Abstract
A decade has passed since we published a comprehensive review in this journal addressing the topic of promoting successful cognitive aging, making this a good time to take stock of the field. Because there have been limited large-scale, randomized controlled trials, especially following individuals from middle age to late life, some experts have questioned whether recommendations can be legitimately offered about reducing the risk of cognitive decline and dementia. Despite uncertainties, clinicians often need to at least make provisional recommendations to patients based on the highest quality data available. Converging lines of evidence from epidemiological/cohort studies, animal/basic science studies, human proof-of-concept studies, and human intervention studies can provide guidance, highlighting strategies for enhancing cognitive reserve and preventing loss of cognitive capacity. Many of the suggestions made in 2010 have been supported by additional research. Importantly, there is a growing consensus among major health organizations about recommendations to mitigate cognitive decline and promote healthy cognitive aging. Regular physical activity and treatment of cardiovascular risk factors have been supported by all of these organizations. Most organizations have also embraced cognitively stimulating activities, a heart-healthy diet, smoking cessation, and countering metabolic syndrome. Other behaviors like regular social engagement, limiting alcohol use, stress management, getting adequate sleep, avoiding anticholinergic medications, addressing sensory deficits, and protecting the brain against physical and toxic damage also have been endorsed, although less consistently. In this update, we review the evidence for each of these recommendations and offer practical advice about behavior-change techniques to help patients adopt brain-healthy behaviors.
Keywords
INTRODUCTION
In 2010, we published an article in this journal entitled, “Promoting successful cognitive aging: a comprehensive review” [1]. A decade has passed, making this a good time to take stock of the field, which has garnered increasing interest. The number of articles on cognitive/brain health and dementia risk reduction has grown substantially. A Pubmed search of papers linked to relevant key words, including “brain health,” “healthy brain aging,” “promoting cognitive health,” “preventing cognitive decline,” “dementia risk reduction,” “dementia prevention,” and “Alzheimer prevention,” went from 917 published in 2001-2010 to 6,243 published in 2011-2020. Many of the suggestions made in 2010 have been supported by additional research and have been increasingly embraced clinically. Several major health organizations have published brain health recommendations based on research studies investigating how various behaviors and lifestyle factors affect the risk of developing cognitive decline and dementia [2–7]. In this update, we will summarize the findings of past and recent research, present them in an accessible format, and offer practical advice about helping patients to adopt brain-healthy behaviors.
An aging population
According to a 2019 United Nations report, the population over the age of 60 is growing faster than other age groups [8]. Average life expectancy has dramatically increased in the last several decades and is predicted to continue increasing on a similar trajectory. In 2019, the global life expectancy at birth reached 72.6 years, an increase of more than eight years since 1990. By 2050, the global life expectancy is predicted to reach 77.1 years [8]. As of 2019, there were an estimated one billion people over the age of 60 globally. This number is expected to grow to 2.1 billion by 2050, and to 3.1 billion by 2100 [8]. Already in Europe, more than a quarter of the population (190 million) is older than age 60 [8]. In the United States (U.S.), the older population is also growing rapidly, as 75 million Baby Boomers (the cohort born between 1946 and 1964) turn 65. Currently, 53 million people over the age of 65 live in the U.S. In the next decade, that number is projected to grow to 74 million [8]. Members of this age group have been particularly concerned about maximizing longevity and quality of life. The prospect of losing memory and independence is among the most feared aspects of aging. In fact, studies have suggested that more than half of adults over the age of 65 have concerns about their memory [9–15].
The many costs of dementia
Dementia is a general term that describes a set of symptoms, including difficulties with thinking and memory, which interfere with the ability to perform activities of daily living [13, 16]. The most common cause of dementia, Alzheimer’s disease (AD), is associated with the abnormal accumulation of the proteins amyloid-β (Aβ) and tau in the brain [13]. Relatedly, mild cognitive impairment (MCI) is a term used to describe the intermediate stage between normal cognitive aging and dementia. Activities of daily living remain intact with MCI, but the brain is often already undergoing early changes due to Alzheimer’s disease and related dementias (ADRDs) [17, 18]. ADRDs are the fifth-leading cause of disability and death among older individuals globally [19]. There are an estimated 50 million people in the world who suffer from ADRDs, and this number is expected to more than triple to 152 million by 2050 [13]. Additionally, 15–20% of adults over 65 have MCI due to ADRDs [13, 18]. It is generally acknowledged that ADRDs are some of the costliest diseases to society, due to many direct and indirect costs associated with caring for patients [20]. Compared to their peers, patients with dementia have worse overall health, increased utilization of healthcare services, higher out-of-pocket healthcare spending, shorter life expectancy, and greater likelihood of dying in hospitals [21–27]. Similarly, the health outcomes and general wellbeing of caregivers and families of patients with ADRDs are notably worse than their age-matched peers, with higher rates of anxiety and depression, worse sleep quality, and poorer overall quality of life [25, 28–30]. The financial impact of cognitive decline on health systems is largest in the transition from MCI to dementia [31, 32]. Projection models demonstrate that delaying the onset of dementia by five years can reduce its prevalence by 50%, substantially decreasing financial and social burdens on patients, families, and healthcare systems [33, 34]. Specifically, some economic models demonstrate that delaying the transition from MCI to dementia by five years could result in a cost-savings of more than $500,000 per patient [32]. Therefore, it is imperative to develop clinical care models that focus on stabilizing patients with MCI and reducing the risk of dementia for healthy older adults. In the aggregate, even small effects on preserving cognitive abilities may have a significant impact on society.
PROMOTING SUCCESSFUL COGNITIVE AGING
Many theories about normal aging have suggested that aging is associated with the risk of declining neurophysiological functions, which often leads to reductions in cognitive processing and capacity [35]. As we [1] and others [36, 37] have suggested, the promotion of healthy cognitive aging can be linked to the dual goals of 1) preventing loss of and 2) facilitating enhancement of brain and cognitive reserve. Brain reserve refers to differences in the “hardware” of the brain, including the number of neurons and synapses, which allow brains to withstand a greater burden of disease or injury before cognitive function is affected [38]. Cognitive reserve includes the ability to use brain networks more proficiently in response to every-day cognitive, emotional, and functional demands, as well as the capacity to utilize alternative cognitive strategies or neural networks in response to cerebral injury or decline [38].
A simple model of brain reserve and deterioration suggests that, when the number of healthy, functioning neurons or their connections falls below a critical level or threshold, individuals manifest symptoms of cognitive impairment and, eventually, dementia [39]. Figure 1 illustrates the trajectory of cognitive decline in individuals with neurodegenerative disease. Additional cerebral insults (e.g., due to strokes) would shift the theoretical trajectory of cognitive decline to the left, causing patients to cross the threshold for dementia earlier in life. Conversely, boosting cognitive or brain reserve (e.g., through education or cognitively stimulating activities) would shift this theoretical curve to the right, allowing patients to cross this threshold later in life.
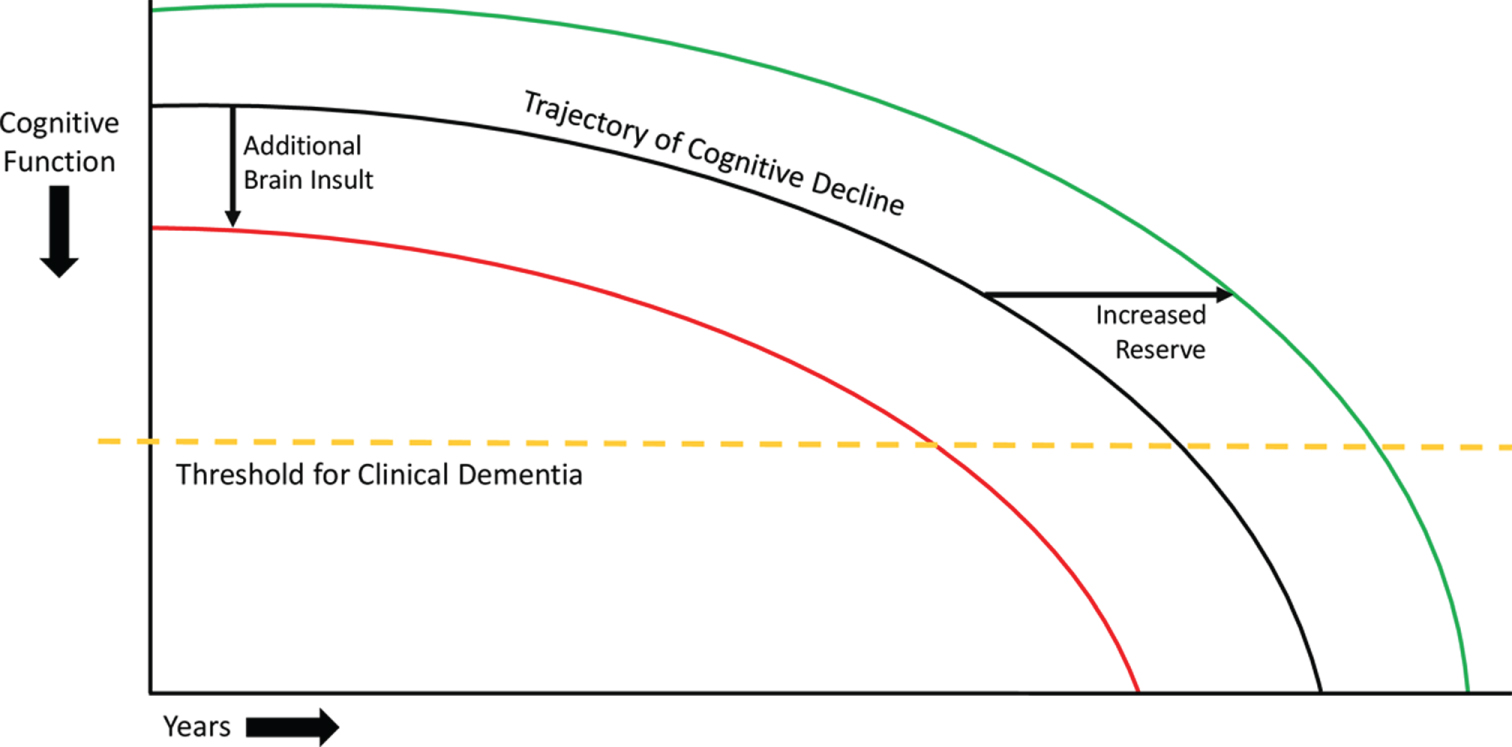
Model of cognitive decline. The black line represents the trajectory of cognitive decline due to neurodegenerative disease. The yellow dashed line represents the threshold for clinical dementia, i.e., the inability to manage activities of daily living. The red line represents the impact of additional injuries to the brain, which can decrease brain reserve and cause a leftward shift of the trajectory of cognitive decline, leading patients to cross the threshold for clinical dementia earlier. The green line represents the effect of increased cognitive and brain reserve, which can cause a rightward shift in the trajectory of cognitive decline, leading patients to cross the threshold for clinical dementia later. These principles involving theoretical shifts in the trajectory of cognitive decline also apply to adults without neurodegenerative disease, though the initial downward trajectory is much less steep.
Blue Ribbon Panels: inadequate evidence that interventions can reduce dementia risk
Over the past decade, several systematic reviews by prestigious academic and expert governmental panels have concluded that there is inadequate high-quality evidence that the risk of cognitive decline and dementia can be modified. For example, in 2010, a 15-member panel at the National Institutes of Health Consensus Development Conference released a statement on preventing AD and cognitive decline, saying that firm conclusions could not be drawn about the association of any potential modifiable risk factor with cognitive decline or AD [40]. In 2011, an eight-member panel for the U.S. Centers for Disease Control completed a systematic review of intervention trials investigating the effect of physical activity and exercise on cognition in older adults. This review acknowledged some positive results but concluded that there was insufficient evidence that physical activity improved cognition in older adults [41]. More recently, in 2017, the Minnesota Evidence-based Practice Center conducted systematic reviews on the effects of physical activity [42], cognitive training [43], and prescription and over-the-counter medications [44] on cognitive decline and dementia; it concluded that no interventions approached the evidence level necessary to make clinical recommendations. In general, these review panels call for large-scale, randomized control trials (RCTs) to address the issue. Ideally investigators would initiate RCT interventions during midlife and follow participants over decades to determine the impact on developing cognitive deterioration and dementia in late life. Unfortunately, such studies would be expensive and impractical to carry out, and financial support for such endeavors seems unlikely. A critical question is whether clinicians or public health officials should wait for the results of large-scale RCTs like these to become available before advising patients or the wider public on behavioral and lifestyle changes to reduce dementia risk
Converging evidence
Despite uncertainties and incomplete information regarding the management of many health risks and medical conditions, clinicians often need to address challenging issues currently facing their patients. Most clinicians are not in a position to simply tell patients that they should come back in 15–20 years when we may have more definitive answers. Rather, clinicians are obligated to offer at least provisional recommendations to patients and policy makers based on the highest quality of evidence available. Healthcare providers can also play an important role in identifying and treating conditions that reduce brain reserve and cognitive capacity, including cardiovascular risk factors, metabolic syndrome, sleep disorders, mood disorders, and side effects from medication. They are well-positioned to serve as advocates for brain health and wellbeing.
Epidemiological evidence suggests that up to 35% of ADRD cases may be due to modifiable risk factors [4], and that healthy lifestyles can offset genetic risk of developing ADRDs [45]. Thus, encouraging ways to promote brain health and reduce risk of cognitive decline is particularly important, especially given the absence of disease-modifying treatments for neurodegenerative diseases.
As detailed in our 2010 review, there are several different ways to evaluate whether a proposed factor or intervention may have an impact on healthy cognitive aging. Four major lines of evidence have been used: 1) epidemiological/cohort studies, 2) animal/basic science studies, 3) human proof-of-concept studies, and 4) human intervention studies. Often, the strongest recommendations are a reflection of converging lines of evidence.
Advantages and limitations of different lines of evidence
Table modified from Daffner 2010 [1].
Consensus recommendations
Based on the available lines of evidence, there is growing consensus among major health organizations that certain healthy behaviors and lifestyles can mitigate cognitive decline and promote successful cognitive aging [2–7]. Regular physical activity and treatment of cardiovascular risk factors have been supported by all of these organizations. Most organizations have also embraced cognitively stimulating activities, a heart healthy diet, countering metabolic syndrome, and smoking cessation. Other behaviors such as regular social engagement, limiting alcohol use, stress management, getting adequate sleep, avoiding medications with anticholinergic properties, addressing sensory deficits, and protecting the brain against physical and toxic damage have received less consistent endorsement. Research has provided evidence that adoption of these brain-healthy behaviors can improve cognition, reduce the risk of brain injury or dysfunction, and augment overall wellbeing and quality of life for cognitively normal older adults. The recommendations of several major organizations are outlined in Table 2.
Recommendations from major health organizations for maintaining brain health and mitigating cognitive decline
For each major recommendation for maintaining brain health and mitigating cognitive decline identified, we have provided a narrative summary of the best available evidence. We relied on high-caliber systematic reviews and meta-analyses, especially of RCTs and long-term observational studies, preferably that included assessments of publication bias and quality of methodology and outcome data. These reviews and meta-analyses were identified by searching for relevant key terms in the PubMed, EMBASE, and PsycInfo databases. We scrutinized individual studies that were highlighted in these reviews and meta-analyses or were identified through additional searches of the same databases for the most recent publications on the subject. We also conducted systematic searches for articles on relevant basic science/animal investigations and human proof-of-concept studies. Our major aim was to synthesize converging evidence from epidemiological, basic science/animal, proof-of-concept, and RCT studies, with the goal of producing a coherent narrative that would be readily accessible to clinicians and researchers in the field.
Physical activity
There is robust evidence that participation in physical activity is associated with a decreased risk of cognitive decline and dementia. The U.S. Department of Health and Human Services recommends that all adults do at least 150 minutes of moderate-intensity aerobic activity, or at least 75 minutes of vigorous-intensity aerobic activity a week, or an equivalent combination of moderate- and vigorous-intensity aerobic activity [46]. These guidelines also call for strength training exercises targeting all major muscle groups at least two times a week for additional health benefits [46]. Between 2006 and 2018, however, only 23% of adults 18 years and older in the U.S. were meeting recommendations for aerobic and muscle-strengthening exercise [47], based on aggregate survey data. This number dropped to 16% of adults between the ages of 65 and 74, and to 10% of adults 75 years and over [47].
Epidemiological studies have demonstrated that high levels of exercise in mid- and late life are associated with increased cognitive performance, decreased rate of cognitive decline, and decreased risk of all-cause dementia as well as dementia subtypes [48–52]. For example, the Nurses’ Health Study of over 18,000 women, aged 70 to 81 years, found that regular, long-term physical activity was associated with better cognitive performance, including tests of general cognition, verbal memory, category fluency, and attention [48]. However, as with many of the factors being discussed, the causal direction of the association is hard to determine. Perhaps, reduced physical activity is due to the early impact of an underlying neurodegenerative process. Against this hypothesis are epidemiological studies that suggest a relationship between earlier-in-life physical activity and the incidence of dementia in older age. For example, a longitudinal study of women, followed for 44 years starting in midlife, found that high fitness during middle age was associated with a decreased risk of dementia compared to medium and low fitness, and a delay of 9.5 years for onset of dementia symptoms compared to medium fitness [49]. These results are consistent with findings from large meta-analyses of prospective studies [53, 54].
Although many cohort studies demonstrate positive outcomes associated with physical activity, others have shown conflicting results. One epidemiological study of the Whitehall cohort (10,308 participants, aged 35–55 years at study inception) found no association between exercise and cognitive decline or exercise and incidence of dementia over 15 and 27 years, respectively [55]. Notably, this study relied on self-report questionnaires to determine level of physical activity, and over 50% of participants reported adhering to consensus recommendations of more than 2.5 hours of moderate to vigorous exercise per week, which seems implausible based on rates in the general population. If the amount of physical activity was inaccurately reported, it would undermine conclusions that could be drawn about the relationship between exercise and dementia risk.
Studies of animals have indicated that exercise promotes neurogenesis, synaptogenesis, and improved learning and memory, through upregulation of proteins including brain-derived neurotrophic factor (BDNF), vascular endothelial growth factor, and insulin-like growth factor [56–60]. In animal models of AD, exercise is also associated with reduced Aβ deposition and tau pathology [61–63].
In humans, physical activity plays a major role in promoting cardiovascular health, which is closely linked to brain health. Managing cardiovascular risk factors, including hypertension, diabetes mellitus (DM), hyperlipidemia, and obesity/metabolic syndrome, can reduce the risk of cognitive decline and dementia. (See “Treat cardiovascular risk factors”, below, for a more detailed discussion on cardiovascular health and brain health.) Exercise has also been shown to diminish the risk of falls, a common cause of head injury, in older adults [64]. In addition to enhancing cardiovascular health and reducing fall risk, exercise has been shown to promote neurogenesis, synaptogenesis, and brain vascularization in humans through upregulation of insulin-like growth factor-1 [65–67] and BDNF [68]. Serum levels of BDNF, which promotes neuroplasticity, synaptic growth and density, and hippocampal neurogenesis [69–72], have been shown to increase both immediately after exercise [73, 74] and following a longer intervention period [75, 76]. One study of 19 older adults found that serum BDNF immediately following a single 35-minute physical activity session was significantly elevated compared to BDNF levels immediately following other potential brain-healthy endeavors (i.e., cognitive activity and meditation) [74]. Aerobic physical activity has also been shown to attenuate loss of brain volume and decrease white matter changes [77–80], which can be seen on magnetic resonance imaging (MRI) scans and have been associated with increased incidence of cognitive impairment and dementia [81]. In one study, 120 older adults were randomized to an aerobic exercise group or a stretching and toning control group. Although the two groups had similar hippocampal volume at baseline, over one year, the control group exhibited an approximate 1.5% decline in hippocampal volume, while the exercise group showed an approximate 2% increase. Within the aerobic exercise group, increased hippocampal volume was associated with improvement on a spatial memory task [77]. Additionally, both aerobic [82] and resistance [83] training have been shown to increase measures of functional neuroplasticity.
Physical activity also may protect against the adverse consequences of cerebral Aβ and tau deposition, which include accelerated cognitive decline and the development of AD. For example, one study of 43 cognitively healthy older adults found that self-reported levels of current physical activity were associated with a decreased positron emission tomography (PET)-quantified tau burden [84]. A recent report from the Harvard Aging Brain Study, a longitudinal investigation of cognitively normal older individuals, indicated that increased baseline physical activity markedly reduced the negative impact of Aβ burden, as measured by Pittsburgh Compound B (PiB)-PET, on cognition and cerebral atrophy over a median of six years [85]. The potential benefits of physical activity also may apply to other neurodegenerative conditions. For example, in a longitudinal study of both mutation carriers and non-carriers in families with autosomal dominant frontotemporal lobar degeneration, greater physical activity was associated with > 55% slowing of clinical decline per year [52].
Aerobic exercise interventions have been associated with decreased age-related cognitive decline [86, 87], improved executive function [88, 89], and improved memory [77, 91] in cognitively normal older adults, and improved memory in individuals with MCI [80]. In one intervention trial, Lautenschlager and colleagues randomized 170 older adults with subjective cognitive decline to a six-month home-based physical activity intervention or an education and usual care control group. Compared to usual care, the six-month program of physical activity provided a modest improvement in cognition, which was sustained over an 18-month follow-up period [92]. Blumenthal and colleagues randomized 160 sedentary older adults with risk factors for dementia to six months of one of four groups: aerobic exercise, Dietary Approaches to Stop Hypertension (DASH) diet nutritional counseling, aerobic exercise plus DASH nutritional counseling, or health education. Participants who engaged in aerobic exercise or aerobic exercise plus DASH nutritional counseling had improved scores on measures of executive function compared to DASH nutritional counseling or health education alone [88]. Additionally, an RCT conducted by de Oliveira Silva and colleagues found that an exercise intervention improved mobility and executive function in those with MCI, but not those with AD dementia, suggesting that exercise interventions may be more effective when implemented earlier in disease progression [93]. Resistance training interventions have also been associated with improved attention [65] and executive function [65, 94] in cognitively normal older adults, and improved attention [82], memory [82, 95], and global cognitive function [96, 97] in individuals with MCI.
Although not all exercise interventions have been associated with improved cognition, a systematic review found that exercising for at least 52 hours during the length of an intervention, regardless of how many weeks the intervention lasted or duration of exercise events, was associated with improved cognitive performance, including global cognition, processing speed/attention, and executive function. All studies associated with no improvement in cognitive outcomes had interventions lasting less than 52 total hours [98].
Cognitive stimulation and education
Converging evidence highlights the importance of remaining cognitively engaged throughout one’s life. In early life, fewer years of education is associated with reduced cognitive reserve and increased risk of dementia in late life [4]. Education may have a greater impact on dementia risk in low- and middle-income countries, where secondary education might not be available to everyone. This risk factor is most effectively addressed by implementing public policy that supports high-quality education for all children.
Although early-life education is critically important, cognitive stimulation during all stages of life has been shown to influence brain health and cognition. Epidemiological studies have shown that cognitive activity in both mid- and late life is associated with improved cognitive performance and reduced risk of cognitive decline and dementia [50, 99–103]. Najar and colleagues found that cognitive activity in midlife was associated with a 34% reduced risk of all-cause dementia and 46% reduced risk of AD over a follow-up period of 44 years, after adjusting for age, education, socioeconomic status, hypertension, body mass index, smoking, DM, angina pectoris, stress, and major depression [50]. For cognitive activity in late life, a meta-analysis of 19 studies in cognitively healthy older adults found that participation in cognitive leisure activities (e.g., crossword puzzles, card games, computer use, arts and crafts) was associated with 31% risk reduction for cognitive impairment and 42% risk reduction for dementia. Cognitive leisure activities were also significantly associated with improved memory, processing speed, and executive function [103]. It is important to note that the studies included in this meta-analysis differed in the confounding variables for which they adjusted and rarely took other lifestyle factors like physical activity or diet into account.
Studies of animals have demonstrated that environmental enrichment, which provides cognitive stimulation, is associated with improved cognitive functioning across the lifespan [104], as well as increased neurogenesis, upregulated neuronal growth factors, and increased synaptic density and plasticity [105, 106]. Environmental enrichment has also been shown to decrease cognitive deficits and Aβ burden in animal models of AD [107, 108].
The mechanisms by which cognitive activity reduces the risk of cognitive decline and dementia in humans are still unclear [109–113]. One hypothesis is that regular cognitive engagement contributes to increased neuroplasticity that partially offsets otherwise expected cognitive losses or the actual accumulation of pathologic changes due to aging or neurodegenerative conditions [114]. The evidence to support or refute this hypothesis is inconclusive. For example, an intervention trial, in which our Center was one of two sites, found that cognitive training for 35 minutes per day, five days per week for five weeks was associated with a 10% increase in serum BDNF [109], a neurotrophic factor that, as noted above, has been associated with neuroplasticity. Another intervention trial, however, found that cognitive training for one hour per day, five days per week for ten weeks was not associated with serum BDNF levels [75]. Other possible mechanisms include reduction in AD pathology or decreased loss of brain volume. A cross-sectional study of 65 healthy older adults, ten AD patients, and 11 young controls demonstrated that cognitive activity, especially in early and midlife, was associated with decreased Aβ deposition, as measured by amyloid PET [110]. Cognitive training for executive function specifically has been linked to attenuated loss of grey matter [115]. Conversely, multiple cross-sectional imaging studies found neither current nor past cognitive activity had any effect on Aβ burden, brain glucose metabolism, or hippocampal volume [111, 112]. More human research is needed to elucidate the relationship between cognitive activity and brain health.
Cognitive training interventions have shown promising results, including increased hippocampal activation during memory tasks [116, 117], improved cognition in several domains, and decreased risk of cognitive and functional decline [118–125]. Some of the most impressive outcomes have been reported for the Advanced Cognitive Training for Independent and Vital Elderly (ACTIVE) study, in which 2,832 older adults (mean age 73.6) were randomized to either a ten-session group training focused on one of three cognitive domains (memory, reasoning, or processing speed) or to a no-contact control group. After ten sessions, all groups showed significant improvement in the domain trained. After two years, cognitive improvement from baseline was maintained for all training groups [118]. Ten years after the ACTIVE intervention, all cognitive training groups reported less functional decline compared to the no-contact control group and measured improvement in the reasoning and processing speed groups was retained [122]. Additionally, individuals randomized to the processing speed training group reportedly had a 29% risk reduction for dementia [123]. The dropout rate (often due to the death of these aging participants) was similar across groups and the attrition rate was similar to that of other studies over the ten-year follow-up period.
Not all cognitive training interventions have shown positive results. For example, one study by Kallio and colleagues found no improvement in cognition after cognitive training for adults with mild to moderate dementia [126], suggesting that cognitive interventions may be more effective when they occur before the onset of dementia. Additionally, many cognitive training interventions have demonstrated enhanced performance on the task trained as well as near-transfer effects (i.e., improvement in an untrained task that involves the same cognitive domain used in the trained task), but have been less successful in showing far-transfer effects (i.e., improvement in an untrained task in a different cognitive realm than had been trained) or an impact on activities of daily living [122–125].
Although it is important to encourage patients to engage cognitively, not all activities will necessarily yield equal cognitive benefit. In the Synapse Project, McDonough and colleagues randomized 39 older adults to High-Challenge (quilting and/or digital photography) or Low-Challenge (e.g., socializing, listening to music) groups. Compared to the Low-Challenge group, the High-Challenge group showed increased ability to modulate brain activity, as measured by functional MRI, in regions associated with attention and semantic processing [127]. Additionally, despite evidence supporting cognitive stimulation for brain health, many popular commercial cognitive or memory training platforms have limited supporting evidence [128, 129]. To date there has been no compelling evidence that computerized cognitive training is superior to other less expensive, readily-available, intellectually-stimulating activities. However, it remains to be determined whether continuously-titrated task difficulty based on real-time performance or having to pay for cognitive training may be motivating influences to achieve the desired outcome. Our view is that if individuals enjoy using these platforms, there is no harm in supporting their use. Perhaps most importantly, existing research suggests that engaging in new, challenging cognitive activities may yield a greater benefit for brain health and cognition in older adults than carrying out familiar, undemanding mental activities.
Social engagement
Epidemiological evidence suggests that reduced social participation, less frequent social contact, and feelings of loneliness are associated with cognitive impairment and incident dementia [100, 130–134]. However, the causal direction of this association is difficult to prove, because more impaired individuals may have fewer social interactions. Decreased social engagement may be a prodromal symptom of dementia, but increasing evidence has demonstrated that it is also an independent risk factor. For example, individuals living with a partner in mid- to late life have a reduced risk of cognitive impairment and all-cause dementia compared to those who are single, separated, or widowed [130, 131]. Maintaining a large social network may also help promote healthy cognitive aging. In a five-year prospective study of 2,249 older women without dementia, social network size was directly associated with reduced risk of dementia and cognitive impairment. The women who had daily contact with friends and family cut their risk of dementia by nearly half [132]. Additionally, a meta-analysis of 51 longitudinal cohort studies examining social network size and social activity found that aspects of social isolation, including low levels of social activity and small social networks, were associated with decreased cognitive function in late life [133].
In animals, social isolation (via individual housing) has been shown to exacerbate memory deficits and promote cognitive decline by increasing systemic inflammation [135, 136], oxidative stress [135], neuroinflammation [137], and Aβ levels [135, 137], and by decreasing BDNF levels and neurogenesis [135, 139].
In humans, decreased social interaction and often attendant feelings of loneliness have been linked to factors that may increase risk of cognitive impairment and dementia, including worsened sleep [140, 141] and increased stress [140], inflammation [141–144], and systolic blood pressure [145]. Conversely, higher levels of social engagement in older adults provide increased opportunities for cognitive stimulation, problem solving, and social support, and have been linked to increased total brain and grey matter volumes [146], greater grey matter integrity in several regions relevant to social cognition [147], and increased neural network function, which boosts cognitive reserve [148]. Social network size and level of social engagement have also been shown to modify the relationship between AD pathology and cognitive function [149, 150]. In a recent report of cognitively normal older adults participating in the Harvard Aging Brain study, Biddle and colleagues observed that the negative impact of high Aβ on cognitive decline was modulated by level of social engagement [150]. Lower baseline social engagement was associated with greater Aβ-related decline in cognitive test scores, while higher baseline social engagement was associated with preserved cognitive test scores over a three-year follow-up period. These results regarding social engagement are reminiscent of the findings suggesting that higher baseline physical activity may modulate the deleterious effect of Aβ cognitive decline, as noted above [85]. Interestingly, baseline Aβ levels were not linked to a decline in social engagement after controlling for relevant co-variates, and did not modulate the negative association between lower baseline cognitive performance and subsequent reduction in social engagement.
Intervention trials aimed at promoting social engagement have also shown promise for boosting brain health and cognition. In one intervention, 149 older adults were randomized to Experience Corps [151], a team-based training group, or the waitlist control group. Experience Corps members trained in teams to work in elementary schools for 15 hours per week for six months, helping students to succeed academically and socially. After completing the program, participants showed improvement in memory and executive function compared to control. Notably, participants with impaired executive function at baseline showed the greatest improvement [151]. A functional MRI study of eight participants in Experience Corps and nine matched waitlist controls found that intervention participants also exhibited increases in prefrontal cortex and anterior cingulate cortex activation compared to controls following the six-month intervention period [152]. A subsequent two-year trial of the Experience Corps intervention found that, compared to control, participation in the program was associated with decreased volume loss in brain regions associated with dementia. In men, participating in the program was associated with a small increase in brain volume in these regions [153]. Another study compared the effects of three different social-cognitive interventions: group-based cognitive training, health promotion classes, and a social engagement-promoting book club. All three interventions resulted in a significant improvement in cognitive performance, with no significant differences between the groups [154].
Heart-healthy diet
Several heart-healthy diets have been associated with a decreased risk of cognitive decline and dementia, including the Mediterranean diet (MeDi) [155], the DASH diet [156], and the Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) diet [157]. Each of these diets includes an abundance of fruits and vegetables and emphasizes consumption of antioxidant-rich foods. DASH differs from the other diets in that it encourages consumption of low-fat dairy products, aims to reduce sodium and fat intake, and does not prescribe nor prohibit wine consumption [156]. The MIND diet is unique in specifying greater intake of leafy green vegetables and berries over other plant-based foods [157]. These diets contrast with a more common diet consumed in the U.S. and other parts of the Western world, which is characterized by excess sodium, saturated fat, refined grains, red and processed meats, and calories from solid fats and added sugar [158].
Epidemiological studies have shown that all three of these heart-healthy diets are beneficial to brain health and cognition. Adherence to the MeDi has been associated with increased cognitive function [159] and decreased risk of cognitive impairment [159, 160] and AD [155, 161]. The DASH diet has been linked to decreased risk of AD [161]. The MIND diet has been associated with better cognitive function [159], decreased rate of age-related cognitive decline [162], and decreased risk of cognitive impairment [159] and AD [161]. One study of 923 older adults followed for an average of 4.5 years found that only high adherence to the MeDi or DASH diet was associated with decreased risk of AD, while moderate to high adherence to the MIND diet was associated with decreased risk of AD [161]. A study of 1,220 older adults determined that the MIND diet, but not MeDi, was associated with decreased risk of cognitive impairment over a twelve-year follow up period [163]. Taken together, these results suggest that the MIND diet may be most effective for promoting brain health and cognition, but more research, including high-quality RCTs, is needed. Meta-analyses of prospective cohort studies have affirmed that a high level of adherence to these diets is associated with reduced risk of developing MCI and AD [164–168]. One meta-analysis examined specific components of the MeDi and demonstrated that increased consumption of unsaturated fatty acids, antioxidants, and B vitamins was associated with decreased risk of dementia, and low levels of vitamin D were associated with cognitive decline [166]. Some of these studies have shown a degree-of-adherence-dependent effect of these brain-healthy diets [159, 160]. In contrast, observational evidence suggests that a more typical Western diet is linked to worsened age-related cognitive decline [169].
Like physical activity, eating a heart-healthy diet plays an important role in managing cardiovascular risk. The MeDi has been linked to beneficial effects on hypertension, DM, obesity, and hyperlipidemia [170]. Additionally, there is evidence that these diets boost cognition and reduce risk of dementia by decreasing vascular inflammation, white matter changes, and Aβ and tau pathology [171, 172]. In both animal and human studies, several components of these heart healthy diets, including omega-3 fatty acids [173, 174], antioxidant vitamins [175], and polyphenols [174, 176], have been linked to decreased neuroinflammation and neuronal death. In humans, increased adherence to the MeDi has also been associated with increased total brain volume [177, 178] and decreased hippocampal atrophy [179]. Additionally, higher MeDi scores, greater consumption of vegetables, vitamin A, and β-carotene, and mild-moderate alcohol consumption (see “Limit alcohol use” section, below) have been linked to decreased Aβ burden [180–182].
Intervention trials have also demonstrated a link between heart-healthy diets and improved cognitive function, including global cognition, memory, and executive function [183–186]. For example, the PREDIMED trial assigned 7,447 cognitively healthy older adults to either MeDi supplemented with olive oil, MeDi supplemented with nuts, or a control diet (general advice to reduce dietary fat). Both of the MeDi groups showed a reduction in myocardial infarction, stroke, and death from cardiovascular events compared to the control group [187]. Subsequent subset analyses of PREDIMED participants found that both MeDi groups also showed improved cognitive function compared to control [183, 184]. RCTs have also examined interventions using specific components of the MeDi, including polyphenol-rich berries [188–190] and omega-3 fatty acids [190–194]. Interventions using berries and berry-based supplements have shown some promising results for improving performance on cognitive tests. For example, in a small study conducted by Boespflug and colleagues, 16 older adults completed a randomized trial in which they received either daily blueberry powder or placebo powder for a 16-week period. After the intervention, participants who received blueberry supplements exhibited increased blood oxygen level-dependent signal activation in the left hemisphere network that serves verbal working memory during an n-back working memory task compared to controls. Results also suggested that participants who received blueberry supplements tended to have increased accuracy on the one-back task [188]. These results are consistent with findings from large, longitudinal cohort studies, which have demonstrated that berry consumption is associated with a decreased rate of cognitive decline [195]. The evidence on omega-3 fatty acids alone is more mixed, with some studies suggesting improved cognition [190–192] and others suggesting no effect [193, 194].
Not all studies have demonstrated positive effects of following a brain-healthy diet. For example, one population-based cohort study found that diet quality in midlife had no effect on subsequent risk of dementia [196]. A six-month RCT of 160 sedentary adults comparing the effects of aerobic activity, DASH diet, and a combination of both found that the DASH diet alone had no significant effects on executive function [88]. Participants assigned to both aerobic activity and the DASH diet, however, showed the greatest improvement in executive function, suggesting a possible synergistic effect [88]. It is important to note that this intervention only lasted for six months, compared to a median of 4.1 years in the PREDIMED trial; it may take a longer study duration to observe the effects of a heart-healthy diet on cognition.
Given the many evidence-based benefits of the MeDi, DASH, and MIND diets for both brain and overall health, patients should be encouraged to adopt manageable, sustainable changes to increase their adherence to a heart- and brain-healthy diet.
Treat cardiovascular risk factors
Cardiovascular risk factors including hypertension, DM, hyperlipemia, obesity, and metabolic syndrome are associated with an increased risk of developing cognitive impairment and dementia. Often these factors may be addressed through adopting healthy behaviors, including physical activity and a heart-healthy diet, as discussed above. In some cases, however, it may also be appropriate to prescribe medications to treat these conditions to diminish risk of both cardiovascular disease and dementia more expeditiously.
Hypertension
The connection between hypertension and cognitive function is well-documented, and controlling hypertension is recommended by all major health organizations for reducing the risk of cognitive decline. Until recently, hypertension was defined as > 140/90 mmHg, but is now separated into stage 1 hypertension (130–139 mmHg systolic, 80–89 mmHg diastolic) and stage 2 hypertension (> 140/90 mmHg) [197]. Epidemiological evidence has demonstrated that midlife hypertension is a major risk factor for all-cause dementia [198], vascular dementia [199], and cognitive dysfunction [200], including poorer executive function, memory, and processing speed [201, 202]. Several meta-analyses of both observational and intervention studies have demonstrated that use of antihypertensive medications can mitigate the increased risk of cognitive decline and dementia [203–205].
While the link between midlife hypertension and dementia risk has been established for several years [206–210], the evidence on late-life hypertension and dementia risk has been less conclusive [4]. Individuals with early dementia often have a decrease in blood pressure [211], which likely reflects the disease rather than a decrease in risk of cognitive decline. Supporting this hypothesis, steep declines in blood pressure between mid- and late life have been associated with increased risk of dementia [209, 212].
Hypertension negatively impacts the brain in a number of ways. In animal models, hypertension has been associated with decreased cognitive function, increased inflammation and oxidative stress, microhemorrhages and infarcts, endothelial dysfunction, increased Aβ accumulation, and disruption of the blood-brain barrier (BBB) [213]. In humans, hypertension has also been linked to neurovascular dysfunction [214], increased risk of stroke [215, 216], increased global and regional brain atrophy [217, 218], increased white matter changes [218–220], increased Aβ [221–224] and tau [216] pathology, and increased BBB permeability [225]. The BBB, a continuous, closed membrane within all cerebral blood vessels, is critical in protecting neurons from circulating neurotoxic debris, cells, and pathogens in the blood, and increased permeability can increase neuroinflammation and trigger different pathways of neurodegeneration [226]. Treating hypertension with medication may at least partially moderate these negative effects on brain health. For example, a recent autopsy study of 96 AD cases and 53 pathological controls found that antihypertensive medication use was associated with a less extensive spread of AD pathology in the brain [227].
Recent RCTs have shown promising results for programs aimed at controlling hypertension [88, 229]. In the SPRINT-MIND trial [228], 9,361 older adults were randomized to either standard or intensive blood pressure control groups (target systolic blood pressures for the groups were < 140 mmHg and < 120 mmHg, respectively). Compared to standard blood pressure control, intensive blood pressure control was associated with a 19% decreased risk of MCI alone and 15% decreased risk of MCI and dementia combined. The 17% reduction in risk of probable dementia did not reach significance, which may have been related to the early termination of the trial due to robust cardiovascular benefits. The SPRINT-MIND trial also showed promising evidence that intensive blood pressure control was well-tolerated across all ages and did not contribute to negative effects on cognition [228, 230]. Intensive blood pressure control in participants over 80 years old was associated with a greater risk of developing hypotension and possibly syncope, but was not associated with a higher risk of falls as compared to the standard blood pressure control group [230]. In most participants over 80 with at least three co-morbidities, intensive blood pressure control was associated with decreased risk of major cardiovascular events, MCI, and death [231].
In a sub-study of the SPRINT-MIND trial, cerebral white matter lesions were measured in a subset of participants who had an MRI scan at baseline and four years after randomization [232]. After four years, the participants in the intensive blood pressure control group had a smaller increase in cerebral white matter lesions compared to the participants in the standard blood pressure control group [232]. Overall, the SPRINT-MIND trial highlights the potential for early and aggressive blood pressure lowering interventions to have a positive impact on the cerebrovascular health of older adults, which may lower risk of cognitive impairment [228, 232].
Evidence varies on the direct effects of blood pressure control on cognitive function or decline. A sub-analysis of the Heart Outcomes Prevention Evaluation (HOPE)-3 trial found that long-term blood pressure lowering with candesartan plus hydrochlorothiazide versus placebo did not affect cognitive decline in older adults, but did significantly reduce their blood pressure [233]. However, in HOPE-3, systolic blood pressure of the intervention group was only a mean of 6.0 mmHg lower than the control group, which was much less than the reduction achieved in SPRINT-MIND. Similarly, the Hypertension in the Very Elderly Trial cognition function assessment (HYVET-COG) also found that antihypertensive therapy in patients 80 years or older did not have a statistically significant effect in reducing incident dementia, but did significantly reduce blood pressure [234]. However, the HYVET-COG study was terminated early due to significant reductions in all-cause mortality and stroke, which could have confounded analysis of the incidence of dementia. Similar to SPRINT-MIND, the HYVET-COG study also found that older participants with higher Frailty Index scores did not have an increased risk of falls or mortality with antihypertensive therapy [235]. Several studies have confirmed that hypertension management does not contribute to negative effects on frailty and mobility, allaying a common worry for clinicians in the use of some antihypertensive regimens in this population [235–237]. Though the HOPE-3 and HYVET-COG studies did not show improvements or changes in cognition directly, the positive, “downstream” effects that reducing blood pressure have on cerebrovascular and neurodegenerative disease risk are sufficiently well-founded to support the practice of antihypertensive therapy for these at-risk, older adults.
Diabetes mellitus
The connection between DM and dementia is well established. Although both type 1 and type 2 DM are associated with increased risk of cognitive dysfunction [238], type 2 is more often studied in the context of dementia. The presence of type 2 DM has been associated with impaired cognition [239, 240], increased risk of falls [239], and an increased risk of all-cause dementia [241–244] and AD [241, 245], likely through repeated fluctuations of blood sugar levels (hyperglycemia and hypoglycemia), and consequent systemic and cerebrovascular complications [246]. A study of 10,316 patients with a new diagnosis of DM and 41,264 age- and sex-matched controls found that DM was associated with a 47% increased risk of dementia [242]. In patients with DM, hypertension and/or hyperlipidemia did not significantly increase dementia risk, suggesting that the increased risk of dementia was primarily driven by DM itself [242]. The Ginkgo Evaluation of Memory Study investigated the relationship between DM and domain-specific cognitive impairments, and found that participants with DM scored worse on cognitive baseline tests and showed greater cognitive declines over a median of 6.1 years in phonemic verbal fluency and executive function compared to their healthy peers [240]. Impaired cognition can lead to poor self-management of DM, creating a vicious cycle of worsened symptoms and worsened cognition, which may be associated with unfavorable health outcomes and decreased quality of life [247].
The mechanisms by which DM likely impacts cognitive function and dementia risk have been reviewed thoroughly [246, 248–250]. Briefly, glucose toxicity, hyperinsulinemia, and vascular damage resulting from DM may contribute to increasing risk of cognitive decline and dementia. In animal models, DM has been associated with cognitive dysfunction, neuroinflammation, vascular injury, atrophy of the hippocampus and prefrontal cortex, increased risk of stroke, and increased AD pathology [246, 250]. In humans, Type 2 DM has been linked to systemic inflammation, neuroinflammation, global and regional atrophy, white matter hyperintensities, decreased functional connectivity, decreased glucose metabolism in the brain, increased AD pathology, increased risk of stroke, and comorbid depression [246, 249]. Some of these changes are also observed in prediabetic populations. For example, in a cross-sectional study of 2,439 middle-aged adults from the Framingham Offspring cohort, both DM and prediabetes were associated with decreased total cerebral brain volume [251].
Some evidence demonstrates that intensive glycemic control may mitigate the increased risk of cognitive decline and dementia. A study by Yaffe and colleagues found that elderly adults with DM had lower cognitive test scores at baseline than participants without DM, and participants with DM and poor glucose control over a nine-year follow-up period had worse cognitive function and a greater rate of cognitive decline than others [252]. In an observational study of 3,433 patients with type 1 DM, individuals with more than 75% of their hemoglobin A1c (HbA1c) measurements at 8.0–8.9% or > 9% were more than twice as likely to develop dementia over a mean follow-up of 6.3 years compared to individuals with minimal exposure (less than 10% of HbA1c measurements) to these levels. Conversely, patients with better glycemic control (i.e., more than 75% of HbA1c measurements at 6.0–6.9% or 7.0–7.9%) had an approximate 60% dementia risk reduction compared to those with minimal exposure to well-controlled levels of HbA1c [253]. A secondary analysis from the Finnish Diabetes Prevention study, which randomized middle-aged adults to a four-year lifestyle intervention or control group, showed that better glycemic control was associated with better cognitive performance nine years post-intervention in individuals with impaired glucose tolerance at baseline [254]. However, most RCTs conducted to date have not found benefits of intensive glycemic control interventions on cognitive function or decline [255–258]. Despite these negative findings, there is ample evidence that untreated DM is harmful to brain health. Patients should be encouraged to adopt healthy lifestyles to prevent or delay the onset of DM. Providers should work with patients in order to detect early signs of DM, obtain an early diagnosis, manage hyperglycemia, and encourage regular monitoring of the disease.
Hyperlipidemia
Hyperlipidemia (total cholesterol > 200 mg/dL [259]) has been associated with dementia risk, although evidence is less conclusive than other cardiovascular risk factors. Some epidemiological evidence has shown that high cholesterol in midlife is associated with increased risk of all-cause dementia [260], AD [208, 260–262] and vascular dementia [261], while other studies have found no link [263]. Late-life cholesterol levels have not been linked to risk of cognitive impairment or dementia [262, 264].
The precise mechanism by which cholesterol impacts dementia risk is still unclear [265]. In a transgenic mouse model expressing human apolipoprotein B-100, hyperlipidemia has been associated with cardio- and cerebrovascular damage, vascular dysfunction, increased permeability of the BBB, and atherosclerosis [266]. In humans, hyperlipidemia increases the risk of cardiovascular disease, atherosclerosis, and stroke [267, 268]. Additionally, Bowman and colleagues investigated the relationship between the BBB and the pathology of AD and found that dyslipidemia was prevalent in 47% of study participants with mild-to-moderate AD and 75% of those who had AD with BBB impairment, suggesting that dyslipidemia may play a role in decreasing BBB integrity [269]. Total cholesterol and low-density lipoprotein cholesterol levels have also been associated with increased Aβ burden in humans [224, 270].
In order to reduce cardiovascular risk, the American Heart Association (AHA) and American College of Cardiology (ACC) recommend keeping total cholesterol levels below 170 mg/dL [259]. The AHA and ACC suggest eating a heart-healthy diet, staying physically active, quitting smoking, and maintaining a healthy weight to reduce cholesterol levels [259]. Patients with elevated cholesterol may also need to be treated with a statin. For most patients, the AHA and ACC recommend initiating statin therapy when low-density lipoprotein cholesterol levels exceed 70 mg/dL [259]. Some evidence suggests that the use of statins may mitigate the risk of all-cause dementia [271] or AD [272]. See “Other medications that may affect cognition” for a more complete review of the current evidence on statin use and risk of dementia.
Obesity and metabolic syndrome
Obesity and metabolic syndrome (or insulin resistance syndrome), which is characterized by abdominal obesity, hyperglycemia, dyslipidemia, and hypertension [273], have been well-established as risk factors for dementia [274–278]. In clinical studies, obesity is most often defined using body mass index (BMI). It is important to note that BMI does not account for differences in body composition, and therefore is not a perfect representation of adiposity. However, the association between BMI and dementia risk is still strong. For example, in a recent study of 6,582 individuals over the age of 50, Ma and colleagues found that obesity (defined as BMI≥30 kg/m2) at baseline was associated with an increased risk of dementia over a mean follow-up period of 11 years [274]. Several studies have demonstrated a positive association between midlife, but not late-life, obesity and risk of developing dementia [279, 280]. Data from the Whitehall II study demonstrated that obesity at age 50, but not at ages 60 or 70, was associated with an increased risk of dementia [280]. The lack of association between obesity in late life and dementia risk, however, may be attributable to the “preclinical” decrease in weight that can often be seen in early cognitive impairment, prior to dementia onset [280].
Obesity (particularly central or abdominal obesity) in both animals and humans has been linked to release of pro-inflammatory cytokines, including interleukin-6 and tumor necrosis factor alpha, and chronic systemic inflammation [281]. Obesity-associated inflammation and hyperinsulinemia contribute to endothelial dysfunction, which leads to increased BBB permeability and decreased insulin transport to the brain [281]. The dysregulation of adipokines, including leptin, adiponectin, and resistin, that occurs in individuals with obesity may also play a role in increasing the risk of dementia, but more research is needed to fully elucidate these relationships [282]. Together, the systemic effects of obesity have deleterious effects on the brain, but losing weight may partially mitigate these negative effects. Prehn and colleagues randomized 37 obese women to a weight-loss intervention following a caloric restriction or control condition [283]. Following the 12-week intervention period and a four-week period where the new weight was maintained, the intervention was associated with increased grey matter volume in the inferior frontal gyrus and hippocampus and augmented hippocampal functional connectivity [283].
Weight loss interventions have shown promise for improving cognition in cognitively normal older adults [284] and older adults with MCI [285–287]. For example, Napoli and colleagues randomized 107 cognitively normal older adults to weight management (diet), exercise, combination weight management and exercise, or a control group [284]. After the 52-week intervention period, participation in any of the three intervention groups was associated with increased scores on the Mini-Mental State Exam compared to control [284]. In another intervention trial, Horie and colleagues randomized 80 older adults with MCI and BMI≥30 kg/m2 to either routine medical care or routine medical care with nutritional counseling [285]. Both groups had significant decreases in BMI over the 12-month intervention period, and decreases in BMI were associated with improvements in cognition, with no difference between groups [285]. The results from these studies suggest that targeting weight loss may be a promising approach for improving cognition or slowing the rate of decline in individuals who already have cognitive impairment.
Stop or reduce smoking
Epidemiological evidence has demonstrated the detrimental effects of smoking on cognitive performance [288, 289], risk of all-cause dementia [290–293] and AD [290–295], and rate of cognitive decline [292]. For example, one cohort study of 21,123 adults followed for 20–30 years, found that smoking more than two packs per day in midlife was associated with an increased risk of dementia of over 100% [291]. Evidence suggests that amount of smoking is positively correlated with risk of dementia. In a meta-analysis of 37 observational studies, for every 20 cigarettes smoked per day, there was a 34% increase in the risk of all-cause dementia [293]. Reducing or quitting smoking, however, may partially or even fully mitigate this increased risk of dementia. Multiple studies have shown that former smokers do not have an increased risk of dementia or AD compared to never-smokers [290, 293].
In animals, exposure to cigarette smoke is associated with increased markers of oxidative stress, depleted cerebral antioxidants, and increased proinflammatory cytokines [296]. In humans, cigarette smoke has been linked to increases in oxidative stress and neuroinflammation, risk of stroke, white matter hyperintensities, subcortical atrophy, and cerebral Aβ deposition [297–299]. Additionally, in cognitively healthy individuals, chronic smoking may lead to atrophy of areas of the basal forebrain that provide cholinergic input to the brain, increasing risk of developing AD [300].
In contrast to most of the literature, a few studies have found no effect or a protective effect of cigarette smoking on dementia risk [301, 302]. However, these results may be misleading due to study participant selection bias, survivor bias, or funding by the tobacco industry [295, 304]. Even after controlling for survivor bias, however, one recent study found that smoking tobacco was not associated with incident dementia, despite strong correlations with other chronic diseases and earlier mortality [305]. Despite a few conflicting results, there is overwhelming evidence that smoking is harmful to brain and overall health, and providers should encourage patients to stop or reduce smoking.
Manage stress and depression
Stress
Stress has been defined as a condition or feeling experienced when a person perceives that demands exceed the personal and social resources the individual is able to mobilize [306]. Managing life’s stressors is often recommended in order to maintain a cognitively healthy lifestyle. Demanding moments can elicit a stress response that varies in intensity, duration, and predictability across each individual experience [307]. This inherent heterogeneity complicates our understanding of the mechanisms by which stress affects cognition. Importantly, not all stress is harmful to cognition. When the stress response is moderate and controlled, it can benefit the way people learn and adapt [308]. For example, mild stress improves task accuracy by facilitating cognitive functions like selective attention [309]. Acute and chronic moderate and severe stress, however, have been shown to be deleterious to cognitive processes.
Generally, acute stress has been found to transiently degrade cognitive capacities such as working memory, processing speed, and attention [310–312]. Coping with acute stress may require cognitive resources to be diverted from tasks at hand to help contain the stressful experience, which can undermine cognitive performance and function [313]. In a meta-analysis of 51 studies, Shields and colleagues found that acute stress exerted a greater effect when working memory load was high. Additionally, acute stress is associated with significantly reduced cognitive flexibility across various conditions [312].
The transition from acute to chronic stress is marked by a prolonged, maladaptive response that can have an even greater impact on cognition. For example, a two-year study of 2,713 older adults revealed that those with higher levels of perceived stress performed worse on tests of processing speed and attention when compared to less stressed peers [314].
Critically, there is a growing body of evidence that has shown chronic life stress to be associated with an increased risk of dementia [315, 316]. In one 35-year prospective study of 1,462 women, frequent or constant stress reported in midlife was associated with an increased risk of dementia [315]. Additionally, smaller studies have found that people who suffer chronic life stressors [317], have current stress disorders [318], or who report a lack of control over their high pressure jobs [319] developed dementia at higher rates than those living less stressful lives. A review of epidemiological evidence showed that increased life stress has been linked to greater risk of MCI or dementia in a number of studies, even if stress is not the primary or direct contributing factor [316].
The precise mechanism by which acute and chronic stress affect cognitive performance and cognitive decline over time is still unclear, although several factors likely contribute. First, recent findings point to the physiologic response of chronic stress having deleterious effects on cortical structures critical to the progression of dementia [320, 321]. The hippocampus is rich in corticosteroid receptors, making it especially vulnerable to increased activity of the hypothalamic-pituitary-adrenal axis [308]. In both animals and humans, high glucocorticoid levels have been linked to increased oxidative stress and subsequent neurodegeneration in the hippocampus [321, 322]. Glucocorticoid exposure in animals has also been linked to decreased hippocampal BDNF expression [323]. In a review of animal studies, both behaviorally-induced stress and mimicry of the stress response via corticosterone treatment significantly altered hippocampal structure and function [324]. In humans, elevated cortisol levels have also been associated with decreased total brain volume [325, 326] and increased white matter changes [327]. Additionally, cortisol release may directly affect AD pathology. In a double transgenic mouse model of AD, the strain that experienced increased oxidative stress showed accelerated amyloid deposition, tau phosphorylation, and gliosis [328]. These findings are consistent with older animal models that showed
Finding ways to manage stress may be helpful as a preventative measure in reducing the risk of dementia. One strategy is practicing meditation or mindfulness, which have been shown to have many health benefits for older adults. They are easily accessible, low-risk methods that can be practiced to reduce stress levels and improve sleep quality and mood, all of which may decrease the risk of developing MCI or dementia [332]. In a recent RCT, adults with memory concerns enrolled in a 12-week meditation program demonstrated significant improvements in measures of cognitive function, with sustained benefits after six months [333]. In another study, 14 patients with MCI reported that a mindfulness-based stress reduction program was helpful in lowering stress levels and promoting wellbeing [334]. Meditation and mindfulness practices have also been shown to diminish release of excess cortisol and increase cerebral blood flow within the frontal lobes, a brain region that is especially important for cognitive functioning [335]. Additionally, yoga has been found to have a positive impact on biomarkers of cellular aging, presumably by regulating stress and inflammatory responses [336]. Yoga has also been found to improve daily attention, memory, and executive function in individuals with MCI [337, 338]. Encouraging patients to manage stress may mitigate risk of cognitive decline.
Depression
There is conflicting evidence about whether depression in older adults is an independent risk factor for or a prodromal symptom of mild cognitive impairment and dementia. We suspect that depression may reflect either, depending on the individual. A review of observational studies reported that depression prior to age 60 was associated with a two- to four-fold increase in dementia risk, while late-life depression was associated with a two- to five-fold increase in dementia risk [339]. Not all studies, however, have found an association between both mid- and late life depression and dementia risk. One epidemiological study found that each self-reported depressive episode until approximately age 51, often of varying duration and sometimes with years separating episodes, was associated with an increased risk of developing dementia, suggesting that depression may be a dementia risk factor [340]. In individuals 51 and younger, those who had one elevated symptom of depression demonstrated an 87% increased risk of dementia [340]. In contrast, a study of 10,189 individuals followed over a 27-year period beginning at age 45 found that depressive symptoms in midlife, regardless of severity or duration, were not associated with a greater risk of dementia [341]. In the same study, however, depressive symptoms in late life, and specifically in the decade preceding the onset of dementia, were associated with an increased risk of dementia [341]. These results suggest that depression may be a feature of the “preclinical” phase of dementia rather than an independent risk factor. It is important to note that studies of late-life depression are highly variable in their timing and length of follow up, making it difficult to define depression as a risk or prodromal factor. Illustrating the complexity of the data, a meta-analysis of 34 studies on depression and dementia suggested that early or midlife depression may be a risk factor, while late-life depression is commonly a prodromal symptom [342].
Despite the conflicting evidence about its connection to dementia risk, depression has been associated with decrements in general cognitive abilities such as processing speed, attention, and executive control [343, 344]. Approximately 30–40% of depressed older adults exhibit signs of executive dysfunction during cognitive examination [345]. When compared to healthy controls, depressed adults over the age of 60 performed significantly worse on measures of episodic memory, language processing, executive function, and processing speed [346]. Notably, those who received psychotherapy for late-life depression exhibited a significant improvement in processing speed after 12 weeks of treatment [347]. Additionally, depression is known to negatively impact sleep integrity. Adults that have shortened or fragmented sleep have been found to report subsequent, suboptimal cognitive processing [348, 349]. See “Maintain adequate sleep” for further discussion on sleep, cognition, and dementia risk.
Proposed pathways for increased dementia risk include the direct physiological effects of depression on stress hormones, hippocampal volume, and neuronal growth factors [342]. In a study of 218 older adults, five years of untreated depression accurately predicted a decrease in left hippocampal volume, which was associated with cognitive impairment [350]. Additionally, Sawyer and colleagues found that depressed individuals had increased atrophy of the right hippocampus over a four-year period. This atrophy was an accurate predictor of decreased scores on the Mini-Mental State Exam [351]. The exact relationship between hippocampal volume loss and depression is unknown, but animal studies suggest that depression may contribute to the pathophysiology of neurotoxic damage [352] or decreased levels of neuronal growth factors, including BDNF [353]. Animal models of depression have demonstrated that the administration of exogenous BDNF can reverse depressive-like behaviors, including reduced exploration and behavioral despair, which are connected to decreased synaptic density in the hippocampus [353].
Despite uncertainty about the causal relationship between depression and dementia, it seems prudent to treat depression as a potential way to help prevent further stress on neurocognitive networks vulnerable to impairment.
Maintain adequate sleep
Adequate sleep is important in order to maintain brain health throughout life. The U.S. National Sleep Foundation recommends 7–9 hours of sleep per night for adults under 65 years of age, and 7-8 hours of sleep for adults 65 years and above [354]. Epidemiological evidence has shown that poor and disrupted sleep are associated with decreased cognitive performance [355], increased rate of cognitive decline [356], and elevated risk of cognitive impairment [357, 358], AD [356–359], and all-cause dementia [359, 360].
Getting enough sleep is imperative for healthy cognitive aging, as it plays a key role in consolidating memories, maintaining cognitive performance, and clearing potentially neurotoxic metabolites, including Aβ, that build up during wakefulness [361, 362]. In animal models, chronic sleep deprivation has been linked to increased inflammation, increased neuronal death, decreased neurogenesis, increased volume loss in the hippocampus and medial prefrontal cortex, and increased Aβ and tau pathology [363]. Studies in humans have shown that shorter sleep duration and lower quality of sleep are associated with increased systemic inflammation [364], decreased amyloid clearance [362, 365], and greater Aβ burden over time [366–368]. An RCT by Ooms and colleagues randomized cognitively healthy middle-aged men to a night of unrestricted sleep or total sleep deprivation. Unrestricted sleep was associated with the expected, normal morning decrease of Aβ42 levels in the cerebrospinal fluid compared to sleep deprivation after a single night, which interfered with this decrease [365]. This result suggests that poor sleep may have direct effects on the homeostatic fluctuations and clearance of aggregated neurotoxins, which may help explain the association of poor sleep with neurodegenerative processes.
Sleep-disordered breathing
Sleep-disordered breathing (SDB) is a common reason for poor and disrupted sleep in adults. Obstructive sleep apnea (OSA) is a type of SDB in which a person has frequent episodes of absent breathing (apneas) and/or slow, shallow breathing (hypopneas) linked to complete or partial blockage of air movement, respectively. These events often result in arousal from sleep and decreases in blood oxygen saturation that interfere with restorative sleep [369]. Globally, nearly one billion adults are estimated to have OSA, though most cases are undiagnosed and untreated [370]. SDB, and particularly OSA, is linked to several factors that may increase risk of cognitive decline, including sleep disruption, increased inflammation and oxidative stress, and cardiovascular comorbidities [371]. Indeed, epidemiological evidence suggests that SDB may increase risk of MCI, all-cause dementia, and AD [358, 372] and decrease the age of onset of MCI and AD dementia [373]. Promisingly, observational studies [373], prospective clinical studies [374], and RCTs [375–377] have demonstrated that use of continuous positive airway pressure (CPAP) seems to at least partially mitigate the negative effects on cognition and dementia risk associated with SDB. Treatment of SDB with CPAP is also associated with improved white matter integrity and decreased Aβ burden in cognitively healthy older adults [374, 378]. Patients should be screened regularly for SDB and other sleep disorders, because diagnosis and treatment may play an important role in reducing risk of cognitive decline for many of them.
Manage hearing and visual impairments
Living with hearing or vision impairment can be difficult and even debilitating, especially if left untreated. Epidemiological evidence has suggested that hearing, vision, or combined sensory impairment can increase the risk of developing cognitive impairment and dementia and increase the rate of cognitive decline [379–388]. One meta-analysis found that hearing impairment in midlife nearly doubled the risk of developing dementia in late life [389]. While most epidemiological studies have found a link between diminished hearing or vison and cognitive impairment or dementia, some have found no significant effect of either sensory limitation [390] or a significant effect of hearing but not vision impairment [391].
The precise mechanism by which hearing and vision impairment affect cognition and dementia risk is still largely unknown. Several possible mechanisms have been presented. One possibility is that sensory deficits may have a direct impact on the brain. A study by Lin and colleagues found that hearing loss in humans was associated with an accelerated rate of brain atrophy in the whole brain and, regionally, in the right temporal lobe (including the superior, middle, and inferior temporal gyri) and the parahippocampal cortex [392]. Animal models of adult deafness have shown that gradual hearing loss is associated with decreased spatial memory, decreased hippocampal plasticity, and changes in neurotransmitter receptor expression [393]. The association between hearing loss and these changes in the brain may be mediated by reduced neural stimulation of the auditory cortex, decreased auditory signal strength over time, or some shared neuropathologic or intrinsic cellular aging process that affects both cochlear and brain aging [392, 394]. Another possibility is that other biological factors related to aging and microvascular pathology underlie the relationship, as these factors can contribute to both sensory deficits and impaired cognition [389, 395]. Our own research suggests that an individual’s efforts to manage degraded sensory signals, as occurs in vision impairment, may deplete limited cognitive processing resources, which likely contributes to increased cognitive difficulties [396]. Finally, hearing and vision loss can undermine an individual’s daily functioning by reducing ability to participate in physical, social, or cognitive activities [395], and can result in depression [397], social isolation, or loneliness [398], which are independent risk factors for worsened cognitive outcomes and dementia. A recent study by Maharani and colleagues found that conditions of depression and loneliness at least partially mediated the effects of hearing loss on cognition [398]. The connection between hearing and vision impairment and dementia is likely due to a combination of several of these factors. Further complicating the evidence, however, a recent study by Brenowitz and colleagues found that impaired hearing was associated with increased neurofibrillary tangle burden prior to, but not after, the onset of cognitive impairment, suggesting that impaired hearing may also be a preclinical marker of tau-related neurodegeneration [399]. More research is needed to fully elucidate the relationship between sensory deficits and dementia risk.
Observational cohort studies have suggested that correcting hearing or vision impairment may boost cognition and counter a patient’s trajectory of cognitive decline [400–403]. RCTs are needed to further investigate these findings and determine exactly what factors might explain treatment responses and impact risk. In the meantime, however, widespread efforts to treat hearing and vision loss have become increasingly important as the population ages. Two of the most easily remediable visual impairments, uncorrected refractive error (URE) and cataracts, make up 75% of the total visual impairments in older individuals (42% for URE and 33% for cataracts) [404]. Recent investigations funded by the National Eye Institute have predicted that the number of individuals in the U.S. with URE will double between 2015 and 2050, increasing from 8.2 million individuals to 16.4 million individuals [405]. Disabling hearing loss is predicted to grow at a similar rate, increasing from 466 million individuals to 900 million individuals globally between 2018 and 2050 [406]. Ensuring patients maintain updated eyeglass prescriptions, undergo cataract surgery if appropriate, and use hearing aids may help to mitigate risk of cognitive decline and dementia. It is our contention that patients should not expend limited cognitive resources on simply trying to decode sounds or decipher visual images.
Avoid medications with anticholinergic properties
Several classes of medications have been investigated to determine their effect on brain health and cognition. Thus far, medications with anticholinergic properties have the most conclusive evidence linking them to impaired cognition and increased risk of cognitive decline. Several epidemiological and cohort studies have demonstrated that use of anticholinergic medications is associated with decreased cognitive performance on tests of memory, executive function, and visual attention and increased risk of dementia [407–415]. A recent study of 688 cognitively normal older adults found that use of anticholinergic medications was associated with increased rates of memory and language decline and elevated risk of progression to MCI. These effects were enhanced in individuals who also had genetic risk of AD or cerebrospinal fluid biomarkers for AD [414]. One common tool used to quantify the effects of anticholinergic medications is the Anticholinergic Cognitive Burden (ACB) scale, a 3-point scale based on the strength of the medication’s anticholinergic properties in the brain as determined by receptor affinity assays and expert-based consensus of clinically relevant cognitive effects [416]. Observational studies that use the ACB scale have confirmed that medications with higher ACB scores are linked to increased risk of dementia [409, 410]. A recent cohort study of 8,216 older adults found that recurrent use of anticholinergic medications with the highest ACB score of 3 (but not 1 or 2) was associated with a 68% increased risk of developing dementia during a ten-year follow-up period [409]. When separated by medication class, antidepressant, antiparkinson, antipsychotic, and antimuscarinic bladder medications with anticholinergic properties, specifically, have been associated with increased risk of dementia [410, 411]. Other classes of drugs with anticholinergic properties, including antihistamines, gastrointestinal antispasmodics, and skeletal muscle relaxants, have not been definitively linked to increased risk of dementia [410, 411], although this does not preclude cognitive risks from use of these medications.
Importantly, not all studies have found a clear link between anticholinergic medication use and dementia risk. For example, a cohort study of 3,690 older adults found that, while strong anticholinergic medications more than doubled risk of MCI, there was no association between anticholinergic medications and dementia risk [417]. As the authors acknowledged, these results were likely impacted by the short duration of the study (one year).
There are several potential mechanisms by which anticholinergic medications may increase risk of cognitive decline and dementia. By definition, anticholinergic medications block cholinergic transmission, which is critical for cognitive processes such as memory and attention. A recent study by Chhatwal and colleagues using functional connectivity MRI in humans found that anticholinergic drugs disrupt neural network connectivity involved in learning and memory, which may have subsequent effects on network-based resistance to neuropathologic change [418]. Limited evidence also suggests that anticholinergic medication use may be associated with increased brain atrophy in humans [415]. Studies of animals and
More research is needed to determine if discontinuing use of anticholinergic medications can improve current cognitive performance or counter the increased risk of cognitive decline and dementia that prior use may have conferred. In one population-based cohort study of 4,128 older adults, discontinuing use of anticholinergic medications was associated with a decreased risk of dementia over a four-year follow-up period [412]. To date, only a few RCTs have investigated the cognitive benefits of interventions to decrease anticholinergic medication burden and results remain inconclusive [421, 422]. In one study conducted by Kersten and colleagues, 87 older adults with no dementia (
In the meantime, current evidence suggests that prescribing medications with strong anticholinergic properties should be avoided when possible, and efforts to reduce or eliminate these medications in older patients already on them seem prudent.
Other medications that may affect cognition
The relationship between dementia risk and other medications like benzodiazepines, proton pump inhibitors (PPIs), and statins has also been investigated. Although the evidence on anticholinergic medications seems to point toward increased risk of dementia, the current evidence on these other medications is less conclusive.
Concerns have been raised about the negative impact of prolonged benzodiazepine use. Some observational evidence suggests that benzodiazepine use is associated with increased risk of dementia [424–427], and that risk increases with exposure duration and strength [426, 427]. A recent meta-analysis of 10 observational studies found that benzodiazepine use was associated with a 51% increased risk of dementia. Risk was higher for long-term benzodiazepine use (> 3 years) and benzodiazepines with a longer half-life (> 20 hours) [428]. Conversely, several observational studies have found no link between benzodiazepine use and dementia risk [409, 429–432]. For example, a recent Danish nationwide-cohort and nested case-control study did not find an association between use of benzodiazepines and risk of dementia, even for cumulative exposure or when divided into short- and long-acting drugs [429]. Notably, even in studies that found no association overall, there was an increased risk of dementia with small amounts of benzodiazepine use (1–30 total standard daily doses over a ten-year period) in older adulthood [431], or new benzodiazepine use in the year prior to dementia diagnosis [430]. Benzodiazepine use during those periods of time could reflect the treatment of anxiety or other prodromal neuropsychiatric symptoms of dementia. More research on benzodiazepine use and dementia risk is needed in order to determine causality and the impact of potentially confounding variables. In the absence of conclusive evidence, expert consensus currently suggests that appropriate benzodiazepine use will not lead to the development of dementia [433]. Still, to moderate risk of potential long-term effects, clinicians should consider prescribing shorter-acting benzodiazepines for limited periods of time when possible.
The evidence on PPIs, such as omeprazole, pantoprazole, and lansoprazole, and risk of dementia is similarly mixed. Some observational studies have found a link between PPI use and risk of dementia [434–436] and AD [435]. In one of these studies, a significant association was also observed between cumulative PPI use and dementia risk [436]. Conversely, many other observational studies have found no association between PPI use and risk of dementia [437–439] or AD [440, 441] regardless of cumulative exposure level or pattern of use. Meta-analyses conducted to date seem to confirm the lack of association [204, 442]. Further complicating the picture, a few studies have found that PPI use is associated with a decreased risk of cognitive decline [443] or dementia [444], suggesting a possible protective effect. For example, a prospective cohort study of 10,486 adults over 50 years old found that continuous or intermittent PPI use was associated with decreased risk of decline in cognitive function and decreased risk of conversion from MCI to dementia compared to never use. Overall, the evidence on PPI use and brain health does not point conclusively in any direction.
Statins, or HMG-CoA reductase inhibitors, are widely prescribed in the treatment of cardiovascular and cerebrovascular diseases. In addition to lowering cholesterol levels, statins have been shown to exhibit many other effects, including improving endothelial function, inhibiting platelet activation, and decreasing inflammation [445], thereby reducing the risk of cardiovascular disease and stroke [446]. Statins have also been associated with decreased cerebral Aβ deposition [447, 448]. Accordingly, several observational studies have linked statin use to decreased incidence of all-cause dementia [449–451] or decreased rate of cognitive decline [452]. Meta-analyses of pooled data seem to support these results [271, 454]. However, intervention trials completed to date have not supported a link between statin use and cognitive status [233, 455]. For example, in the HOPE-3 study (as discussed above in the section on “Hypertension”), 2,361 older adults were randomized to receive blood-pressuring-lowering medication (candesartan plus hydrochlorothiazide) or placebo and lipid-lowering medication (rosuvastatin) or placebo once daily until study completion, an average of 5.7 years later. Neither blood pressure lowering, lipid lowering, or their combination had a significant effect on cognitive decline over the 5.7-year follow-up period [233]. Some observational studies have also found no link between statin use and risk of dementia [456], rate of cognitive decline [457], or neuroimaging biomarkers of AD [458]. Additionally, a 2017 retrospective cohort study of 3,500 adults, ages 51 to 100, found that statins increased the risk of cognitive impairment and dementia [459]. As discussed, however, data from most studies conducted to date do not seem to support negative effects of statins on brain health. As with the other medications reviewed here, more research, including additional RCTs, is needed to further clarify the effects of statins on cognition and dementia risk. Despite the results of some epidemiological studies that suggest a correlation between statin use and reduced cognitive decline, there is currently no compelling reason to initiate treatment with a statin for the purpose of improving cognitive status or preventing decline. Likewise, there is no persuasive evidence that statin use leads to a deterioration of cognitive abilities.
Nutraceuticals and supplements
Nutraceuticals and supplements have been the subject of a number of recent reviews [460–462]. Although they fall outside the scope of this article, nutraceuticals and supplements are worth briefly mentioning due to the rapid growth of the industry in recent years. While no official legal or regulatory definition exists to date, a “nutraceutical” is generally defined as a food or food-based product that provides medical or health benefits [463]. Nutritional supplements are dietary add-ons, like vitamins, proteins, or minerals, that are also often found naturally in foods. These products are not regulated by agencies such as the U.S. Food and Drug Administration, and their safety and efficacy is often based on testimonials rather than scientific research [464]. A 2018 meta-analysis of 38 studies examined the human evidence of various supplements, including omega-3 fatty acids, soy, gingko biloba, B vitamins, vitamin D plus calcium, vitamin C or beta-carotene, multi-ingredient supplements, and other over-the-counter interventions, and found insufficient evidence that any of these supplements improved cognitive performance in a clinically meaningful way or reduced the risk of cognitive decline or dementia [461]. Despite the minimal scientific evidence supporting their efficacy, nutraceuticals and supplements targeting memory have become increasingly popular due to the lack of disease-modifying treatment options for AD and other dementias. By marketing their products as memory- or cognition-enhancing, this multibillion-dollar industry seems to be taking advantage of aging individuals eager to retain their mental capacities [465].
Limit alcohol use
Alcohol use has been identified as a risk factor for cognitive decline and dementia, but some evidence suggests that certain levels of alcohol consumption may have a protective effect. Major health organizations recommend limiting alcohol use to a moderate level, defined as up to one drink per day for women and up to two drinks per day for men [158]. Exceeding this recommended amount is considered heavy alcohol use [158]. Epidemiological evidence on alcohol consumption points toward a U-shaped dementia risk curve, with mild to moderate drinking associated with the lowest risk of dementia compared to either abstention or heavy drinking [4, 466–470]. A non-linear relationship has also been observed between alcohol consumption and cognitive function, whereby alcohol abstinence is associated with a higher risk of impaired memory and executive function compared to moderate alcohol intake [471].
Alcohol use disorder (AUD) specifically has been associated with a particularly strong risk of all-cause dementia. A nationwide, retrospective cohort study involving 31.6 million adults in France found that AUD more than tripled risk of all-cause dementia. The association was particularly clear for early-onset dementia (before age 65), although underlying dementia pathology was not determined [469]. These results are especially concerning given the prevalence of AUD. As of 2018, an estimated 5.8% of adults had AUD in the U.S. alone [472].
The risks of excessive alcohol consumption on the nervous system are well established. Longstanding evidence has demonstrated heavy alcohol use is associated with structural and functional abnormalities of the brain [473]. In animals, long-term consumption of alcohol has been linked to increased neuroinflammation via microglial activation and upregulation of pro-inflammatory cytokines [474], decreased BBB integrity [475], and increased expression of AβPP and elevated Aβ deposition [476]. In humans, heavy drinking is linked to increased risk of cardiovascular disease and stroke [477, 478], increased neuroinflammation via microglial activation and upregulation of pro-inflammatory cytokines [474], thiamine deficiency (which can contribute to cognitive and neurological dysfunction) [479], and alcohol-related dementia or alcohol-induced persisting amnestic syndrome [479]. Neuroimaging and neuropathological studies have also shown widespread loss of white matter in individuals with diagnosed AUD [479].
Conversely, mild to moderate alcohol consumption in humans has been linked to decreased risk of cardiovascular events [478, 480] and decreased Aβ deposition in older adults [180], suggesting possible reduced risk of AD. The neuroprotective effect of mild to moderate alcohol consumption may be due to antioxidant polyphenols or ethanol itself, which have each been associated with decreased neuroinflammation and oxidative stress [480]. The Mediterranean diet, as discussed in the “Heart-healthy diet” section, allows for regular, mild- moderate alcohol intake and especially emphasizes the consumption of wine. Red wine, specifically, is abundant with antioxidant polyphenols such as resveratrol and has been linked to the best cardiovascular and brain health outcomes [480, 481].
It is important to note that there is conflicting evidence about both the benefits of mild-moderate alcohol consumption and the risks of heavy alcohol consumption. For example, a recent cohort study of 3,021 older adults found that alcohol consumption within recommended limits (i.e., 7.1 to 14.0 drinks per week) was not associated with decreased risk of dementia in cognitively normal individuals or individuals with MCI compared to drinking less than 1.0 drinks per week [482]. Exceeding 14.0 drink per week, however, was associated with the most severe cognitive decline in individuals with MCI [482]. A meta-analysis found that light to moderate drinking was associated with a 25% to 28% decrease in risk of AD, vascular dementia, and all-cause dementia compared to abstinence, but heavy drinking had no impact on dementia risk. The authors acknowledge that there could be a bias due to sampling or survival effects, as heavy drinkers are less likely to participate in or finish these studies and more likely to have comorbid health conditions [483]. More research is needed to understand the effect of various levels of alcohol consumption on brain health.
Despite outstanding questions about the effect of alcohol consumption on risk of cognitive decline and dementia, the general health risks of heavy alcohol use are widely acknowledged. Patients should be encouraged to limit their alcohol consumption to a mild to moderate level, especially if they are already experiencing cognitive difficulties. Evidence suggests that the best drinking pattern for cardiovascular and brain health may be one glass of red wine per day.
Protect the brain from physical and toxic injuries
Physical injury
Traumatic brain injury (TBI) results from an impact to the head that disrupts normal brain function [484]. The most common causes of TBI are falls and motor vehicle accidents [484]. TBI has been associated with an increased risk of cognitive decline and dementia. Physical injury to the brain reduces brain reserve, thus lowering an individual’s capacity to compensate for the development of neurodegenerative processes. Whether TBI directly promotes neurodegenerative processes, however, remains an area of ongoing research.
Many epidemiological and cohort studies suggest that physical injury of the brain of any severity can increase the risk of dementia [4, 485–489], or decrease age of onset of MCI [490] or AD [491]. A nationwide cohort study in Denmark found that a history of TBI, regardless of severity, was associated with a 24% increased risk of all-cause dementia diagnosis and a 16% increased risk of a clinical AD diagnosis. The risk of dementia was also increased with multiple events (22% increased risk for one TBI versus 183% increased risk for five or more TBIs) [485]. In a retrospective cohort study of 357,558 veterans with and without TBI, even mild TBI without loss of consciousness (LOC), was associated with a doubled risk of dementia [487]. Consistent with these results, a recent meta-analysis found that history of TBI of any severity was associated with an 84% increased risk of all-cause dementia [4].
In addition to lowering brain reserve, TBI may increase dementia risk by increasing neuroinflammation, white matter changes, and AD-related pathology, although the exact etiology varies depending on the nature, frequency, and severity of injury [492, 493]. Data suggest that, in some patients, TBI initiates long-term neuroinflammation and neurodegenerative processes [494, 495]. For example, an autopsy study of 52 individuals with moderate-severe TBI and 44 healthy, age-matched controls found evidence of ongoing TBI-initiated inflammatory processes including activated microglia, axonal degeneration, and atrophy of the corpus collosum for up to 18 years following a single TBI [494]. Additionally, TBI has been linked to increased AD pathology in the brain. In animal models of TBI, brain injury is associated with increased expression of AβPP [496, 497] and accumulation of Aβ [498, 499] and tau [496]. Similarly, imaging and autopsy studies in humans have shown that TBI is linked to increased Aβ and tau accumulation [500]. The
Not all studies have found a clear effect of TBI on the development of dementia. One study of 706 older adults from the National Alzheimer’s Coordinating Center found that a history of TBI with LOC did not impact the course of cognitive decline in participants with or without AD [502]. Another recent study by Sugarman and colleagues found that, in 4,761 deceased participants from the National Alzheimer’s Coordinating Center, remote TBI with LOC was not associated with AD neuropathology in cases of autopsy-confirmed AD or with late-life cognitive performance in individuals with or without autopsy-confirmed AD [503]. It is worth noting that both of these studies relied on self-report data to determine history of TBI. Given these conflicting findings, more research is needed to understand the connection between TBI, cognitive decline, and AD pathology.
Even while research continues, it is wise to protect our brains from loss of cognitive capacity and brain reserve. It is important to encourage patients to wear protective equipment, especially seat belts and bicycle helmets, when participating in activities that are associated with the risk of head strike, and to reduce their risk of falls by staying physically active.
Toxic injury
Air pollutants nitrogen dioxide (NO2), carbon monoxide (CO), and fine ambient particulate matter (PM2.5) from traffic and wood burning [4, 504] as well as acute CO poisoning [505, 506] have been linked to increased risk of cognitive decline and dementia. Cohort studies have shown that living in an area with traffic-related air pollution, specifically, is associated with increased incidence of dementia [507–510]. For example, in a Canadian population-based cohort study of approximately 2.2 million older adults, individuals living within 50 meters of a highly-trafficked road had a 7% increased risk of dementia compared to individuals living greater than 300 meters away [507].
Air pollutants have also been associated with worsened cognitive performance. PM2.5 exposure has been associated with decreased performance in working memory and orientation [511], verbal learning [512], and visuospatial ability [513]. NO2 has been linked to decreased memory and executive function [512] as well as visuospatial ability [513]. A recent study of two cohorts in New York City investigated the link between long-term exposure to ambient air pollution and cognitive decline. Among 5,330 participants in the larger cohort, living in areas with higher levels of air pollution was associated with lower cognitive scores at baseline and more rapid rates of cognitive decline over time, after adjusting for sociodemographic factors, including race and ethnicity, education, and socioeconomic status. No association was found, however, in the smaller cohort with fewer repeat measures of cognition [514].
Air pollutants likely increase the risk of cognitive decline and dementia through several mechanisms [515]. Exposure to pollutants is linked to increased inflammation, oxidative stress, and Aβ accumulation in both animals [516–518] and humans [519–521]. An autopsy study of 47 clinically-healthy individuals who died suddenly, aged two to 45 years, found that exposure to high levels of air pollution was already associated with increased inflammation and Aβ accumulation in children and young adults [520]. Additionally, exposure to air pollution has been linked to increased risk of medical conditions that may worsen cognition. Long term exposure to PM2.5 has been associated with increased risk of acute coronary events [522] and cardiovascular mortality [523]. Short term exposure to NO2 and PM2.5 have been associated with increased risk of stroke and mortality due to stroke [524].
The effects of neurotoxic metals (e.g., lead, mercury, aluminum, etc.) and pesticides on dementia risk have also been investigated, but results remain inconclusive [515]. More research is needed to understand the connection between these toxic substances and dementia risk.
Although it may be difficult to eliminate pollution- and heavy metal-related exposures, it is important for clinicians and patients to understand the likely connection between these environmental hazards and cognitive decline and dementia. It is also important to understand that communities with overall lower socioeconomic status are more likely to be exposed to a higher concentration of air pollutants, and are therefore more likely to experience the health risks associated with air pollution [525]. Establishing public policies aimed at reducing air pollution in these communities is of paramount importance for mitigating this disparity. On an individual level, ways to reduce risk of toxic injury to the brain include installing a carbon monoxide detector at home and wearing protective equipment when working with toxic materials.
Multimodal interventions
Epidemiological studies have demonstrated that a combination of brain-healthy behaviors and lifestyles may yield a greater benefit than participation in any single activity [526–528]. For example, findings from two Chicago-based longitudinal studies indicated that a composite healthy lifestyle score, including moderate to vigorous physical activity, non-smoking, light to moderate alcohol consumption, MIND diet adherence, and late-life engagement in cognitive activities, was associated with a 27% decreased risk of AD for each additional healthy lifestyle factor [528].
In recent years, interventions that combine multiple behavior changes have also demonstrated a positive impact on cognition [529, 530] and a possible synergistic effect on dementia risk [531, 532]. For example, the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) study, a two-year study of 1,260 older individuals at risk for cognitive impairment, investigated a multimodal intervention that combined diet, exercise, cognitive training, and management of cardiovascular risk factors. Participation in the intervention was associated with improved BMI, dietary habits, physical activity, and cognition, including executive functioning and processing speed. Although the effect sizes measured were very small, the intervention was also associated with a 24% decreased risk of cognitive decline [531]. Not every multimodal behavioral trial, however, has shown such promising results. In the three-year Multidomain Alzheimer Prevention Trial (MAPT), a multimodal intervention for older adults, which included physical activity, nutrition, and cognitive training and consumption of polyunsaturated fatty acids, in combination or individually, had no significant effect on cognitive decline [533]. A recent report by Yu and colleagues, which analyzed many of the evidence-based suggestions for AD prevention, found insufficient evidence to draw any robust conclusions about multimodal interventions due to intervention heterogeneity and small sample sizes [534]. More clinical trials are needed to determine whether multimodal interventions are effective in preventing or delaying cognitive decline. Ongoing global research efforts, including the U.S. Study to Protect Brain Health Through Lifestyle Intervention to Reduce Risk (US-POINTER) and the Multimodal Interventions to Delay Dementia and Disability in Rural China (MIND-CHINA) study, are aiming to replicate the results of FINGER, and test the generalizability of a similar multimodal intervention to improve brain health [535, 536].
Promoting behavior change
As described in this review, there is growing consensus among health organizations about what to recommend to older adults in order to promote successful cognitive aging and reduce the risk of cognitive decline. However, a substantial gap remains between what healthcare providers and public health specialists think patients should do and what they are actually doing. For example, only 16% of adults between the ages of 65 and 74 and 10% of adults 75 years and over in the U.S. regularly meet the physical activity guidelines established by the Department of Health and Human Services [47]. Evidence that dementia risk is modifiable will not make a difference unless individuals actually change their behavior.
There are many barriers to translating knowledge about factors that may promote healthy cognitive aging into real-world practice. Changing human behavior often requires complex, multidimensional interventions, blending elements of psychology, social science, and medicine. Standard clinical practice does not systematically promote or monitor improvement of modifiable lifestyle changes, but a growing field of research is seeking to address this limitation. Recent efforts to characterize and standardize behavior change techniques (BCTs) have allowed researchers to define and replicate behavior change interventions across different populations and target behaviors [537]. Several techniques that have relied on principles derived from motivational interviewing (MI), behavioral activation (BA), and behavioral economics (BE) have shown promise in encouraging behavior change across many medical specialties and conditions.
MI focuses on reflecting and reinforcing the participant’s intrinsic motivation through a non-judgmental empathic alliance, increasing optimism, intent, and self-efficacy in patients [538–540]. Principles of MI are widely used in psychiatry and addiction medicine, where successful interventions have been developed to address several behaviors and conditions, such as smoking cessation [541] and alcohol use disorder [542]. In patient populations with or at risk for chronic health conditions, the incorporation of MI principles into behavior change counseling has effectively increased physical activity [543], weight loss [544, 545], and management of cardiovascular risk factors [546, 547]. MI has been shown to be effective even in brief or time-limited interventions [542, 548].
BA uses tangible, actionable goal setting to increase accountability and self-efficacy [549]. BA is a collaborative process between health providers and patients that accounts for individual goals and obstacles to increase positive activities and improve patients’ ability to self-monitor activity levels [550, 551]. BA also has been used to promote brain-healthy behaviors, including cognitive, physical, and social activity, relying on action plans with visual cues, written schedules, step-by-step sequencing, and procedural memory to account for cognitive deficits [549].
BE combines principles of psychology and economics to optimize behavior change. More traditional clinician education and counseling tends to appeal to patients’ slow, logical, rational cognitive system by providing facts and evidence, whereas behavior is often driven by fast, automatic, unconscious thinking [552]. BE focuses on the latter; it accounts for individuals’ often limited willpower and rationality and their use of mental shortcuts in decision-making [553]. BE-guided interventions often involve making changes to individuals’ environments or daily structures to try to develop positive, automatic, habit-based behaviors [554]. This approach has been employed in numerous public health settings, including primary care physical activity counseling [555], nutritional counseling [556], sleep interventions [557], and heart failure clinical care [558].
Table 3 offers examples of motivational strategies using each of these three principles and ways to implement them into one’s own medical practice or a program across a health system.
Behavior change toolbox
BA, behavioral activation; BCT, behavior change technique; BE, behavioral economics; MI, motivational interviewing.
Recent studies have used BCTs to change behavior in the service of promoting brain health [549, 559]. A study by Rovner and colleagues randomized Black individuals with MCI to a two-year intervention of BA for cognitive, physical, and social activities versus supportive therapy. Results demonstrated that BA participants engaged in more cognitive activities and exhibited less memory decline and greater functional stability than supportive therapy participants [549]. In the Brain Health Champion study, our team demonstrated the efficacy of a low-cost, health-coaching intervention in older adults with MCI or mild dementia. Using evidence-based BCTs, including MI and BA, the health-coaching intervention was associated with increased adherence to brain-healthy behaviors and improved self-reported quality of life [559]. The results of these studies demonstrate the promise of employing models of evidence-based BCTs to encourage the adoption of brain-healthy behaviors. More research is needed to investigate the most effective and efficient ways for clinicians to help patients to implement behavior change, including the use of mobile health platforms and other innovative technologies.
CONCLUSION
The converging evidence from epidemiological, basic science, human proof-of-concept, and available RCT studies provides a strong justification for lifestyle-modification- and brain-health-promoting interventions to help prevent and delay cognitive decline. Table 4 provides a brief summary of lifestyles and behaviors reviewed in this paper, the main evidence supporting their role in fostering brain health, and emerging consensus recommendations. The factors discussed hold promise for decreasing the risk of cognitive impairment and dementia. Of particular importance is physical activity, cognitive and social stimulation, and management of cardiovascular risk factors.
Summary of evidence and consensus recommendations for different lifestyle/behavior interventions
ACC, American College of Cardiology; AD, Alzheimer’s disease; AHA, American Heart Association; BBB, blood-brain barrier; BDNF, brain derived neurotrophic factor; BMI, body mass index; CDC, Centers for Disease Control and Prevention; DASH, Dietary Approaches to Stop Hypertension; DM, diabetes mellitus; IGF-1, insulin-like growth factor 1; MeDi, Mediterranean diet; MIND, Mediterranean-DASH Intervention for Neurodegenerative Delay; OSA, obstructive sleep apnea; RCT, randomized controlled trial; VEGF, vascular endothelial growth factor.
It is important to acknowledge that none of the recommendations made in this article have been definitively proven in the way that some experts insist is required for evidence-based treatment guidelines. In fact, as noted in the introduction, “gold-standard” large-scale RCTs that follow participants over years to decades may never be conducted for many of these recommendations.
Thus, these guidelines are best viewed as provisional ones, based on the highest-quality evidence currently available. In this context, it is worth considering the negative consequences of being wrong about specific lifestyle recommendations or medical interventions that turn out not to reduce the risk of cognitive decline and dementia. There are potential opportunity costs associated with following these guidelines. The time, effort, and resources individuals devote to following them could be invested in carrying out other activities or making different choices. Moreover, many of the recommendations involve putting restrictions on one’s life, for example, reducing access to certain enjoyable foods, alcohol, or cigarette smoking, or feeling constrained by having to wear seat belts and helmets or by having to get seven or more hours of sleep at night. Additionally, individuals may be embarrassed by wearing hearing aids or seeking treatment for depression, or may feel ashamed about being overweight or not exercising enough. Individuals may also feel uncomfortable carrying out challenging cognitive activities or doing aerobic exercise. We recognize the potential costs of these actions, many of which recapitulate the reasons why some of our patients do not embrace them.
Of note, even if these lifestyle choices and healthcare efforts do not prove to directly mitigate cognitive decline or delay dementia, they are often beneficial for other reasons. For example, management of cardiovascular risk factors, such as hypertension, DM, and elevated cholesterol, reduces the chances of suffering from a stroke or myocardial infarction.
Cardio- and cerebrovascular risk can be lowered by following a heart-healthy diet or participating in regular physical activity, the latter of which also diminishes the incidence of falls. Smoking cessation reduces the risk of heart disease, stroke, and lung disorders. Adequate sleep has been linked to improved mood, sense of wellbeing, and cognitive performance. Use of seat belts and helmets helps to diminish the severity of traumatic brain injury due to accidents. Thus, even if the recommendations reviewed in this paper do not directly result in reduced risk of cognitive deterioration and dementia, there are other very compelling reasons for providers and health systems to promulgate them. On balance, we are comfortable with the risk/benefit profile of this set of recommendations. However, clinicians will need to review the data and decide for themselves. Clinicians are in an optimal position to educate their patients about available evidence, explain why they favor certain lifestyle choices, and use behavior-change techniques to assist patients in achieving their goals.
Given the epidemiological evidence that more than a third of ADRD cases may be due to modifiable risk factors, helping individuals make brain-healthy behavior changes could improve the lives of millions of older adults around the world and reduce the social and economic burdens associated with cognitive decline and dementia. The lifestyles and behaviors examined in this review are not exhaustive, and ongoing research continues to refine our understanding of the factors that promote successful cognitive aging. We expect that work in this field will continue to expand rapidly and anticipate that our next update will need to be completed in much less than a decade.
Footnotes
ACKNOWLEDGMENTS
Funding for this work came from the Wimberly Family Clinical Care Research Fund, the Gaudet Family Research Fund, and the Alzheimer Innovation Fund, Brigham and Women’s Hospital. In addition, the Laboratory of Healthy Cognitive Aging at Brigham and Women’s Hospital has been strongly supported by the Muss family, the Mortimer/Grubman family, and the Herman F. Woerner Trust. The Patti Piper Memorial Fund has also supported this work and related research endeavors. The authors would like to acknowledge Hope Schwartz for her work on several prior brain health research efforts, including the ongoing Brain Health Champion Study.
