APOE ε4 carriers with high pTau181 and low education have worse functional performance than non-carriers. B) APOE ε4 carriers with both high pTau181 and high education have similar functional performance to non-carriers.
Hurdle models using years of education (secondary analysis)
Applying the hurdle model to the high pTau181 group, increasing years of education was associated with reduced odds of having a CDR-FUNC score greater than 0 (OR = 0.89, 95% CI [0.79, 0.99], p = 0.049), adjusting for age at exam and sex. Among those with non-zero CDR-FUNC, years of education were not associated with the severity of functional impairment (β = 0.00, 95% CI [−0.03,0.05], p = 0.9). A summary of the hurdle model results for this group is provided in Table 3.
Hurdle model, functional status by years of education, age, and sex within high pTau181 group.
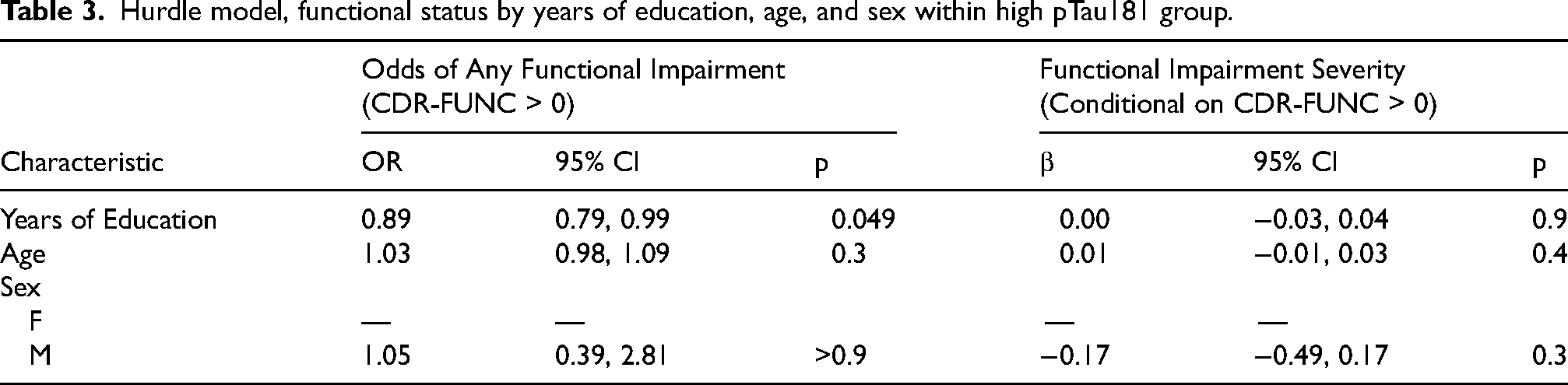
In the low pTau181 group, years of education showed a weaker, statistically non-significant association with the odds of having a CDR-FUNC score above 0 (OR = 0.92, 95% CI [0.84, 1.02], p = 0.11). Among those with non-zero CDR-FUNC scores, years of education were not associated with severity of functional impairment (β = −0.05, 95% CI [-0.12, 0.01], p = 0.2). The results for the low pTau181 group are shown in Table 4.
Hurdle model, functional status by years of education, age, and sex within low pTau181 group.

CI: confidence interval; OR: odds ratio.
The addition of an interaction term between APOE ε4 carrier status and years of education did not provide evidence of an interaction effect when assessing the odds of having a CDR-FUNC > 0 (ORinteraction = 1.03, 95% CI [0.81, 1.3], pinteraction = 0.8). In the gamma component, the interaction between APOE ε4 carrier status and education was negative (βinteraction = −0.04, 95% CI [−0.11–0.03], pinteraction = 0.2), indicating that the effect of carrying ε4 decreased with increasing years of education. Specifically, each additional year of education was associated with an approximately 4% reduction in the expected impairment between ε4 carriers and non-carriers. However, this trend did not reach statistical significance. These results are provided in Table 5.
Hurdle model with APOE ε4 carrier by education interaction among individuals with high pTau181.
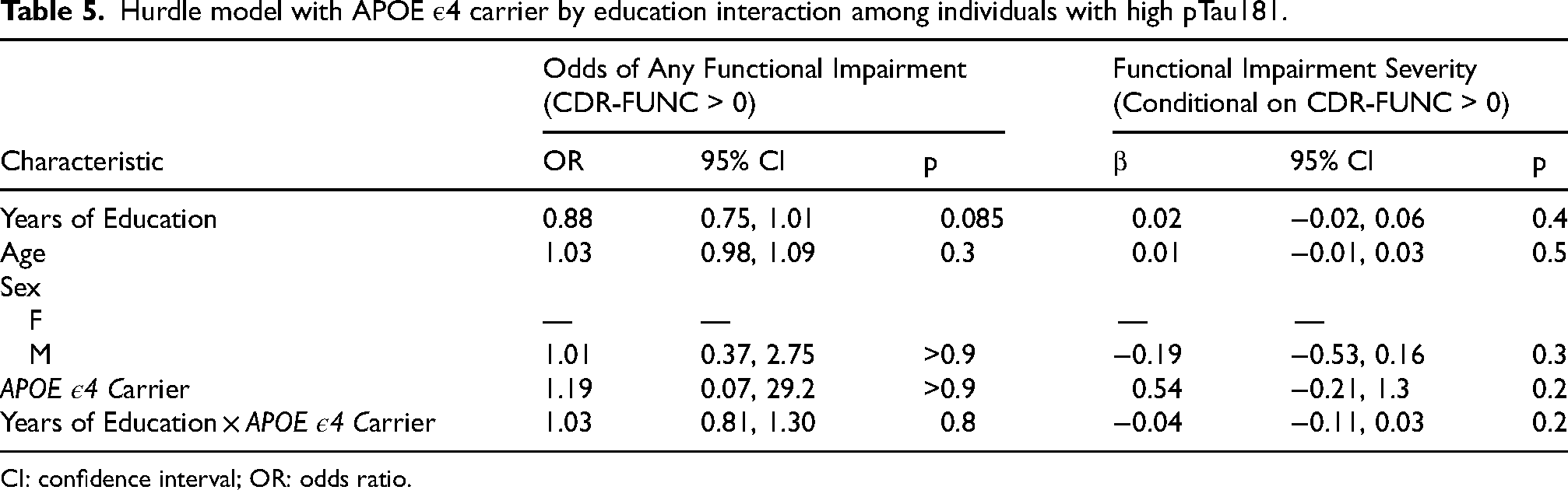
CI: confidence interval; OR: odds ratio.
Discussion
Across both the categorical (EA) and continuous (years of education) analyses, greater education was associated with functional preservation among older PR adults. This relationship was present in both low and high pTau181 groups, but was more pronounced in the high pTau181 group, suggesting that greater education may enhance CR and promote resilience to ADP in the PR population. Additionally, among those with high pTau181, APOE ε4 carriers tended to have worse functional performance (higher median CDR-FUNC) than non-carriers within the high EA group. In contrast, among those with low EA, the median CDR-FUNC difference between ε4 carriers and non-carriers was more modest. This pattern may indicate that higher EA partially offsets additional risk conferred by ε4 in the presence of elevated pTau181.
Education may independently contribute to variability in cognitive test performance among unimpaired individuals (e.g., improved literacy, test-taking skills), and to the overall risk of cognitive impairment through CR. In the context of functional abilities, as estimated by the CDR-FUNC, higher levels of education may promote skills that support daily independence—such as finance management, social engagement, and the pursuit of hobbies—even in the absence of ADP. The findings from our primary analysis reflect this possibility, as individuals with low pTau181 and high EA demonstrated modestly better functional performance than those with low EA. Nevertheless, the relationship between EA and functional performance was notably more pronounced in the high pTau181 group, suggesting that by promoting CR, higher EA may help mitigate the deleterious effects of ADP. The effects observed in the secondary analysis support this trend, although increasing education was more strongly associated with reduced odds of any functional impairment (CDR-FUNC > 0) than with impairment severity. Furthermore, these results align with the established theory of CR, which posits that greater CR reduces the brain's susceptibility to injury by engaging compensatory mechanisms that help maintain functional integrity.10,40
APOE ε4 is among the strongest risk factors for AD dementia, and identifying therapeutic strategies that mitigate its deleterious effects—whether through pharmacologic or lifestyle mechanisms—remains a central goal in AD research. 41 Our primary analysis examined whether APOE ε4 carriers with elevated pTau181 faced additional risk of functional decline compared to non-carriers, across EA strata. Notably, only APOE ε4 carriers with low EA exhibited worse functional performance than their non-carrier counterparts, suggesting a possible interplay between APOE status, EA/CR, and functional performance. The inclusion of an APOE ε4 × years of education interaction term in the secondary analysis did not yield statistically significant effects in either the binary or gamma components. However, in the gamma model, the negative direction of the interaction suggested that the detrimental effect of ε4 on functional impairment severity may modestly weaken with increasing education, consistent with the pattern observed in the non-parametric results.
While one of the major pathobiological mechanisms by which APOE ε4 increases AD risk is promoting Aβ aggregation and inhibiting its clearance, growing evidence indicates that APOE ε4 also exerts a pathogenic effect through several Aβ-independent pathways, including glucose metabolism, immune response, synaptic integrity, and cerebrovascular function. 42 The observation that ε4 carriers with low EA and high pTau181 exhibited the poorest functional outcomes raises the possibility that diminished CR may exacerbate the effects of ε4 across multiple biological domains. It is worth noting, however, that fewer individuals comprised the low EA/high pTau181 group than the high EA/high pTau181 group (25 versus 55, respectively). As more data are collected, it is important to follow-up on these results to determine whether the observed effect persists in a larger sample.
This study has limitations that warrant consideration. First, although plasma pTau181 is a well-established biomarker that correlates strongly with tau burden in the brain, it serves as an indirect measure of neuropathology and may introduce classification error when assigning individuals to ADP strata. 43 Additionally, while the observed association suggests that greater education/CR may protect against functional decline, the cross-sectional study design necessitates careful interpretation of these effects. Observational studies are inherently susceptible to statistical biases such as confounding and reverse causality. Isolating education's independent contribution is particularly challenging, as it interacts with interrelated factors such as income, healthcare, and lifestyle, all of which likely influence functional preservation. Greater premorbid cognitive ability or genetically driven aptitude may also facilitate higher educational attainment and contribute to CR, potentially confounding the observed association. 44 The cross-sectional design further limits our ability to distinguish CR from resilience (e.g., sustained resistance to decline over time), which typically require longitudinal cognitive measures. Moreover, randomized longitudinal studies designed to establish causality between sociodemographic factors like education and health outcomes are often infeasible due to ethical and logistical constraints. 45 Lastly, our modest sample size limits power for subgroup analysis—particularly higher-order interactions involving APOE, EA, and pTau181—which should therefore be interpreted with caution. Nevertheless, specialized causal inference techniques, such as instrumental variable analyses, have been used to identify casual links between education and aging traits, including cognitive performance and dementia risk, suggesting that education may directly influence these outcomes.46,47 The neurobiological influence of education on developing CR may be partially driven by increasing synaptic density and enhancing interneuronal connectivity during early development. 48
The present study provides insights into the dynamics between education, CR, and functional preservation in PR older adults, advancing our understanding of these processes in one of the largest Hispanic subpopulations in the United States. Previous estimates indicate that Hispanics—particularly Caribbean Hispanics including PRs—experience a higher prevalence and severity of AD compared to other non-Hispanic populations. 49 This increased burden may reflect structural factors such as educational quality, economic circumstances, and quality of healthcare, which could influence the development of CR. Our findings align with our previous research in an African American cohort, which also identified a general protective effect of EA against functional decline in the presence of ADP (pTau181). 25 Together, these studies suggest that while EA may confer protective benefits across populations, the magnitude and mechanism of this effect may vary based on contextual factors that influence educational opportunities and other life-course exposures. Given the limited availability of treatments for conditions driving cognitive decline, prioritizing modifiable lifestyle factors such as EA in public health strategies could be crucial for mitigating risk and reducing incidence rates.
Supplemental Material
sj-docx-1-alz-10.1177_13872877261415933 - Supplemental material for Educational attainment is associated with reduced functional decline in Puerto Ricans with elevated pTau181
Supplemental material, sj-docx-1-alz-10.1177_13872877261415933 for Educational attainment is associated with reduced functional decline in Puerto Ricans with elevated pTau181 by Daniel A. Dorfsman, Dingtian Cai, Kara L. Hamilton-Nelson, Larry D. Adams, Pedro R. Mena, Vanessa C. Rodriguez, Jose Javier Sanchez, Glenies S. Valladares, Mariangelie Lopez, Patrice L. Whitehead, Michael B. Prough, Patricia Manrique, Anisley Martinez, Sabrina M. Mas, Carolina Scaramutti, Heriberto Acosta, Concepcion Silva-Vergara, Katalina McInerney, Anthony J. Griswold, Briseida E. Feliciano-Astacio, Michael L. Cuccaro, Brian W. Kunkle, Christiane Reitz, Giuseppe Tosto, William S. Bush, Jonathan L. Haines, Joshua O. Akinyemi, Rufus O. Akinyemi, Adesola Ogunniyi, Goldie S. Byrd, Jeffery M. Vance, Katrina Celis, Azizi Seixas, Margaret A. Pericak-Vance and Farid Rajabli in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
This work was supported by the National Institute on Aging (NIA) grants AG070864 and AG074865, as well as the Coins for Alzheimer's Research Trust (CART). We also thank the participants for their contributions to this study.
Ethical considerations
Study protocols were approved by the Institutional Review Boards of the University of Miami (protocol no. 20070307) and Universidad Central del Caribe (protocol no. 2016-26).
Consent to participate
All participants provided informed consent in accordance with the guidelines of the Institutional Review Boards of the University of Miami and Universidad Central del Caribe.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institute on Aging (NIA) grants AG070864 and AG074865; Coins for Alzheimer's Research Trust (CART).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Rufus Akinyemi is an Editorial Board Member of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer-review.
The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data referenced in this manuscript are available upon request and with principal investigator approval by reaching out to the primary author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
