Abstract
Background:
The negative effects of periodontitis on systemic diseases, including diabetes, cardiovascular diseases, and Alzheimer’s disease (AD), have been widely described.
Objective:
This systematic review aimed to gather the current understanding of the pathophysiological mechanisms linking periodontitis to AD.
Methods:
An electronic systematic search of the PubMed/MEDLINE, Scopus, and Embase databases was performed using the following PECO question: How can periodontitis or periodontal bacteria influence Alzheimer’s disease features?”. Only preclinical studies exploring the biological links between periodontitis and AD pathology were included. This study was registered at the International Prospective Register of Systematic Reviews (PROSPERO), and the Syrcle and Camarades protocols were used to assess the risk of bias.
Results:
After a systematic screening of titles and abstracts (
Conclusions:
Systemic inflammation and brain metastatic infections induced by periodontal pathogens contribute to neuroinflammation, amyloidosis, and tau phosphorylation, leading to brain damage and subsequent cognitive impairment.
Keywords
INTRODUCTION
Alzheimer’s disease (AD) is a neurodegenerative disorder that affects more than 35 million individuals worldwide and more than 10 million individuals in Europe [1]. AD is a multifactorial disease characterized by early synaptic and neuronal dysfunction, which induces amnesia, cognitive impairment, and subsequently dementia [2]. The AD brain physiopathology involves lesions characterized by the presence of extracellular insoluble senile plaques formed by amyloid-β (Aβ) peptide and the presence of intraneuronal neurofibrillary tangles (NFTs) formed by hyperphosphorylation of the tau protein (p-Tau) (tauopathy) [1, 4]. Moreover, neuroinflammation is considered a key factor in both the onset [5] and progression of AD [6].
Periodontal disease (PD) is a chronic bacterial inflammatory disease [7, 8] that affects 20–50% of the overall population [9]. The disease is characterized by colonization by gram-negative bacteria, which induce local inflammation inside the periodontium, with progressive destruction of the supporting tissues around the teeth [10–12]. Second, owing to mastication and tooth brushing [13], periodontal bacteria or their byproducts can be released into the bloodstream, resulting in transient or prolonged bacteremia that influences systemic diseases or conditions, such as cardiovascular pathology or diabetes [14]. Furthermore, recent systematic reviews of clinical studies have shown a positive association between periodontitis and AD [15–17]. On the one hand, the presence of PD is associated with the presence of AD [15], while on the other, patients with dementia show significantly worse clinical periodontal variables than healthy individuals do [16]. Additionally, Nadim et al. [17] provided evidence that PD increased the risk of incident dementia and speculated that a 50% reduction in the current prevalence of PD in the population could save 850,000 patients from dementia worldwide while preventing and treating PD could contribute to controlling the global dementia epidemic.
Inflammation is a risk factor shared by these two diseases, influencing their onset and progression [18, 19]. Indeed, in AD, the brain accumulation of Aβ peptide activates glial cells, lymphocytes, and macrophages, which release inflammatory mediators such as cytokines, chemokines, neurotransmitters, and reactive oxygen species (ROS) [20, 21]. In addition, the tauopathy induces the synthesis of pro-inflammatory cytokines as tumor necrosis factor (TNF-
However, the pathophysiological mechanisms that link AD and periodontitis remain poorly investigated [30, 31]. Therefore, the present systematic review aims to explore and gather the identified mechanisms linking the PD to AD pathology, throughout
MATERIALS AND METHODS
Study design
The present study was designed as a systematic review focusing on the relationship between AD and PD. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA-SR) checklist and guidelines were followed. This study was initially registered as CRD42022257306 in the Prospective International Registry of Systematic Reviews (PROSPERO).
Focus question and search strategy
The focus question and PECO framework (P: population, E: exposure, C: control, O: outcome) were used in order to answer the following research question: “How periodontitis or periodontal bacteria can influence Alzheimer’s disease features?” This question was addressed according to the following criteria.
Population: Animals with AD pathology Exposure: Exposed to periodontitis or periodontal bacteria Control: Not exposed to periodontitis or periodontal bacteria Outcome: In the brain: neuroinflammation, expression of pro-inflammatory cytokines (TNF- In the blood: CRP and pro-inflammatory cytokines (TNF- Cognitive behavior.
The search strategy (Fig. 1) consisted of each database’s controlled vocabulary (MeSH terms) and free keywords. Supplementary Table 1 presents the search algorithm. An electronic search of the literature was performed using PubMed/MEDLINE, Scopus, and Embase databases. Selected publications were collected using the Mendeley Reference Manager software, and the Cochrane tool (COVIDENCE) was used for screening and data extraction.
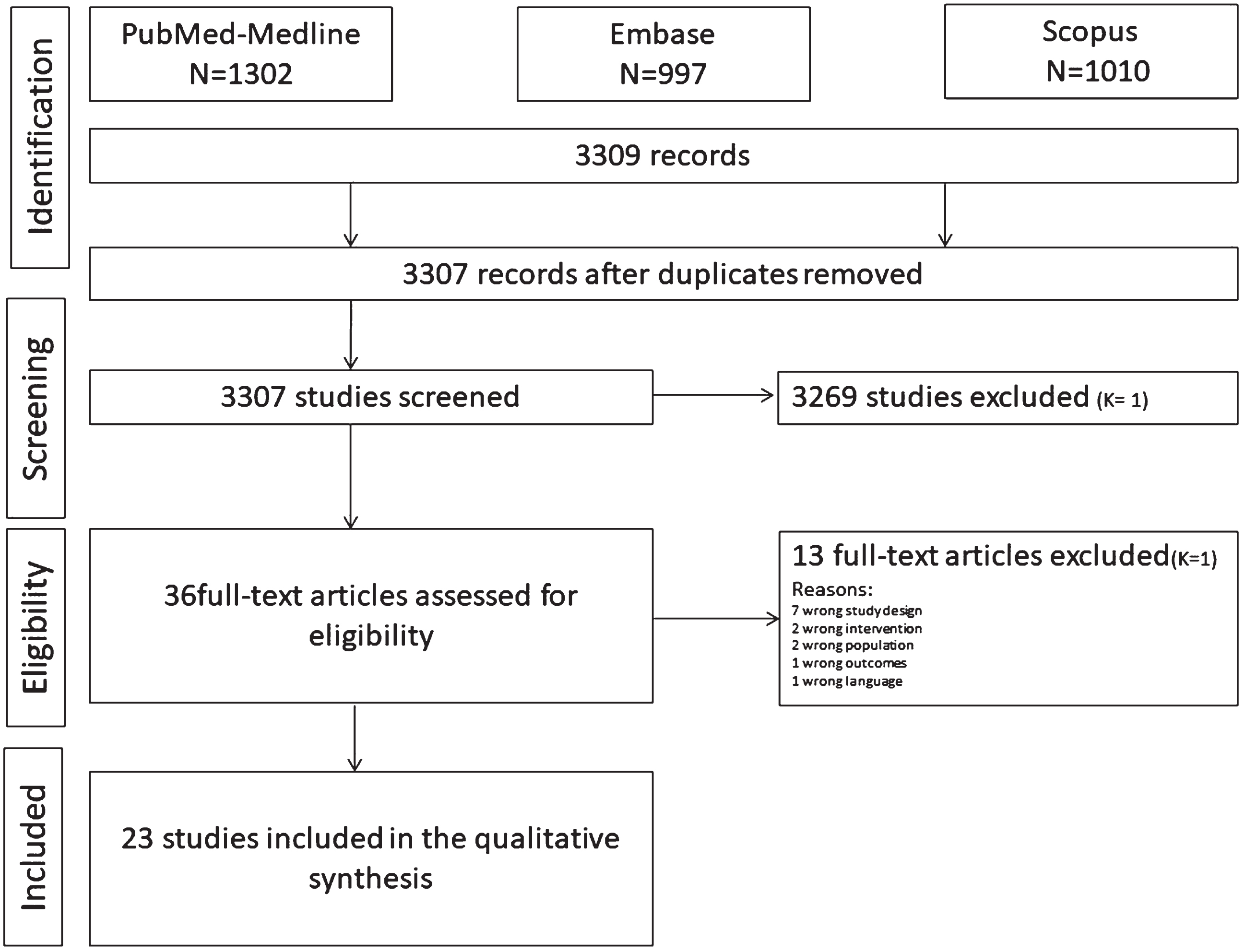
Search strategy. Diagram of article selection process.
Study characteristics
AD, Alzheimer disease; Aβ, amyloid-β peptide; AβPP, amyloid-β protein precursor; BA, butyric acid; BBB, blood-brain barrier; Cat, cathepsin; CFU, colony forming unit; CSF, cerebrospinal fluid;
Study selection and data extraction
Procedure for study selection
Two independent blinded reviewers (LS and YA) performed study selection using the online Cochrane platform (COVIDENCE). The reviewers (LS, YA,) first screened titles, then abstracts, and finally full texts of the 3307 articles identified by electronic and manual searches. Agreement between examiners was generated using Cohen’s kappa (κ) coefficient at each level of selection. Records were excluded only to enhance sensitivity if both reviewers excluded them based on their titles. Disagreements were resolved by discussion with a third reviewer (FL) resolved the issue.
Inclusion and exclusion criteria
The inclusion criteria were the following: Animal studies (preclinical studies) Species: mice and rats Studies focusing on the relationship between periodontitis or periodontal bacteria and AD features. The studied animals must suffer from both AD pathology and periodontitis The experimental animals must display one or more of the following AD characteristics: Cognitive impairment Pro-inflammatory cytokines in the blood or brain (IL-1β, IL-6, and TNF- Neurodegeneration Microgliosis and astrogliosis Aβ peptides levels in blood or in the brain NFTs and tau protein phosphorylation in the extracellular area of the brain Experimental animals must display periodontitis manifested through a ligature or by oral or systemic exposure to: Periodontal bacteria or periodontal genetic materials (DNA, RNA) Byproducts such as lipopolyssacharides (LPS) and endotoxins The exclusion criteria were the following: Human study Review articles Studies in other species Study not involving AD pathology and periodontitis Any outcomes not related to both AD pathology and periodontitis Animals with systemic disease (cardiovascular disease, diabetes) Use of medical drugs (antibiotics)
Methods for data extraction
All selected studies were extracted from the three main databases and sorted according to each database using Mendeley software, a bibliographic tool. The data were then sent from Mendeley to the COVIDENCE platform for extraction. All information extracted from the articles (see the list below) was grouped, filled in, and checked by two authors to verify inconsistencies and ensure data accuracy using Microsoft Office Excel 2022.
In the case of missing data, the authors were contacted by e-mail to collect the required information.
Data to be extracted
Authors and year of publication Aim/hypothesis Studied animals (specie, age, gender, background) Study design (sample size, treatment groups) Methods for AD features assessment and AD model induction Methods for periodontitis induction and characterization (intervention) Effects of periodontitis on both AD-related systemic inflammation and neuroinflammation Consequences of systemic and brain inflammation on animals’ cognitive behavior Conclusion of the authors
Statistical analysis
The level of agreement between the reviewers was calculated using the Cohen κ coefficient at each level of selection. The Kappa test was performed directly using the Covidence tool. The selection was performed according to inclusion and exclusion criteria.
Assessment of study quality
To assess the risk of bias and consistency of the studies, two reviewers rated each article independently based on its quality by 2 reviewers, using the criteria outlined in the Systematic Review Centre for Laboratory Animal Experimentation Office of Health Assessment (SYRCLE) bias tool. This tool contains 10 entries related to selection, performance, detection, attrition, reporting, and other biases (Supplementary Table 2).
Methodological quality assessment was also performed using the collaborative approach to meta-analysis and review of Animal Data from Experimental Studies (CAMARADES) checklist items. This checklist comprised 22 independent items (see Supplementary Table 3). To evaluate study quality, scores were calculated as 1 (V = fulfilling the criterion) or 0 (N = not fulfilling the criterion) for each item on the checklist. After providing scores for study quality, the scores of each study were summed (Supplementary Table 3).
RESULTS
Search and selection
The search strategy and characteristics of the included studies are shown in Fig. 1 and Table 1. The Embase, Scopus, and PubMed search strategies resulted in 3,307 articles. After discarding duplicates, two calibrate reviewers (YA and LS) screened the titles, and 3,271 articles were excluded by the first (LS) and second reviewers (YA), resulting in a Cohen κ coefficient of 1. Thirty-six titles were selected for the abstract reading, of which 13 were excluded (k = 1). After the full-text assessment, no additional articles were excluded. Ultimately, both reviewers agreed to include 23 articles in the systematic review. Studies were excluded, and the reasons for exclusion are shown in Fig. 1.
Assessment of study quality
Using the CAMARADES tool, 15 studies showed a low risk of bias, two exhibited a moderate risk of bias, and six displayed a high risk of bias.
Regarding the SYCRLE assessment, 15 studies were associated with a low risk of bias, 1 study presented a moderate risk of bias, and 7 studies demonstrated a high risk of bias.
For both tools, most studies (65.3%) had a low risk of bias.
Study characteristics
The relationship between AD pathology and periodontitis was explored by evaluating the effects of periodontal inflammation and periodontal bacteria on systemic inflammation (
Descriptions of the included articles are summarized in Table 1. The effects of periodontitis on systemic inflammation (SI), brain tissue damage, and their consequences on cognitive behavior are described in Table 2.
Periodontitis assessment
Periodontitis was established using experimental oral infection models ( Experimental oral infection models
Oral infection models were established either by gingival injection of periodontal bacteria ( Systemic injection of periodontal bacteria or their byproducts
The injections of periodontal bacteria
Alzheimer’s disease induction models and assessment
To establish an AD model, animals were either genetically modified (
AD pathology was assessed using cognitive function tests ( Cognitive function
The cognitive function tests included the open field test (
Neuroinflammation
The neuroinflammatory state was assessed by quantification of pro-inflammatory molecules expression in the brain, such as TNF- Blood quantification
The concentration of Aβ peptides and the ratio of Aβ1 - 40/1 -42 were investigated in the serum ( Brain tissue
Aβ peptide concentration was investigated in the hippocampal neurons, cerebral cortex, and periventricular area (
Effect of periodontitis on AD physiopathology
The effects of periodontal inflammation or bacteria on the increase in systemic inflammation and brain tissue damage and the impairment of cognitive behavior were investigated in 13, 21, and six studies, respectively. Considering the heterogeneity of the models and data, a meta-analysis could not be performed. The study results are presented in Table 2. Effect on systemic inflammation
Effect of periodontitis on systemic inflammation, brain inflammation, and cognitive behavior: results, proposed mechanisms, and conclusion
AD, Alzheimer’s disease; Aβ, amyloid-β; AβPP, amyloid-β protein precursor; BBB, blood-brain barrier; Cat, cathepsin; CFU, colony forming unit; CNS, central nervous system; CRP, C-reactive protein; DG, dentate gyrus; ER, endoplasmic reticulum; ERK, extracellular signal-regulated kinase; exRNAs, extracellular RNAs; IL, interleukin; IFN, interferon; LPS, lipopolysaccharide; MMP, matrix metalloproteinases; NF-κB, nuclear factor-κB; NFTs, neurofibrillary tangles; OMVs, outer membrane vesicles; PAM, plaque-associated microglia; PAR, protease-activated receptor; PD, periodontal disease; Pg,
Gingival injection of
Hence, the authors concluded that periodontitis infection or periodontal pathogens could increase AD-specific and-nonspecific biomarkers of systemic inflammation. Effect on brain tissue inflammation/damage
The effects of periodontitis and periodontal pathogens on the brain tissue involve neuroinflammation and neuronal cell damage. Periodontal infections are also associated with hippocampal microgliosis and astrogliosis [35–37, 52]. Indeed, significantly increased expression of interleukins (IL-6,-17) [35, 52], TNF- Effect on cognitive behavior
Chronic systemic exposure to
DISCUSSION
The present systematic review of preclinical studies raises hypotheses regarding the pathophysiological mechanisms linking periodontitis-induced systemic inflammation and metastatic infection with AD progression.
Indeed, it has been widely demonstrated that Gram-negative periodontal bacteria and their byproducts can invade the blood circulation and subsequently increase SI and/or colonize vulnerable sites called
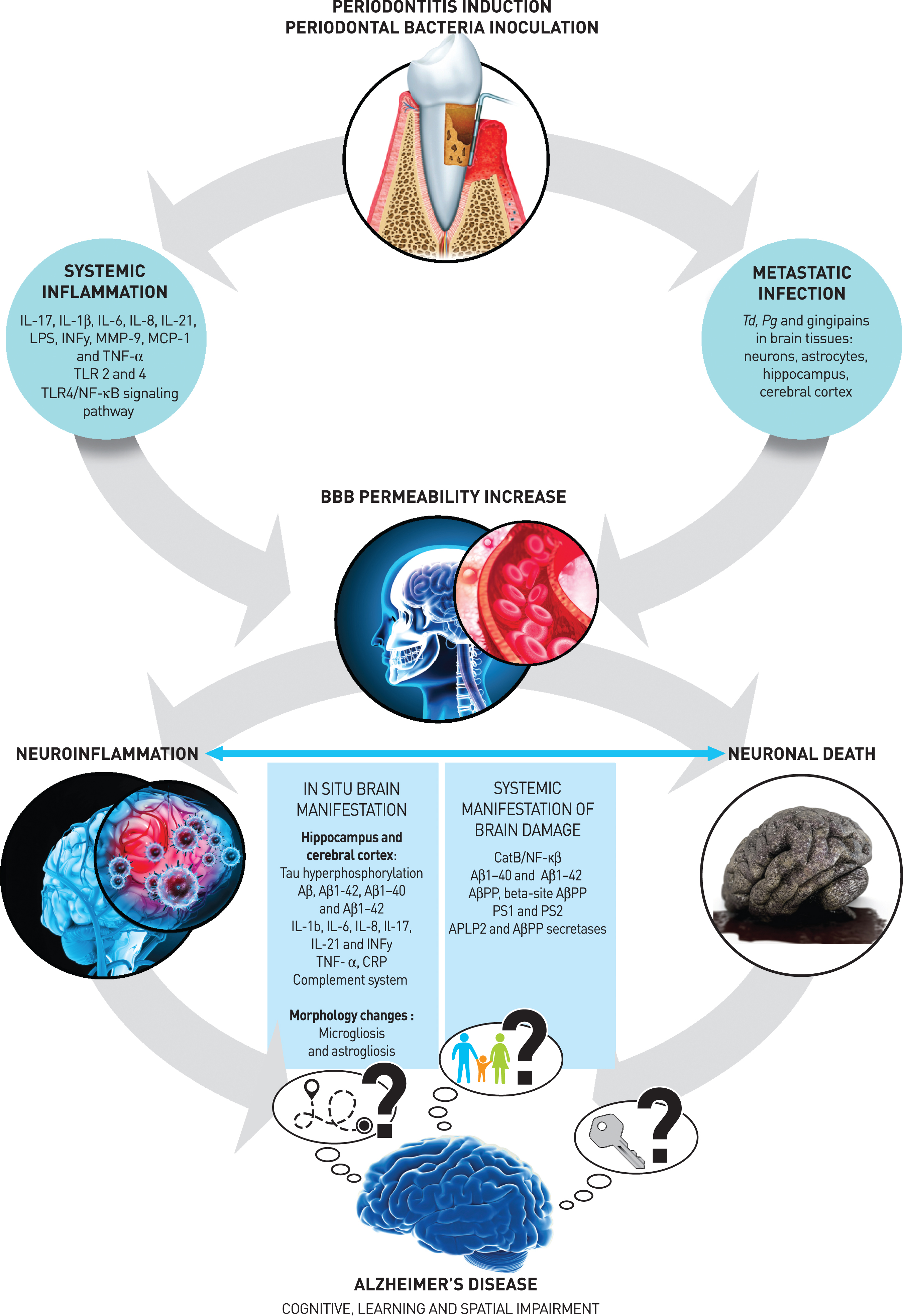
Hypothetical physiopathological mechanisms linking periodontitis and Alzheimer’s disease.
Effect of the periodontal pathogen on blood-brain barrier permeability
Several studies included in the present review have demonstrated in preclinical models that SI [33–35, 53] and/or brain MI [49–51] occur after periodontal induction or periodontal bacterial inoculation. Two studies demonstrated that both SI and MI induce the permeability of the blood-brain barrier (BBB), namely through a decrease in claudin 5 levels [46] and the destruction of collagen and fibronectin [39]. The resulting destruction of the components of the basal membrane of brain endothelial cells enhances vascular permeability [39] and therefore facilitates the penetration of other bacteria.
Effect of the periodontal pathogen on in situ brain manifestation
This review also highlights how periodontal pathogens may lead to in situ brain manifestations at the cellular and molecular levels. Periodontal bacteria (
Neuroinflammation (1)
The molecular mechanisms of periodontitis-related neuroinflammation are linked to the activation of the NF-κB pathway, the involvement of CatB, the secretion of pro-inflammatory cytokines (as TNF-
LPS [45], gingipains [39, 51], capsules, and fimbriae of
CatB is another molecule that plays a role in periodontitis-related neuroinflammation. Indeed, this molecule is involved in the NF-κB upregulation and further increases the expression of pro-inflammatory cytokines inside microglia and hippocampus [47] and that of RAGE in cerebral endothelial cells [52]. Additionally, Han et al. [36] suggested that the periopathogen could directly increase the brain expression of TNF (without the activation of NF-kB or CatB). Notably, extracellular RNAs located in the outer membrane vesicles of the periodontal pathogen are able to increase the brain expression of TNF-
Finally, neuroinflammation-related periodontitis can also be explained by complement system activation [50]. Indeed,
Amyloidogenesis (2)
Several studies have demonstrated that periodontitis authors [35, 53] that periodontitis infection can contribute to brain amyloidogenesis. Chronic systemic exposure to
Tau protein (3)
Finally, it was demonstrated that the presence of periodontal bacteria or virulence factors [38, 43] induces hyperphosphorylation of the tau protein (p-Tau) [38, 44] and the formation of NFTs. Consequently, microglial changes (microgliosis and astrogliosis) [34, 42–45] and neuronal death [40, 50] lead to the progression of AD. The suggested molecular mechanism is that
Systemic manifestation of brain damage
The
Effect of the periodontal pathogen on cognitive behavior
In addition to the above-mentioned biological and biomolecular findings, this systematic review also supports that chronic systemic exposure to
This systematic review had some limitations. A meta-analysis was not possible because of the heterogeneity of the studies, which had different aims and analyzed different mechanisms/markers and different outcomes. Therefore, the establishment of standardized and validated AD models in future studies would yield more consistent conclusions regarding the influence of periodontitis on AD progression. Moreover, the present data remain preclinical, and further clinical data are required to validate this hypothesis regarding the effects of periodontitis and periodontal treatment on AD progression. However, the preclinical study analyses in the present systematic review have the advantage of excluding confounding factors, such as diabetes and tobacco, which are often encountered in human clinical data.
Conclusion
Within the limits of preclinical models, the present systematic review contributes to the understanding of the potential mechanisms linking periodontal bacteria to AD. Systemic inflammation and brain metastatic infections induced by periodontal pathogens contribute to neuroinflammation, amyloidosis, and tau phosphorylation, leading to brain damage and subsequent cognitive impairment.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available within the article and/or its supplementary material.
