Abstract
Background:
Deterioration of the oral environment is one of the risk factors for dementia. A previous study of an Alzheimer’s disease (AD) model mouse suggests that tooth loss induces denervation of the mesencephalic trigeminal nucleus and neuroinflammation, possibly leading to accelerated tau dissemination from the nearby locus coeruleus (LC).
Objective:
To elucidate the relevance of oral conditions and amyloid-β (Aβ) and tau pathologies in human participants.
Methods:
We examined the number of remaining teeth and the biofilm–gingival interface index in 24 AD-spectrum patients and 19 age-matched healthy controls (HCs). They also underwent positron emission tomography (PET) imaging of Aβ and tau with specific radiotracers, 11C-PiB and 18F-PM-PBB3, respectively. All AD-spectrum patients were Aβ-positive, and all HCs were Aβ-negative. We analyzed the correlation between the oral parameters and radiotracer retention.
Results:
No differences were found in oral conditions between the AD and HC groups. 11C-PiB retentions did not correlate with the oral indices in either group. In AD-spectrum patients, brain-wide, voxel-based image analysis highlighted several regions, including the LC and associated brainstem substructures, as areas where 18F-PM-PBB3 retentions negatively correlated with the remaining teeth and revealed the correlation of tau deposits in the LC (
Conclusions:
Our findings with previous preclinical evidence imply that tooth loss may enhance AD tau pathogenesis, promoting tau spreading from LC to the hippocampal formation.
Keywords
INTRODUCTION
The number of people with dementia worldwide is estimated to increase from 57.4 million in 2019 to 152.8 million by 2050 [1]. Alzheimer’s disease (AD) is neuropathologically characterized by depositions of abnormal amyloid-β (Aβ) and phosphorylated tau aggregates in the brain. AD has recently been regarded as a continuum among three distinct clinically defined entities consisting of preclinical AD, prodromal AD (mild cognitive impairment [MCI]), and AD dementia [2]. To date, some advances in disease-modifying treatments of AD have been achieved, as represented by the review and approval of a few Aβ-targeting drugs by the US Food and Drug Administration [3–5]. However, there is still controversy regarding their effectiveness, adverse events, and costs in real-world clinical settings [6–8]. Hence, there is still an urgent requirement to identify modifiable risk factors for AD, such as education, vascular issues, and depression [9, 10] to prevent disease onset.
Deterioration of the oral environment, including tooth loss and periodontal disease, has been suggested as a factor predisposing to the development of dementia in several cohort studies [11–17]. Tooth loss in humans has been linked to the onset of dementia via various pathways [18], such as nutritional imbalances resulting from impaired masticatory function [19] and decreased cerebral blood flow in the prefrontal cortex [20]. In a nonclinical study using transgenic AD mice, tooth loss caused denervation of the trigeminal mesencephalic nucleus (Vmes), with its nerve endings at the root apex. This alteration was followed by degeneration of the locus coeruleus (LC) located near the Vmes, conceivably via activation of inflammatory microglia [21]. LC is also known as a region presenting the earliest tau pathology that occurs in youth [22, 23], and tau deposits in this location are likely to spread transsynaptically to the hippocampal formation at an initial tau pathology stage proposed by Braak and Braak [24]. However, a long-term gap exists between the occurrence of tau lesions and neuronal loss in the LC [25], and its pathophysiology is still unknown. In the aforementioned study using a mouse model, hippocampal degeneration and dementia-like behavior were also found following degeneration of Vmes and LC, suggesting the impact of tooth loss on the pathological progressions and neurofunctional deteriorations in AD [21].
Periodontal disease is an infectious inflammatory condition caused by periodontopathogenic bacteria, chronic exposure to which has been associated with an increased risk of the development of AD [26]. The potential link between periodontitis and AD can be direct, via the invasion of bacteria and its products into the brain, or indirect, via the systemic inflammation caused by the presence of periodontal bacteria [27]. Regarding the association with AD pathologies, studies have suggested that, by using model mice and postmortem brains, periodontal pathogens and their enzymes could promote the Aβ depositions and tau phosphorylation [28–30]. In humans, associations between the biofilm–gingival interface (BGI) index [31] and cognitive impairment have been reported in four studies [32–35]. Two of these studies indicated an association of the BGI index with the Mini-Mental State Examination (MMSE) score [34, 35], while the others reported no such links [32, 33].
A number of clinical studies using neuroimaging have examined the relationship between tooth loss, periodontal disease, and AD pathogenesis. In magnetic resonance imaging (MRI) studies, the number of remaining teeth was negatively correlated with gray matter volume [36, 37], particularly in the parahippocampal gyrus of AD patients [38, 39]. The magnitude of periodontal disease in cognitively normal participants was associated with the brain Aβ load in a positron emission tomography (PET) study [40], whereas a prospective cohort study did not show any significant association between periodontal disease and Aβ PET positivity [41]. Furthermore, no tau PET studies have explored the relationship between the number of remaining teeth and the severity of periodontitis and tau lesions in the brain. Thus, the association between the oral environment and AD pathogenesis in humans remains elusive.
The recent development of PET ligands has enabled us to assess tau deposits in humans [42]. Several tau PET ligands have been shown to quantify and visualize tau pathology with AD and aging, and their clinical application as diagnostic and possibly prognostic tools is anticipated [43]. 18F-PM-PBB3 (APN-1607/florzolotau) is one of the novel tau PET ligands, which captures tau lesions with high contrast [44], and its potential has already been demonstrated in several clinical studies of AD [45–48]. In the present study, we hypothesized that tooth loss could provoke neuronal damage in the brainstem and subsequent acceleration of AD neuropathologies, while periodontal disease has also been suggested to be linked with abnormal protein deposits in the brain. Consequently, we evaluated the number of remaining teeth and the severity of periodontitis in participants who underwent Aβ (11C-PiB) and tau (18F-PM-PBB3) PET scans in order to correlate these oral statuses with AD pathogenesis.
METHODS
Participants
We enrolled 51 participants aged over 65 in this study between January 2020 and June 2022. Participants were recruited from affiliated hospitals and the National Institutes for Quantum Science and Technology volunteer association. The subjects were comprised of 19 AD and 13 MCI cases diagnosed according to the National Institute of Neurological and Communicative Disorders and Stroke/Alzheimer’s Disease and Related Disorders Association criteria [49] and Petersen’s criteria [50], respectively. Nineteen participants who did not meet these criteria and had no history of neurologic and psychiatric disorders were included as healthy controls (HCs). All participants underwent screening to exclude those with any organic brain complications or disorders, such as altered states of consciousness, prior head injuries requiring hospitalization and treatment, or a history of apparent cerebral infarction or hemorrhage.
Participants subsequently underwent a battery of neuropsychological evaluations, MRI examinations, and Aβand tau PET scans as part of the clinical trials described below. The neuropsychological evaluations assessed cognitive, executive, and functional deficits using the MMSE, Frontal Assessment Battery (FAB), and Clinical Dementia Rating (CDR) scales. For participants experiencing difficulties understanding test instructions due to cognitive deficits, we accommodated the guidance by providing explanations in simpler language. Furthermore, we requested caregivers to accompany these subjects, which facilitated each participant’s successful completion of almost all testing procedures. The examination findings were analyzed to assess their consistency with the diagnostic criteria. Besides, three experienced PET experts (K.T., H.E., Y.T.) also assessed the presence or absence of Aβ deposition through visual inspection of 11C-PiB-PET images [51]. Seven MCI and one AD patient with no evidence of Aβ deposition were excluded from the study, and 24 subjects in the AD-spectrum patients (six MCI and 18 AD) with Aβ positivity and 19 HCs with Aβ negativity were included in the subsequent analyses. No participants had undergone tooth extraction, incision and drainage of a periodontal abscess, or tooth preparation between the PET scans and the oral examinations.
This study was conducted according to the Declaration of Helsinki on Biomedical Studies Involving Human Subjects and approved by the National Institutes for Quantum Science and Technology Certified Review Board (19-034) and the Institutional Review Board of the Tokyo Dental College (967). Written informed consent was obtained from all participants and spouses or other close family members when participants were cognitively impaired. This study was registered with UMIN Clinical Trial Registry (UMIN-CRT; number 000038858).
Remaining teeth and periodontal condition of the participants
A dentist (H.M.) carried out comprehensive dental data acquisition. The dentist counted the number of remaining teeth, including the erupted third molars and residual roots, because the loss of these teeth providing no occlusion contact could also induce the neurodegeneration of Vmes [21]. The periodontal tissues were assessed using pocket probing depth (PPD, mm) and bleeding on probing (BOP, %). PPD is the distance from the base of the periodontal pocket to the gingival margin, which becomes deeper due to gingival swelling and irrevocable loss of connective tissue attachment due to tissue destruction associated with periodontal disease. BOP refers to the finding of bleeding from the periodontal pocket at the time of PPD measurement, and teeth without this finding are considered to be at low risk for periodontal disease progression. PPD was recorded at the worst site around each tooth, and teeth with BOP were recorded as positive. Additionally, the extent of BOP was calculated as a percentage of all teeth. With PPD and BOP, we determined each participant’s biofilm–gingival interface (BGI) index and classified it into the following five levels. BGI-H: biofilm–gingival interface-healthy (PPD≤3 mm, BOP extent scores < 10%) BGI-G: BGI-gingivitis (PPD≤3 mm, BOP extent scores > 10%) P1: BGI-deep lesion/low bleeding (PPD≥4 mm, BOP extent scores < 10%) P2: BGI-deep lesion/moderate bleeding (PPD≥4 mm, BOP extent scores 10% to 50%) P3: BGI-deep lesion/severe bleeding (PPD≥4 mm, BOP extent scores≥50%)
The BGI index is a categorization of periodontal disease developed to reflect an individual’s inflammatory, microbial, and immune response statuses and is assessed using a combination of PPD and BOP [31].
PET and MRI data acquisition
Aβ and tau deposits in all participants were assessed using PET scans with 11C-PiB and 18F-PM-PBB3, respectively, as part of the clinical trials (UMIN-CRT; number 000026385, 000029608, 000030248, and 000043458). The scan protocols follow the procedures described elsewhere [44, 48]. Briefly, each PET scan was performed over 20 min. 11C-PiB PET images were acquired at 50–70 min after the radioligand injection (injected dose, 508.2±125.9 MBq; molar activity, 83.5±20.7 GBq/μmol), and 18F-PM-PBB3 PET images were obtained at 90–110 min after the injection (injected dose, 194.5±55.3 MBq; molar activity, 250.9±89.3 GBq/μmol). PET scans were mainly performed using a Biograph mCT flow system (Siemens Healthcare, Erlangen, Germany), and a Discovery MI (GE Healthcare, Chicago, IL, USA) was also used for some cases (five 11C-PiB scans and three 18F-PM-PBB3 scans in AD-spectrum patients and five 11C-PiB scans in HCs). The acquired PET images were reconstructed using the filtered back projection method with a Hanning filter (4.0 mm full-width at half-maximum). MRI examination was conducted along with PET scans by a 3-tesla scanner (MAGNETOM Verio; Siemens Healthcare, Erlangen, Germany). Structural three-dimensional T1-weighted images were acquired by magnetization prepared using a rapid acquisition gradient-echo sequence (echo time [TE], 1.95 ms; repetition time [TR], 2300 ms; inversion time [TI], 900 ms; field of view [FOV], 250 mm; flip angle, 9°; acquisition matrix, 512×512; and axial slice thickness, 1 mm).
Image analysis
All images were preprocessed using PMOD software (version 4.0, PMOD Technologies Ltd, Zürich, Switzerland), FreeSurfer 6.0 (https://surfer.nmr.mgh.harvard.edu/), and Statistical Parametric Mapping software (SPM12, Wellcome Department of Cognitive Neurology, London, UK). PET images were applied to motion correction and were coregistered with individual anatomical T1-weighted MR images. We generated parametric PET images showing the standardized uptake value ratio (SUVR), which is a ratio of the radioactivity concentration between each voxel and a reference region devoid of target elements such as Aβ and tau deposits. In this study, we defined the reference in the cerebellar cortex. PET images were then spatially normalized to the Montreal Neurologic Institute space using the Diffeomorphic Anatomical Registration Through Exponentiated Lie Algebra algorithm [52]. Normalized PET images were also smoothed with a Gaussian kernel at 8-mm full-width at half maximum in voxel-wise analyses.
We investigated the relationship between oral parameters and Aβ and tau deposits in the brain by conducting a voxel-wise, brain-wide analysis. Oral parameters were introduced as an explanatory variable in the SPM regression model. Then a T-map was generated to explore clusters of voxels showing significant correlations of local SUVR values for Aβ and tau PET tracers with oral parameters. Subsequently,
Statistical analyses
Demographic data, dental status, and VOI-based SUVR values were statistically analyzed using IBM SPSS (version 28, IBM Corp., Armonk, NY, USA) and GraphPad Prism (version 9, GraphPad Software, San Diego, CA, USA). Group comparisons were conducted using the Mann-Whitney U test and Pearson’s chi-squared test (sex and BGI index). Spearman’s rho test was used to evaluate correlations (
RESULTS
Demographic and dental data
The demographics of the participants are shown in Table 1. These values are listed as median [1st quartile, 3rd quartile]. No significant differences in sex, age, and years of schooling were found between the diagnostic groups. The AD-spectrum group showed significant cognitive deterioration as assessed by MMSE and FAB (
Demographic data
Mann–Whitney U test, Pearson’s chi-squared test (sex). Values are listed as median [1st quartile, 3rd quartile]. AD, Alzheimer’s disease; CDR, Clinical Dementia Rating; FAB, frontal assessment battery; HCs, healthy controls; MMSE, Mini-Mental State Examination.
Dental status
Mann–Whitney U test (remaining teeth). Values listed as median [1st quartile, 3rd quartile], Pearson’s chi-squared test (BGI index). AD, Alzheimer’s disease; BGI, biofilm–gingival interface; HCs, healthy controls.
PET assessments of Aβ and tau deposits in the brain
Figure 1 shows representative axial Aβ and tau PET images in HCs and AD-spectrum patients (two individuals in each group). Retentions of both PET tracers were enhanced in extensive cerebral cortical areas of AD-spectrum patients. Additionally, tau PET tracer retention in the brainstem was increased in a subset of AD-spectrum patients with poor oral condition, as exemplified by a case displayed in the rightmost column of Fig. 1, with 10 remaining teeth and BGI index of 4. Statistical parametric maps of the brain-wide group comparison illustrated Aβ and tau deposition in the AD-spectrum group. These data demonstrated significantly enhanced retentions of the two tracers in the frontal cortex, temporo-parietal junction and cingulate cortex of AD-spectrum patients. Furthermore, the tau PET tracer retention was increased in the medial temporal cortex, including the hippocampal formation and inferior and lateral temporal cortices of the AD-spectrum group compared with HCs (
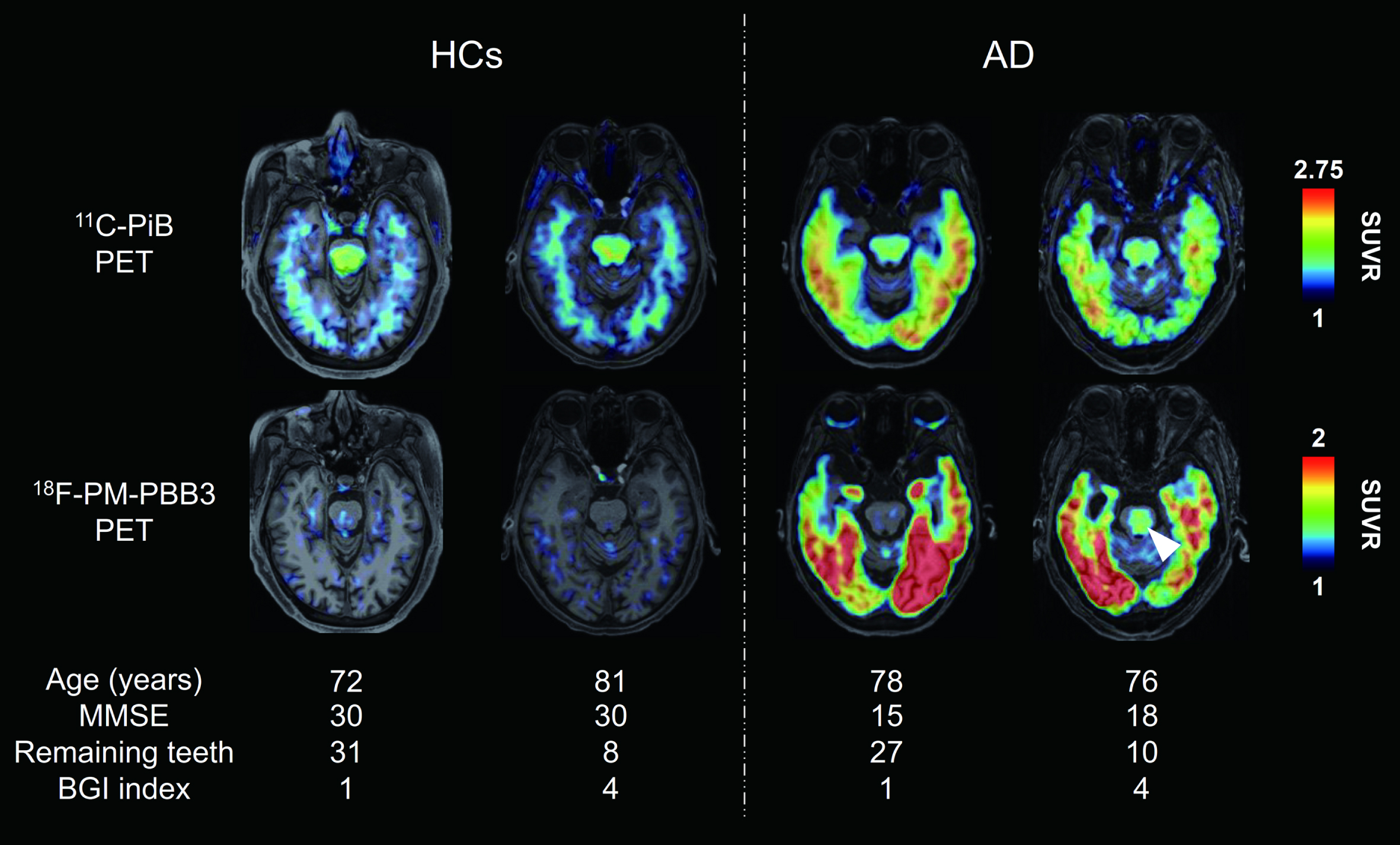
Representative PET images of Aβ and tau in HCs and AD-spectrum patients. Axial 11C-PiB (Aβ) and 18F-PM-PBB3 (tau) PET images of individual participants. PET images were coregistered with the individual anatomical magnetic resonance images. Representative PET images were shown according to participant group, and good and poor oral condition, respectively. White arrowhead indicates tau PET tracer binding in the brainstem. Tau PET tracer retention was increased in the brainstem in a subset of AD-spectrum patients with poor oral parameters compared with HCs. AD, Alzheimer’s disease; BGI, biofilm–gingival interface; HCs, healthy controls; MMSE, Mini Mental State Examination; PET, positron emission tomography; SUVR, standard uptake value ratio.
Correlations between oral parameters and PET tracer retention in the brain
We explored brain areas showing correlations of Aβ or tau depositions with the number of remaining teeth or the BGI index in each diagnostic group. A voxel-based analysis revealed that the number of remaining teeth in AD-spectrum patients showed a negative correlation with tau PET tracer retentions in the brainstem and frontal cortex. The brainstem cluster consisted mainly of the midbrain and pons, including the LC, while the frontal cortex cluster included the bilateral middle frontal, superior frontal, and inferior frontal gyri (see Supplementary Table 2). In contrast, no brain regions displayed associations between Aβ depositions and tooth number in these cases (Fig. 2). Neither Aβ nor tau lesions in AD-spectrum patients were correlated with the severity of periodontal status in the voxel-wise assay. In the brains of HCs, no areas exhibited associations of Aβ and tau PET signals with oral parameters.
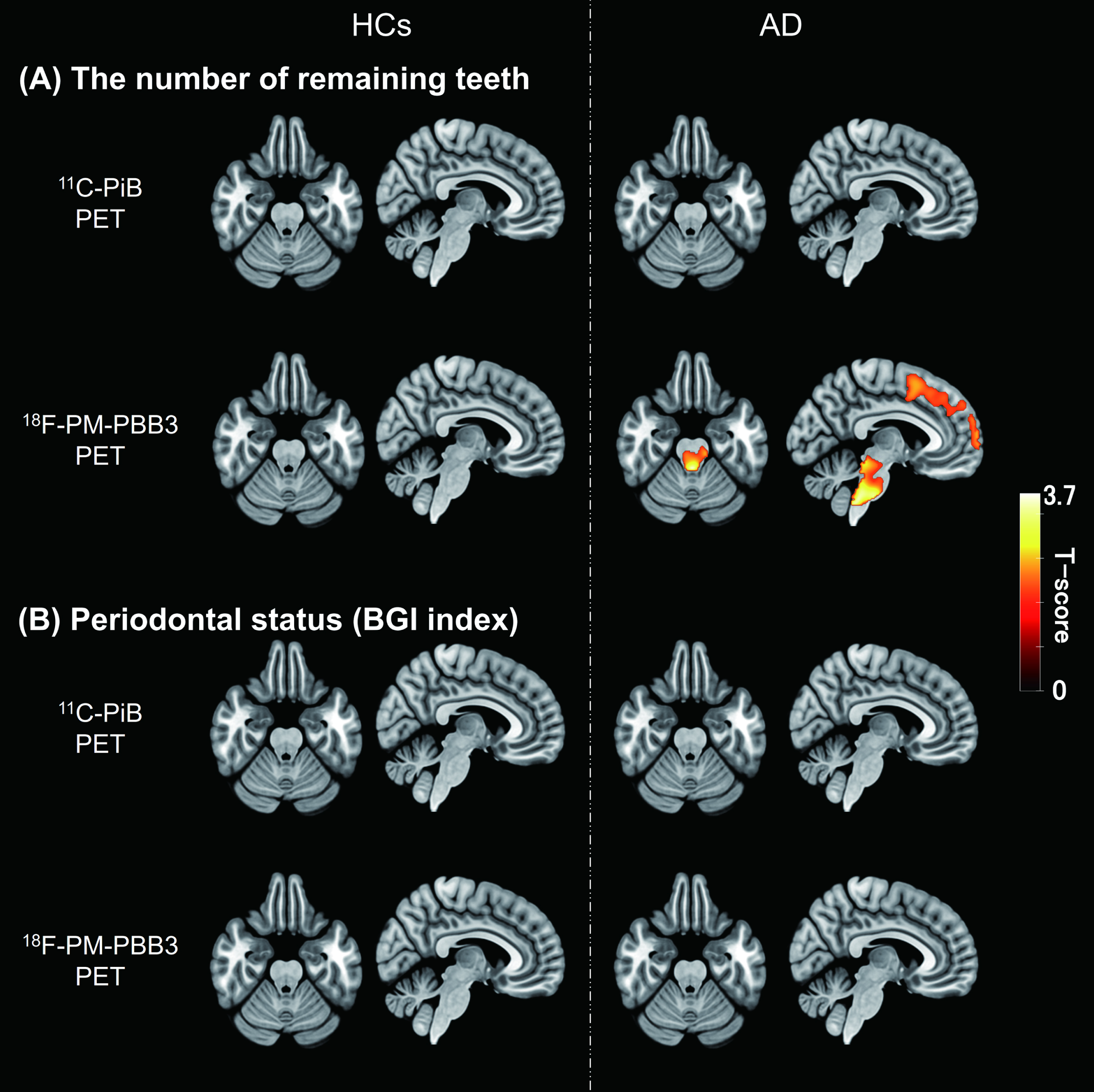
Correlations between oral parameters and PET tracer retention. A) T-score maps of correlation analyses between the number of remaining teeth and each PET tracer retention in HCs and AD-spectrum patients. B) T-score maps of correlation analyses between periodontal status (BGI index) and each PET tracer retention in HCs and AD-spectrum patients. Regions with significant correlations indicated an increase of tau PET tracer accumulation with a decrease in the number of remaining teeth (uncorrected,
VOI-based examination of correlations between the number of remaining teeth and tau depositions
Since the brainstem, including the pons and the medial middle frontal cortex, was highlighted in the voxel-wise investigation, we further assessed the links between tau accumulations in the LC and frontal cortex, and tooth loss by defining VOIs in these anatomical structures, as depicted in Fig. 3A and Supplementary Figure 3, respectively. The group comparison of tau PET SUVR values in the LC did not show significant contrast between HC and AD-spectrum groups (
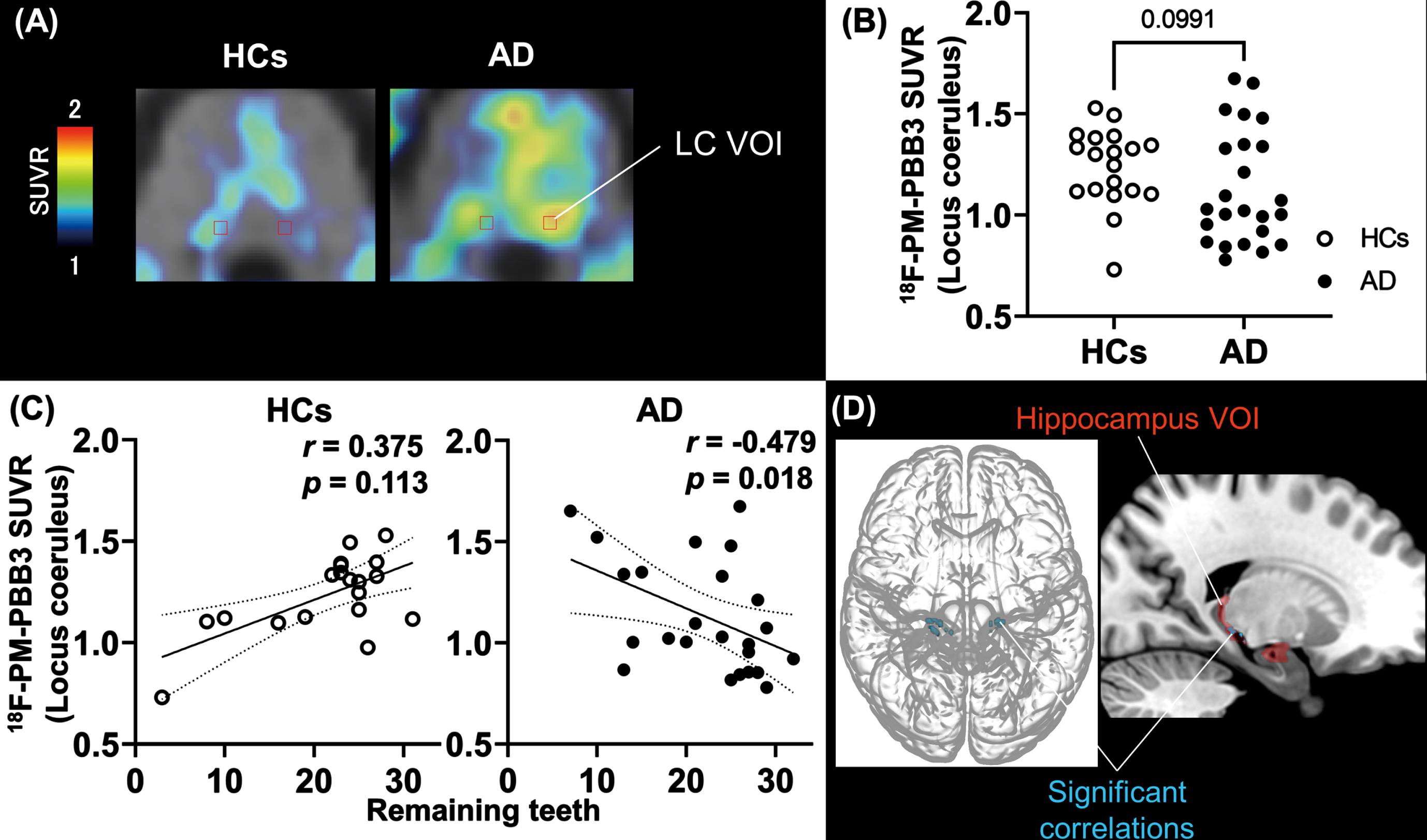
Number of remaining teeth and correlation with tau lesions in the locus coeruleus and hippocampal region. A) Anatomical volume of interest of locus coeruleus (LC-VOI) on normalized PET and MRI images of HCs and AD. B) Group comparison of tau PET SUVR values in LC-VOI between HCs and AD groups. The
Correlations between tau loads in the LC and hippocampus
As tau tracer SUVRs in the LC but not frontal VOI presented a notable correlation with the tooth number in the AD-spectrum group, we sought brain areas with tau burdens closely associated with the LC tau load. SPM analysis with LC SUVRs as explanatory variables picked up hippocampal subregions (Fig. 3D), even after correction for multiple comparisons (Table 3). We also placed a VOI in the hippocampus, but correlations between local SUVR and tooth number were seen in neither AD nor HC group (AD,
Cortical regions correlated with 18F-PM-PBB3 binding in locus coeruleus
Statistical significance threshold was set as
DISCUSSION
To our knowledge, this is the first clinical study to investigate associations between oral parameters and AD pathology using both Aβ and tau PET. Our results demonstrated that the number of remaining teeth in AD-spectrum patients was negatively correlated with tau accumulation in the brainstem (Figs. 2A, 3C) based on both whole-brain and VOI-based analyses. Additional whole-brain analysis revealed a correlation between tau burdens in the LC and hippocampal formation was also found (Table 3, Fig. 3D). The conjunction between the current clinical findings and previous preclinical evidence [21] suggests that tooth loss may enhance tau pathogenesis in AD patients, promoting the spread of tau from the LC to the hippocampal formation. Meanwhile, there was no significant difference in tau PET tracer accumulation in the LC between AD-spectrum patients and HCs (Fig. 3B), nor was a periodontal disease associated with retentions of Aβ and tau tracers.
Besides the lack of associations between Aβ depositions and periodontal disease in HCs and AD-spectrum patients, which were in agreement with findings in a previous cohort study [41], the current work has provided new findings on the absence of links between Aβ pathologies and tooth number in the two diagnostic groups. Correspondingly, previous
Unlike Aβ depositions, tau fibril formations in specific brain areas were tightly related to the tooth number in AD cases. While postmortem study reported no association between the progression of Braak tau stages and the number of remaining teeth [57], an AD model mouse study has shown Aβ and phosphorylated tau accumulation in Vmes before the appearance of AD pathology in the cerebral cortex [58]. Our data imply the contribution of tooth loss to the tau pathogenesis in the brainstem at prodromal and early clinical stages of AD, although there were no correlations of the tooth number with Aβ PET tracer retentions in this region (Supplementary Figure 3). This notion is in line with the enhanced tau accumulations in LC and hippocampal formation as initial events of tau pathogenesis.
Likewise, an AD mouse model exhibited a loss of neurons in the hippocampal region following tooth extraction, presumably as a consequence of neurodegenerative tau pathologies in the brainstem, including Vmes and LC [21]. Both structures are located in the dorsal brainstem, where Aβ and tau lesions could occur in humans [59]. Vmes, the only primary sensory neuron located within the central nervous system [60, 61], receives synaptic and diffusible transmissions of norepinephrine from the LC [62, 63], which may act as a suppressor of inflammatory changes [64]. Microglial activation might be provoked in Vmes by tooth extraction in the presence of Aβ [21, 65] and may expand to the neighboring LC, leading to hyperphosphorylation and aggregation of tau [66, 67] in these regions. Degenerative changes in LC neurons and consequent declines of the norepinephrine release could further accelerate inflammatory microgliosis in the Vmes and LC. Furthermore, the propagation of tau pathologies from the LC of AD patients may be reinforced in the presence of Aβ deposits [68] indirectly by tooth loss.
It should also be noted that norepinephrine neurons in the LC have long and widespread projections to the cerebral cortex, and the degeneration of LC neurons elicited by tooth loss may trigger disruptions of axonal cytoskeletons, accumulations of microtubule-unbound tau species, which are prone to aggregation around the axonal terminals. This process could be boosted in a brain area with a heavy Aβ burden, such as the frontal cortex, but may not necessarily be coordinated with the intensification of fibrillar tau assemblies in the somatodendritic compartment of LC neurons, as indicated by the lack of relationships between tau tracer retentions in the LC and frontal cortex VOIs (Supplementary Figure 4). Additionally, tau accumulation in the frontal cortex may involve tau hyperphosphorylation arising from cerebral hypoperfusion, as supported by numerous animal model studies and clinical investigations [69–71], where tooth loss leads to reduced masticatory function and cerebral blood flow in the frontal cortex [72, 73]. Nevertheless, we found only a tendency to the correlation between the middle frontal tau accumulations and missing teeth in the
By contrast, intimate correlations between tau depositions in the LC and hippocampal formation might stem from the dissemination of tau pathologies through the axonal pathway connecting these two areas. It should also be noted that the number of remaining teeth was correlated with atrophy of several brain areas enriched with tau deposits from an early stage of AD, including the parahippocampal gyrus and olfactory cortex (Supplementary Figure 5), consistent with previous studies [38, 39]. Despite this volumetric finding, there was a lack of direct correlation between tau tracer retentions in these regions and the remaining tooth number (Supplementary Figure 6). It is presumable that robust estimation of this association might be impeded by the radioactivity spillover from adjacent choroid plexus [44] and atrophy-induced partial volume effects on the apparent radio signals in the target area. While the correlation between SUVRs of these regions and tooth number was not significant even after the VOI-based partial volume correction (Supplementary Figure 6), the technical issues would be more precisely addressed by implementing deconvolution-based, voxel-wise partial volume correction as applied elsewhere [74].
In the present study, there were no differences in LC tau PET signals between HCs and AD-spectrum patients (Fig. 3B), which could be related to the fact that tau lesions in the LC could emerge in HCs as pretangles [22], neurofibrillary tangles, and neuropil threads [75]. Declines in the tooth number do not affect this aging-related tau accumulation but may promote the formation and dissemination of tau fibrils in the presence of Aβ deposits in AD. The Aβ species involved in this synergy at an early stage of AD pathologies might be oligomers rather than fibrils, as oligomeric Aβ assemblies could be located in LC neurons [76].
The present study had several limitations. First, the sample size was small, leading to the difficulty in conducting a stratified analysis of MCI and AD to reveal stage-specific insights and focus on different sub-types of cognitive impairments. Second, the timing of tooth loss was unknown. In patients with dementia, the oral environment could deteriorate due to poor hygiene associated with cognitive decline, and the temporality of the association with the onset of AD was unclear. However, because most AD-spectrum patients in this study had mild cognitive deterioration, it is conceivable that deterioration of the oral environment associated with cognitive decline was not substantial. Third, the number of remaining teeth and periodontal health were maintained in both groups compared with people of the same generation [77] and did not significantly deteriorate in the AD group. In particular, most participants had no or only mild periodontal disease (Table 2). Thus, further investigation is still needed to elucidate the impact of periodontal disease. Conversely, the present study allowed us to examine the effect of the number of remaining teeth on brain lesions in a group in which the influence of periodontal disease was controlled. Finally, as a technical limitation, the small size of the LC might make it challenging to assess tracer retention accurately within the resolution of the current PET scanner. VOI-based partial volume correction did not modify the outcomes (Supplementary Figure 7), and validation utilizing neuromelanin-sensitive MRI may be advantageous, analogous to other studies [78, 79] in the future.
Despite these limitations, the first evaluation of oral parameters and AD pathologies using both Aβ and tau PET imaging in humans revealed a correlation between the number of remaining teeth and tau lesions in the brain. The current findings were in line with the hypotheses established by a preclinical study [21], which suggested that preventing tooth loss might be a practical approach to decelerating the progression of AD pathogenesis.
Footnotes
ACKNOWLEDGMENTS
The authors thank all patients and their caregivers for participation in this study, as well as researchers Keisuke Takahata, Kosei Hirata, and Masaki Oya, and radiochemists Kazunori Kawamura and Ming-Rong Zhang, and research ethics advisers at QST. We thank APRINOIA Therapeutics for kindly sharing a precursor of 18F-PM-PBB3. The authors acknowledge support with advice on statical analyses of the present study by Mutsumi Takagiwa at the Laboratory of Mathematics, Tokyo Dental College, and the recruitment of patients by Hitoshi Shinotoh at the Neurology Clinic Chiba, Shunichiro Shinagawa at the Department of Psychiatry, Jikei University School of Medicine, and by Shigeki Hirano at the Department of Neurology, Chiba University.
FUNDING
This study was supported in part by MEXT/JSPS KAKENHI grant numbers 22K19636 to T.K.G., M.H., and T.G., JP16H05324 to M.H., JP18K07543 to H.S., and JP22K15776 to K.T., by AMED under grant numbers JP18dm0207018, JP19dm0207072, JP18dk0207026 to M.H., by J.S.T. grant numbers JPMJMS2024 to M.H., by the Private University Research Branding Project from MEXT of Japan to Tokyo Dental College to H.M., and by Biogen Idec Inc. and APRINOIA Therapeutics.
CONFLICT OF INTEREST
Hitoshi Shimada and Makoto Higuchi hold patents on compounds related to the present report (JP 5422782/EP 12 884 742.3/CA2894994/HK1208672).
DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author on reasonable request.
