Abstract
BACKGROUND:
The aim of the study was to longitudinally investigate the serum levels of mesothelin, sestrin1, hyaluronan synthase 2 (HAS2), midkine, and high mobility group box 1 (HMGB1) before and after chemotherapy and at the time of relapse in malignant pleural mesothelioma (MPM) patients treated with chemotherapy and to compare the changes in biomarker levels with radiological treatment outcome.
METHODS:
A total of 64 MPM patients treated with chemotherapy were enrolled in the study and longitudinally followed for changes in biomarker levels in response to treatment. Biomarkers levels were measured in serum using a human ELISA kit. Relative and absolute changes in biomarker levels were compared with the best radiological overall response at each time point.
RESULTS:
Median survival was 20.0
CONCLUSION:
Biomarkers may be useful in the longitudinal monitoring of response to treatment in MPM. However, the results of our study should be validated in larger groups with sufficient case numbers from multicenter institutions.
Introduction
The incidence of malignant pleural mesothelioma (MPM) is not expected to decrease due to increasing environmental exposure to asbestos worldwide and occupational exposure in developing countries [1, 2, 3, 4]. In addition to the increased risk, MPM has an aggressive course and poor prognosis with a median survival time of approximately 12 months. It is usually diagnosed at an advanced stage and treated with chemotherapy [5]. At this point, the most important issues in the follow-up of patients treated with chemotherapy are the prediction of which patients will respond to chemotherapy, the correct determination of response to chemotherapy to continue treatment, and the early detection of relapse.
Response to chemotherapy in MPM is assessed according to the modified response evaluation criteria in solid tumors (mRECIST) criteria using thoracic computed tomography (CT), a morphologic imaging modality [6]. MPM typically causes diffuse pleural involvement and has a unique growth pattern. At the onset of the disease, tumor usually arises from several different foci simultaneously as small lesions on the visceral and parietal serosal surfaces. As the disease progresses, the lesions spread and fuse, surrounding the lung like a thick layer and invading adjacent tissues. The distinct morphology and nonspherical growth may cause some problems in measuring and evaluating the tumor response to chemotherapy in MPM. In addition to its growth pattern, concomitant tumor pathologies such as pleural fluid, atelectasis, and fibrosis cause difficulties in the radiologic evaluation of MPM. Proposals for the definition of response measures were presented by mRECIST 1.1 [6, 7]. However, these definitions depend on the observer. Difficulties remain regarding interobserver variability and reproducible assessment of response [8]. It is more appropriate to evaluate the response with a method that can represent the entire tumor. The most appropriate approach would be a volumetric assessment for all pleural and non-pleural lesions. One of the two problems in this regard is that no feasible method (automatic measurement) has yet to be developed for routine clinical practice. Second, ideal volumetric response criteria have not yet been developed [9].
In this context, further studies are needed to evaluate the tumor response in addition to the morphologic assessment of the MPM. Biomarkers may represent the entire lesion. We thought that by determining biomarker levels, we could evaluate the whole tumor, such as volumetry, and eliminate the disadvantages of morphologic measurements. In the study, we examined five protein biomarkers, mesothelin, midkine, sestrin1, and hyaluronan synthase 2 (HAS2) and, high mobility group box 1 (HMGB1), to test their relevance to chemotherapy response. Mesothelin is encoded by the MSLN gene. It is a glycosylphosphatidylinositol-anchored cell-surface protein that may function as a cell adhesion protein. Mesothelin, a well-known biomarker that has been extensively studied in the diagnosis and differential diagnosis of MPM. Midkine is encoded by the MDK gene and promotes cell growth, migration, and angiogenesis, especially during tumorigenesis. Sestrin1 is encoded by the SESN1 gene and mediates cell growth inhibition by p53, leading to inhibition of the Mammalian Target of Rapamycin protein. Hyaluronan synthase 2 (HAS2) is encoded by the HAS2 gene and a multipass transmembrane protein. These three markers, midkine, sestrin1, and HAS2, are associated with MPM and were the subject of our previous studies [10, 11, 12]. High mobility group box 1 (HMGB1) is encoded by the HMGB1 gene, regulates transcription and is involved in DNA organization. It plays a role in several cellular processes, including inflammation, cell differentiation, and tumor cell migration. HMGB1 also plays a role in the pathogenesis of mesothelioma [13]. This study aimed to determine biomarker levels before and after chemotherapy and the time of relapse in MPM patients treated with chemotherapy and to compare the change in biomarker levels at each time point.
Material and methods
Patients
The study was prospectively conducted in patients with MPM diagnosed, treated, and followed to end of life between December 1, 2014, and March 31, 2019, at the Chest Disease Department, a referral center for mesothelioma. Written informed consent was obtained from all patients. The Ethics Committee of Eskisehir Osmangazi University approved the study (No: 26/01/2017-11).
Inclusion criteria for the study were age
The patient’s demographic, clinical, laboratory, treatment, and median survival data were recorded in the mesothelioma database of Lung and Pleural Cancers Research and Clinical Centre of Eskisehir Osmangazi University. The TNM classification was used for staging [14]. Platinum-based pemetrexed was administered as chemotherapy in the study. Radiologic response to chemotherapy was determined by mRECIST 1.1 [6]. Computed tomography of the chest was performed before treatment, every two cycles of chemotherapy until the end of therapy or disease progression, and every three months during follow-up until relapse. Complete response is disappearance of all pleural and non-pleural disease. Partial response is a summed measurement decrease by at least 30% from the baseline scan summed measurement. Progressive disease is a summed measurement increase by at least 20% from the nadir of the summed measurements from all prior scans. Stable disease is a decrease in the summed measurement that does not qualify as partial response, or an increase in the summed measurement that does not qualify as progressive disease. The best overall radiological response was recorded for each patient. Overall survival time was calculated from diagnosis to death or the last visit in surviving patients.
Biomarker assays
Serum samples were collected from all patients before and after two cycles of chemotherapy, from continued chemotherapy after every two cycles of chemotherapy up to six cycles, and from patients with partial/com-plete response and stable disease at recurrence. Samples were stored at
Statistical analysis
SPSS (version 15.0) and MedCalc Statistical Software (version 19.1.16, Medcalc Software Ltd, Ostend, Belgium) were used to analyze the data. Patients were categorized according to their best overall radiologic response: objective response (partial and complete response), stable disease, or progressive disease.
The agreement of the data with the normal distribution was checked using graphs and the Kolmogorov-Smirnov test. Nonparametric tests were used in the analysis because the data were not normally distributed. The Wilcoxon signed-rank test was performed for each response group to determine whether the median differences and relative changes in biomarker levels at the corresponding time points were consistent. Continuous variables were expressed as median and 25th to 75th percentile. Kaplan-Meier curves and the log-rank test were used to compare survival differences between groups.
A panel was formed based on the positivity of at least one of the biomarkers relevant to the determination of objective response and recurrence. Sensitivity, specificity, accuracy, and 95%CI were calculated.
Results
This study analyzed 64 MPM patients who received chemotherapy, and in whom changes in biomarker levels were longitudinally followed to treatment response. The descriptive characteristics of the study group are shown in Table 1.
Characteristics of the study group
Characteristics of the study group
SD: Standard deviation.
Baseline characteristics such as age, sex, histologic subtype, stage, and KPS of patients in relation to radiologic response were assessed and compared. The mean age of patients in the progressive disease, stable disease, and objective response groups was 61.1
Kaplan-Meier survival curves of the study group according to radiological response to chemotherapy.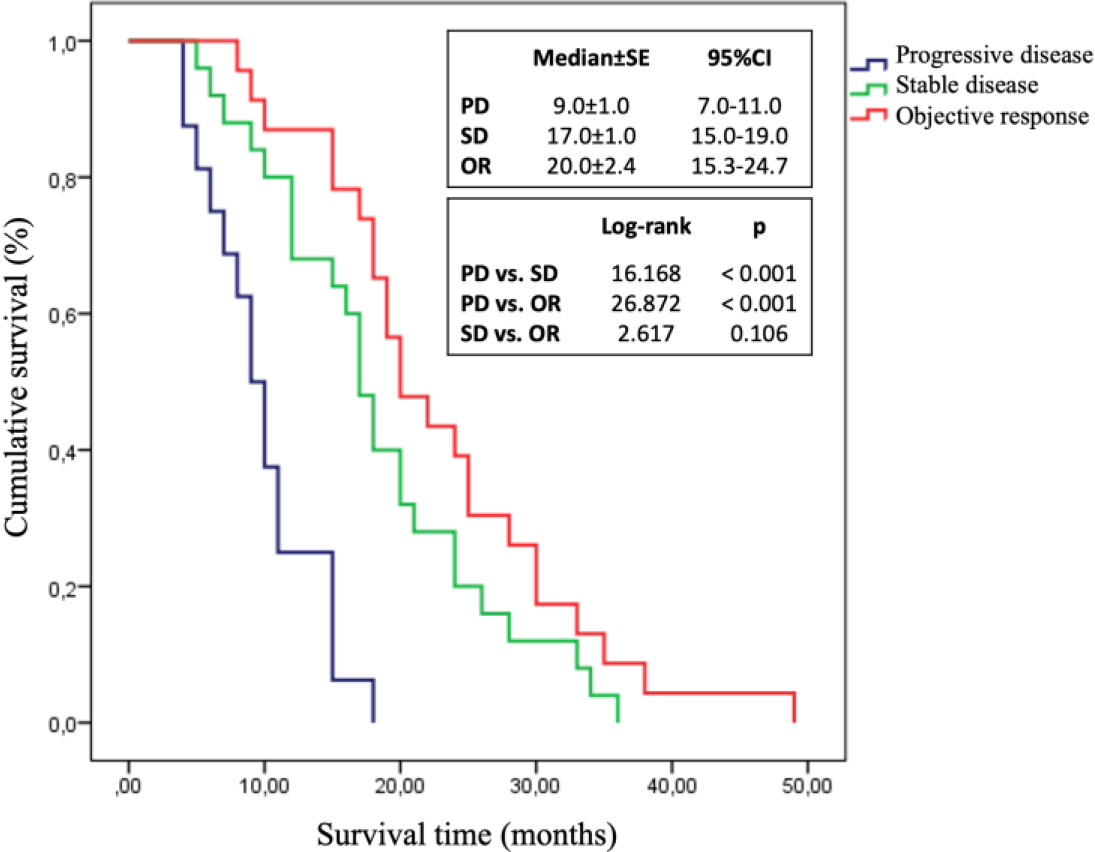
The survival times of the patients were compared according to radiologic response to chemotherapy. The median survival time
Biomarker levels as a function of response to chemotherapy in serum samples collected from patients before and during response measurement are shown in Table 2, and biomarker level courses are shown in Fig. 2.
Serum biomarker levels of the study group in relation to radiological response
HMGB1: high mobility group box 1; HAS2: hyaluronan synthase 2.
Biomarker levels in patients with MPM were monitored longitudinally at each time point. a) mesothelin; b) midkine; c) HMGB1; d) sestrin1. 1: before treatment; 2: during response measurement; 3: at recurrence.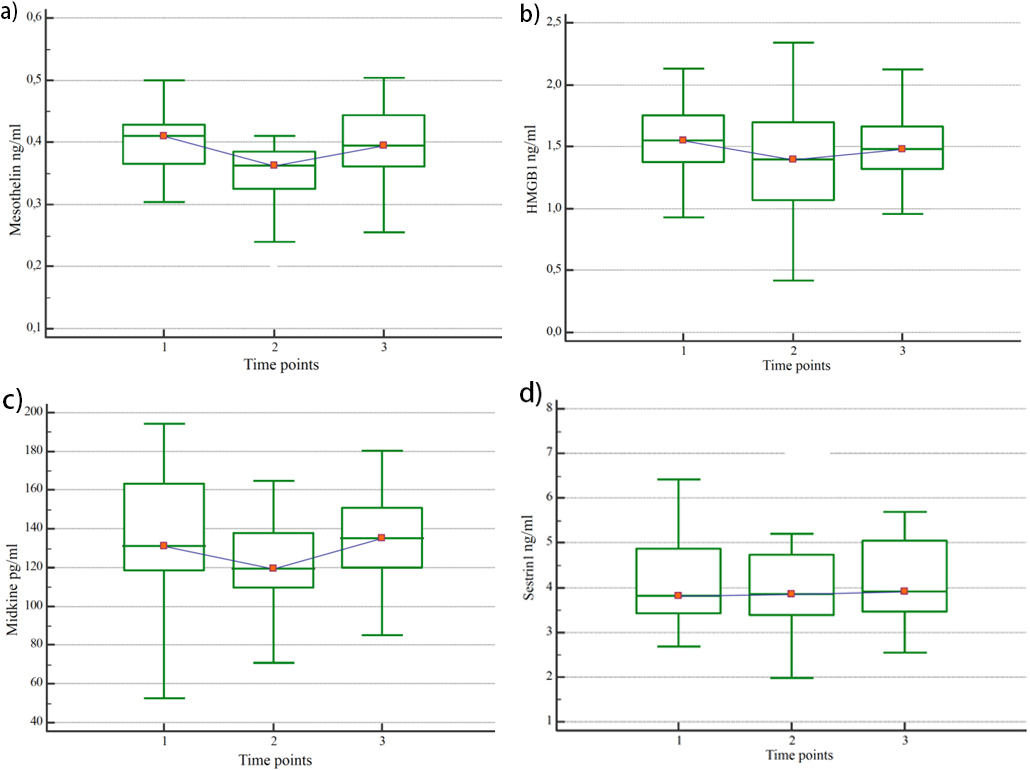
A significant decrease in serum levels of mesothelin, midkine, and HMGB1 was observed in patients with radiologically objective responses to chemotherapy. In these patients, mesothelin levels decreased by 15%, midkine levels by 7%, and HMGB1 levels by 15%. In addition, HMGB1 serum levels were found to increase significantly by 15% in patients with radiologically progressive responses to chemotherapy compared with pretreatment serum levels (Table 2).
The sensitivity, specificity, and accuracy (95%CI) of a panel of biomarkers consisting of a significant decrease in mesothelin, midkine, and HMGB1 to determine objective response were 73.9% (95%CI: 51.6–89.8), 61.0% (95%CI: 44.5–75.8), and 65.6% (95%CI: 52.7–77.1), respectively.
Biomarker levels in serum samples at the end of treatment and disease recurrence in patients with objective response and stable disease to chemotherapy are shown in Table 3, and the courses of biomarker levels are shown in Fig. 2.
Biomarker levels at the end of treatment and disease recurrence in patients with objective response and stable disease to chemotherapy
HMGB1: high mobility group box 1; HAS2: hyaluronan synthase 2.
In patients with objective response to chemotherapy, mesothelin levels increased by 15%, midkine by 12%, and sestrin1 by 8% when the disease recurred (Table 3).
The sensitivity, specificity, and accuracy (95%CI) of a panel of biomarkers consisting of a significant increase in mesothelin, midkine, and sestrin1 to determine recurrence were 81.3% (95%CI: 54.4–96.0), 61.5% (95%CI: 31.6–86.1), and 72.4% (95%CI: 52.8–87.3), respectively.
In the treatment of MPM, accurate and timely determination of treatment response in patients receiving chemotherapy and disease recurrence in patients after treatment may allow timely reassessment of treatment, thereby prolonging patient survival and reducing costs. Biomarkers appear promising to overcome the problems posed by the growth characteristics of MPM in morphologic assessment of response to chemotherapy by CT [15, 16, 17]. It is conceivable that the total tumor burden could be assessed by biomarkers, which would eliminate the disadvantages of morphologic measurements. Several biomarkers have been proposed, but no biomarker is routinely used to monitor patients during and after chemotherapy for MPM [17].
In this study, we examined five biomarkers compared to the radiological response in determining the chemotherapy response for MPM patients. Some of these biomarkers are useful in our previous studies, some of which have been presented in the current literature as potentially useful in MPM patients mentioned below in detail. We measured biomarker levels in MPM patients treated with chemotherapy before, after best response, and at tumor recurrence by radiological assessment. We compared the change in biomarker levels at each time point. We found that mesothelin, midkine, and HMGB1 were effective in treatment monitoring and mesothelin, midkine, and sestrin1 effectively detect recurrence.
Mesothelin is a well-known biomarker that has been extensively studied in the diagnosis and differential diagnosis of MPM [17, 18]. Mesothelin has a high specificity of 88% to 100% and a moderate sensitivity of 19% to 68% in the differential diagnosis of MPM [19]. The moderate sensitivity may be due to the early stage of disease, sarcomatoid subtype, and some other factors such as age, chronic renal failure, or obesity, and limits the use of mesothelin in the diagnosis and differential diagnosis of mesothelioma [18, 20]. Over time, studies of mesothelin in mesothelioma have focused on determining its importance in prognosis. Fontana et al. reported that higher soluble mesothelin-related protein (SMRP) plasma concentrations in MPM were associated with poor prognosis after adjustment for prognostic factors and pointed to SMRP as a potential biomarker to improve the prognostic ability of the European Organisation for Research and Treatment of Cancer (EORTC) index [21]. However, the prognostic value of serum mesothelin is currently controversial and remains unclear.
In addition, some studies investigated the relationship between the change in serum level of mesothelin and tumor response, which is also the subject of the present study. A prospective study of 41 MPM patients showed that a 10% increase in serum mesothelin level predicted radiological progression with 96% sensitivity. In addition, increasing mesothelin levels at six months were associated with a significantly worse objective response to chemotherapy compared with stable or decreasing levels [22]. Serial measurements of mesothelin were performed in 40 patients with MPM. Patients who responded to treatment or were stable were observed to have mesothelin levels fall below baseline after two months and return to baseline six months later. Authors suggested that stable or decreased mesothelin levels were ultimately related to treatment response [23]. In patients with mesothelioma receiving chemotherapy, it was found that differences in serum mesothelin levels before treatment and in various responses to treatment may be useful biomarker for assessing response to treatment and determining disease progression [24]. Similar results have been reported in other related studies [25]. A systematic review showed serum mesothelin levels correlated with radiologic progression and survival [26]. On the other hand, Linch et al. found no correlation between pre-chemotherapy mesothelin levels and response in 18 MPM patients treated with chemotherapy [27].
In addition, studies were performed in which serum mesothelin levels were determined along with other biomarkers and compared with the radiologic response. In a study of 62 MPM patients, a 15% change in mesothelin, megakaryocyte-potentiating factor (MPF), and osteopontin levels at the time point corresponding to overall radiologic response was found to be associated with response to chemotherapy, and the relationship between mesothelin and MPF levels and disease progression was better than for osteopontin [28]. Katz et al. showed that using SMRP and fibulin-3 as serological biomarkers in the immunotherapy trial was not useful in following tumor response longitudinally [29]. Wheatley-Price et al. evaluated mesothelin and osteopontin in 21 patients with MPM and found that a change in mesothelin of greater than 10% was associated with radiological response, and an increase in mesothelin was observed in all patients with radiological progression of disease [30].
Most studies in the literature report that mesothelin levels are assessed by the radiological response and are related to response to treatment, decreasing in patients who respond to treatment and increasing in those who do not respond or have a progressive course. However, no generally accepted criteria for the degree of decrease or increase in mesothelin levels have yet been developed. In our study, we found that the level of mesothelin significantly decreased by about 15% in the partial and complete response group to chemotherapy, which is consistent with other studies. Conversely, we found that mesothelin levels increased by approximately 15% in patients who had relapsed. We found an 8% increase in the progressive group, but this was not statistically significant. This may be due to our study’s relatively small number of patients. An important drawback of the studies is the limited number of patients. If the number of patients increases, it is likely that the change in biomarker levels will be more pronounced. Although the changes in mesothelin levels seem relatively small in our study and other studies, it can be said that the biomarkers may be useful for guiding treatment, considering that they represent the whole tumor, as claimed for volumetric measurements. On the other hand, the lack of change in biomarker levels during relapse in the group with stable disease suggests that this group is a slowly progressive group that does not respond to treatment.
Midkine is a newly studied biomarker in mesothelioma. Ostroff et al. examined more than 1000 serum proteins from mesothelioma patients and asbestos-exposed individuals using a proteomic assay. They identified midkine as one of 13 markers in a panel useful for differential diagnosis of mesothelioma [31]. In a study performed at our center, the median survival time of patients with high midkine expression in MPM tumor tissue was six months, whereas the median survival time of patients with low midkine expression was ten months. In a multivariate analysis, in addition to histopathology, stage, and performance score, midkine gene overexpression was found to be independently and significantly associated with survival [11]. In another study conducted at our center, the significance of serum midkine level in the differential diagnosis and prognosis of MPM was compared with serum mesothelin level. The study included 95 cases with MPM, 56 with cancer metastasized to the pleura, 27 with benign pleural disease, and 20 with benign asbestos pleurisy. The cut-off value was set at 1.5 nmol/L for mesothelin and 421 pg/mL for midkine. At the end of the study, midkine was shown to be ineffective in the differential diagnosis of MPM compared with other diseases, but it is associated with prognosis, and the prognosis is worse in cases with a serum midkine level above 421 pg/mL [10].
In our current study, midkine levels were significantly decreased by 7% in the partial and complete response groups. Conversely, midkine levels increased by 12% in patients who relapsed. Although these levels appear low, it should be considered that midkine may be useful for guiding treatment because it represents the entire tumor, as we hypothesized above.
Recently, attention has been drawn to the importance of HMGB1 in asbestos-induced carcinogenesis in mesothelioma. It was found that HMGB1 serum levels were higher in patients with MPM than in the healthy individual exposed to asbestos and that HMGB1 is a sensitive and specific biomarker to distinguish individuals with malignant mesothelioma [32, 33, 34]. In a study of 170 MPM patients, HMGB1 protein and gene expression levels were assessed by immunohistochemistry and reverse transcription-polymerase chain reaction in biopsy specimens. It was found that HMGB1 may be a useful prognostic biomarker in MPM when detected by immunohistochemistry. Conversely, it was concluded that HMGB1, which is also expressed in normal and reactive mesothelial cells, cannot be used as a diagnostic biomarker in MPM [35].
A study evaluating the response to chemotherapy with HMGB1 has not yet been performed. Our study found that HMGB1 serum levels decreased by approximately 15% in the partial or complete response group. Conversely, HMGB1 levels increased by 15% in patients with progressive disease. In patients who relapsed during follow-up after chemotherapy, a 5% increase in HMGB1 levels was observed, but this was not statistically significant. This may be due to the small number of patients.
In the only existing study of Sestrin1 in mesothelioma, the median survival of patients with elevated Sestrin1 gene expression in tumor tissue at diagnosis was longer than the others (11 months vs. 7 months) [11]. Subtype analyzes were also performed in this study, and the prognosis was better in patients with high Sestrin1 gene expression in epithelioid subtype. The results of the study suggest that Sestrin1 is a potent regulator of cell growth and proliferation. In the current study, we investigated the change in Sestrin1, the protein encoded by the Sestrin1 gene, during the treatment of patients with MPM based on previous findings. We found that serum levels of Sestrin1 did not change in the same direction as the radiological response. However, in tumor recurrence in patients who responded to chemotherapy, the 8% increase in Sestrin1 levels was statistically significant. Further studies of Sestrin1 are needed to evaluate the response to chemotherapy in mesothelioma.
In a study, serial measurements of hyaluronan were performed during treatment with methotrexate in 37 patients with mesothelioma, and a significant increase in serum hyaluronan levels was observed in patients with progressive disease. On the other hand, in three patients with high hyaluronan serum levels at the beginning of treatment, a significant decrease in hyaluronan levels was observed in parallel with the reduction in tumor mass, and hyaluronan levels were consistently low when the response persisted [36]. A previous study conducted in our clinic investigated the association between the HAS2 gene expression level and the survival of MPM patients receiving chemotherapy. Median survival was longer in patients with gene expression above the cut-off (11 months vs. 7 months) than in patients below the cut-off. The same relationship was found in patients with epithelioid subtype [11]. In our study, the association between HAS2 and response to chemotherapy or relapse was investigated based on the previous study mentioned above, and it was found that serial HAS2 measurements in serum showed no change in agreement with radiological assessments.
A panel consisting of mesothelin, midkine, and HMGB1 to determine the objective response showed high sensitivity and an acceptable level of specificity. That is, the above panel was able to predict objective response in three of the four patients. Similarly, the panel of mesothelin, midkine, and sestrin1 showed high sensitivity and an acceptable level of specificity in determining recurrence. In other words, this panel was able to predict recurrence in four of the five patients, and we can say that the use of biomarkers together may lead to better clinical outcomes [34].
In conclusion, determining the response to chemo-therapy in mesothelioma with more objective, rapid, and cost-effective methods is an important and emerging topic. The results of our study suggest that biomarkers detectable in serum will be useful in longitudinal monitoring of response to treatment in mesothelioma. However, in this regard, the markers identified so far should be evaluated in larger groups with sufficient case numbers and multicenter studies.
Footnotes
Acknowledgments
This study has been supported by Eskisehir Osmangazi University Scientific Research Projects Coordination Unit under grant number 201811036.
Author contributions
Conception: G.A., S.M, M.M.
Interpretation or analysis of data: G.A., S.M., F.B., A.A., M.M.
Preparation of the manuscript: G.A., S.M., F.B.
Revision for important intellectual content: M.M., S.M., G.A.
Supervision: S.M., G.A., M.M., A.A.
