Abstract
OBJECTIVE:
Multiparametric magnetic resonance imaging (mpMRI) –Ultrasound- fusion guided biopsy of the prostate (FBx) is the new gold standard for the detection of prostate cancer. Hallmark studies showing superior detection rates of FBx over randomized biopsies routinely excluded patients≥75 years and information on outcome of FBx on this patient cohort is sparse. As a large referral center, we have performed FBx on a substantial number of patients this age. By evaluating outcome of FBx of patients over the age of 75 years we wanted to close the gap of knowledge on this patient cohort.
MATERIALS AND METHODS:
Between 2015 –2022, 1577 patients underwent FBx at our department and were considered for analysis. Clinical and histopathological parameters were recorded. Clinical data comprised age at FBx, serum level of Prostate-specific antigen (PSA), prostate volume, PSA-density, history of previous biopsies of the prostate, result of the digital rectal examination (DRE) and assessment of the indexlesion of mpMRI according to the Prostate Imaging and Reporting Data System (PI-RADS). Univariate analysis and multivariable logistic regression was used to identify age barrier of 75 years as a potential risk factor of detection of clinically significant prostate cancer by FBx.
RESULTS:
379/1577 patients (24%) were≥75 years and 1198/1577 (76%) patients were < 75 years, respectively. Preoperative PSA was significantly higher in patients≥75 years compared to patients < 75 years (9.54 vs. 7.8, p < 0.001). Patients≥75 years presented significantly more often with mpMRI target lesions classified as PI-RADS 5 compared to patients < 75 years (45% vs. 29%, p < 0.001). Detection rate of clinically significant prostate cancer was significantly higher in patients≥75 years compared to patients < 75 years (63% vs. 43%, p < 0.001). Aggressive prostate cancer grade ISUP 5 was significantly more often detected in patients≥75 years compared to patients < 75 years (13% vs. 8%, p = 0.03). On multivariable logistic regression model adjusted for PSA and PI-RADS score, age barrier of 75 years was identified as a significant risk factor for the detection of clinically significant prostate cancer by FBx (OR: 1.77, 95% CI: 1.36 –2.31, p < 0.001).
CONCLUSION:
After evaluation of a large patient cohort, we show that age≥75 years represents a significant risk factor for the detection of clinically significant prostate cancer. Further studies on mid- and long term outcome are necessary to draw conclusions for clinical decision making in this patient cohort.
Background
Prostate cancer is the most common malignancy in men [1]. Incidence of prostate cancer is strongly related to age and reaches a peak in the age group of 75 –79 [1, 2]. Multiparametric magnetic resonance imaging (mpMRI) –Ultrasound- fusion guided biopsy of the prostate (FBx) emerged as a new cornerstone in the diagnostic pathway of prostate cancer [3–7]. Several randomized controlled studies showed superior detections rates of clinically significant prostate cancer, defined as grading > 1 according to the International Society of Uropathologists (ISUP), compared to randomized biopsy [8–12]. European guidelines adapted these results, recommending FBx over randomized biopsy [13]. However, many of the hallmark trials excluded patients over 75 years [9, 10]. In fact, guidelines also recommend not to screen for prostate cancer in patients with a life expectancy of 10 years or less[14]. According to epidemiological statistics in Germany, this would account for patients who are 75 years or older[15]. Therefore, information on assessment and outcome of FBx on this age group is sparse. As a high-volume referral center for FBx for private urological practices, we have been performing FBx on 1577 patients for the past seven years. A substantial number of patients during this period were referred to us even though being 75 years of age or older. By evaluating outcome of FBx in these patients we want to close a gap of knowledge which was left out by large trials before.
Material and methods
We retrospectively reviewed our prospectively maintained database of patients undergoing Fbx at the outpatient clinic of the Department of Urology at LMU Klinikum in Munich, Germany. Between March 2015 and August 2022, 1577 patients underwent Fbx and were considered for analysis. Patients with clinical signs of metastasis on mpMRI were excluded from this study. Patients were referred by either their office urologist or by the outpatient clinic of the Department of Urology of the Ludwig-Maximilian-University of Munich, Germany. 398/1577 patients (25%) underwent mpMRI at the Department of Radiology of Ludwig-Maximilian-University of Munich, Germany, while 1179/1577 (75%) underwent FBx with mpMRI assessed by a heterogeneous group of radiology offices, including university hospitals, private practices and of radiology offices in foreign countries. Altogether, patients underwent FBx with mpMRI from 111 different radiology offices. mpMRI is not reviewed by a local domestic radiologist at our department before FBx. Clinical and histopathological parameters were recorded. Clinical data comprised age at FBx, serum level of Prostate-specific antigen (PSA), prostate volume, PSA-density (calculated by PSA divided by prostate volume [ng/ml/ccm3]), history of previous biopsies of the prostate, result of the digital rectal examination (DRE) and assessment of the indexlesion of mpMRI according to the Prostate Imagig and Reporting Data System (PI-RADS) [16]. Histopathological data comprised the number of cancer positive biopsy cores, grading of prostate cancer according to the International Society of Uropathologists (ISUP) [17] and the ratio of tumor infiltration per cancer positive biopsy core. Histopathological examination was assessed by the Department of Pathology at LMU Klinikum in Munich, Germany. Documentation of clinical data is inconsistent throughout the study and recorded as follows: Prostate volume in 1318/1577 (84%), history of previous biopsies of the prostate in 1351/1577 (86%) patients, result of DRE in 817/1577 (52%) and localization of index lesion in preoperative mpMRI 1419/1577 (90 %) patients. Surgeons conducting FBx comprised a group of ten urologists with different levels of experience, but at least 100 independently performed FBx altogether. In every patient an additional systematic biopsy (SBx) comprising of 6 cores from the left and right side respectively (base, mid and apical gland) was performed according to current guidelines [14]. Fusion of the mpMRI and real-time ultrasound was performed using plane wise fusion. The axial T2-weighted-MRI-sequence was used for image fusion. Software and Hardware for assessment of FBx was provided by Epiq7, Philips Percunav®, Philips Medical Systems, Bothell, WA. FBx of up to 3 mpMRI-lesions was performed and a mean of three cores per targets were obtained. Univariate analyses were performed using Mann-Whitney U Test for continuous variables and Chi-square test for categorical variables. Multivariable logistic regression analysis was performed on clinical parameters PSA, PI-RADS score (linear regression PI-RADS 3 –5) and≥75 years vs.<75 years to identify potential influence on the detection of clinically significant prostate cancer by FBx. A p value of < 0.05 was considered statistically significant. All calculations were carried out using the R statistical program (version 3.6.3) and Prism 6 (GraphPad Software, San Diego, CA). This study was approved by the local ethics committee (22-0318).
Results
Patient characteristics
Patient characteristics are displayed in Table 1. Overall, 1577 patients underwent FBx at the outpatient clinic of the Department of Urology at LMU Klinikum in Munich, Germany, between March 2015 and August 2022. Median age was 68 years (Interquartile range IQR 62 –74). Median PSA was 8.05 ng/ml (IQR 5.7 –12.1). Median prostate volume was 48 ccm3 (IQR 34 –69). Median PSA density was 0.17 ng/ml/ccm3 (IQR 0.11 –0.26). 41% of patients had a prior biopsy of the prostate. 38% of patients had a DRE suspicious of prostate cancer. Concerning PI-RADS score of the mpMRI targets, 17% of mpMRI targets were classified as PI-RADS 3, 50% as PI-RADS 4 and 33% as PI-RADS 5, respectively. Splitting the patient cohort according to age,≥75 years versus < 75 years, we saw significant differences in clinical parameters at clinical presentation before FBx.
Demographic and clinical patient characteristics. Continuous values are presented as median and inter-quartile-range (IQR); categorical values are given as number (n; %). PSA: Prostate specific antigen; P-Vol: Prostate Volume; PSAd: PSA-density; DRE: Digital rectal examination; PI-RADS: Prostate imaging and reporting Data System
Demographic and clinical patient characteristics. Continuous values are presented as median and inter-quartile-range (IQR); categorical values are given as number (n; %). PSA: Prostate specific antigen; P-Vol: Prostate Volume; PSAd: PSA-density; DRE: Digital rectal examination; PI-RADS: Prostate imaging and reporting Data System
Detailed patient characteristics adjusted to age group is displayed in Table 2. In detail, 1198 patients were < 75 years old and 379 patients were≥75 years, respectively. PSA of patients≥75 years old was significantly higher compared to patients < 75 years, respectively (9.54 ng/ml (IQR 6.6 –14.7) vs. 7.8 ng/ml (IQR 5.49 –11.6), p < 0.001). Prostate volume of patients≥75 years was significantly higher compared to patients < 75 years, respectively (52 ccm3 (IQR 36 –70) vs. 47 ccm3 (IQR 34 –67), p = 0.03). PSA density of patients≥75 years was significantly higher compared to patients < 75 years, respectively (0.18 ng/ml/ccm3 (IQR 0.12 –0.3) vs. 0.16 ng/ml/ccm3 (IQR 0.11 –0.25), p = 0.002). Concerning history of prostate biopsy, we saw no significant difference comparing patients≥75 years and < 75 years (37% vs. 42%). Patients≥75 years showed significantly more often a suspicious DRE compared to patients < 75 years (45% vs. 35%, p = 0.01). Concerning PI-RADS score of the mpMRI targets, mpMRI targets of patients≥75 years were significantly less classified as PI-RADS 3 compared to patients < 75 years (11% vs. 19%, p < 0.001). Likewise, mpMRI targets of patients≥75 years were significantly less classified as PI-RADS 4 compared to patients < 75 years (44% vs. 52%, p = 0.004). However, mpMRI targets of patients≥75 years were significantly more frequently classified as PI-RADS 5 compared to patients < 75 years (45% vs. 29%, p < 0.001).
Age adjusted demographic and clinical patient characteristics:<75 years and≥75 years old. Continuous values are presented as median and inter-quartile-range (IQR); categorical values are given as number (n; %). Bold values indicate p-values<0.05 and were considered statistically significant. PSA: Prostate specific antigen; P-Vol: Prostate Volume; PSAd: PSA-density; n.s.: not significant; DRE: Digital rectal examination; PI-RADS: Prostate imaging and reporting Data System
Histopathological outcome after FBx is displayed in Table 3. Overall, 755/1577 patients (48%) were diagnosed with clinically significant prostate cancer by FBx. Ratio of patients with clinically significant prostate cancer detected by FBx was significantly higher in patients≥75 years compared to patients < 75 years (63% vs. 43%, p < 0.001). Patients≥75 years were significantly less often diagnosed with prostate cancer ISUP 2 compared to patients < 75 years (38% vs. 47%, p = 0.02). While detection of prostate cancer ISUP 3 and 4 did not differ between patients≥75 years and patients < 75 years, patients≥75 years were significantly more often diagnosed with prostate cancer ISUP 5 compared to patients < 75 years (13% vs. 8%, p = 0.03). Number of cancer positive biopsy cores and median of highest tumor infiltration per cancer positive biopsy core did not differ significantly between patients≥75 years and patients < 75 years.
Overall and age-adjusted clinical and histopathological characteristics of patients with clinically significant prostate cancer detected by mpMRI-targeted prostate biopsy:<75 years and≥75 years old. Continuous values are presented as median and inter-quartile-range (IQR); categorical values are given as number (n; %). Bold values indicate p-values<0.05 and were considered statistically significant. PSA: Prostate specific antigen; P-Vol: Prostate Volume; PSAd: PSA-density; DRE: Digital rectal examination; PI-RADS: Prostate imaging and reporting Data System; ISUP: International Society of Uropathologists
Overall and age-adjusted clinical and histopathological characteristics of patients with clinically significant prostate cancer detected by mpMRI-targeted prostate biopsy:<75 years and≥75 years old. Continuous values are presented as median and inter-quartile-range (IQR); categorical values are given as number (n; %). Bold values indicate p-values<0.05 and were considered statistically significant. PSA: Prostate specific antigen; P-Vol: Prostate Volume; PSAd: PSA-density; DRE: Digital rectal examination; PI-RADS: Prostate imaging and reporting Data System; ISUP: International Society of Uropathologists
We evaluated the clinical parameters PSA, PI-RADS score and age barrier of 75 years before FBx in a univariate and multivariable logistic regression model in order to assess the influence of age barrier of 75 years as a risk factor for the diagnosis of clinically significant prostate cancer by FBx (Table 4 and Fig. 1). In univariate analysis we identified age barrier of 75 years as a significant risk factor for the diagnosis of clinically significant prostate cancer by FBx (Odd ratio OR: 2.20, 95% Confidence Interval CI: 1.71 –2.83). After adjusting for PSA and PI-RADS score, age barrier of 75 years remained a significant risk factor for the diagnosis of clinically significant prostate cancer by FBx (OR: 1.77, 95% CI: 1.36 –2.31, p < 0.001).
Univariate and multivariable logistic regression models for clinically significant prostate cancer. Bold values indicate p-values<0.05 and were considered statistically significant. OR: Odd-ratio; 95% CI: 95% Confidence interval; PSA: Prostate specific antigen; PI-RADS: Prostate imaging and reporting Data System
Univariate and multivariable logistic regression models for clinically significant prostate cancer. Bold values indicate p-values<0.05 and were considered statistically significant. OR: Odd-ratio; 95% CI: 95% Confidence interval; PSA: Prostate specific antigen; PI-RADS: Prostate imaging and reporting Data System
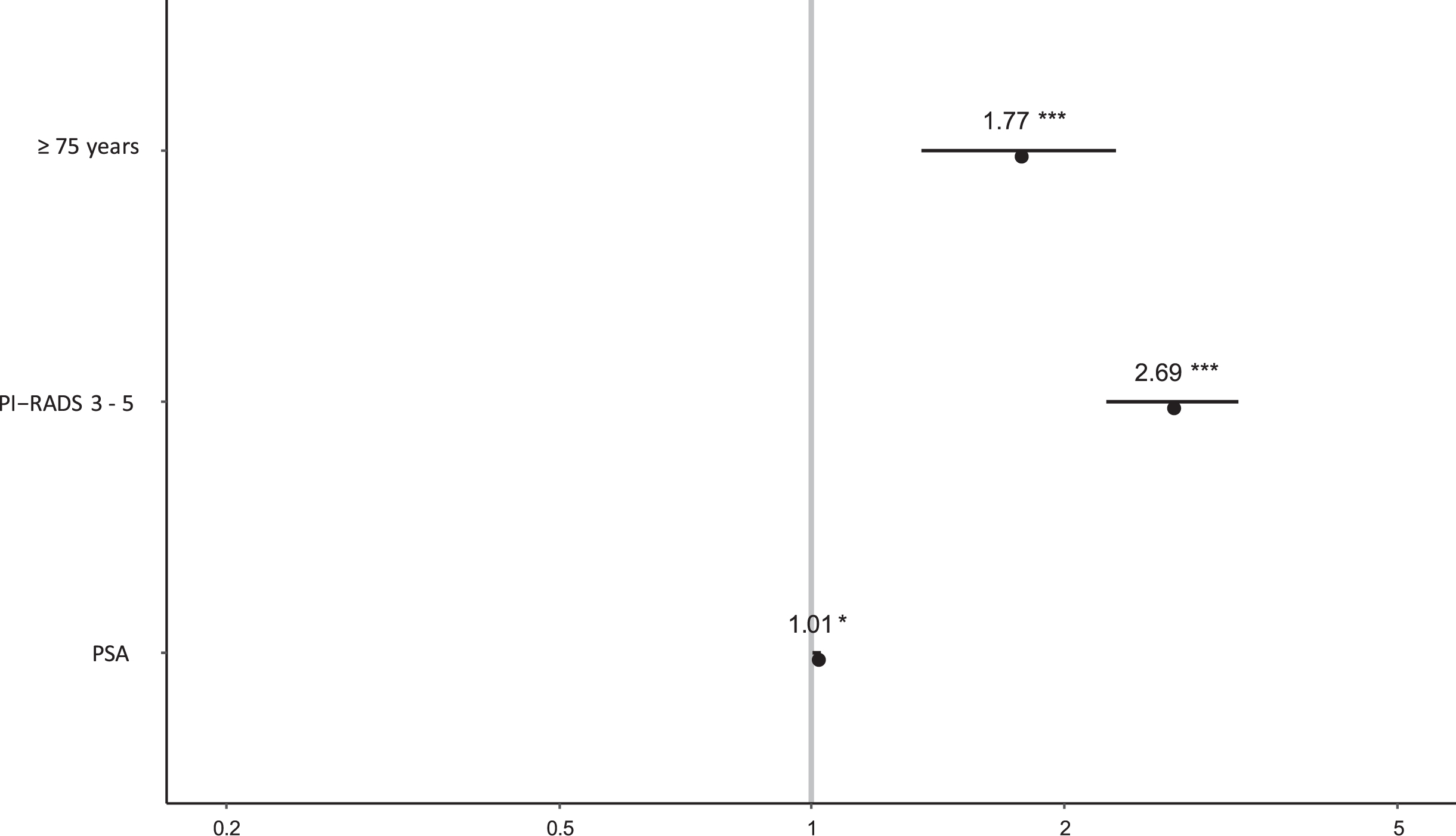
Age barrier of 75 years as a significant risk factor for the diagnosis of clinically significant prostate cancer.
Patients who are 75 years or older represent an age group, which was often excluded in large trials assessing mpMRI-targeted biopsy. We therefore aimed to investigate clinical outcome of FBx in this patient cohort. We saw that patients≥75 years presented more often with significantly more severe clinical parameters indicating prostate cancer compared to patients < 75 years. Furthermore, we saw that detection rate of clinically significant prostate cancer in patients≥75 years was significantly higher compared to patients < 75 years (63% vs. 43%, p < 0.001). Patients≥75 years were significantly more often diagnosed with highly aggressive prostate cancer ISUP 5 compared to patients < 75 years (13% vs. 8%, p = 0.03). On multivariable analysis adjusted for PSA and PI-RADS score, age barrier of 75 years represented a significant risk factor for the diagnosis of clinically significant prostate cancer (OR: 1.77, 95% CI: 1.36 –2.31, p < 0.001).
Reasons for restricting prostate cancer screening and diagnostics to patients with a life expectancy of 10 years or more are inherent to the slow progression of the disease [14]. According to guidelines, the goal of screening and diagnostics of prostate cancer is the identification in an organ-confined state with curable intent [14]. However, studies also show that clinical progression of aggressive prostate cancer occurs also within 10 years or less. In a screening study, Rud et al. performed lumbar column and pelvis MRI after diagnosis of prostate cancer in 390 treatment naïve men [18]. They could show that 23% of patients diagnosed with ISUP 5 prostate cancer showed lymph node metastases [18]. 15% of patients diagnosed with ISUP 5 prostate cancer showed bone metastasis [18]. In our study, patients≥75 years were significantly more often diagnosed with ISUP 5 compared to patients < 75 (13% vs. 8%, p = 0.03). Even though these patients might not represent a curable state, they might benefit from systemic treatment in terms of cancer-specific and progression-free survival. In a retrospective observational study with a median follow-up of 24 years, Albertsen et al. showed that men with high-grade prostate cancer (ISUP 4 and 5) had a high probability of cancer-related death within ten years of diagnosis (121 deaths per 1000 person-years; 95% CI: 90 –156) [19]. In an analysis of the Surveillance, Epidemiology and End-Results (SEER) program, Clark et al. identified 116.796 patients diagnosed with prostate cancer between 1992 and 1997 and followed them for 20 years throughout the registry [20]. The authors found that risk of prostate cancer related death was 17% for men younger than 70 years and 21% for men older than 70 years [20]. Men with Gleason score≥7 showed a peak in mortality 2 –3 years after diagnosis [20]. The authors concluded that high grade prostate cancers are aggressive from the outset [20]. Godtman et al. evaluated the influence of age on prostate cancer diagnosis throughout the screening arm of the Göteborg-1 screening trial [21]. The authors found that for each year increase in age, risk of being diagnosed with clinically significant prostate cancer increase by 11% (95% CI: 4.7 –17) [21]. Recent developments in androgen deprivation therapy proofed to significantly prolong progression-free survival and improve health-related quality of life compared to placebo [22, 23]. Therefore, we believe our data support the referral of patients≥75 years for FBx to avoid clinically symptomatic prostate cancer in patients with high suspicion of the disease. Comparing clinical presentation of patients≥75 years and < 75 years, one can assume that peripheral urologists assess a threshold in referring patients over 75 years to our department. In our study, patients≥75 years presented with a significantly higher PSA compared to patients < 75 years (9.54 IQR 6.6 –14.7 vs. 7.8 IQR 5.49 –11.6, p < 0.001). Patients≥75 years presented significantly more often with PI-RADS 5 target lesions compared to patients < 75 years (45% vs. 29%, p < 0.001). Other data on histopathological outcome of FBx in elderly patients is sparse. Recently, Luan et al. assessed the outcome of FBx on 43 patients≥80 years, with a PSA of≥100 ng/ml and/or a target lesion of PI-RADS 5. The authors showed a detection rate of 90.7 % of clinically significant prostate cancer. Complication rate was moderate with haematuria in 10.5% of patients and urinary retention in 2.6% of patients, respectively [24]. The detection rate in this study was much higher compared to the detection rate of patients≥75 years in our study (63%). However, PSA levels of the included patients differed substantially between our study and Luan et al. Huh et al. retrospectively reviewed the feasability of FBx in 216 elderly patients with high levels of serum PSA. Median age was 79.5 (Range 75 –87) and median PSA was 81.3 ng/ml (Range 20.0 –869.0). Detection rate of clinically significant prostate cancer was 80%. Unfortunately, the authors did not provide detailed information on perioperative mpMRI and postoperative histopathology according to ISUP. Again, differences in detection rates of our study compared to Huh et al. are probably due to substantial differences in inclusion criteria. To our knowledge, none of the existing studies explicitly assessed an age barrier of life expectancy under 10 years as a risk factor for detection of clinically significant prostate cancer by FBx.
Our study is limited by its retrospective, monocentric design. However, since FBx is performed using mainly externally assessed mpMRI at our department, we fulfil a multicentric pattern in terms of assessment of preoperative mpMRI. Another limitation is the lack of preoperative clinical data on prostate volume in 16% of patients, history of previous biopsies of the prostate in 14% of patients, result of DRE in 48% of patients and localization of index lesion in preoperative mpMRI in 10% of patients. Further, lack of mid-and long-term survival of the included patients might limit clinical implication of our results.
Conclusion
On retrospective evaluation of FBx in 1577 patients over the past 7 years we saw that patients≥75 years presented with significantly more severe clinical parameters indicating high risk prostate cancer compared to patients < 75 years. Furthermore, detection rate of clinically significant prostate cancer by FBx was significantly higher in patients≥75 years compared to patients < 75 years. In a multivariable logistic regression model adjusted for PSA and PI-RADS score, age barrier of 75 years was a significant risk factor of detection of clinically significant prostate cancer by FBx. Further studies on mid- and long-term survival of this patient cohort are required to implement our results in clinical decision making.
