Abstract
BACKGROUND:
Numerous evidence have suggested the vital role of lncRNAs in human tumorigenesis. And lncRNA APAP1-AS1 has been proved to act as an oncogene.
OBJECTIVE:
Nevertheless, the molecular process underlying ARAP1-AS1 for the lymphoma progression has not been well studied.
METHODS:
RT-qPCR was used to ascertain the miR-6867-5p and ARAP1-AS1 in lymphoma cells and tissues. The localization of ARAP1-AS1 was determined via subcellular fractionation analysis. A xenograft model was used to investigate the influence of ARAP1-AS1 in formation of tumor in vivo. In addition, interactions between ARAP-AS1 and miR-6867-5p were tested by bioinformatics analysis, RIP assay, luciferase reporter and Pearson’s correlation analysis. Combined with loss-of-function experiments, MTT assays and flow cytometry were performed to evaluate the function of miR-6867-5p and also ARAP-AS1 in proliferation and apoptosis of lymphoma cells, respectively.
RESULTS:
ARAP1-AS1 was remarkably upregulated in lymphoma cells and tissues, while miR-6867-5p expression was downregulated. Furthermore, high ARAP1-AS1 expression suppressed miR-6867-5p expression in lymphoma cell lines (Raji and CA46), and Pearson’s analysis showed negative correlation between ARAP1-AS1 expression and also miR-6867-5p expression. In addition, knockdown of ARAP1-AS1 resulted in weakened cell viability and uplifted apoptosis rate of lymphoma cells (Raji and CA46) as well as a delay in the tumor growth in vivo. Further investigations illustrated that miR-6867-5p inhibitor reversed all above biological activities.
CONCLUSIONS:
LncRNA ARAP1-AS1 served as a tumor-promoter in lymphoma cells by sponging with miR-6867-5p, which may help to provide potential therapeutic target gene for lymphoma patients.
Introduction
Lymphoma is a cancer that affects human lymph nodes, which are small bean-shaped bumps under the skin. In 2008, lymphomas were classified as Hodgkin (HL), 10%, or non-Hodgkin (NHL), 90% by World Health Organization (WHO) [1]. Statistically, the estimated new cases of lymphoma are 90,390, which including 21,680 estimated deaths in the USA in 2021 [2]. Especially, in adolescents and young adults, lymphomas represent approximately one quarter of all cancers in this age group [3]. Although Lymphoma can be effectively treated in recent years, there are unwanted side effects from medicines and radiation [4]. Since lack the early clinical symptoms, lymphoma is usually diagnosed during the advance stages. Hence, useful bio-indicators in diagnose of lymphoma are needed.
lncRNA i.e., Long and non-coding RNA is defined as a part of noncoding RNAs, who are responsible for the transcription of more than 200 nucleotides in length [5]. Previous research has established that the lncRNA played a vital role in the human tumorigenesis and for development via serving as tumor oncogenes or inhibitors [6, 7], suggesting that lncRNA might function as a biomarker or potential therapeutic target in human cancers. In lymphoma, lncRNA are proved to regulate the development of lymphomas [8, 9, 10]. For instance, lncRNA, SNHG14, was stated to accelerate the cell proliferation, invasion and epithelial-to-mesenchymal in the lymphoma development [11]. Moreover, lncRNA SMAD5-AS1 was shown that downregulation of SMAD5-AS1 enhanced lymphoma cell proliferation in vitro and in vivo [12]. Long noncoding RNA ARAP1 cluster antisense RNA1 (lncRNA ARAP1-AS1), as a novel lncRNA, was published a crucial oncogene in the development of various human cancers, like colorectal cancer [13], lung cancer [14], cervical cancer [15, 16], bladder cancer [17]. Furthermore, the molecular mechanism by which ARAP1-AS1 contributes to Lymphoma progression is yet unclear.
MicroRNAs (miRNAs), a 19–25 nt single-stranded small noncoding RNAs, have been reported as a significant regulator in mediating the post-transcriptional of messenger RNA (mRNA) to regulate gene expression [18]. As a matter of fact, recent investigators have examined that lncRNA could work as sponges through inhibiting microRNAs (miRNAs) expression to facilitate proliferation and metastasis of human tumors [19, 20]. Notably, the function of miRNA regulation in diagnose and therapeutics for lymphoma has been widely disclosed (reviewed by [21, 22]). For instance, Wang et al. studied that miRNA-584 inhibited the development of lymphoma via regulating FOXO
The aim of this paper is to study and explore the regulation between ARAP1-AS1 and miR-6867-5p for the development and survival of lymphoma. Our research will provide a new perspective for lymphoma and propose a target gene which will help in the diagnosis and treatment of lymphoma.
Materials and methods
Patient samples
The specimens with tumors and specimens with their adjacent non-tumor which is 20
Related sequences for primers were provided
Related sequences for primers were provided
Normal human lymphocyte cells and lymphoma human cell lines (Raji, CA46, Ramos) taken from American Type Culture Collection (ATCC) from Maryland (USA). All cell lines were recultured in PRMI 1640 medium i.e., supplemented with 10% (v/v) of fetal bovine serum (FBS; PAN-Biotech, Aidenbach, Germany) and 100 U/ml streptomycin/penicillin (Invitrogen, Carlsbad, USA) in humidified atmosphere containing 5% of CO
Cell transfection
GenePharma Corporation (China) developed ARAP1-AS1-targeting siRNA (si-lnc) as well as a negative control siRNA (si-NC). The lentiviral vector of sh-ARAP1-AS1 (sh-lnc) and negative control (sh-NC) were purchased from TsingKe (Wuhan, China). MiR-6867-5p mimic and mimic control (miR-NC), miR-6867-5p as inhibitors and its corresponding negative control (inhibitor-NC) were also obtained by GenePharma Corporation (Shanghai, China). Cell transfection was carried out using Lipofectamine 2000 according to the manufacturer’s recommendations, with cell convergence exceeding 70%. After 48 hours, the rate of the transfection was observed by RT-qPCR.
Xenograft tumor formation
For in vivo experiments, BALB/c nu/nu thymus-deficient mice (nude) were obtained from Guangdong medical laboratory of animal center, China. All mice were allowed to grow in SPF animal experiment centers. After the end of adaptive feeding, the mice (nude) were categorized in two groups, first group was injected with Raji cells that were transfected with sh-ARAP1-AS1 and second group with sh-NC, respectively. The density of cells was 1
Reverse transcription-quantitative PCR (RT-qPCR)
To extract RNA from all cells and tissues, Trizol reagent (Invitrogen) was utilized. The isolated RNAs were then transcribed into cDNA using a Reverse Transcription Kit (Vazyme, China). Gene expression was measured using the SYBR Green PCR Kit (Japan). The ABI 7500 Fast Real-Time PCR instrument was used in this experiment (Applied Biosystems, USA). Expression of lncRNA/mRNA was normalized upto GAPDH, while miRNA expression levels were normalized upto U6. On basis of the sample threshold cycle (Ct) values of trplicates replications, data were analysed. Tsingke (Wuhan China) developed all of the primers for the qRT-PCR experiment. All sequences of primers were provided in Table 1.
MTT Reduction assay/3-(4,5-Dimethylthiazol- 2-yl)-2,5-diphenyltetrazolium bromide Reduction assay
In elisa plates of 96-wells 3
RNA immunoprecipitation (RIP) assay
MagnaRIP, RNA-Binding, Protein Immunoprecipitation Kit (Bedford, USA) was used to perform the RIP experiment [11]. RIPA cell lysis buffer was used to incubate and extract Raji and CA46 cells. The lysates are then processed in RIP buffer with magnetic beads coated with AGO2 antibodies or IgG as a control. The protein was digested with Proteinase K, and the immunoprecipitated RNA was extracted. Then RT-qPCR was performed to analyze ARAP1-AS1 and miR-6867-5p expression levels.
ARAP1-AS1 is upregulated in lymphoma cells and tissues, while APAP1-AS1 knockdown inhibits lymphoma cell proliferation and accelerates cell apoptosis. (A) ARAP1-AS1 expression levels in normal lymphocyte cells and lymphoma cell lines were measured using RT-qPCR. 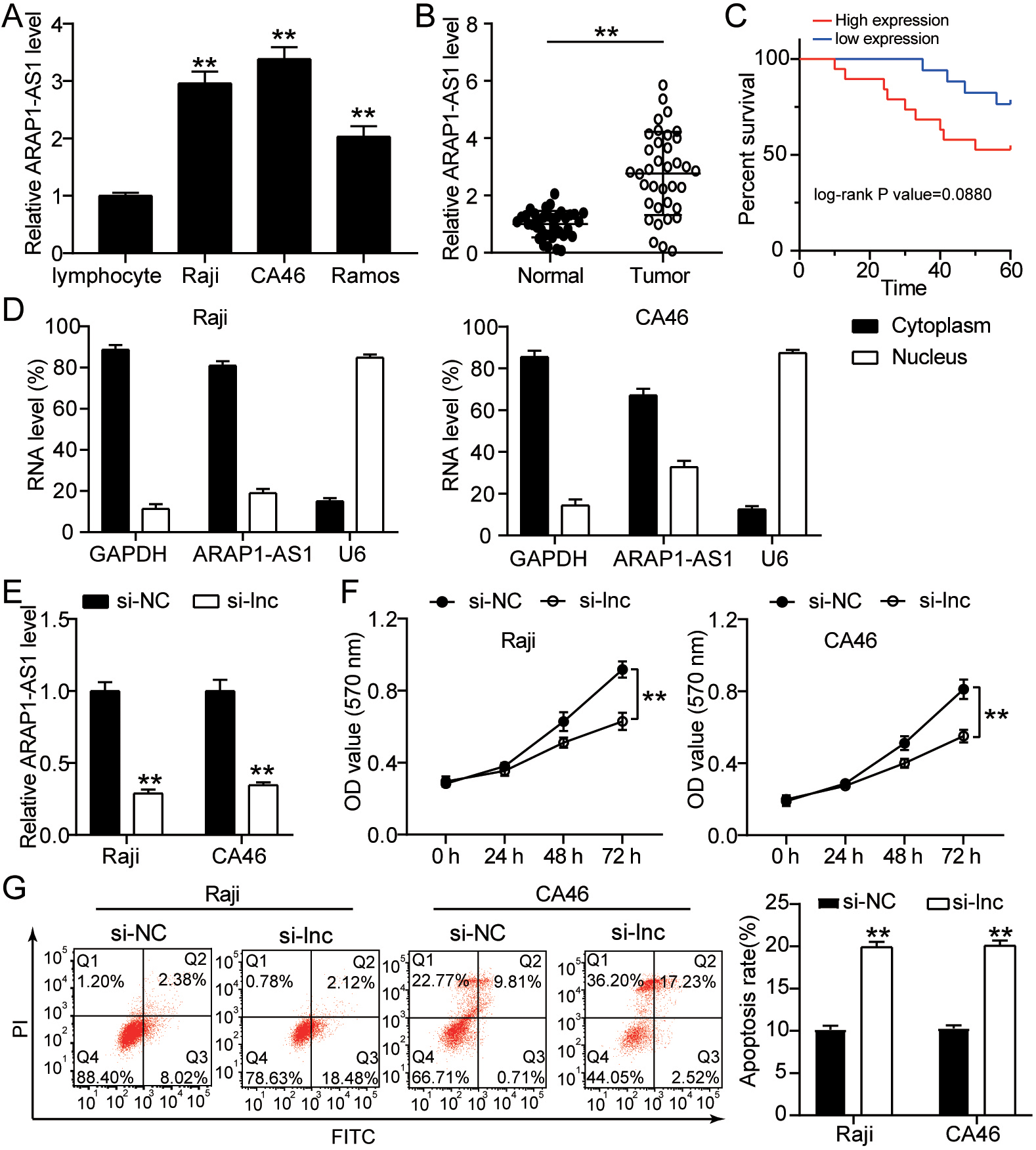
ARAP1-AS1 (full length) and its mutant (lacking the miR-6867-5p binding sites) were produced, and they were then ligated into pGL3 vectors (Promega, USA). ARAP1-AS1-WT and ARAP1-AS1-Mut are the two variants. Using Lipofectamine 2000 (USA: Invitrogen), these recombinant plasmids were transfected with miR-6867-5p and miR-NC into Raji cells or the CA46 cell line. Dual-Luciferase Reporter Assay System (Promega, USA) was used to determine the luciferase activities after 48 hours transfection [11].
Flow Cytometry (FC)
Knockdown of ARAP1-AS1 inhibits the lymphoma cell growth in vivo. (A) Raji cells transfected with sh-ARAP1-AS1 or sh-NC developed a tumor, which was detected. (B-C) Tumor volume and weight were measured in response to ARAP1-AS1 knockdown. 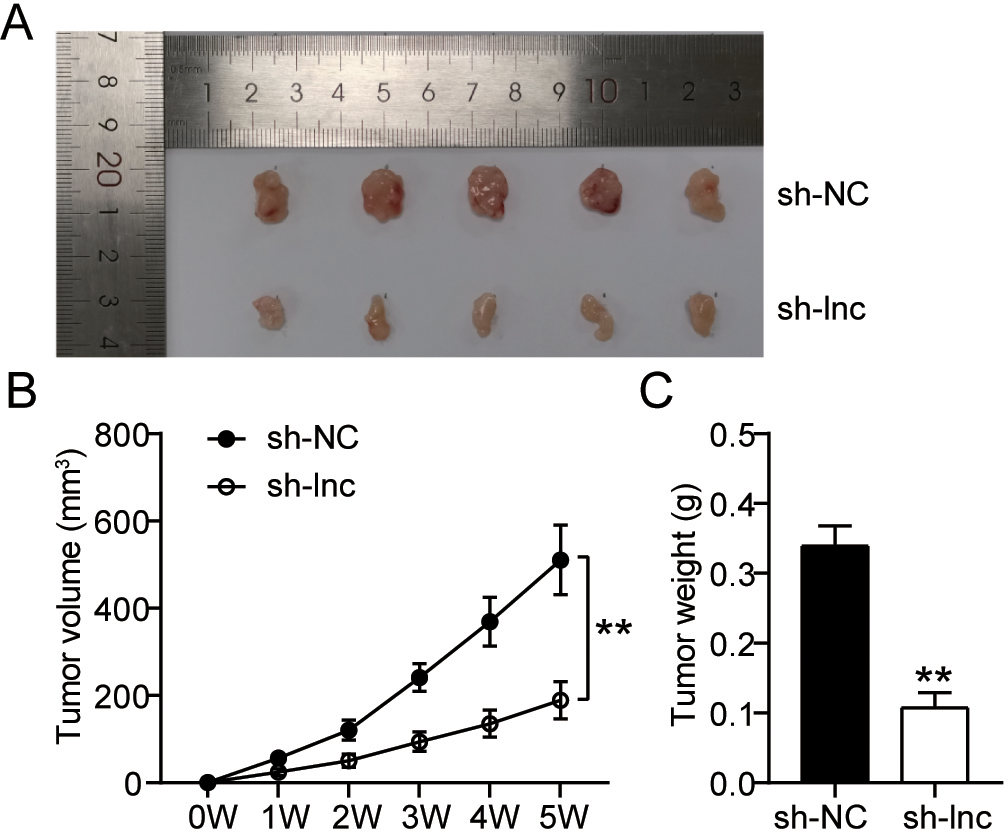
The apoptosis was determined using the Annexin V-FITC Apoptosis Detection Kit (Beyotime, China). Manufacturers’ instructions were followed as the previous study [28]. In brief, Raji and CA46 cells lines were collected 48 h after transfection and trypsin was used to remove cells, then centrifugation was done and washing two times with cold PBS. Cells were then suspended in binding buffer at a concentration of 1
MiR-6867-5p is a target gene of ARAP1-AS1. (A) The binding sequence between the ARAP1-AS1and miR-6867-5p was found and illustrated by miRDB. (B) The combination of ARAP1-AS1 and miR-6867-5p was confirmed using a luciferase reporter experiment. 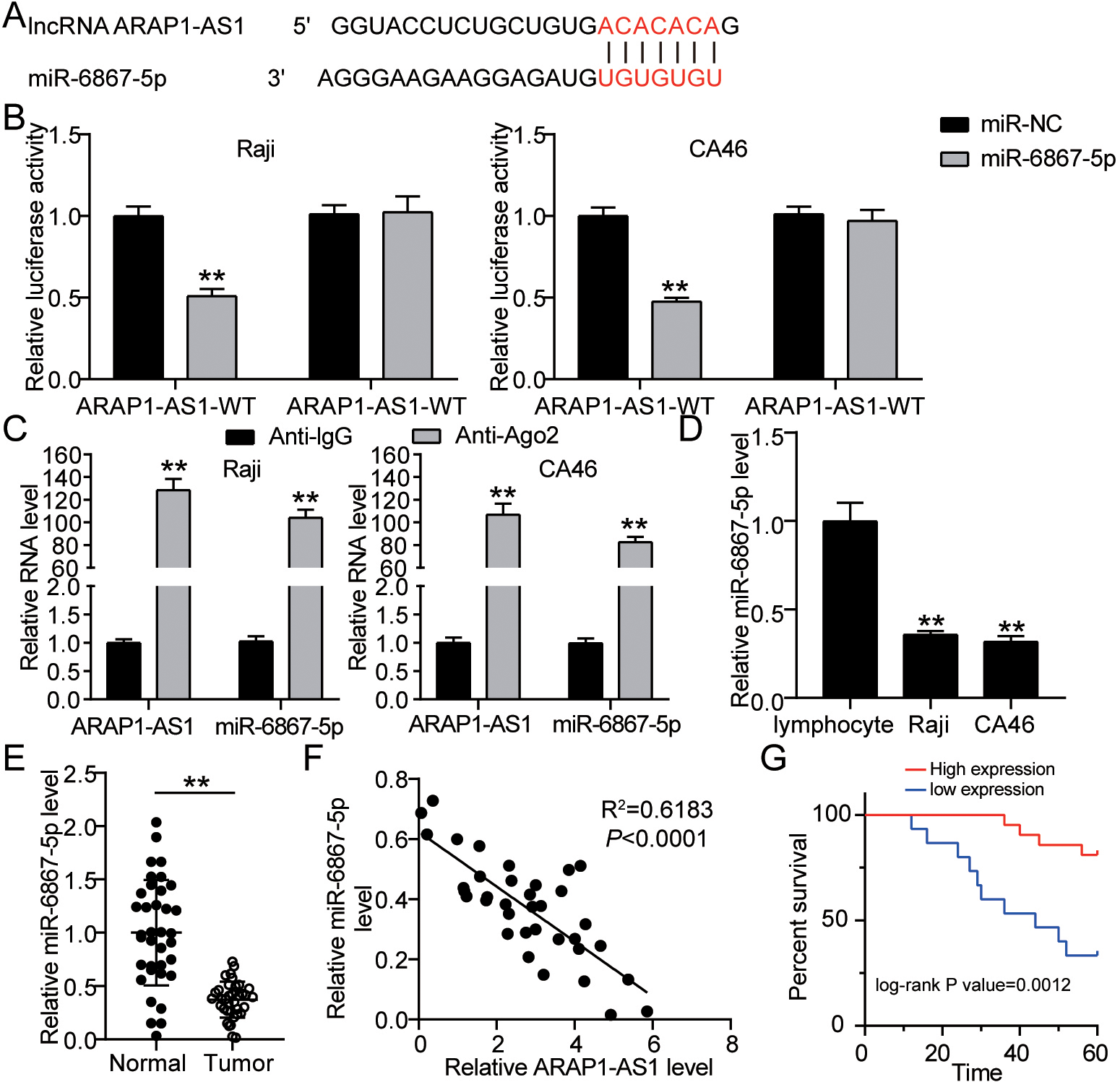
ARAP1-AS1 accelerates the proliferation and impairs apoptosis by regulating miR-6867-5p. (A) The expression level of miR-6867-5p in Raji cells, after transfected with si-NC, inhibitor-NC, si-lnc, inhibitor and si-lnc+inhibitor. (B) Cell proliferation ability of indicated Raji and CA46 cells was examined by applying rescue assays. (C) Cell apoptosis rates of Raji and CA46 cells were tested by performing rescue assays. Three different trials were used to get the data, which represents the mean 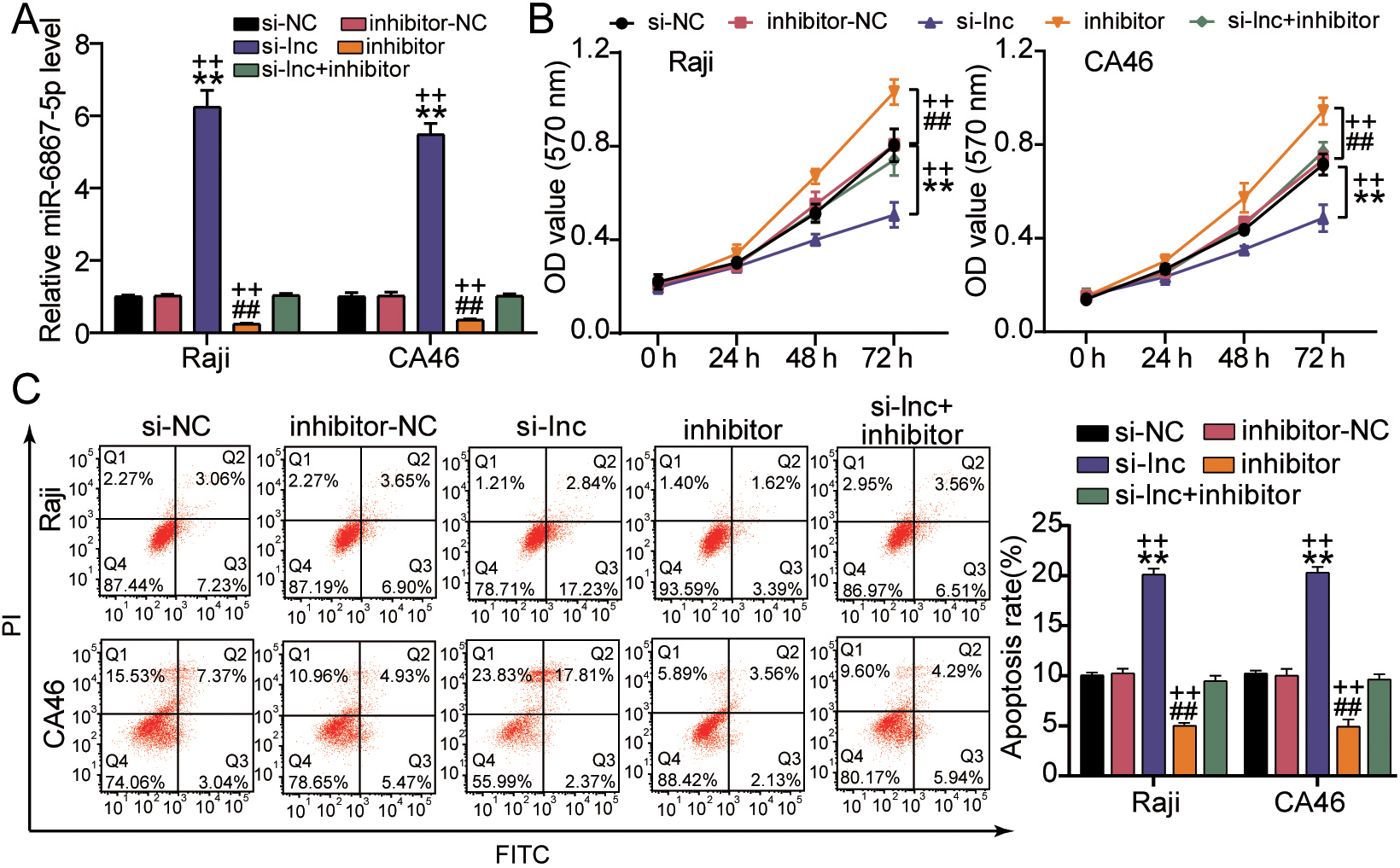
According to previous study [29], we use NE-PER cytoplasmic and nuclear extraction reagents (Thermo Fisher, USA), for the removal of the cytoplasmic and nuclear fractions of Raji and CA46 cell lines. RT-qPCR was used to evaluate the RNAs. The cytoplasmic and nuclear controls were GAPDH and U6, respectively.
Statistical analysis
Quantitative data was presented as a mean
Results
ARAP1-AS1 is upregulated in lymphoma cells and tissues, while ARAP1-AS1 knockdown inhibits lymphoma cell proliferation and accelerates cell apoptosis
RT-qPCR was used to evaluate ARAP1-AS1 expression in several lymphoma cell lines. This finding showed that in comparison with normal lymphocyte cell in these three lymphoma cell lines (Raji, CA46 and Ramos), ARAP1-AS1 was overexpressed (Fig. 1A). According to an RT-qPCR analysis of its expression pattern in 36 pairs of lymphoma and neighboring tissues, the expression level of ARAP1-AS1 in tumor tissues was substantially greater than in surrounding tissues (Fig. 1B). By survival analysis, ARAP1-AS1 with high expression showed the poor survival (Fig. 1C). Furthermore, a sub-cellular fractionation investigation verified the higher concentration of ARAP1-AS1 in Raji and CA46 cells’ cytoplasm (Fig. 1D). According to the results, ARAP1-AS1 may operate as a competitive endogenous RNA (ceRNA) in the development of lymphoma. To explore the further role of ARAP1-AS1 in lymphoma cells, we performed loss of function assays in the Raji and CA46 cells. The results of RT-qPCR demonstrated that ARAP1-AS1 expression was knocked down after transfection with si-ARAP1-AS1 plasmid (Fig. 1E). As a consequence, MTT assay showed that low expression of ARAP1-AS1 assisted to the obviously reduced of Raji and CA46 cell viability (Fig. 1F). Furthermore, knocking down ARAP1-AS1 meaningly improved the apoptosis rate of Raji and CA46 cells (Fig. 1G). ARAP1-AS1 jointly had a critical function in lymphoma cell proliferation and survival.
Knockdown of ARAP1-AS1 inhibits the lymphoma cell growth in vivo
To corroborate the participation of ARAP1-AS1 in development of lymphoma cells in vivo, xenograft tumor formation experiments were carried out. According to the findings, Raji cells transfected with sh-ARAP1-AS1 developed tumors that were much shorter than Raji cells transfected with sh-NC (Fig. 2A). In proportion, both volume and weight of tumors derived from Raji cells with low expression of ARAP1-AS1 were getting smaller with time than negative control (Fig. 2B–C). These results illuminated that ARAP1-AS1 was positively correlated with lymphoma progression.
MiR-6867-5p is a target gene of ARAP1-AS1
To corroborate the further molecular mechanism of the ARAP1-AS1 in lymphoma, the bioinformatics analysis was executed to seek potential target gene of ARAP1-AS1. As a result, we found that ARAP1-AS1 confined a promising binding site for miR-6867-5p (Fig. 3A). Furthermore, luciferase reporter experiments exposed that only miR-6867-5p mimics, rather than miR-NC, were able to inhibit the luciferase activity of the wide-type ARAP1-AS1 sequence in Raji and CA46 cells, while having no effect on the mutant ARAP1-AS1 sequence in both cell lines (Fig. 3B). RIP experiments consistently revealed that Ago2 antibody increased ARAP1-AS1 and miR-6867-5p in lymphoma cells when compared to IgG, showing a link between ARAP1-AS1 and miR-6867-5p in lymphoma cells (Fig. 3C). Next, miR-6867-5p expression at mRNA level was examined in lymphoma cells and normal lymphocyte cell by RT-qPCR. The results indicated that compared with normal lymphocyte cell, miR-6867-5p was generally declined in lymphoma cells (Fig. 3D). In cancer tissues, miR-6867-5p mRNA expression was suppressed, as expected (Fig. 3E). In lymphoma cells, ARAP1-AS1 and miR-6867-5p have a negative association, according to Pearson’s correlation analysis (Fig. 3F). Moreover, survival analysis displayed that the lymphoma patients with miR-6867-5p low expression had less survival time (Fig. 3G). ARAP1-AS1 inhibited the expression of miR-6867-5p in lymphoma cells, according to all of these studies.
ARAP1-AS1 accelerates the proliferation and impairs apoptosis by regulating miR-6867-5p
We first validated the influence of ARAP1-AS1 on miR-6867-5p in lymphoma cells to further delineate the contribution of ARAP1-AS1 to lymphoma development was regulated by a miR-6867-5p-dependent mechanism. As expected, the mRNA expression of miR-6867-5p were promoted when transfected si- ARAP1-AS1 in both Raji and CA46 cells and the inhibitory impact of miR-6867-5p expression was alleviated after co-transfecting si- ARAP1-AS1 and miR-6867-5p inhibitor (Fig. 4A). Meanwhile, MMT assays suggested that the inhibitor miR-6867-5p reduced the repressive impact of si-ARAP1-AS1 on cell viability (Fig. 4B). Inhibition of miR-6867-5p also reduced the apoptosis rate that was promoted in ARAP1-AS1-depleted cells (Fig. 4C). In other words, through down-regulating miR-6867-5p, the lncRNA ARAP1-AS1 boosted lymphoma cell growth and reduced apoptosis.
Discussion
There is a mounting body of literature that recognizes the vital role of long-coding RNAs in human cancers by regulating genes expression at various levels, such as epigenetic, transcription and post-transcription [6, 30, 31]. In present, the lncRNA ARAP1-AS1, as a new oncogene, has been demonstrated in a number of malignant tumors. For instance, Li et al. [32] discovered that the lncRNA ARAP1-AS1 targeted the miR-4735-3p/PLAGL2 pathway to produce malignant phenotypes in ovarian cancer cells. ARAP1-AS1 detected cell migration and proliferation in breast cancer through the miR-2110/HDAC2/PLIN1 axis, according to Lu et al. [33]. Furthermore, Zhou et al. [34] discovered that ARAP1-AS1 aided cervical cancer growth by partly controlling POU2F2 expression through miR-149-3p.
ARAP1-AS1 was shown to be elevated in tumor tissues and lymphoma cell lines in this study, which was verified using the RT-qPCR technique. Corresponding with other studies [35, 36, 37], ARAP1-AS1 was also shown to be preferentially localized in the cytoplasm, suggesting that it might be a competitive endogenous RNA (ceRNA) in lymphoma cells. In addition, loss-of-function assays confirmed that ARAP1-AS1 played an important role in survival of the lymphoma cells, by showing that knockdown of ARAP1-AS1 reduced the Raji and CA46 cell viability, while increased the apoptosis of the Raji and CA46 cells. Consistently, the Raji with low expression of ARAP1-AS1 led to remarkable smaller tumor volume in nude mice than the negative control. In conclusion, the lncRNA ARAP1-AS1 might serve as an important biomarker of lymphoma cells.
To further investigate the molecular mechanisms of lncRNA ARAP1-AS1 in enhancing the development of lymphoma cells, we carried out bioinformatics analysis using miRDB database and established that miR-6867-5p had the putative binding site for ARAP1-AS1. MicroRNA (miRNA) is a kind of endogenous and small noncoding RNA, which can regulate cell proliferation, differentiation, apoptosis, cell cycle and tissue development [38]. In addition, prior studies that have noted the importance of microRNA therapeutics in cancer [39, 40]. MiR-6867-5p, as a novel miRNA, has been found that lncRNA MACC1-AS1 functions via sponging miR-6867-5p in human brain microvascular endothelial cells [26]. Moreover, miR-6867-5p was also reported to serve as a target miRNA of Quercetin (3, 3
In this study, we mainly focus on the effect of the interaction between ARAP1-AS1 and miR-6867-5p on lymphoma growth. Further work needs to be carried out to validate the more complicated mechanisms of ARAP1-AS1 in promoting the development and proliferation of human lymphoma, such as more experiments in vivo and clinical trials.
In a word, our results it was recommended that the lncRNA ARAP1-AS1 played a substantial role in enhancing the multiplication and development of the lymphoma cells through functioning as a miR-6867-5p sponge. This discovery will lead to a new gene target for human lymphoma diagnosis and therapy.
Funding
Funding information is not available.
Ethics approval
The present study was approved by the Ethics Committee of the Sixth Hospital of Wuhan, Affiliated Hospital of Jianghan University (Wuhan, China). The processing of clinical tissue samples is in strict compliance with the ethical standards of the Declaration of Helsinki. All patients signed written informed consent.
This animal experiment was conducted in accordance with the ARRIVE guidelines and was authorized by the Animal Care Committee at the Sixth Hospital of Wuhan, Affiliated Hospital of Jianghan University.
Consent to participate
All patients signed written informed consent.
Consent for publication
Consent for publication was obtained from the participants.
Availability of data and material
All data generated or analyzed during this study are included in this article.
Authors’ contributions
Conception: BP.
Interpretation or analysis of data: BP, YFG, and JW.
Preparation of the manuscript: SFQ.
Revision for important intellectual content: BP, YFG, and JW.
Supervision: BP and YFG.
All authors read and approved the manuscript.
Footnotes
Acknowledgments
None.
Conflict of interest
The authors declare that they have no conflict of interests.
