Abstract
BACKGROUND:
ALK receptor tyrosine kinase (ALK) aberrations have an established role in pathogenesis of many neoplasms, but their clinical significance in high grade serous ovarian carcinoma (HGSOC) is unclear.
OBJECTIVE:
To analyse the frequency of ALK overexpression, molecular abnormalities of ALK, and their impact on the progression-free survival (PFS) and overall survival (OS) in HGSOC.
METHODS:
Protein expression was examined by immunohistochemistry (IHC) using three different clones of anti-ALK antibody. The presence of translocations was analysed using fluorescent in situ hybridization. Next-generation sequencing was used for studying the copy number variation, as well as point mutation and translocations involving other commonly rearranged genes.
RESULTS:
ALK overexpression was demonstrated in up to 52% of tumours, whereas ALK copy gains in 8.2%, with no clear impact on survival. ALK point mutations were identified in 13 tumours (8.9%), with 3 belonging to the class IV showing significantly better OS. A trend suggesting better PFS was also noticed in these cases. Additionally, three gene fusions were found: ERBB2-GRB7, PRKCA-BRCA1 and SND1-BRAF, none of which has been previously described in HGSOC.
CONCLUSIONS:
HGSOC harbouring activating ALK mutations might be associated with a better survival, while ALK overexpression and ALK amplification does not impact the prognosis.
Introduction
Every year, over 300,000 women worldwide are diagnosed with ovarian cancer [1], and the overall survival (OS) remains low despite the combination of surgical and systemic treatment, with most of the patients dying within 3 years of diagnosis [2]. Understanding the underlying molecular abnormalities seems to be crucial for selection of the most appropriate therapy and to explore the possibility of new therapeutic options.
ALK receptor tyrosine kinase (ALK, anaplastic lymphoma kinase) is a tyrosine kinase receptor with an established role in the pathogenesis of different malignant neoplasms, including non-small cell lung carcinoma (NSCLC) and neuroblastoma [3]. Depending on the specific tumour type, ALK overexpression as well as ALK translocations and point mutations were proven to have various impact on clinical behaviour. For example, in anaplastic large cell lymphoma (ALCL) the response to classic chemotherapy and overall prognosis is more favourable if a tumour harbours ALK rearrangements. This resulted in the separation of this entity into ALK-positive and ALK-negative ALCL in recent WHO classifications, despite otherwise similar morphology and phenotype of these tumours [4]. In other neoplasms, however, such as neuroblastoma or colorectal carcinoma, ALK amplifications, point mutations and translocations result in worse prognosis and response to treatment [5, 6], or lack of response to certain treatment modalities [7].
The ALK overexpression in ovarian cancer was first described in 2012 by Ren et al. [8]. In a cohort of 69 malignant ovarian neoplasms the phosphorylated form of ALK was found in 3 cases of serous ovarian carcinomas. Another report on the ALK significance in the ovarian cancer documented ALK overexpression identified immunohistochemically in 27% of serous ovarian carcinomas [9]. In the last year an additional report suggesting potential negative clinical implications of ALK overexpression was published, based on in vitro studies and immunohistochemical analysis [10].
The difference in incidence of ALK overexpression in previously published studies indicated the necessity to perform additional tests to establish the actual incidence of ALK aberrations in ovarian carcinoma. As differences in the frequency of various molecular aberrations of ALK may also depend on patients’ ethnicity, as shown in NSCLCs [11] and renal cell carcinoma [12], further analyses seem justified. Prior studies in ovarian carcinoma were based mainly on Asian population, potentially narrowing the genetic landscape of examined tumours, and stipulating the need to perform a study on European patients. The incidence of ALK-positivity in specific populations seems to be particularly relevant in the context of the promising results in the preclinical studies on the antitumor activity of the crizotinib combined with cisplatin in the ovarian cancer cell lines [13].
The main goal of this study was to determine the frequency of ALK overexpression and various ALK aberrations in high grade serous ovarian carcinomas in the Polish population, as well as to examine their role as a potential prognostic and predictive factor for standard chemotherapy. The secondary goal of the study was to establish the value of immunohistochemistry as a screening method in ovarian cancers with various molecular aberrations of ALK.
Materials and methods
Ethical committee approval
The study was approved by the Bioethical Committee of the Medical University of Gdansk (Poland).
Clinical data collection
A total of 150 high-grade serous ovarian carcinomas were selected for the study, 115 of which came from the archives of University Clinical Centre (UCC) of Gdansk (2009–2015), and 35 were obtained from the Military Institute of Medicine in Warsaw (MIM) (2004–2014). Clinicopathological data including date of birth, surgery date, clinical stage, applied therapy, biochemical response based on Ca125 levels and clinical PFS were obtained from hospital documentation. Selection of patients was based on the availability of the clinical data and the cancer tissue in the paraffin blocks. If applicable, date of death was obtained from the PESEL Registry of Polish Ministry of Digital Affairs.
Microarray preparations
Tissue microarrays (TMA) consisting of core sections (diameter: 2 mm) of primary tumors were prepared using the Manual Tissue Arrayer MTA-1 (Beecher Instruments, Inc., Sun Prairie, WI, USA) for the purpose of immunohistochemical staining and hybridization in situ. Non-neoplastic tissue fragments were used as a control and location markers in each microarray.
Immunohistochemistry
4
Next-generation sequencing (NGS)
A custom-designed KAPA HyperChoice Enrichment Kit (Roche Holding, Basel, Switzerland) was used for the detection of single-nucleotide variants (SNVs), insertions, deletions, InDels and Copy Number Variation (CNV) of 21 exons of ALK. Automated target enrichment library preparation was performed by the i7 Robot (Beckman Coulter GmbH, Krefeld, Germany). Sequencing was done on the Illumina platform using NovaSeq 6000 (Illumina, San Diego, U.S.A.). Libraries were sequenced at 2 x 150 cycles and achieved a quality score of
Pathoclinical data of all patients
Pathoclinical data of all patients
No. – number of patients, PFS – progression-free survival (months). OS – overall survival (months).
Results of immunohistochemical analysis
FISH assay for ALK rearrangements was performed on TMAs by using dual-colour break apart ALK FISH probe set (Empire Genomics, Williamsville, NY, USA) according to the standard protocol. At least 100 cells were analysed. The cut-off for potential rearrangement of ALK was set at 10% of cells showing a split or isolated 3’ pattern, and in these cases the hybridization was to be repeated on the whole sections.
FISH assay for ALK amplification analysis was performed on 3
Statistical analysis
PFS was defined as the time from the first diagnosis to the first documented progressive disease (PD) or censored at the last available follow-up. OS was calculated from the date of first diagnosis to the death of any cause or censored at the date of access to the PESEL Registry (consulted the 19.05.2022) for patients living at that time. For survival analysis, only patients treated with the standard chemotherapy regimen of paclitaxel and carboplatin, and with clinical follow-up available, were included.
Statistical analysis was performed using Statistica, version 13 (TIBCO Software Inc., San Francisco, CA, USA). Correlations were assessed using the Kendall
Immunohistochemical (IHC) and fluorescent in situ hybrydization (FISH) analysis. A and B – weak to moderate IHC staining with clone 5A4; C – moderate IHC staining with clone D5F3; D – increased number of ALK signals (green) both independently and in relations to centromer signal (red) in FISH, signifying ALK amplification.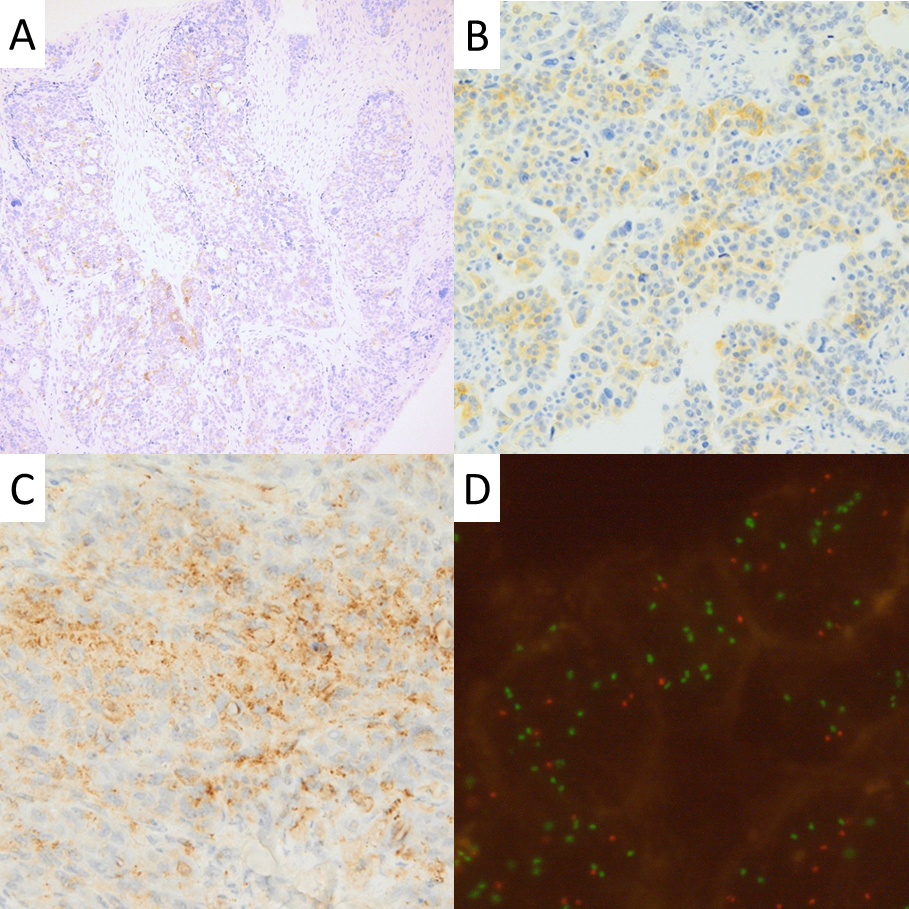
Correlations between immunohistochemistry, copy gain and pathoclinical data
PFS – progression-free survival, OS – overall survival.
Pathological re-evaluation and clinical data analysis
After pathological re-evaluation (A.G. and P.C.) 4 cases were removed from the study as representing entities other than HGSOC. The summary of the clinicopathological data for the remaining 146 cases is shown in Table 1. The median age at admission was 63 years (range 31–82). Data on disease progression was available in 104 cases, with the average time to progression of 12 months and the average time of observation in patients without progression of 91 months. In all these patients, first-line treatment included platinum derivatives-based chemotherapy. Six patients were removed from the survival analysis, four because of the first-line chemotherapy differing from the standard paclitaxel-carboplatin regimen (cisplatin, docetaxel, and capecitabine in one case, paclitaxel-cisplatin in another and cisplatin in monotherapy in two others), one due to missing information on treatment in the documentation and one due to the lack of data on overall survival in the PESEL database.
ALK overexpression
The immunohistochemical staining was completely negative with clone ALK1, which was excluded from the statistical analysis, and showed positivity ranging from 0 to 55% in two other clones, with most of the cases showing weak and focal cytoplasmic staining (Fig. 1a–c; Table 2), with only single cases presenting with concomitant membranous positivity. Statistical analysis showed a weak negative correlation between D5F3 positivity and disease progression – both during 5 years of observation (Kendall’s
ALK rearrangements and amplification
FISH analysis with break-apart probe did not show any translocations including ALK locus. NGS results showed ALK copy gains in 11 cases, with CNV varying between 2.07 and 2.81. Statistical analysis showed weak correlation between the positive staining with 5A4 clone and the copy gain presence (Kendall’s
ALK point mutations
NGS target coverage was at least 99% at 200x. ALK point mutations were found in 13 tumours (Table 4, patients 1–13), including three categorized as class IV variants, all of which came from stage IIIC HGSOC. The average PFS in these patients was longer than in other patients with progression (30 vs. 11 months). One of the patients harbouring the class IV mutation was still alive after 138 months of observation. The OS of the two other patients was also longer than the average (49 and 72 months vs. 38 months). Survival analysis confirmed that for patients with class IV ALK mutations OS was significantly longer than for the other patients from the similar stage group (Fig. 2a, Table 5). A similar trend was visible, albeit with no statistical significance, for OS in all patients regardless of the stage, as well as in PFS analysis (Fig. 2b–d, Table 5).
ALK point mutations
Fusion and breakpoint detection using FACTERA has shown rearrangements in three cases: ERBB2-GRB7 (break support 154/59), SND1-BRAF (break support 23/97 and 54/22) and PRKCA-BRCA1 (break support 33/62). None of these tumours showed any ALK aberrations by FISH or by NGS. Clinicopathological data on these patients are summarized in Table 4 (patients 14–16).
Discussion
This study is the first showing better prognosis in patients with potentially pathogenic ALK point mutations. Previous reports, based on in vitro, IHC and genetic profiling, suggested more aggressive behaviour of ALK-positive tumours [10], and worse overall prognosis and response to treatment in patients with ALK point mutations [17, 18]. This, however, was not confirmed in our research, which was based on methodology routinely used in tumour diagnostics.
It is worth noticing that in previously published studies the impact of ALK mutations on prognosis and treatment efficacy was analysed without discriminating between mutation classes. It also included mutations with little to no influence on pathogenesis [18]. It cannot be ruled out that most of those mutations appear as late and secondary events in advanced tumours with instable genome. If this is the case, the notion that they negatively impact the prognosis was already confirmed by the studies of ovarian tumours with high mutation burden [19]. In further studies, it seems appropriate to focus on the impact of aberrations with potentially pathogenic significance, excluding the tangential passenger mutations. Since ALK overexpression is an established positive prognostic factor in ALCL, both with and without ALK translocations [20], and might be associated with a better response to treatment in NSCLC [21], we cannot exclude potentially better prognosis and treatment response in HGSOC associated with pathogenetically relevant ALK-point mutations. Considering the genetic diversity of tumours in different populations, it cannot be completely ruled out that the obtained results may differ from the previous ones also due to the fact that the study was conducted on a different ethnic group.
Summary of pathoclinical data of patients with molecular abberations
ChTH – chemotherapy, P-C – paclitaxel-carboplatin regimen; Patients 1–13 with ALK point mutations, Patients 14–16 with fusions involving genes other than ALK.
Statistical analysis of ALK overproduction and ALK aberations
PFS – progression-free survival, OS – overall survival.
Kaplan-Meier curves of survival for patients with ALK class IV mutations vs other patients. A – Overall survival for stage IIIC HGSOC, B – Overall survival for all stages, C – Progression-free survival for stage IIIC HGSOC, D – Progression-free survival for all stages. OS – overall survival, PFS – progression-free survival.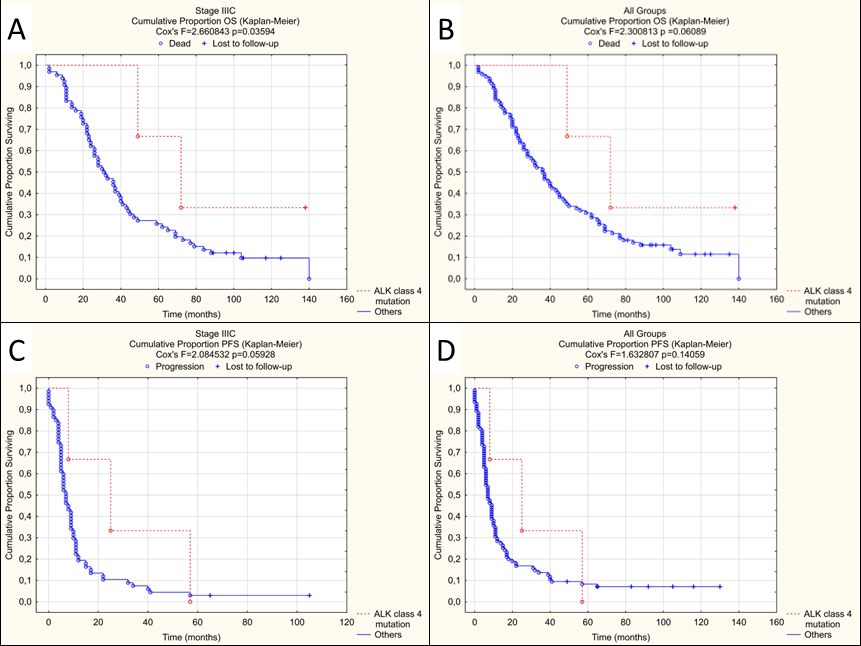
Our analysis showed that three out of 146 examined tumours (2%) harboured novel ALK mutations classified as potentially pathogenic. All these patients were diagnosed in stage IIIC, but their OS was significantly better (
The weak correlation between the positive IHC and increased copy numbers in ALK 5A4 with complete lack thereof in two other anti-ALK clones and negative staining in all 3 clones in cases with FISH confirmed amplification indicates that overexpression of ALK and ALK amplification are independent events, neither of which had a strong impact on patients’ survival. This also decreases the value of IHC as a screening method for testing molecular aberrations. The weak correlation between positive IHC and the lack of progression as well as 5-year overall survival is worth mentioning, however, as it contradicts the previously mentioned data on the negative impact of ALK overexpression in ovarian carcinoma. It is further corroborated by the fact that in the univariate analysis, despite not showing statistical significance, all of the factors potentially resulting in the increased ALK activity (ALK copy gain, overexpression visible as IHC positivity, class III/IV point mutations) were associated with a lower risk of dying (RR
In this study, we showed the presence of fusions involving other genes encoding protein kinases as demonstrated in three tumours having fusions of genes other than ALK (Table 4). In a 65-year-old female patient with stage IIIC HGSOC, who died 10 months after diagnosis, ERBB2-GRB7 fusion resulting from the deletion of the segment of 17q chromosome was discovered. Such an aberration has not been described so far in ovarian cancer. In colorectal cancer, it is associated with a lack of response to treatment with HER2 inhibitors, and a negative ERBB2 immunohistochemistry results even in cases with accompanying ERBB2 amplification in FISH [22]. It has also been reported in lobular breast carcinoma [23] and urothelial carcinoma [24].
Another fusion, PRKCA-BRCA1, occurred in a 54-year-old patient diagnosed with stage IV HGSOC with progression 16 months, and death 32 months after initial diagnosis. Such rearrangement has not been described in the literature so far. Both partner genes are known to be found in other fusions, including NF1, FAM134C, BECN1 and LSM12 for BRCA1 [25] and TANC2, SLC44A1, CDH8 and KIRREL for PRKCA [26], where their potentially pathogenic nature was indicated. Aberrant BRCA1 expression may affect the response to PARP inhibitors [27], which may indicate a potential clinical implication of such discovery in ovarian cancer.
The inversion resulting in SND1-BRAF was detected in an advanced (stage IV) tumour of a 79-year-old woman who progressed quickly and died within 2 months of surgery. It was previously reported in pancreatic acinar carcinoma [28] and NSCLC [29]. The research indicated, inter alia, the potential benefit of BRAF inhibitors and MEK inhibitors treatment in this group of patients. Our results expand the group of neoplasms with SND1-BRAF fusion, underlying the significance of further studies on potential efficacy of targeted therapeutics in those patients.
To summarize, the results of this study point out the potential role of ALK point mutations as an important factor for better response to chemotherapy and overall survival in a significant subset of locally advanced ovarian carcinomas. It also provides information on the frequency of ALK overexpression in the European population and expands the scope of tumours harbouring potentially targetable genetic fusions.
Author contributions
Conception: Adam Gorczynski, Wojciech Biernat, Piotr Czapiewski, Jörg Gabert, Karsten Neumann.
Interpretation or analysis of data: Adam Gorczynski, Kevin Miszewski, Piotr Czapiewski, Wojciech Biernat, Yann Gager, Sonja Koch, Jane Potschke, Dimitar Ugrinovski, Agata Pospieszynska, Bartosz Szymanowski, Szczepan Cierniak, Karsten Neuman, Katarina Mirkov.
Preparation of the manuscript: Adam Gorczynski, Piotr Czapiewski, Irene Kruecken.
Revision for important intellectual content: Yann Gager, Renata Duchnowska, Jörg Gabert, Wojciech Biernat.
Supervision: Piotr Czapiewski, Wojciech Biernat, Jörg Gabert, Renata Duchnowska, Dariusz Wydra.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-230117.
sj-xlsx-1-cbm-10.3233_CBM-230117.xlsx - Supplemental material
Supplemental material, sj-xlsx-1-cbm-10.3233_CBM-230117.xlsx
sj-xlsx-2-cbm-10.3233_CBM-230117.xlsx - Supplemental material
Supplemental material, sj-xlsx-2-cbm-10.3233_CBM-230117.xlsx
Footnotes
Acknowledgments
This project was financed from the PRELUDIUM 13 grant 2017/25/N/NZ4/00558 funded by National Science Centre, Poland.
The authors would like to thank dr. Magdalena Ratajska, dr. Victoria Tomkowicz, dr. Karolina Mollin, dr. Rafal Peksa, and dr. Michal Kunc for their invaluable input in designing and conducting the study.
