Abstract
Alzheimer’s and Parkinson’s diseases (AD, PD) have a pediatric and young adult onset in Metropolitan Mexico City (MMC). The SARS-CoV-2 neurotropic RNA virus is triggering neurological complications and deep concern regarding acceleration of neuroinflammatory and neurodegenerative processes already in progress. This review, based on our MMC experience, will discuss two major issues: 1) why residents chronically exposed to air pollution are likely to be more susceptible to SARS-CoV-2 systemic and brain effects and 2) why young people with AD and PD already in progress will accelerate neurodegenerative processes. Secondary mental consequences of social distancing and isolation, fear, financial insecurity, violence, poor health support, and lack of understanding of the complex crisis are expected in MMC residents infected or free of SARS-CoV-2. MMC residents with pre-SARS-CoV-2 accumulation of misfolded proteins diagnostic of AD and PD and metal-rich, magnetic nanoparticles damaging key neural organelles are an ideal host for neurotropic SARS-CoV-2 RNA virus invading the body through the same portals damaged by nanoparticles: nasal olfactory epithelium, the gastrointestinal tract, and the alveolar-capillary portal. We urgently need MMC multicenter retrospective-prospective neurological and psychiatric population follow-up and intervention strategies in place in case of acceleration of neurodegenerative processes, increased risk of suicide, and mental disease worsening. Identification of vulnerable populations and continuous effort to lower air pollution ought to be critical steps.
Keywords
INTRODUCTION
We are watching with great preoccupation the number of SARS-CoV-2 patients with a wide spectrum of neurological complications, while worsening of cognitive deficits are being recognized in exposed individuals residing in air polluted cities. Neurological manifestations range from the common anosmia and ageusia to encephalopathy, Guillain-Barré syndrome, acute cerebrovascular events resulting in cerebral ischemia, intracerebral hemorrhage, and interestingly ischemic strokes in vascular territories seldom seen on daily practice, i.e., vertebrobasilar system [1–5]. Intriguingly, endotheliopathies and bleeding are described in patients with high morbidity and mortality [1], while patients with delayed recovery of consciousness or severe agitation had bleeding in corpus callosum, internal capsule, and middle cerebellar peduncles attributable to endothelial damage related to viral direct effects or hypoxemia [3]. It is clear SARS-CoV-2 patients exhibit worsening of depression, anxiety, and suicide mortality on one hand [6, 7], while researchers are documenting mental health crises in health systems and communities [8]. Mexico’s national poverty line included 52.4/127 million Mexicans as of 2018, a number that will be augmented by 10 million as a result in part of the contraction of the economy associated with the COVID-19 global pandemic [9, 10].
Metropolitan Mexico City (MMC) is a 25 million densely populated city with high sustained concentrations of air pollutants, high rates of poverty and marginalization, and limited public health, housing, and public transportation capacity, where violence is epidemic and widespread and depression and suicide are increasing in the middle of the pandemic event [11–15]. Jayaweera et al. [16] have stated about COVID-19 “Airborne transmission is highly virulent and represents the dominant route to spread the disease”; this citation is a serious call to all in MMC, where millions of people live in overcrowding conditions, the slums spreading over large areas with little or no basic services like running water available.
METROPOLITAN MEXICO CITY FINE PARTICULATE MATTER PM2.5 AND OZONE
Across the world, in both urban and rural areas, millions of people are exposed to concentrations of PM2.5 above the United States Environmental Protection Agency (USEPA) [17]. Most PM2.5 in urban areas is associated with incomplete internal and external combustion and vehicular traffic, including diesel trucks. High traffic roads close to residencies carry a higher risk of cardiovascular and chronic neurodegenerative diseases, including Alzheimer’s disease (AD) [18–28]. Traffic-related air pollution is a major air pollution contributor [29, 30], with low-income subjects and minorities at higher risk [31].
MMC is a prime example of traffic-related air pollution [29], and residents are exposed to high ozone and PM2.5 that exceed health limits set forth by the USEPA air quality standards and the World Health Organization (WHO) guidelines [14, 33]. This issue is important because within PM2.5, we find the combustion and friction-derived nanoparticles associated with early and progressive development of AD and myocardial damage [34–42].
Figure 1 shows the concentrations of PM2.5 from 2004 to date in MMC; note PM2.5 has been above current USEPA standards during the entire 16-year period. Zhang et al. [43] discussed airborne transmission of SARS-Cov-2 representing the dominant route to spread the disease. Setti et al. [44] have presented preliminary evidence that SARS-CoV-2 can be deposited on outdoor PM and have suggested that under appropriate conditions of atmospheric stability and elevated levels of outdoor PM, SARS-CoV-2 could be able to create clusters with PM. However, these authors recognize that further studies are needed to confirm that PM might work as a “carrier” for the viral aerosol nuclei when adsorbed on the suspended matter.
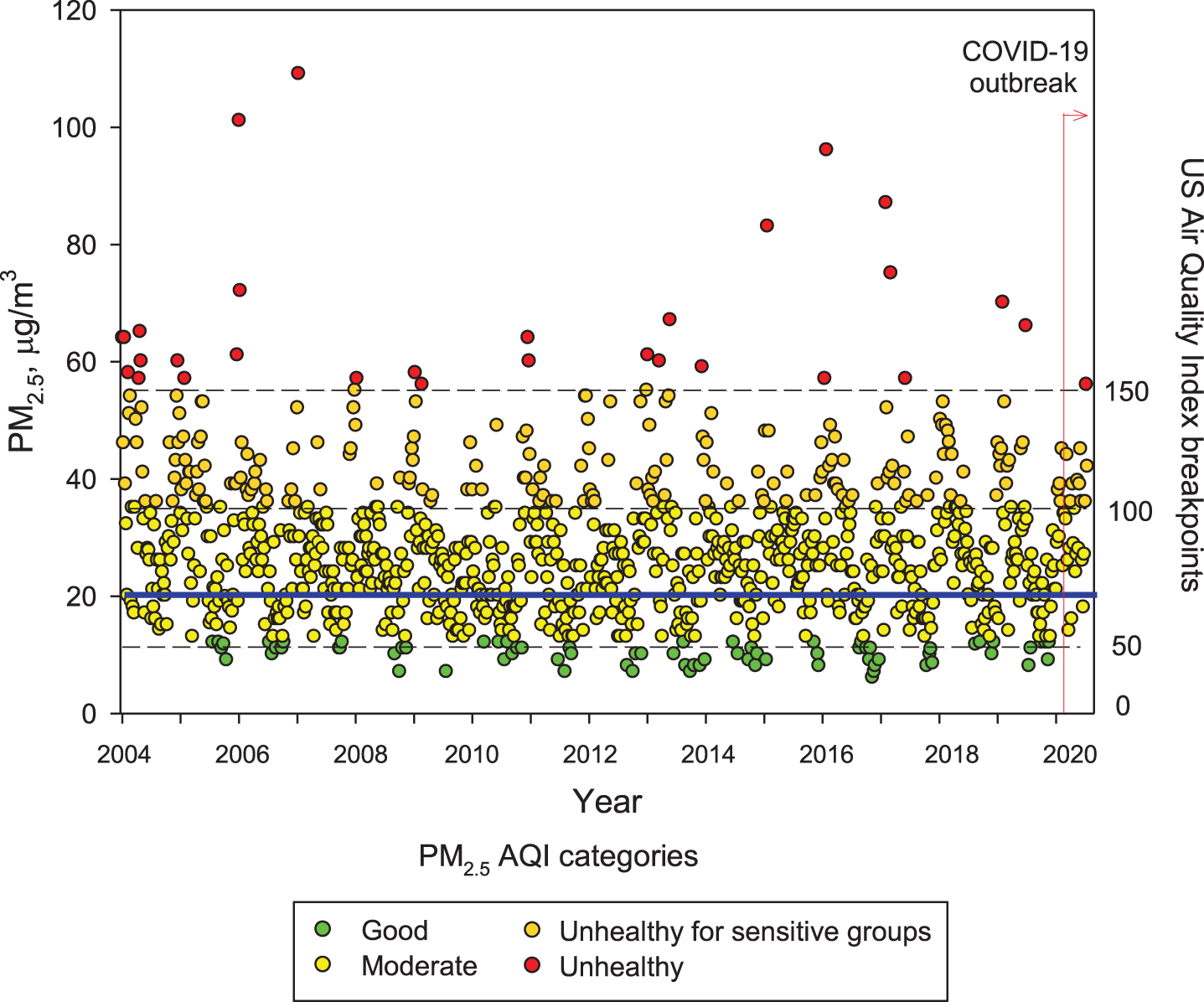
Trend of maxima PM2.5 24-h average concentrations registered in monitoring stations of the MMC from January 2004 to May 2020 and their comparison against the WHO daily mean average guideline (blue solid line) and the US AQI categories. Data correspond to measurements from the manual PM network of the SEDEMA under a 6-day sampling schedule, except data from the period March to May 2020 which were obtained from the automatic monitoring network following the same schedule for manual sampling. Source: SEDEMA at: http://www.aire.cdmx.gob.mx/default.php#
Understanding the seriousness of the exposures to air pollutants is key to surveil health consequences. Independent of everyday high PM2.5 exposures, MMC residents are also exposed to frequent meteorological transport of air pollution plumes from regional uncontrolled wildfires [45, 46] that bring PM2.5 peaks of hourly averages up to ≥200μg/m3. MMC residents are also exposed to high black carbon and organic aerosols (around 10% and 30% of the PM2.5, respectively) [47–54].
Unfortunately, little is known about ultrafine and nanoparticles in MMC; research needs to be done to determine if these particles behave like gases, and if they may have the capacity to remain in suspension almost indefinitely. These nano-size particles have the potential of carrying toxic metals, spores, viruses, and bacteria [55]. In MMC, particles with an aerodynamic diameter smaller than 1μm (PM1) represent around 75% of the concentration of PM2.5 [52]. On the other hand, while PM2.5 in urban areas can be directly emitted mainly from combustion sources and formed from complex atmospheric reactions, surface ozone (O3) is mainly produced from photochemical reactions involving volatile organic compounds and nitrogen oxides. MMC suffered from very high ozone levels from the late 1980s the early 2000s [56]. However, a weak declining trend for O3 has been observed since 2004, despite several ozone strategy plans implemented by local authorities [57, 58]. Due to the MMC geographical situation, March-May is usually warm, sunny, and rainless, therefore ideal for the chemical production of ozone and other secondary pollutants [56]. Typically ground-level ozone reaches its highest concentrations during these months, leading to the so-called term “MMC ozone season”. Figure 2 shows composite trends of maxima O3 8-h averages for MMC’s ozone seasons in 2005, 2010, 2015, and 2020 as indicators of the ozone behavior during the last two decades. Ozone concentrations have been documented above both the WHO guidelines and the USEPA standards for the last 16 years and remain high in the COVID pandemic months (Fig. 2, 2020 panel).
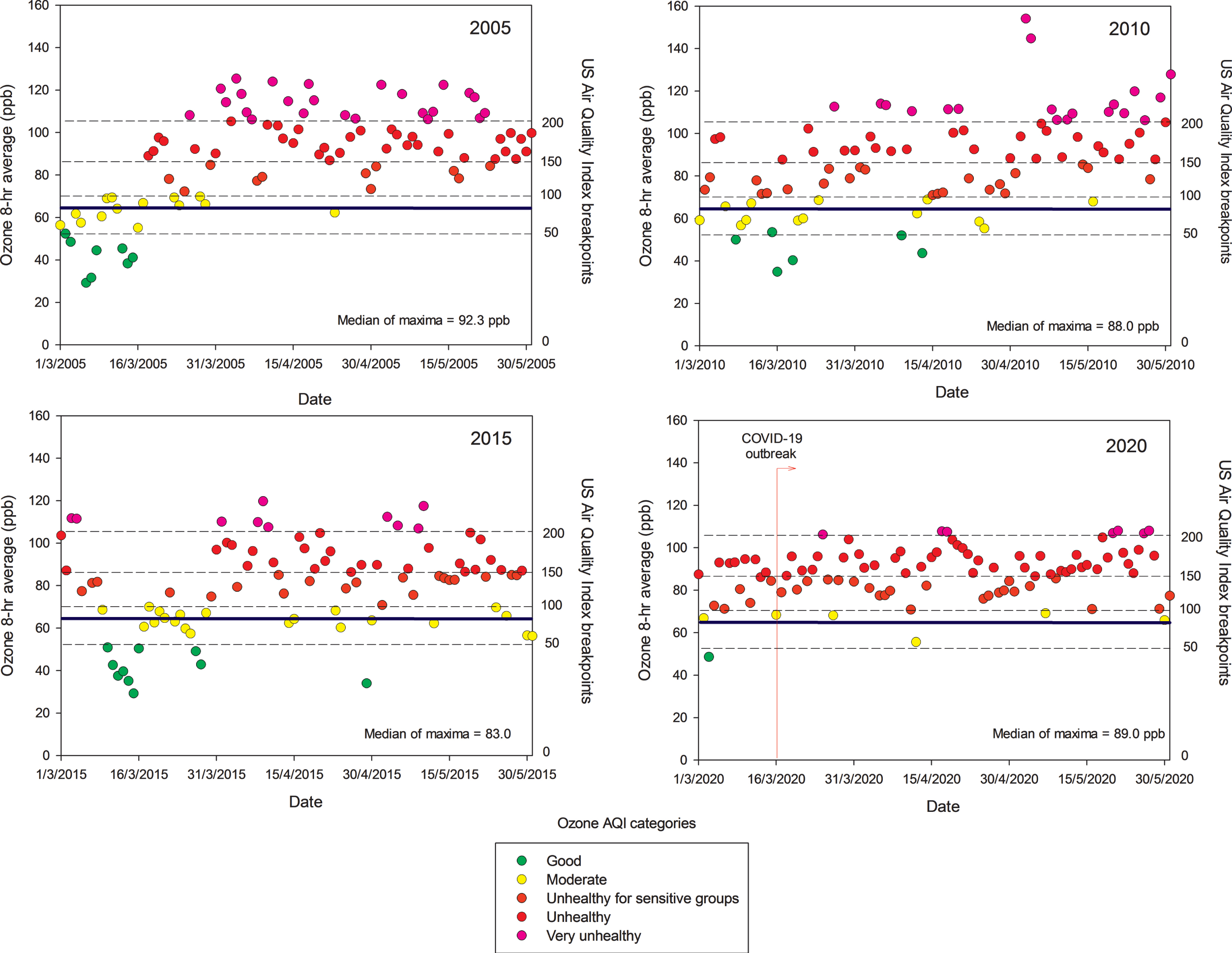
Trend of daily maxima O3 8-h average concentrations registered in all monitoring stations of the MMC from March to May, 2005, 2010, 2015, and 2020 and their comparison against the WHO daily mean average guideline (blue solid line) and the US AQI categories. MMC residents are chronically exposed to sustained yearlong high O3 concentrations. Source: SEDEMA at: http://www.aire.cdmx.gob.mx/default.php#
It is worth mentioning that despite strong official restrictions implemented to vehicular traffic in MMC once the COVID-19 was officially declared in the middle of March 2020, neither ozone nor the PM2.5 levels went down. Unexpectedly, they kept relatively constant, with ozone showing an increase, indicating how complicated the atmospheric chemistry behavior and the strong reactivity of the complex pollutant mixtures in the region. Without question, MMC residents have been exposed to sequential high concentrations of air pollutants—PM and gaseous toxic pollutants—almost every day for the last 20 years [14, 59].
NANOPARTICLES ARE CRITICAL FOR BROAD SPECTRUM BRAIN PATHOLOGY AND SARS-COV-2 PENETRATES ALREADY DAMAGED PORTALS OF ENTRY
The term ultrafine particles applies to airborne particulates of less than 100 nm in aerodynamic diameter, i.e., diesel exhaust particles, products of cooking, heating, and wood burning [60]. Nano-size particles have different sizes and shapes and can be organic, inorganic, and carbon-based [61]. Metals are a key component associated with nanoparticles (NPs) and Fe-bearing NPs including exhaust emissions (both diesel and gasoline), brake wear, tire and road surface wear, resuspension of roadside dust, underground, train and tram emissions, and aircraft and shipping emissions [18].
NPs are crucial because of their small size, specific physicochemical properties, their protein corona characteristics allowing the immune system to detect them and in the case of metal oxide-based NPs: their magnetic properties [61–66].
Neural and vascular cells are directly and indirectly damaged by NPs and abnormal proteins are evidence of cell damage (Fig. 3). Nanoparticles can travel free in blood and cross any biological barrier, key information to understand their toxicity [42, 63–69].
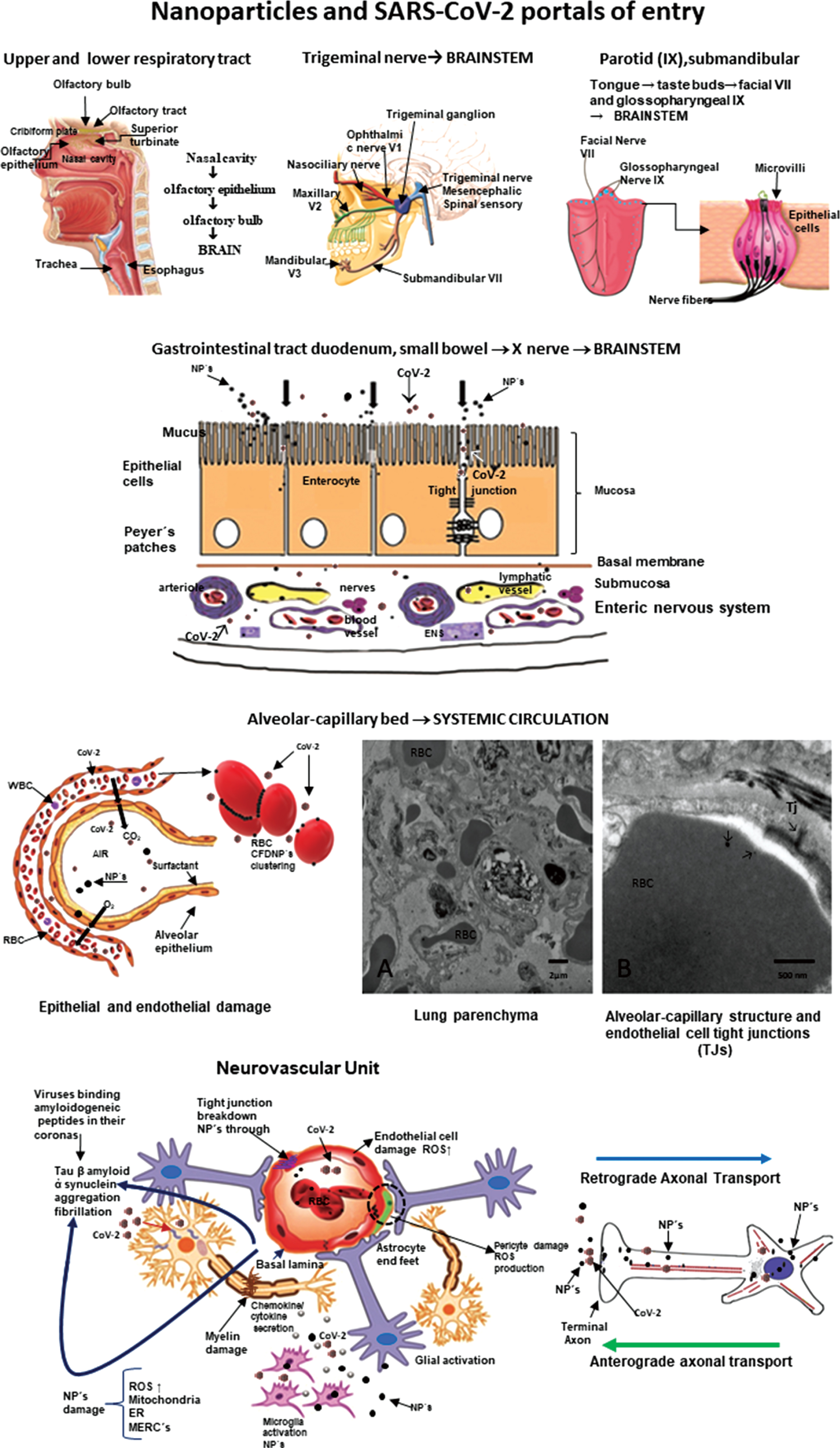
Nanoparticles and SARS-CoV-2 portals of entry. The different portals of entry shared by nanoparticles and the RNA virus. The electron microscopy of the lung parenchyma in a 23-year-old male non-smoking resident in Mexico City illustrates a common finding: activated endothelial cells in the alveolocapillary barrier and tight junctions loaded with nanoparticles, also seen in red blood cells in close contact with the endothelial cell.
Once they cross the alveolar/capillary barrier, nanoparticles are in the major capillary bed in the body and quickly reach the systemic circulation either free in the blood flow or hitchhiking on red or white blood cells [42, 70–72]. Red blood cell transportation of NPs is a very effective way to deliver nanocarriers [73] and along with white blood cells, a key delivery platform to the brain, heart, and gastrointestinal tract [67]. The nasal pathway is also extraordinarily effective and direct to the brain through the olfactory bulb, and since there is an extensive vasculature, access to the systemic circulation is also greatly successful for both NPs and viruses [38, 75]. The nasal portal of entry is critical since it is an early target of NPs damage (young children have extensive olfactory bulb damage), olfaction deficits are early signs of neurodegenerative diseases, including AD and Parkinson’s disease (PD) [36, 76–78] and anosmia and ageusia are described in coronavirus infections [79–84]. The trigeminal nerve has to be considered in terms of SARS-CoV-2 ocular involvement [85–87] and certainly plays a key role in the transport of NPs directly to the brainstem: the ophthalmic and maxillary branches are portals of entry and the trigeminal nerve has a few fibers ending in the olfactory bulb [88–91]. The gastrointestinal tract is often ignored as a portal of entry of NPs—we swallow a significant amount of the inhaled NPs—and it is an invasive portal for SARS-CoV-2 [92–94]. MMC residents have already significant small bowel mucosa damage and there is a deep concern SARS-CoV-2 could have an easy access to the enteric nervous system [95, 96]. We have shown NPs reach preganglionic parasympathetic fibers and the vagus nerve, and immunoreactivity of stomach and vagus to phosphorylated α-synuclein Ser129 and hyperphosphorylated tau are documented in young MMC urbanites [96]. Lundquist and Artursson’s review [92] emphasized two key entry routes for NPs, which are certainly applicable to viruses: the paracellular route via tight junctions and the transcellular transcytosis route.
NANOPARTICLES AND THE SARS-COV-2 DUO HAVE GREAT ADVANTAGES TO DAMAGE THE BRAIN THROUGH THE NEUROVASCULAR UNIT (NVU)
NVU integrity guarantees the coupling between neural activity and blood flow [97]. Multiple cells are engaged in distinct signaling pathways and effector systems across the entire cerebrovascular network already compromised in MMC residents [97, 98]. MMC young residents≤40 years show NVU abnormalities including 1) endothelial cells (EC) tight junction abnormalities with NPs deposition; 2) EC, pericytes, and smooth muscle cells with abnormal mitochondria and endoplasmic reticulum with numerous NPs; 3) passage of NPs from the capillary wall to the neural spaces, and 4) expansion of the Virchow spaces with breakdown of the perivascular neuropil and leaking of serum and red and white blood cells. Every NVU component is compromised, and mitochondria, mitochondria-associated endoplasmic reticulum, and mitochondrial matrix and cristae are targets of NPs. Abnormal generation of reactive oxygen species, mitochondrial dysfunction, and altered mitochondrial quality control are the direct result of NVU damage [99–109]. The NP biomolecular corona, the result of the adsorption of biomolecules on the NP surface, ultimately determines the NVU and neural cells severity damage [110–114]. Thus, the NVU damage will impact at least in part, the capacity of SARS-CoV-2 to invade neural tissues and extend throughout the brain. Understanding of protein function following particle and virus binding is necessary to properly evaluate pathophysiological events [112, 115–117]. Viruses, i.e., respiratory syncytial virus and herpes simplex virus type 1 (HSV-1), can make protein coronas, information relevant to Parkinson’s disease (PD), influenza, and now SARS-CoV-2. Ezzat and coworkers emphasized a critical piece of information: viruses share many biophysical properties with NPs. The authors discussed respiratory syncytial virus and HSV-1 as capable of accumulating distinctive protein coronas in different biological fluids and corona pre-coating differentially affecting viral infectivity and immune cell activation. Moreover, authors demonstrated that viruses bind amyloidogenic peptides in their corona and catalyze amyloid formation via surface-assisted heterogeneous nucleation. Also, key for this review, Berardi et al. [118] discussed non-sticky viral NPs are capable of crossing most non-specific protein and glycoprotein barriers encountered, when administered through mucosal and non-mucosal routes.
The endothelium is a critical barrier and damage by both NPs and viruses will alter the regulation of molecular and cellular traffic across this barrier [119]. SARS-CoV-2 direct and indirect endothelial damage certainly contributes to mortality [120, 121] through diverse mechanisms, endothelial dysfunction, production of extracellular vesicles and thromboembolism, oxidative stress, dysregulated immune responses, abnormal coagulation, and hypoxia [122, 123]. Lymphocytic vasculitis with EC swelling, fibrinoid necrosis, and thrombosis, along with viral particles in endothelium are described in children’s skin chilblain biopsies with clinical purpura [124]. Damage to the myocardium and bowel are also related to microvascular thrombosis and diffuse EC injury [125, 126]. EC damage contributes significantly to multiple organ failure.
Thus, NVU damage predicts brain dysfunction and neurodegeneration and SARS-CoV-2 could use a damaged NVU to facilitate neural invasion and extent the damage further. The biologically plausible association between nanoparticles and viral protein coronas will impact viral infectivity and immune cell activation. Viruses bind amyloidogenic peptides in their corona and catalyze amyloid formation via surface-assisted heterogeneous nucleation. Initiation of amyloid formation could have devastating consequences in highly exposed air pollution populations.
NEUROINFLAMMATION, NANOPARTICLES, AND SARS-COV-2
Nanoparticles per se induce neuroinflammation [64, 127–131]. Titanium dioxide NPs induced an age-dependent modulation of blood-brain barrier integrity associated with interleukin-1β and interferon-γ increases in aging rats’ brain [127]. Diesel exhaust NPs produced olfactory bulb and hippocampal oxidative stress and inflammatory responses in adult mice in conjunction with anxiety and depression [128]. Strikingly, there is evidence exposure to metal and metal oxide NPs, such as Fe2O3, CuO, and ZnO, plays a potential role in the pathophysiology of neurodegenerative diseases such as AD through neuroinflammation [131]. A low dose of CuO NPs activates the NFκB signaling pathway and increases amyloid-β protein precursor (AβPP) expression, a pathway directly related to Aβ amyloidosis, a hallmark of AD [131]. Portals of entry such as the tongue constitute a direct pathway to the brain and neuroinflammation via the taste nerve translocation pathway (VII and IX cranial nerves) as described using ZnO and TiO2 NPs [129, 132]. Binding of NPs to activated endothelial cells, astrocytic feet and choroid plexus damage the NVU and the blood-cerebrospinal fluid barrier contributing to the inflammatory process [133–136].
It is important to remember that NPs have endothelial cells as first line targets and ubiquitous particles such as zinc oxide nanoparticles very efficiently reduced the continuity of tight junction proteins claudin-5 and zonula occludens-1 (ZO-1) at the cell junctions [136]. ZnO NPs induced adherens junction protein VE-cadherin internalization from membrane to cytosol and dissociation with β-catenin, leading to reduced and diffused staining of VE-cadherin and β-catenin at cell junctions. Chen et al. [136] discussed how disrupted tight and adherens junctions lead to inflammatory cellular infiltrates.
Neuroinflammation plays a central role in the pathogenesis of AD along mitochondrial dysfunction, excitotoxicity, abnormal innate immunity, and the activation of inflammasomes and microglia [137–146]. Mexico City subjects exhibit neuroinflammation, as measured by upregulation of genes, very soon after birth followed by increased neuroinflammation associated with progression of AD [20, 137]. Neuroinflammation is an intrinsic part of AD, thus activation of astrocytes and microglia, neuronal and glia death, and upregulation of proinflammatory and anti-inflammatory cytokines, chemokines, and inflammasomes are expected [141, 147–157]. Chronic inflammation, a dysfunctional immune system, and the onset and progression of AD go hand in hand [149, 154].
Parkinson’s disease (PD) shares with AD the neuroinflammation and immunologic central and peripheral responses key to a-synuclein pathology [59, 158–165]. Lewy neurites are detected in the olfactory bulb and brainstem of MMC children and teens concomitantly with the AD neuropathological hallmarks [38].
Coronaviruses invade, replicate, and proliferate in neural cell lines and inflammation is a well know mechanism of their neural devastation [74, 166–175]. Coronaviruses are lipid enveloped and spherical viruses in the nanosize range 100–120 nm. The nucleocapsid contains a single-stranded non-segmented positive-sense RNA genome [176]. The name Coronavirus comes from the crown-like appearance of the virus—large petal-shaped spikes protruding from the envelope—the spike glycoprotein, 20–40 nm long. The M protein is the most abundant protein and provides structural support, while the E protein is a small membrane protein essential for the assembly and release of virions [176]. Specific chemotactic cytokines such as Chemokines CXCL10 (interferon-inducible protein 10 kDa) and CXCR2 play a role in host defense and give rise to demyelination and neurodegeneration, both well documented in Coronavirus models of acute encephalomyelitis [177–181]. Host inflammatory responses activate T and B lymphocytes expressing CXC chemokine receptor 3 (CXCR3) and in turn determine the persistence of the virus in white matter tracts, chronic neuroinflammation, and demyelination [180]. Coronaviruses recognize host receptors via spike glycoprotein (S protein, with a key role in receptor (ACE2) binding and virus entry) and other proteins, including Orf 3, Orf 8, and Orf 10, invade host cells by membrane fusion, replicate through large replication-transcription complexes, and increase proliferation by interfering, modulating and suppressing the immune response in the host [174, 182]. The presence of furin-like cleavage site in SARS-CoV-2 facilitates the S protein priming and could increase the spread efficiency of SARS-CoV-2 [183]. An extended structural loop containing basic amino acids at the interface of the receptor binding (S1) and fusion (S2) domains was identified by Jaimes et al. [184]. The authors suggested this loop could be a key component in the evolution of SARS-CoV-2, affecting virus stability and transmission [184]. The presence of point mutations in the spike glycoprotein (rOC/Us183 - 241) increases the neurovirulence in mice and induces cell death in murine and human neuronal cells [185]. Meessen-Pinard and coworkers [185] emphasized that as necroptosis disrupts cellular membranes and allows the release of damage-associated molecular patterns, the cascade of proinflammatory cytokines may represent a proinflammatory cell death mechanism that contributes to excessive neuroinflammation and neurodegeneration and eventually to neurological disorders after a coronavirus infection.
Twenty years ago, the discovery of angiotensin-converting enzyme (ACE2), a zinc metallopeptidase able to hydrolyze Ang I to Ang 1–9 and Ang II to Ang 1–7, marked the critical role of the enzyme as a negative regulator of the renin-angiotensin system and later, as a SARS-CoV-2 receptor (the major spike glycoprotein S1 binds to the N-terminal region of ACE2) [174, 187]. The issue of SARS-CoV-2 binding to its receptor ACE2 is of utmost importance for residents in polluted environments [188–196]. Briefly, ACE2 is extensively present in the brain in areas associated with cardiovascular function, specifically the area postrema, dorsal motor nucleus of the vagus, nucleus of tractus solitarius, nucleus ambiguous; primary motor cortex, frontal cortex, and numerous other nuclei extending from the cerebral cortex [197–202]. In the excellent ACE2 review by Gheblawi et al. [194], the authors made it clear that the balance of the renin-angiotensin system (RAS) is vital and that loss or gain of function are involved in heart failure, systemic and pulmonary hypertension, myocardial infarction, and cardiovascular disease complications of metabolic diseases. The cell invasion of SARS-CoV-2 is facilitated by the interaction between the spike glycoprotein S1 with the extracellular domains of the ACE2 transmembrane proteins, followed by downregulation of surface ACE2 expression [194]. Since both S-proteins and ACE2 are proteolytically modified in the process, the baseline host cell proteases including transmembrane serine protease 2 are key for virulence [203]. The depletion of ACE2 is important in the pathogenesis of SARS-CoV-2 because the virus is endocytosed by cells in conjunction with ACE2. ADAM-17, a type 1 transmembrane Zn-dependent metalloprotease, comes to play because it also mediates proteolysis and ectodomain shedding of ACE2. Upregulation of ADAM 17 is a key factor in inflammation associated with heart failure and coronary artery disease [204]. Of deep interest for MMC residents, ADAM17 is also involved in neuroinflammation through the cleavage of TNFα and interleukin-6 receptor [205], two key proinflammatory cytokines upregulated in young MMC urbanites’ brain [22, 35]. SARS-CoV-2 gives rise to an imbalance of Th17/Treg cell function, overactivation of immune cells, a proinflammatory cytokine storm enhancing the RAS imbalance, and the loss of ACE2 resulting in severe inflammation [194]. Strikingly, ACE2 expression is reduced in specific brain areas (i.e., area postrema, dorsal motor nucleus of the vagus) in animal models with hypertension and heart failure [201]. On the other hand, ACE2 inhibition elevates blood pressure and increase the sympathetic cardiac tone. Thus, autonomic mechanisms play a key role in the impact of ACE2 as a negative regulator of the renin-angiotensin system [206]. Indeed, SARS-CoV-2 may downregulate ACE2, which increase Angiotensin II with detrimental consequences [207].
Of deep concern is the capability of SARS-CoV-2 to reach the brain using the systemic circulation and the endothelium of the brain capillaries [191, 208] given that aging per se decreases ACE2 and results in oxidative stress and endothelial dysfunction in cerebral arteries—a scenario present in MMC young residents in relation to exposure to PM2.5 and combustion and friction-associated nanoparticles [39]. The review by Baig et al. [209] shows pathways facilitating the brain invasion: the interaction with ACE2 receptors in cerebral capillaries would be an ideal portal of entry, followed by direct entrance through the olfactory bulb. Other pathways facilitating neural invasion include SARS-CoV-2 receptor ACE2 exploiting species-specific interferon-driven upregulation of ACE2 [210] and upregulation of transmembrane serine protease 2, an androgen-responsive protease that cleaves SARS-CoV-2 spike protein [211].
Chronic neuroinflammation and a dysfunctional immune system go hand in hand with the onset and progression of AD and PD in young urbanites exposed to air pollutants. Both nanoparticles and SARS-CoV-2 produce neuroinflammation and use the same portals of entry. The downregulation of ACE2 resulting in oxidative stress and endothelial dysfunction likely contribute to the SARS-CoV-2 neuroinflammatory capabilities. An extensive neuroinflammatory and neurodegenerative process with an abnormal neurovascular unit set the optimal conditions for SARS-CoV-2 brain invasion.
SARS-COV-2 AND THE RENIN ANGIOTENSIN SYSTEM
The role of the central RAS in the pathophysiology of neurological and psychiatric diseases including AD and PD has been discussed by a number of researchers across the world [202, 212–225]. A key issue is the use of drugs acting via the renin-angiotensin system in the prevention and treatment of AD. Kehoe et al. (2016) and Walker et al. (2020) described the significant reduction of ACE2 in patients with AD and the inverse correlation with Aβ and p-tau, the two prime neuropathological markers of AD [202, 226]. They also showed in AD patients the reduction of the ratio Ang II to Ang 1–7, as a proxy for ACE2 activity. The RAS system impacts blood pressure, cerebral blood flow, neuroinflammation, and oxidative stress. On the one hand, Angiotensin (Ang) II, via angiotensin type 1 receptors promote brain tissue damage, while Ang-1–7 seems to elicit neuroprotection [220]. RAS-targeting drugs as ACE inhibitors and angiotensin receptor blockers seem to exert beneficial effects on AD [220, 227].
Evans et al. [225] have shown that increased brain ACE2 activity, a major effector of rRAS, by intraperitoneal administration of an activator of ACE2, lowered hippocampal Aβ and restored cognition in mid-aged (13-14-month-old) symptomatic Tg2576 mice. The authors concluded that “ACE2 enhancement protects against and reverses amyloid-related hippocampal pathology and cognitive impairment in a preclinical model of AD” [225]. Wright and Harding [218] summarized in a highly recommended review paper how an unbalanced RAS system contributes to “neuroinflammation, oxidative stress, reduced cerebral blood flow, tissue remodeling, and disruption of memory consolidation and retrieval.” We fully agreed with Verdecchia et al. [228] and Perrota and co-workers [229] that RAS is a double-edged sword in the SARS-CoV-2 world. On one hand, the ACE2 upregulation suggest a potential role against lung damage in SARS-CoV-2 infected patients. On the other hand, the controversy sets on risk of increase susceptibility to infection linked to ACE2 upregulation associated with RAS inhibitors. At this time, however, a number of Cardiology, Hypertension and Pharmacology Societies strongly recommend against ACE inhibitors or angiotensin receptor blocker drugs discontinuation in patients with COVID 19. We agreed with Barilla et al. [230] on their statement: “Their discontinuation can increase Ang II activity and induce injury to the lungs or cardiovascular system.”
The issue of particulate matter and increased susceptibility to SARS-CoV-2 is raised in the relationship between PM2.5 concentrations and incidence, mortality rate and case fatality risk of COVID-19 by Borro et al. [231]. One hundred and ten Italian provinces were categorized by PM 2.5 concentrations and differential SARS-CoV-2 spread in the period 20 February-31 March 2020. Strikingly, bioinformatic analysis of the DNA sequence encoding the SARS-CoV-2 cell receptor ACE2 identified positive correlations between PM2.5 levels and the incidence (r = 0.67), mortality rate (r = 0.65), and death rate (r = 0.7) through nine putative consensus motifs for the aryl hydrocarbon receptor. The hypothesis that pollution-induced over-expression of ACE2 on human airways may favor SARS-CoV-2 infectivity is in place in this Italian study. Wu and co-workers [232] in a preprint USA work (as of August 21, 2020), covering 3,000 counties up to April 22, 2020 found that an increase of PM2.5 of 1μg/m3 was associated with 8% increase in COVID-19 death rate. This is not an unexpected result in view of Borro et al.’s work [231] and brings to public light the absolute need to control PM2.5 .
Tobacco smokers [233, 234] are exposed to high PM2.5 concentrations and tobacco or marijuana smoke and vaping exposures trigger the expansion of goblet cells with a concomitant increase in ACE2 expression. Smokers should be considered high risk for SARS-Cov-2 especially if they already have chronic obstructive pulmonary disease [235]. Usman et al. [236] wrote about the controversial issues related to smoking: the claims of protective effects (i.e., the anti-inflammatory effect of nicotine) versus the extensive respiratory damage directly related to smoking and worsening the response to COVID infections.
NEUROLOGICAL MANIFESTATIONS OF COVID 19 AND NEURODEGENERATION
The list of neurological manifestations keeps growing, with anosmia and ageusia on top of the list [5, 238–240]. The long-term development of neurological and neurodegenerative disorders is of deep concern for a number of researchers [7, 241–243]. PD is certainly a possibility since viruses, i.e., H1N1 replicate in neuronal cells and could induce seeds of aggregated α-synuclein, key factors in the pathogenesis of synucleinopathies [244]. Marreiros and co-workers [244] support the concept of aberrant proteostasis induced by viruses is an underappreciated factor in initiating protein misfolding. We absolutely agreed with Naughton et al. [245] on their statement “Immune response and excessive inflammation in COVID-19 may also accelerate the progression of brain inflammatory neurodegeneration”, and their inclusion of high risk for diabetic patients, African-Americans, and Hispanics is warranted. Heneka et al. [246, 247] and Ising et al. [139] discussed a key inflammation player that is very relevant for SARS-CoV-2 patients: the NLRP3 inflammasome-mediated inflammation inducing or aggravating neurodegenerative processes that lead to functional impairment in AD.
There is no question the viral neurotropism is important along factors intrinsic to the host, including genetics, innate immunity, the hyperactivation of the immune system, and the development of cytokine storm syndrome along with the immune previous status of the host to the COVID-19 encounter [75, 248–250]. Thus, the approach suggested by Winkler et al. [251] with the Global COVID-19 Neuro Research Coalition and the work of Ferrarese and coworkers [252] about establishing a retrospective and prospective observational study aimed to detect neurological manifestations of COVID-19 will yield an enormous opportunity to define in targeted populations the range of neurological manifestations and the associated risk factors. Long-term neurodegenerative diseases ought to be in the mind of every neurologist across the world, with aberrant proteostasis, neuroinflammation, and abnormal immune responses being key factors for accelerating AD and PD pathology.
PSYCHIATRIC DEMANDS, SUICIDE, AND COVID-19
There is no doubt in the minds of physicians and health workers across the globe of the multiple factors associated to the pandemic playing a decisive role in the mental health of millions of people. Problems ranging from massive unemployment, contraction of labor market, economic insecurity, domestic violence, uncertainty, and social isolation have a detrimental impact on mental health. It is also clear that some people are more vulnerable than others, including patients surviving severe COVID-19 disease, patients with pre-COVID serious mental illness, and those with alcohol use disorder and alcohol-associated liver disease may be among the most severely impacted [6, 253–261].
There are three scenarios of deep concern in terms of psychiatric support at this time: Psychiatric manifestations such as depression related directly to virus impacting the brain of exposed subjects. Lyons and co-workers [262] discussed the possibility of literally having a tsunami of post-viral depression, a biological plausible scenario knowing the neurotropic features of SARS-CoV-2. The relationship between peripheral inflammatory markers in COVID-19 patients and higher levels of depression (p < 0.001), anxiety (p < 0.001), and post-traumatic stress symptoms (p < 0.001) discussed in the work of Guo et al. [263]. MMC residents with symptomatic or asymptomatic COVID-19 are fully expected to accelerate their progression of AD and PD, based on their synergistic systemic and neuroinflammatory increases related to the virus itself and the ongoing air pollution related process. Children, young adults, and the elderly could present with progression of pre-existing or increased neuropsychological pathological outcomes [264–266].
Suicide ought to be a significant concern in patients exposed to SARS-CoV-2 [261]. McIntyre and Lee [253] have discussed preventing suicide in the context of the COVID-19 is a critical public health priority. It is also a priority to identify the most vulnerable populations. We discussed patients with serious mental problems and persons experiencing features of post-traumatic stress disorder or drug/alcohol abuse are at higher risk.
MMC subjects carrying an Apolipoprotein 4 allele have a higher risk of suicide in the pre-COVID era, i.e., 4.92 times higher suicide odds versus APOE3, and strikingly, 23.6 times higher odds of NFT V (p < 0.0001) (acceleration of the AD staging) versus APOE4 non-carriers-obligating us for identification of this high-risk population [37]. In Mexico, suicide is linked to unemployment [267]. Wang and coworkers [267] performed an analysis of suicides in Mexico in the period 1981–2016 and it was clear there was a positive bidirectional causality between suicide rate and the unemployment rate in the preCOVID-19 period. In the 18 months pre-COVID era, we already had a major reduction of Mexico’s growth, now projected as minus 10.5% in 2020 according to the International Monetary Fund [268]. According to the International Monetary Fund for economies struggling to control infection rates (i.e., Mexico), a lengthier lockdown will inflict an additional toll on economic activity. As discussed by Valeria Moy, the “country’s ability to grow in the future is being destroyed, bit by bit, mostly unperceived” [269]. Mexico economic performance added to the COVID-19 crisis will result in a significant increment in the number of suicides [11, 12], especially in the hard-hit cities, those with a great number of unemployed, poor people, without access to health care. Health crisis built on economic disasters result in serious health impacts. Suicide is one consequence of this complex crisis and identifying the vulnerable populations and helping them, should be a key health priority.
SUMMARY OF THE PROPOSED CASCADE OF PATHOLOGICAL EVENTS IN METROPOLITAN MEXICO CITY RESIDENTS
The starting point is the fact that SARS-CoV-2 is a neurotropic virus, capable of accessing the CNS through the same portals of entry documented for combustion and friction associated NPs [39]. These pathways include: the nasal olfactory epithelium with a direct access to the olfactory bulb and the trigeminal (V), facial (VII), and glossopharyngeal (IX) nerves, and each of these providing direct access to the brainstem. Thus, the nasal, ocular, salivary glands, and tongue are effective portals for SARS-CoV-2 and the access to the brain is direct. All sites are indeed effective transmission sites and portals of entry of NPs [270–274].
The gastrointestinal tract is also very effective portal of entry for both NPs and SARS-CoV-2. MMC young residents have duodenal barrier damage and autoimmune responses to tight junctions and neural proteins [95]. The intestinal breakdown barrier has an impact on the neuroenteric system, the vagus, and the brainstem [36, 96]. Trans-synaptic transmission of NPs or viruses reaching preganglionic parasympathetic vagus motor neurons is a plausible biological pathway to reach the brain. Changes in microbiota composition, increased permeability of the gut barrier, and immune activation leading to systemic inflammation, which in turn may impair the blood-brain barrier and promote neuroinflammation, neural injury, and ultimately neurodegeneration [275–285]. Millions of people are being exposed to many common consumer products with NPs, including food additives/supplements and packaging materials in contact with foods [92, 286–290]. The direct effects of NPs upon the gut microbiome and the changes in the intestinal barrier supportive of bacterial changes are well known [91, 291–296].
SARS-CoV-2 ACE2 is highly expressed on differentiated enterocytes as shown in human small intestinal organoids [297], and indeed Venkatakrishnan and coworkers [298] have discussed the gut as the “putative hotspot of COVID-19, where a maturation correlated transcriptional signature is shared in small intestine enterocytes among coronavirus receptors (ACE2, DPP4, ANPEP).”
MMC residents have a disrupted intestinal barrier related to NPs and thus are at higher risk of quick intestinal invasion with all detrimental consequences [43, 299–301], including persistent viral shedding in feces, a serious problem in a country where fecal contamination in the urban environment due to inadequate sanitation is common, running water to wash peoples’ hands is scarce, and where public schools do not have readily available water on school grounds. Deeply worrisome is the presence of SARS-COV-2 RNA on PM10 collected in Northern Italy supporting the virus can create clusters with PM [44] and thus PM is a Trojan horse for the virus. The presence of SARS-CoV-2 in sewage thus is indeed not a surprise [302] and complicates things for MMC residents getting their vegetables from areas using sewage water [303].
The alveolar-capillary bed is a target of SARS-CoV-2. Diffuse alveolar damage is characterized by intra-alveolar edema, fibrin deposition, hyaline membranes, and interstitial inflammation, followed by Type II pneumocyte proliferation, focal hemorrhage, granulation tissue formation, and collagen deposition [304]. The lung pathology associated with NPs depends on the chemical composition, dose, and host responses [305]. Experimentally, Fe2O3NPs and AgNPs cause significant DNA base oxidation as indicated by the elevated DNA content of 8-hydroxy-2-deoxyguanosine in the heart and lungs. Moreover, significant decrease in antioxidant enzymes, total antioxidant capacity, and reduced glutathione in heart and lung are striking. A dose-dependent increase in production of TNF-α and IL-6 are seen along with synergistic effects if more than one type of NP is administered [305, 306].
MMC residents showed significant oxidative damage, rupture of the alveolo-capillary barrier with passage of NPs through the barrier and pick up of NPs by intracapillary macrophages [307]. Interestingly, the lungs even in small children have ample anthracosis patches on the pleural surfaces. These children were from households with no history of smoking [307]. Once the NPs go to the systemic circulation, either free or in red or white blood cells, their journey can take them to the brain. The alterations seen in Fig. 4 include enlargement of the Virchow space, breakdown of the EC cell junctions, and NPs traversing the NVU barrier. Axonal transport of NPs can be retrograde and through synapsis, all documented in MMC residents.
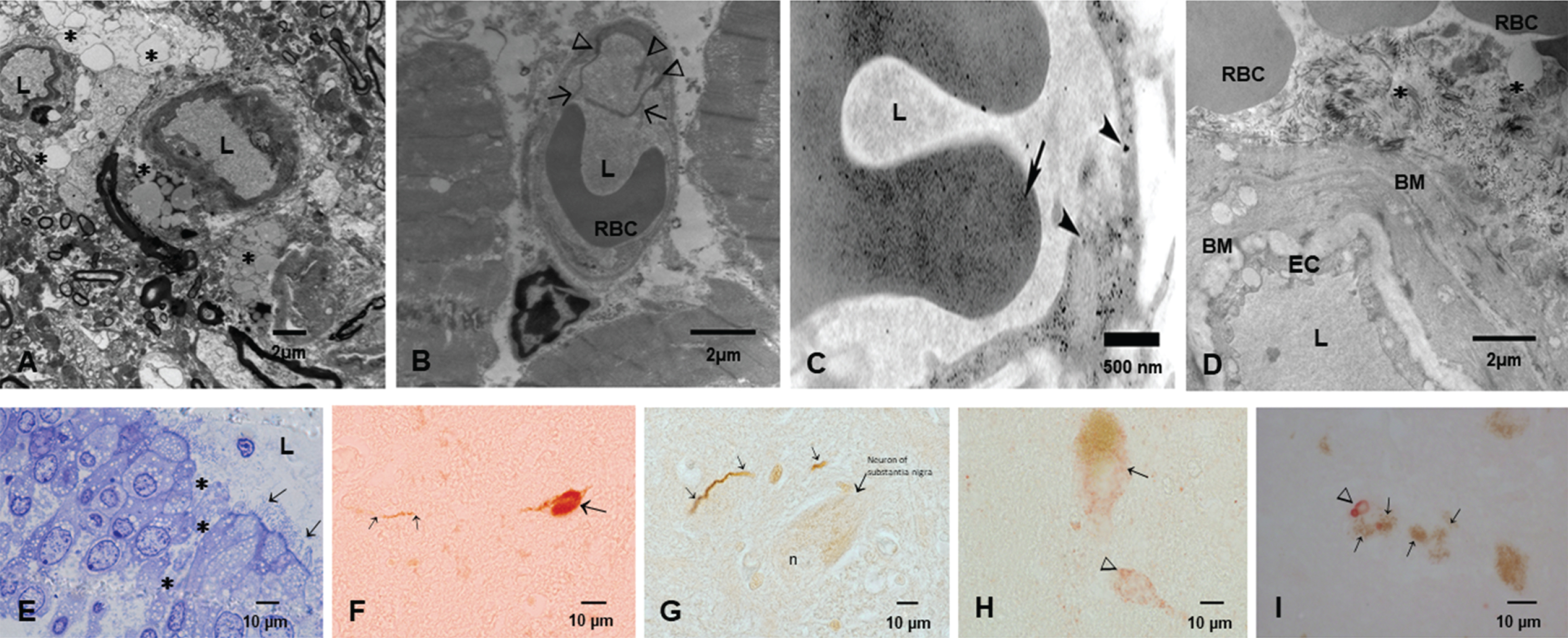
The key early neural damage in Mexico City residents is the endothelium of the brain capillaries with an abnormal neurovascular unit. The damage is present in early childhood and it gets worse with age in the absence of any comorbidities. A) Brain neurovascular unit showing a central capillary with an extensive damage to the perivascular neuropil, including vacuolization (*) and fragmentation of the neuropil with abnormal extravascular accumulation of lipids, the result of leaking vessels. B) A left ventricular blood vessel with marked endothelial activation as evidenced by multiple endothelial intraluminal extensions C) A capillary in the myocardium showing extensive caveolar activity and location of nanoparticles inside endothelial cells (arrowheads), while the red blood cell shows also nanoparticulates (long arrow) D) A chronically damaged olfactory bulb vessel with thick basement membranes (BM) A significant amount of fibrillary material is seen in perivascular location (*). Basically, Mexico City residents have no normal neural or heart blood vessels from childhood. E) One micron toluidine blue section of nasal respiratory epithelium, extensive areas of breakdown of the barriers are common findings (*), while ciliated epithelial cells show a decreased number of cilia (arrows). F) A lower cervical pτ positive neuron in an 11-year-old female. G) Hyperphosphorylated tau neurites in the substantia nigra of a female teen. H) Alpha-synuclein is positive in substantia nigra neurons in a young adolescent. I) An older subject shows Lewy bodies in substantia nigra neurons. The early spectrum of both AD and PD pathology is seen from pediatric ages.
SARS-CoV-2 arrives to MMC residents that have opened barriers for easy access to the brain. Figure 4 shows electron microscopic pictures with the typical lesions in≤40 year old MMC subjects. The extensive NVU and nasal and olfactory bulb damage and the hallmarks of AD and PD are seen in childhood and progress as the subject ages in MMC. SARS-CoV-2 takes advantage of the endothelial activation and the organelle damage and the opening capillary gaps to the neural space. Brain hemorrhages and strokes likely relate to the baseline host vascular damage and expectedly, subjects with morbidities such as hypertension, will have the highest risk for brain acute involvement. ACE2 status would be critical here since the depletion of ACE2 is key in the pathogenesis of SARS-CoV-2. ACE2 is depleted in MMC residents, in keeping with the AD literature [202].
CONCLUSIONS AND FUTURE DIRECTIONS
The SARS-CoV-2 virus has caused a complex serious health global problem and in Mexico a pre-COVID disastrous economic performance for 2020 with a contraction in gross domestic product worsen the situation. According with the National Council for the Evaluation of Social Development Policy (CONEVAL, [10]) the number of Mexicans in 2018 living in poverty was approximately 53.4 million with projections of 10 million more associated with the COVID crisis. Bello-Chavolla et al. [308] proposed a predictive score for SARS-CoV-2 lethality including age≥65 years, diabetes, early-onset diabetes, obesity, chronic kidney disease, hypertension, and immunosuppression significantly discriminated lethal from non-lethal cases (C-statistic = 0.823). Strikingly, obesity mediated 49.5% of the effect of diabetes on COVID-19 lethality. Héctor Hiram Hernández Bringas [309], researcher at the Universidad Nacional Autónoma de México (UNAM), has clear information about the vulnerable affected groups: 84% of SARS-CoV-2 deaths are poor and unemployed citizens, including housewives (28.1%), street vendors, police, factory workers, brick layers (16.8%), retired workers and pension workers (12%), government workers (including health workers) (11.7%), and public transportation workers (8.5%). The fact 71.2% of affected Mexican citizens only had elementary education or they never attended school reflects their lack of access to preventive medicine, healthy diets, and steady jobs. Only 13.1% of deaths included citizens that attended college or had graduate school education. For MMC, the epicenter of the country’s pandemic, the situation is grim; the highest number of COVID 19 cases and the highest mortality rate (∼32%) are located in the Metropolitan area coinciding with the highest numbers of unemployed people, lower SES, violence, drug addition, food insecurity, lack of water, and no access to health coverage. Mexico ranks near the bottom in testing among Latin American countries: 99 tests per 100,000 residents as of May 15, 2020 [310].
This is our reality as health providers, in a city where development of AD starts in childhood and we can stage AD in 99.25% of 134 consecutive autopsies≤30 years and where 66% of≤30 years urbanites have cognitive impairment. SARS-CoV-2 infections represent a significant threat for our population.
These are our needs: 1) To accurately surveil the number of individuals with exposure to SARS-CoV-2. Understanding the patterns of viral distribution among our population is key for interventions to control virus spread and dissemination coupled with implementation of protocols to detect neurological and psychiatric changes in exposed subjects.
2) Defining the role of exposures to air pollution, occupation, age, and gender, and the higher risk of SARS-CoV-2 should be prioritized. We know a priori air pollution is a risk factor for the development of both AD and PD in MMC residents, thus adding the risk of SARS-CoV-2 to the equation will translate in a potential acceleration of neuroinflammatory and neurodegenerative processes.
3) MMC children are historically a group characterized by significant systemic and neural inflammation, immunosuppression, cognitive impairment, and brain spectroscopic and magnetic resonance abnormalities [311–321]. Because of the mild or asymptomatic clinical SARS-CoV-2 infections in pediatric populations and their capacity to transmit the disease within the family, school, and play environments, we need to test our seemingly health pediatric populations and explore their response to the virus. Our major concern in pediatric cohorts is the development of neurological and/or psychiatric short and long-term complications resulting from the synergistic action of environmental air pollution and SARS-CoV-2. Acceleration of their neuroinflammation and progression of AD and PD pathology are particularly troublesome in children.
4) Forensic autopsies surveillance for SARS-CoV-2 is a must. We need to know where the virus can be localized and do exhaustive brain sampling to look for viral pathology and AD/PD neuropathology including staging for both and sampling ocular conjunctiva, cerebrospinal fluid, supra and infratentorial brain anatomical regions, and the neuroenteric system.
There is no doubt that millions of Mexicans and millions of people across the world are exposed to complex mixtures of air pollutants and to the new SARS-CoV-2 virus and their responses to both will be an interplay of genetics, systemic and neural inflammatory responses, insulin resistance, hyperleptinemia, diabetes, APOE4, lifestyle, socioeconomic factors, stress, violence, etc., signaling the trajectory of young people toward an unknown scenario. Are we prepared for the short- and long-term health effects of two major problems air pollution and viral neurotropic viruses? Are we prepared to surveil behavioral, cognitive, and neurological manifestations in children, teens, and young adults exposed to SARS-CoV-2 and air pollution? Are we ready to identify vulnerable populations and protect them? We are witnessing in MMC an accelerated AD process with striking progressive timelines starting in the pediatric ages. It is imperative to understand we also have disease modulators at work and equally important several abnormal folding proteins are identified in young urbanites. Nanoparticles are key for the strong damage to critical neural, heart, gastrointestinal, and neuroenteric structures and damage to mitochondria, endoplasmic reticulum, mitochondria-associated endoplasmic reticulum, chromatin, and nuclear and cytoplasmic membranes along the early and sustained oxidative stress complete the cellular havoc we are documenting in children and teens. The cognition deficits are literally devastating [321].
MMC is a megacity with specific, well-defined PM problems: heavy diesel trucks are a major pollution source and excluding them from regulation is a major error, severely affecting 25 million people. For physicians, infectologists, pediatricians, neurologists, toxicologists, and neuropathologists, our goal is to learn how SARS-CoV-2 manage to enter the brain at ease, and how NPs contribute to the process. Is the neural damage synergistic? Are we accelerating the neurodegenerative process? Are we on verge of significant increments in suicides related to the neuroinflammation and advanced AD stages? We also need early non-invasive tools to diagnose AD [318, 322] and how exposures to ubiquitous radiofrequency electromagnetic radiation, mostly microwave radiation generated for wireless communication and surveillance technologies [323, 324] is making havoc of our brain’s organelles. The complexity of AD and PD is now accompanied by the neurotropic capabilities of an unexpected guest and clinical challenges of diagnosing them at early stages await all of us on Earth.
DISCLOSURE STATEMENT
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/20-0891r1).
