Abstract
Background:
Emerging evidence indicates a central role of gliosis in Alzheimer’s disease (AD) pathophysiology. However, the regional distribution and interaction of astrogliosis and microgliosis in association with amyloid-β (Aβ) still remain uncertain.
Objective:
Here we studied the pathological profiles in autopsy AD brain by using specific imaging tracers.
Methods:
Autopsy brain tissues of AD (
Results:
Conclusion:
The use of multi-imaging tracers revealed different regional pattern of changes in autopsy AD brain with respect to amyloid plaque pathology versus astrogliosis and microgliosis.
Keywords
INTRODUCTION
Alzheimer’s disease (AD) involves complex pathophysiology, featured by amyloid-β (Aβ) deposits, neurofibrillary tangles, gliosis, and neural loss. Emer-ging evidence implies that Aβ, astrogliosis, and mi-crogliosis play important roles at early phases of AD [1–5]. However, the regional distribution, consequence of gliosis in relation to Aβ deposits, whether beneficial or harmful, as well as accurate imaging biomarker for gliosis remains elusive.
Autopsy analysis and
Accurate detection of neuroinflammation and gliosis
Microglia activation has been assessed by PET using translocator protein (TSPO) tracers [31, 32], such as first-generation 11C-PK11195 [33], second-generation 11C-DAA1106 [34], 11C-PBR28 [35], 18F-FEMPA [36], 11C-GE180 [37], and 18F-DPA-714 [38]. Results from TSPO imaging in MCI and AD have been inconclusive: increase or no change in TSPO retention comparing AD to control group has been reported [33, 39–41]. This could be due to several reasons, including 1) tracer specificity [42]; 2)
To understand the regional distribution of fibrillar Aβ deposits in relation to astro- and microgliosis, we determined the binding of 3H-florbetaben/3H-PIB, 3H-L-deprenyl, and 3H-PK11195/3H-FEMPA in autopsy brain tissues from AD and control cases by using homogenate binding assays, autoradiography, enzyme-linked immunosorbent assay (ELISA) for astrocyte marker glial fibrillary acidic protein (GFAP). This study demonstrates a clear regional correlation between amyloid plaque deposition and astrogliosis in AD.
MATERIALS AND METHODS
AD and control autopsy brains
Fifteen AD cases (mean age 70.4±8.5 years; mean postmortem delay 5.1±0.9 h), each with a clinical diagnosis confirmed by pathological examination (NINCDS-ADRDA criteria), and twelve control cases (mean age 76.6±10.9 years; mean postmortem delay 7.0±3.8 h) were included in this study (Table 1). Early-onset AD (EOAD) and late-onsetAD (LOAD) were classified based on the age of on-set of the clinical symptoms (before or after 65 years of age). Autopsy brain tissues from the frontal, parietal and temporal cortics, hippocampus, caudate nucleus, and cerebellum were obtained from the Netherlands Brain Bank (NBB), Netherlands. The disease relevant cortical and subcortical brain regions, as well as the reference brain region in amyloid PET imaging (cerebellum) were selected. All materials had been collected from donors or from whom a written informed consent for a brain autopsy and the use of the materials and clinical information for research purposes had been obtained by the NBB. Frozen brain tissues from the left hemisphere (all cases) were homogenized in ice-cold 5×0.32 M sucrose containing 10μl/ml protease inhibitor; brain tissues from 4 AD and 4 control cases were cryostat sectioned at 10μm and stored at –80°C. Protein concentration was determined using DC protein assay (Bio-Rad Laboratories AB, Sweden). The apolipoprotein E (
Demographics
EOAD, early onset Alzheimer’s disease; LOAD, late onset Alzheimer’s disease; PM, postmortem.
The study was conducted according to the principles of the Declaration of Helsinki and subsequent revisions. All experiments on autopsied human brain tissue were carried out in accordance with ethical permission obtained from the regional human ethics committee in Stockholm (permission number 2011/962/31-1), the medical ethics committee of the VU Medical Center for the Netherlands Brain Bank tissue (permission no. 1998-06/5).
Materials
3H-florbetaben ([N-
Characterization of 3H-florbetaben, 3H-L-deprenyl, 3H-FEMPA, and 3H-PK11195 binding properties in AD and control brains
Saturation binding assays with 3H-florbetaben (SA 63.0 Ci/mmol) were performed in the frontal cortex homogenates of AD (
Saturation binding assays with 3H-L-deprenyl (SA 80.0 Ci/mmol) were performed in the frontal cortex homogenate of AD cases (
Measurements of regional 3H-florbetaben (5 nM), 3H-L-deprenyl (6 nM), 3H-PK11195 (2.5 nM), and 3H-FEMPA (2.5 nM) binding were performed in the frontal, parietal, temporal cortex, hippocampus, caudate nucleus, and cerebellum of 12 AD and 12 control cases. The incubation time, incubation buffer, and washing buffers were same as in aforementioned saturation assay respectively. Measurement of 3H-PIB (1 nM, SA 85.0 Ci/mmol) binding was performed in the frontal and parietal cortex of the same 12 AD and 12 control cases by incubating in 0.1 M PBS + 0.1% BSA buffer (pH 7.4) for 2 h at RT. NSP binding was determined in the presence of 1μM BTA-1. The termination and counting procedure for 3H-PIB is the same as that for 3H-florbetaben assay. The 3H-PIB specific binding was calculated as the difference between total and NSP binding and expressed as pmol/g tissue.
ELISA measurement of GFAP level
The level of GFAP expression was determined by using ELISA as described previously [46] in the frontal, parietal, temporal cortex, hippocampus, caudate nucleus of 12 AD and 12 control cases. Brain tissue homogenates were sequentially incubated with monoclonal anti-GFAP antibody (Merck KGaA, Germany), alkaline phosphatase-conjugated anti-mouse IgG (Vector, USA), AP and substrate (p-nitro-phenyl-phosphate; Bio-Rad) in a rabbit anti-GFAP antibody pre-coated (Dako, Denmark) microtiter plate. Optical density was assessed in a microtiter plate reader (Tecan, Switzerland) at 405 nm. The level of GFAP was analyzed using Soft Max Pro Plus software (Molecular Devices, USA).
Autoradiography using 3H-florbetaben, 3H-L-deprenyl and 3H-FEMPA in AD brain tissue slices
Adjacent slices (10μm) from the frontal cortex (
Statistical analyses
Data was analyzed using GraphPad Prism software 7.0 (GraphPad Software Inc, USA). Kd and Bmax values from saturation binding curves were determined using a one-site non-linear fitting model. Non-parametric
RESULTS
Demographic information
Fifteen AD (9 female/6 male) and twelve control cases (6 female/6 male) were included in this study (Table 1). Among the AD cases, 8 were EOAD and the other 7 were LOAD cases. Among the control cases, 2 were younger than 65 years old, and 10 were older than 65 years old. Age and gender did not significantly differ between AD and control groups analyzed by using
Binding characterization
Saturation binding studies with 3H-florbetaben at 0.01–40 nM revealed a high-affinity binding site of Kd = 11.8±1.4 nM in the frontal cortex of AD by non-linear fitting (Table 2). Saturation binding studies with 3H-L-deprenyl at 0.01–25 nM revealed a high-affinity binding site of Kd = 10.2±0.8 nM in the frontal cortex of AD cases (
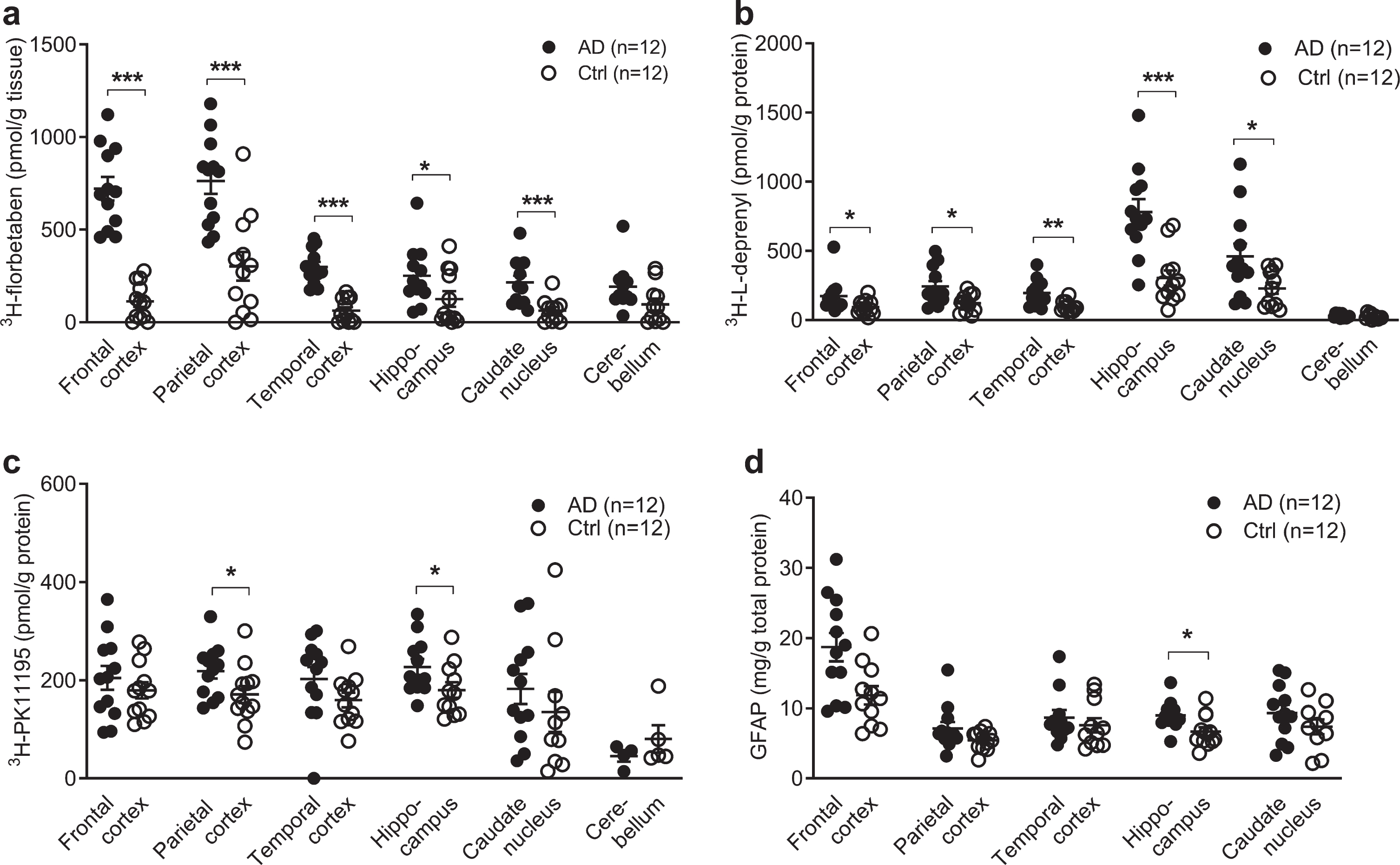
Regional 3H-florbetaben, 3H-L-deprenyl, 3H-PK11195 binding and level of GFAP expression in AD and control brain. a) 3H-florbetaben (5 nM) binding in the brain homogenate from AD and control cases. b) 3H-L-deprenyl (6 nM) binding in brain tissue homogenates from AD and control cases. c) 3H-PK11195 (2.5 nM) binding in brain tissue homogenates from AD and control cases. d) Level of GFAP expression in the same AD and control cases; Significant differences between AD and control groups are indicated by *
Binding properties for 3H-florbetaben, 3H-L-deprenyl, 3H-FEMPA, and 3H-PK11195 in the frontal cortex of AD cases
Kd, dissociation constant; Bmax, maxium number of binding sites. Results are expressed as mean±standard deviation.
Different regional 3H-florbetaben, 3H-L-deprenyl, 3H-PK11195, and GFAP distribution in AD and control brain
3H-florbetaben (5 nM) showed significant increa-ses in the frontal cortex (
The difference between EOAD, LOAD, and control group was analyzed using two-way ANOVA comparison (Table 3). High 3H-florbetaben binding was observed in in all three cortical regions in EOAD and LOAD compared to control. The 3H-L-deprenyl binding was higher in the hippocampus while lower in the caudate nucleus of LOAD compared to EOAD. No difference was observed in the regional 3H-florbetaben, or 3H-PK11195 binding between EOAD and LOAD group.
Comparison of 3H-florbetaben, 3H-L-deprenyl, 3H-FEMPA, and 3H-PK11195 binding between EOAD, LOAD and control
CN, caudate nucleus; Ctrl, control; EOAD, early onset Alzheimer’s disease; HIP, hippocampus; LOAD, late onset Alzheimer’s disease; ns, not significant; PC, parietal cortex; TC, temporal cortex. Values represent Mean±standard deviation.
Autoradiography showed high 3H-florbetaben in the cortex, and 3H-L-deprenyl signal in the dental gyrus
Autoradiography using 3H-florbetaben (2.5 nM) showed increased signal in the grey matter in the frontal cortex slices from AD compared to control cases (
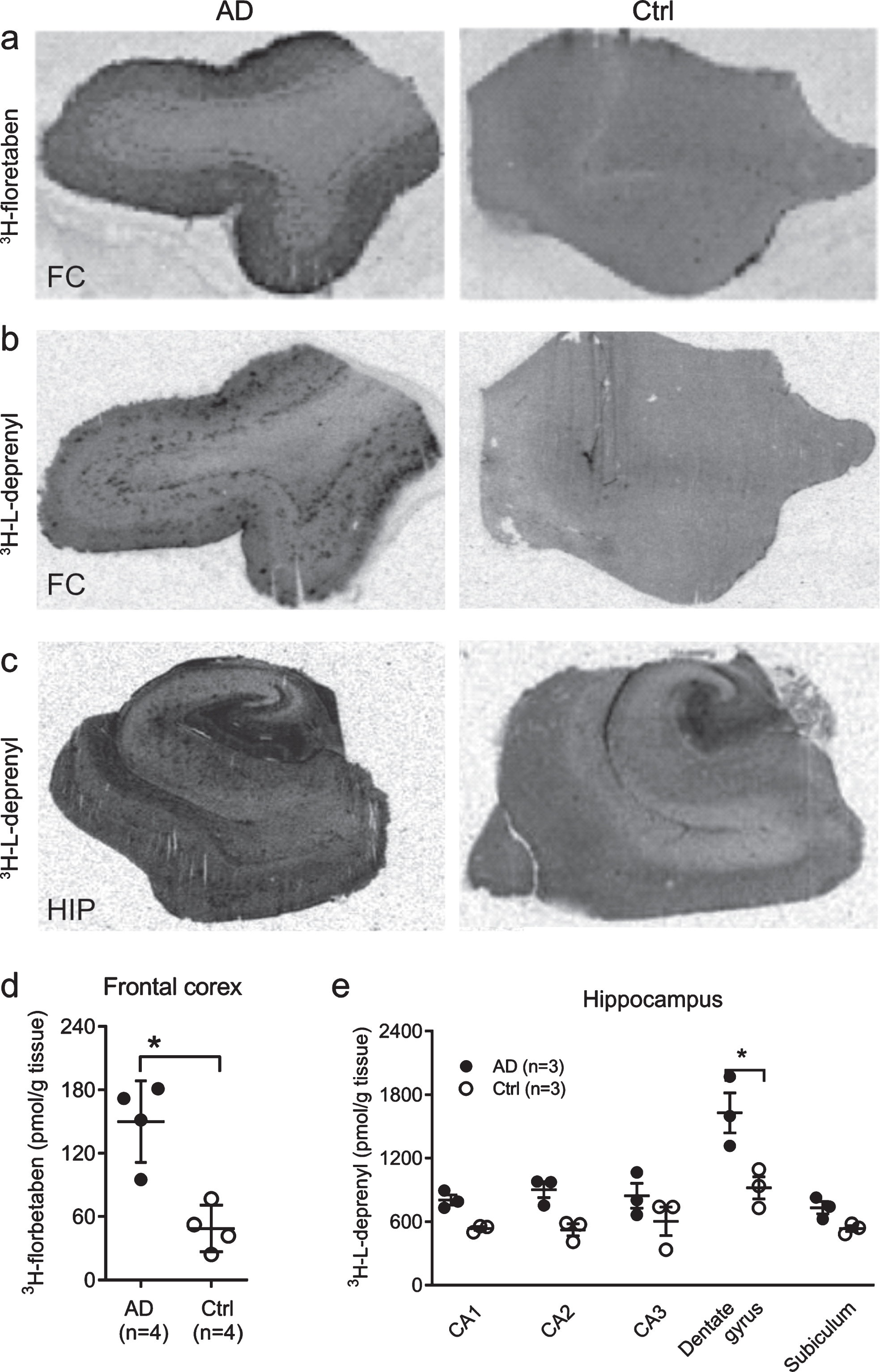
Autoradiography using 3H-florbetaben and 3H-L-deprenyl in AD and control brain. Autoradiography and quantification in AD (
Correlation analysis
To investigate the relation between the levels of different readouts in AD and control, Pearson’s r correlation analysis was performed. The 3H-florbetaben binding positively correlated with 3H-L-deprenyl binding in the hippocampus, parietal and temporal cortex of AD and control cases (Fig. 3a-c). No significant correlation was observed between 3H-florbetaben and 3H-PK11195 binding in the hippocampus (data not shown). 3H-PIB (1 nM) binding measured in the frontal cortex and parietal cortex showed significant increases in AD (
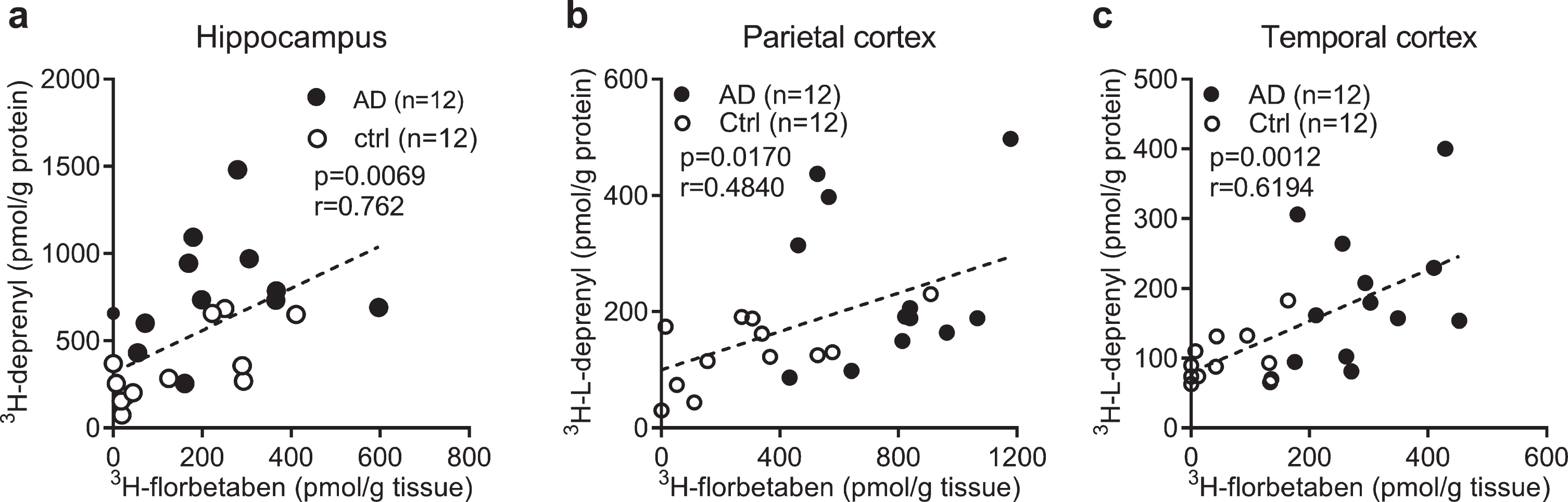
Correlation between regional 3H-florbetaben, 3H-L-deprenyl binding and level of GFAP expression in AD and control brain. a-c) Correlation between 3H-L-deprenyl (6 nM) and 3H-florbetaben (5 nM) binding in the hippocampus, parietal and temporal cortex of AD and control cases.
We analyzed the correlation between different readouts across the whole brain. Correlations were observed between 3H-PK11195 and 3H-L-deprenyl binding (
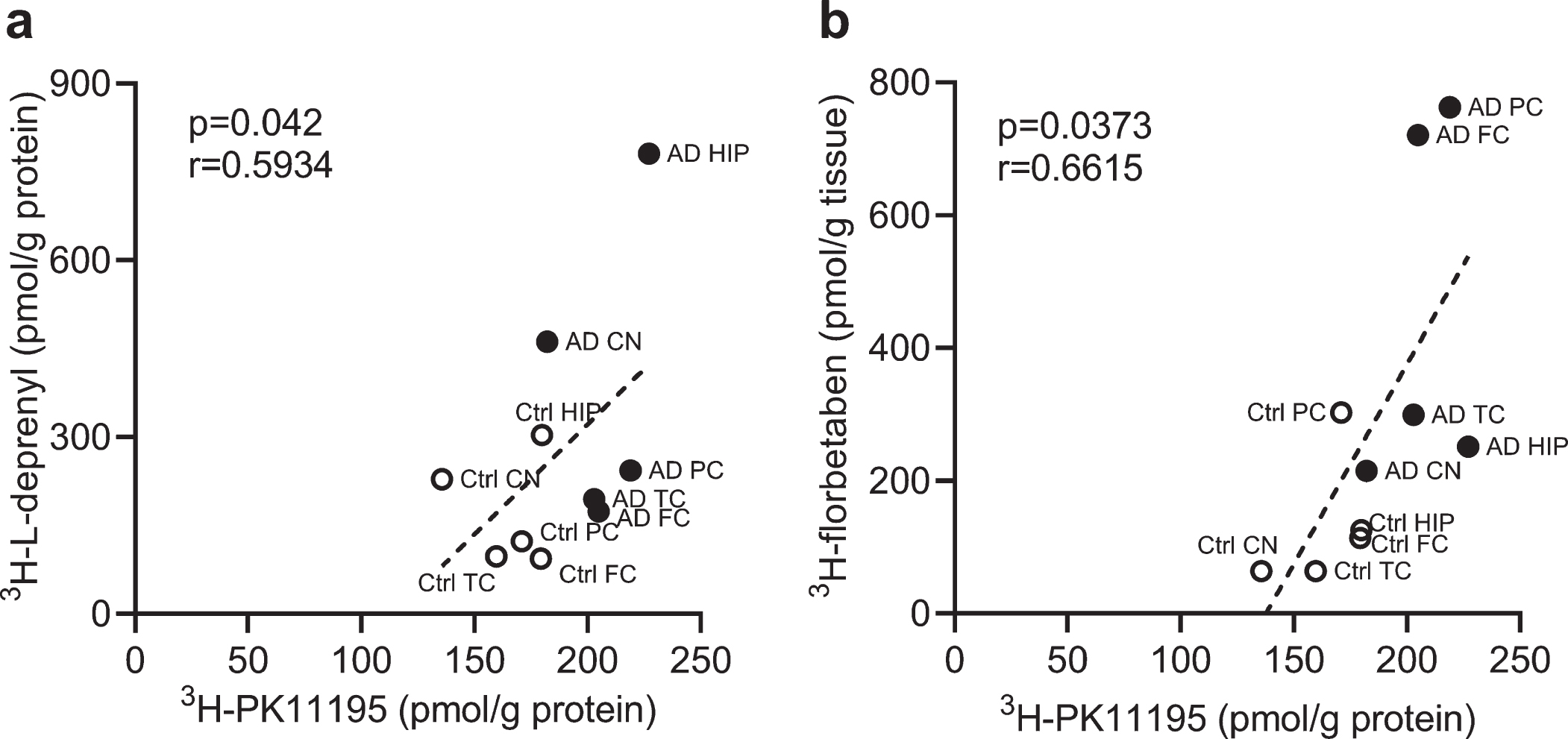
Correlation between regional 3H-PK11195, 3H-florbetaben, and 3H-L-deprenyl binding in AD and control brain. a) Correlation between 3H-PK11195 (2.5 nM) and 3H-L-deprenyl (6 nM) binding across the six brain regions: frontal cortex (FC), parietal cortex (PC), temporal cortex (TC), hippocampus (HC), caudate Nucleus (CN), and cerebellum (CN). b) Correlation between 3H-PK11195 (2.5 nM) and 3H-florbetaben (5 nM) binding across the five brain regions: FC, PC, TC, HC, and CN.
DISCUSSION
Cellular events such as abnormal Aβ and tau ac-cumulation, dystrophic neurites, reactive astrocytes, and microglia contribute to the pathogenesis of AD. Different pathological subtypes of AD, with/without abnormal Aβ accumulation have been documented [2, 48]. Regional characterization of different molecular pathology profiles in autopsy AD brain thus provides insights into the disease mechanism [49] as well as validation for imaging biomarkers. Here we demonstrated distinct region-spe-cific distributions of Aβ deposits (3H-florbetaben), astrogliosis (3H-L-deprenyl, GFAP) and activated microglia (3H-PK11195) in AD brain.
We showed that regional 3H-florbetaben binding corresponded to the known high cortical and low hippocampal Aβ deposits pattern in AD brain. Good
Astrocytes play an important role in the brain physiology, as well as in learning and memory formation [59]. Reactive astrogliosis are dynamic and heterogeneous in their location, subtypes, hypertrophy/proliferation, and upregulation of different markers such as GFAP, vimentin, nestin, MAO-B, and gamma-amino butyric acid [60, 61]. Reactive astrocytes have been shown play an important role early in the development of AD [62, 63]. Elevated 3H-L-deprenyl (MAO-B) measures of astrogliosis was observed in the hippocampus especially in the dentate gyrus, followed by the cortical regions of AD, consistent with autopsy [50, 65] and
Regional GFAP expression seems to differ from 3H-L-deprenyl binding in AD brains. In comparison to 3H-L-deprenyl binding, the GFAP expression is most elevated in the frontal cortex of AD and a positive correlation with 3H-L-deprenyl was only observed in the hippocampus. Different distribution patterns of GFAP and 3H-L-deprenyl were also observed in brain of Tg2576 mouse model of AD [66]. These two methods might detect different status/types of astrogliosis, as 1) GFAP is expressed non-uniformly on subtypes of astrocytes [67]; 2) change in glia phenotype profiles but not number of GFAP + astrocytes in AD brain; 3) MAO-B expressed in astrocytes as well as in dopaminergic neurons mainly in subcortical regions.
We found similar high affinities of 3H-PK11195 and 3H-FEMPA in the cortex of AD, in line with reported high-affinity binding sites of 3H-PK11195 [43] and 18F-FEMPA [36]. The presence of subjects with mixed binding sites of 3H-FEMPA may lead to the observed larger variation in binding distribution compared to 3H-PK11195. 3H-PK11195 shows highest binding in the temporal cortex and hippocampus that is markedly affected by tau aggregates as shown by histopathology [68] and in PET investigations [69, 70]. 11C-PK11195 is the first PET tracer for TSPO imaging. Despite its limitations including a short half-life, relatively low brain uptake, suboptimal metabolic profile, high non-specific binding, 11C-PK11195 is still currently the most used TSPO tracer in AD research. The level of 11C-PK11195 and amyloid load measured by 11C-PIB in AD patients were positively correlated within the frontal, parietal and temporal cortices [71, 72]. Recent study using autopsy brain tissues suggest an overlap in the levels of TSPO protein and mRNA between AD and healthy-control, and a limited influence by
We observe a similar higher regional 3H-florbetaben binding in EOAD and LOAD cases, compared to control cases. This is in line with previous observation by
Positive correlations between 3H-florbetaben-GFAP and 3H-florbetaben-3H-L-deprenyl were observed in the hippocampus, parietal cortex, and temporal cortex in the current study. This corresponds to the reported 3H-PIB-GFAP correlation in sporadic AD autopsy brain [19]. However, negative Aβ40-GFAP correlation in both sporadic and familial AD have also been reported [49]. In addition, 3H-PK11195 binding positively correlates with 3H-florbetaben in five brain regions and 3H-L-deprenyl binding in all six brain regions. This corresponds to the reported positive 11C-PIB-11C-PK11195 correlation in AD cases [88]. The regional link between 3H-florbetaben-3H-L-deprenyl appears stronger than 3H-florbetaben-3H-PK11195 (Fig. 3). Reactive astrocytes and activated microglia were observed in the vicinity of Aβ plaques in the AD frontal cortex [89]. Microglial innate immune responses are highly versatile in AD, producing an array of proinflammatory cytokines and mediators in response to Aβ. These proinflammatory changes activates astrocytes, which in turn secretes cytokines such as interleukin-1 and TNF-alpha. Thus, a vicious neuroinflammatory cycle occurs that initiates and propels disease forward to widespread circuits that undergo dysfunction at a later stage.
Regions within the default-mode network (DMN) are highly overlapping with the spatial distribution of both Aβ and tau pathologies detected by using PET [90–94]. The degree of alterations to DMN connectivity has also been found relating to disease progression [92]. The spread of amyloid pathology from the medial temporal lobe has been found associated with glucose metabolism measured by 18F-FDG PET [95, 96].
This study presents the binding properties of different PET tracers for detecting different AD pathologies. We have to conclude that investigations in autopsy AD brain tissues represent the status at final stage of AD. This is in contrast to
There are several limitations of the study. Firstly, additional
CONCLUSIONS
Our results showed a different regional profile of Aβ plaque deposits versus astrogliosis and microgliosis; and supported the specific measurement by 3H-florbetaben, 3H-L-deprenyl, GFAP expression, and 3H-PK11195 binding in AD and control brains. A clear relationship was observed between Aβ plaque deposition and astrogliosis in AD hippocampus which was not observed for microgliosis. Developing of more specific tracers for disease-relevant gliosis will improve the development of new PET tracers for studies of neuroglia in AD and thereby provide a further understanding of early disease mechanisms important early detection and diagnosis and new treatment targets in AD.
Footnotes
ACKNOWLEDGMENTS
This work was financially supported by a research agreement between Karolinska Institutet and Bayer AG, Pharmaceuticals, Berlin, Germany, the Swedish Foundation for Strategic Research (RB13-0192), the Swedish Research Council (project 05817, 2017-0295, 2017-06086), the Stockholm County Council-Karolinska Institutet regional agreement on medical training and clinical research (ALF grant), the Swedish Brain Foundation, the Alzheimer Foundation in Sweden, and Center for Innovative Medicine, Karolinska Institutet (CIMED).
