Abstract
There are a number of potential implications for the field of Alzheimer’s disease (AD) stemming from the global spread of SARS-CoV-2. Neuroinflammation is known to be a prominent feature of neurodegeneration and plays a major role in AD pathology. Immune response and excessive inflammation in COVID-19 may also accelerate the progression of brain inflammatory neurodegeneration, and elderly individuals are more susceptible to severe outcomes after SARS-CoV-2 infection. Individuals with type 2 diabetes (T2D) are at an increased risk for AD as well as severe outcomes after SARS-CoV-2 infection. Genetic and socioeconomic factors influencing the rates of T2D, AD, and COVID-19 severity may create an exceptionally high-risk profile for certain demographics such as African Americans and Hispanic Americans. Type I interferon response plays an important role in both host response to viral infection, as well as AD pathology and may be a sensible therapeutic target in both AD and COVID-19.
The novel coronavirus SARS-CoV-2 emerged in China in late 2019 and its origin has been traced to a market in Wuhan, China [1]. The virus responsible for COVID-19 subsequently spread throughout the world and was declared to be a global pandemic by the World Health Organization on March 11, 2020 [2]. As of May 3, 2020, there were a total of 3,349,786 COVID-19 cases and 238,628 fatalities globally [3]. As our knowledge of the COVID-19 has progressed, advanced age has become recognized as one of the predominant risk factors associated with severe outcomes after infection with those over 59 years of age being 5 times more likely to die from COVID-19 than younger people [4]. The predominance of additional risk factors such as cardiovascular disease and metabolic disorders indicates that biological age (as opposed to chronological age) may be the single biggest risk factors toward severe or fatal outcomes [5]. Diabetes is considered a major comorbidity factor for COVID-19 related fatalities [6]. Type 2 diabetes (T2D), the most common form of diabetes, has also been linked with an increased predisposition to Alzheimer’s disease (AD) through shared genetic etiologies [7]. Genetic and socioeconomic factors have compounded the risk of AD, T2D, and COVID-19 in certain populations. African Americans and Hispanics had 1.5–2 times higher incidence of both T2D and AD compared to non-Hispanic whites [8, 9]. These racial and ethnic disparities are further exacerbated in COVID-19. For example, in Wisconsin and Louisiana, African Americans account for 70% of COVID-19 deaths despite accounting for only 26% and 32% of those populations, respectively [10]. In New York City, African Americans account for 24.3% of the population but accounted for 35.4% of COVID-19 related hospitalizations and 30% of COVID-19 related deaths. Similarly, Hispanics account for 29.1% of the population but accounted for 31.3% of COVID-19 related hospitalizations and 30.4% of COVID-19 related deaths [11]. Given the parallels between higher rates of T2D, AD, and COVID-19 along with the fact that diabetes has been identified as a major comorbidity factor for COVID-19, these demographics are at an increased risk of severe outcomes related to COVID-19.
Severe outcomes after SARS-CoV-2 infection are often associated with a “cytokine storm” of pronounced inflammation [12]. One of the reasons that elderly individuals are at a higher risk after infection may be due to a higher existing baseline of inflammation. Inflammation steadily increases with age in a process referred to as “inflamm-aging” [13]. The “cytokine storm” associated with COVID-19 has been associated with an increase of pro-inflammatory cytokines such as tumor necrosis factor-alpha, interleukin-1 (IL-1), and IL-6 all of which have been identified to be increased in the elderly [13, 14]. Thus, rendering the elderly particularly vulnerable to severe outcomes resulting from COVID-19. Interferon regulatory factor 5 (IRF5) plays a key role in facilitating exaggerated immune response (cytokine storm) after Influenza A virus infection, and is regulated by the glucose sensitive upstream factor O-GlcNAc transferase [15]. The potential for elevations in blood glucose to increase inflammation through IRF5 activity, along with the overlapping pathology of T2D and AD may create a “perfect storm” of excessive immune response after infection with SARS-CoV-2 in AD patients (Fig. 1). Increased IRF5 activity resulting from T2D comorbidity may also synergize with amyloid-stimulated Type I interferon (IFN) response (described below).
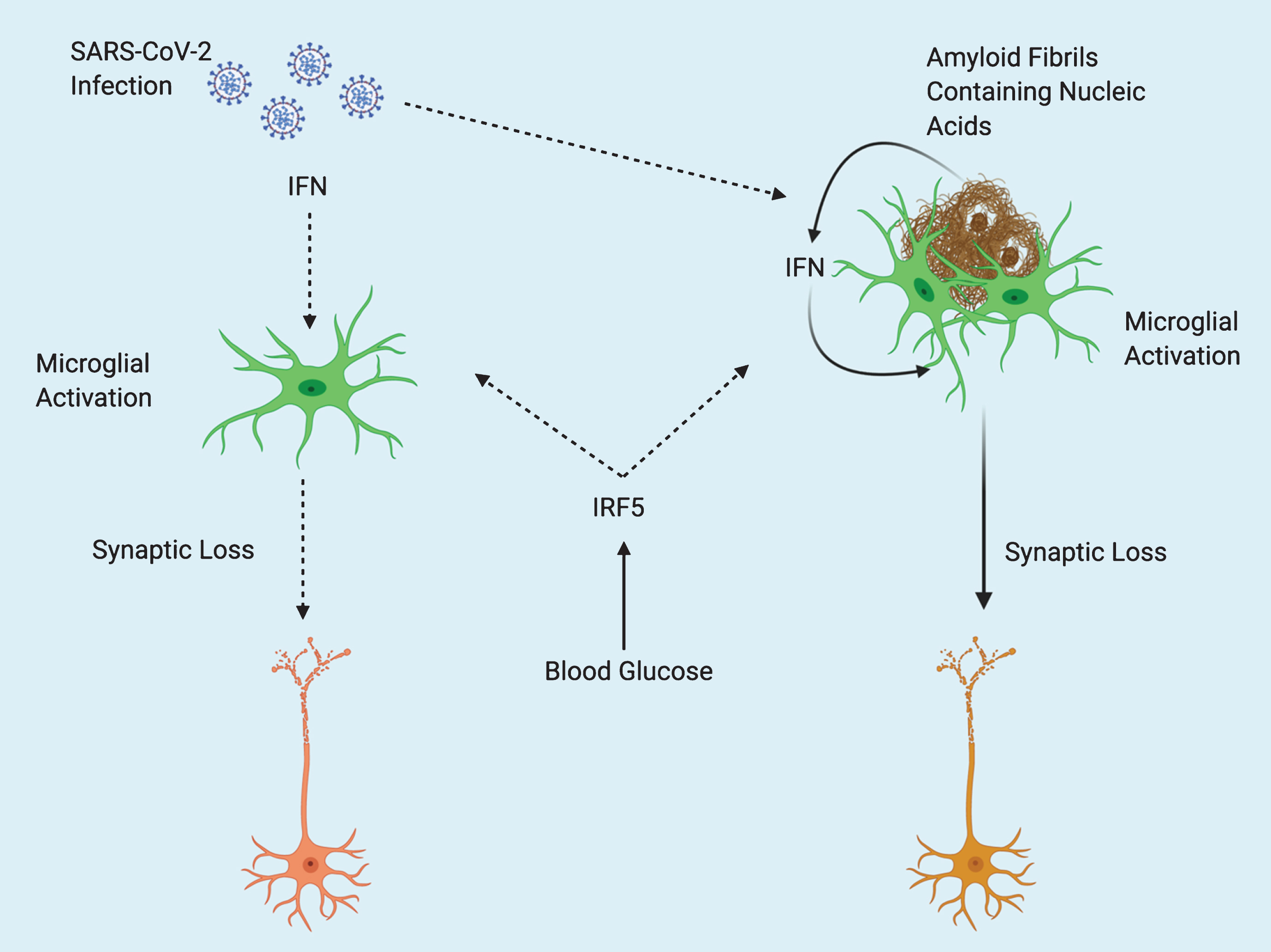
Potential Interactions Between SARS-CoV-2 and Alzheimer’s Disease. Type 2 diabetes (T2D) acts as predisposing factor for Alzheimer’s disease (AD). Elevations in blood glucose resulting from T2D may exacerbate pathology in both AD and COVID-19 or comorbidity of the two, through increased activity of Interferon regulatory factor 5 (IRF5). Type I interferons (IFN) mediate inflammation after viral infection, and in response to nucleic acid containing amyloid fibrils, eventually leading to synaptic loss. Amyloid fibrils may entrap viral particles, leading to further enhancement of IFN response. Solid line arrows indicate proven mechanisms, dotted line arrows indicate theoretical mechanisms.
IFNs are a group of cytokines that play an important role in mediating host response to viral infection [16]. Roy and colleagues recently demonstrated the role of IFN in mediating AD pathology. Hippocampal tissues from multiple different transgenic mouse models of AD were all shown to have increased expression of genes known to be induced by IFN. Furthermore, nucleic acid containing amyloid fibrils were shown to stimulate the expression of genes known to be involved with IFN response both
While the potential for IFN response in AD and COVID19 to synergize and exacerbate pathology has yet to be explored, IFN response may also play a role in the immediate effects of COVID-19 on the brain. The acute neurological complications resulting from SARS-CoV-2 infection include meningitis and acute necrotizing encephalopathy, as well as gustatory and olfactory impairments [20 , 23]. IFN has previously been associated with viral encephalitis as well as the inherited encephalopathy Aicardi–Goutières syndrome [24, 25].
Suppression of IFN response in both AD and COVID-19 (or comorbidity of the two) may be an advantageous strategy for controlling excessive immune response. Additionally, it is interesting to speculate on how AD therapeutics might interact with SARS-CoV-2 infection. Currently, the approved treatments donepezil, rivastigmine, and galantamine all act by increasing acetylcholine levels by inhibiting its degrading enzyme acetylcholinesterase [26]. While not yet explored, the cholinergic anti-inflammatory pathway may be a viable target for controlling inflammation and preventing a “cytokine storm” after SARS-CoV-2 infection, and acetylcholinesterase (AChE) inhibitors could potentially be repurposed as an adjunctive therapy. Interestingly, the AChE inhibitor galantamine has been shown to decrease serum cytokines and chemokines such as TNF-
It is also important to recognize the indirect effects that the COVID-19 pandemic will likely have on AD patients. Because elderly individuals are at a higher risk for mortality after SARS-CoV-2 infection, it is necessary to isolate these individuals and limit their contact with other people during this pandemic. However, time spent with caretakers and family, as well as social interaction and daily activity are generally considered to help prevent cognitive decline in the elderly. Thus isolation, while necessary may unfortunately carry increased risk of cognitive decline in elderly individuals. While the sum total effects of the COVID19 pandemic are not yet known it will undoubtedly affect many AD patients either directly or indirectly. Only large-scale retrospective analysis in addition to preclinical research will reveal the full range of implications for aging and neurodegeneration.
Footnotes
ACKNOWLEDGMENTS
This study was supported by Grant Number P50 AT008661-01 from the NCCIH and the ODS. Dr. Pasinetti holds a Senior VA Career Scientist Award. We acknowledge that the contents of this study do not represent the views of the NCCIH, the ODS, the NIH, the U.S. Department of Veterans Affairs, or the United States Government. ![]() was made using Biorender.com.
was made using Biorender.com.
