Abstract
BACKGROUND:
Although exosomal microRNAs (exo-miRNAs) regulate angiogenesis, they are not sufficient for the development of anti-vascular drugs for tongue squamous cell carcinoma (TSCC). miR-205-5p is an exo-miRNA that is highly expressed in the saliva of patients with oral SCC.
OBJECTIVE:
We aimed to clarify the role and molecular mechanism of exosomal miR-205-5p in regulating TSCC angiogenesis.
METHODS:
Effect of exosomes derived from TSCC cells on human umbilical vein endothelial cell (HUVEC) function was determined using the CCK-8, Transwell, Transwell-Matrigel, and Matrigel-based tube formation assays. Protein levels were detected by western blot. The binding between miR-205-5p and the 3
RESULTS:
Exosomal miR-205-5p (exo-miR-205-5p) promoted the proliferation, migration, and invasion of HUVECs, increased the number of tubes formed by HUVECs, and increased the vascular endothelial growth factor receptor 2 levels in HUVECs. Exo-miR-205-5p downregulated the AMOT level in HUVECs. Results of the luciferase reporter assay showed that miR-205-5p could bind to the 3
CONCLUSION
: Exo-miR-205-5p derived from TSCC regulates the angiogenic activity of HUVECs by targeting AMOT and might be a new molecular target for the development of anti-vascular drugs for TSCC.
Introduction
Oral cancer, mainly tongue, lip, and gum cancers, is one of the most common malignant tumors of the head and neck [1]. Oral cancer is the sixth most common cancer worldwide [2]. Tongue cancer is a malignant tumor that develops in the tongue and ranks first among oral cancer types. Tongue squamous cell carcinoma (TSCC) is the most common type of tongue cancer [1]. The incidence of TSCC is increasing every year. TSCC features high rates of regional lymph node metastasis and distant metastasis rate. Its prognosis is relatively poor [3, 4]. Currently, there are no effective cancer markers for the clinical diagnosis and treatment of TSCC. Knowing key factors in the occurrence and development of TSCC and clarifying the important mechanisms would be valuable in discovering new markers for diagnosing TSCC and therapeutic targets.
Tumor metastasis results from the interaction of multiple factors, including tumor vessel growth, tumor microenvironment, and cell-cell fusion [5, 6, 7]. Angiogenesis is an important factor in tumor metastasis and has been intensively investigated. Tumor microvessel density is related to the growth and metastasis of tumors, suggesting that it could be an important indicator of clinical prognosis [8, 9]. Anti-angiogenesis therapy for tumor growth and metastasis has become a unique method for tumor treatment [9]. Ongoing experimental studies are investigating the therapeutic effects of angiogenesis inhibitors in different tumors. Additionally, anti-angiogenesis is a successful treatment for some types of solid tumors [10]. The clinical efficacy of existing angiogenesis inhibitors in treating patients with TSCC is not satisfactory, and further studies are needed to identify more effective anti-vascular targets [11].
Exosomes are lipid bilayer membrane vesicles with a diameter of approximately 30–150 nm, which can be secreted by almost all cells and are widely distributed in all body fluids [12]. Exosomes carry a variety of substances, such as proteins, nucleic acids, and lipids, which play crucial roles in the occurrence, development, and metastasis of tumors and are important markers in the early clinical diagnosis of cancer [12, 13]. Exosomal microRNAs (exo-miRNAs) may serve as biomarkers for the diagnosis and prognosis of various cancer types [14]. Exo-miRNAs play various roles in the cellular uptake of exosomes [14]. The role of exo-miRNAs in regulating angiogenesis has been previously reported [14]. Angiogenesis is a key component of tumor growth, invasion, metastasis, and recurrence. Therefore, exo-miRNAs that regulate angiogenesis in TSCC may be an effective therapeutic target for TSCC. Identification of exo-miRNAs will facilitate the development of anti-vascular drugs for TSCC.
miR-205-5p is an exo-miRNA that is highly expressed in the saliva of patients with oral SCC [15]. Exosomal miR-205-5p (exo-miR-205-5p) induces angiogenesis in other types of cancer, such as nasopharyngeal carcinoma and ovarian cancer [16, 17]. However, whether exo-miR-205-5p induces angiogenesis in TSCC is unknown. Hence, we aimed to clarify the role and molecular mechanism of exo-miR-205-5p in the regulation of TSCC angiogenesis.
Materials and methods
Cell culture and transfection
The TSCC cell line SCC-25 was cultured in a 1:1 mixture of Dulbecco’s modified Eagle’s medium (Hy-clone, Logan, UT, USA) and Ham’s F12 medium containing 1.2 g/L sodium bicarbonate, 2.5 mM L-glutamine, 15 mM HEPES, and 0.5 mM sodium pyruvate and supplemented with 400 ng/ml hydrocortisone and 10% fetal bovine serum (Hyclone). Another TSCC cell line, HSC-3, was cultured in Eagle’s minimal essential medium (Hyclone) with 10% fetal bovine serum (Hyclone). All cells were cultured at 37
The miR-negative control (NC), miR-205-5p, miR-NC inhibitor, and miR-205-5p inhibitor were synthesized by GenePharma (Suzhou, China). To construct the angiomotin (AMOT) overexpression vector, the coding sequence of
Extraction and identification of exosome derived from TSCC cells (exo-TSCC)
After transfection for 48 h, the supernatant was collected, and the exosomes were extracted using supercentrifugation at 4
Quantitative reverse transcription-polymerase chain reaction (PCR)
Total RNA was isolated using TRIzol reagent (Invitrogen), and genomic DNA was removed using RQ1 RNase-Free DNase (Promega, Madison, WI, USA) following the manufacturer’s instructions. To analyze miR-205-5p expression level, RNA was transcribed into cDNA using reverse transcription primers (CTCAACT GGTGTCGTGGAGTCGGCAATTCAGTTGAGCAGACTC) and Moloney murine leukemia virus reverse transcriptase (Promega). The PCR system was prepared using the SYBR Green qPCR SuperMix kit (Invitrogen). The PCR primer sequences used were 5
HUVEC culture with different groups of exo-TSCC
To investigate the effect of exo-miR-205-5p on HUVEC function, HSC-3, and SCC-25 cells were transfected with miR-NC, miR-205-5p, miR-NC inhibitor, and miR-205-5p inhibitor. The exosomes extracted from these TSCC cells (exo-miR-NC, exo-miR-205-5p, exo-miR-NC inhibitor, and exo-miR-205-5p inhibitor, respectively) were added to the HUVEC culture medium. To investigate whether AMOT overexpression blocks the effect of exo-miR-205-5p on HUVECs function, HUVECs were transfected with pcDNA-AMOT or empty vector pcDNA3.1, and incubated with the exo-miR-205-5p after transfection for 6 h (treated HUVECs were termed as exo-miR-205-5p
Measurement of cell proliferation rate CCK-8 assay
After culturing in conditioned medium with different groups of exo-TSCC for 0, 1, 2, 3, and 4 days, 10
Transwell assay of cell migration and cell invasion
After culturing in conditioned medium with diff-erent groups of exo-TSCC for 24 h, the HUVECs were harvested and resuspended in a culture medium without fetal bovine serum. One hundred microliters of the cell suspension was added to the inner surface of the upper Transwell insert, and 600
Matrigel-based tube formation assay
After culturing in conditioned medium with different groups of exo-TSCC for 24 h, the HUVECs were harvested and resuspended in the culture medium. Fifty microliters of the suspension containing 1
Predication for target genes of miR-205-5p related to angiogenesis
The target genes of miR-205-5p were obtained using TargetScanHuman 7.1 (
Luciferase reporter assay
Total RNA isolated from SCC-25 cells was transcribed to cDNA using oligdT as a reverse transcription primer using reverse transcriptase M-MLV (Promega). The cDNA was used as the template to amplify the fragment of the
Western blot analysis
Total protein was isolated from each group of HUVECs or exosomes. Proteins denatured by boiling were separated using sodium dodecyl-sulfate polyacrylamide gel electrophoresis. The proteins were transferred to polyvinylidene fluoride (PVDF) membranes. After blocking with 5% nonfat-dried milk, the membranes were incubated with primary antibodies against CD63 (ab134045, abcam, Cambridge, MA, USA), CD81 (ab155760), TSG101 (#72312, Cell Signaling Technology, Danvers, MA, USA), VEGFR2 (#9698, Cell Signaling Technology), AMOT (#43130, Cell Signaling Technology) or GAPDH (KC-5G5, KangChen Bio-tech, Shanghai, China). After washing with Tris-based immunoblot wash buffer (TBST) three times, the membrane was incubated with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (H+L) (4050-05, SouthernBiotech, Birmingham, AL, USA) and then washed with TBST three times. Finally, the HRP content was assessed using a western chemiluminescent HRP substrate (Millipore, Billerica, MA, USA), and the signal was exposed to X-rays. Western blot analysis was repeated three times.
Statistical analysis
Statistical analysis was performed using SPSS software (version 19.0; IBM, Chicago, IL, USA). The results were analyzed using one-way analysis of variance followed by a post-hoc least significant difference test. The results from three independent experiments were expressed as mean
Results
Identification of extracted exosomes. A: Exosome examined by transmission electron microscopy. The black scale bar indicates 100 nm. B: Diameter of exosomes measured by a flow nanoanalyzer. C: The levels of typical exosome markers CD63, CD81, and TSG101 in extracted exosomes detected by western blot.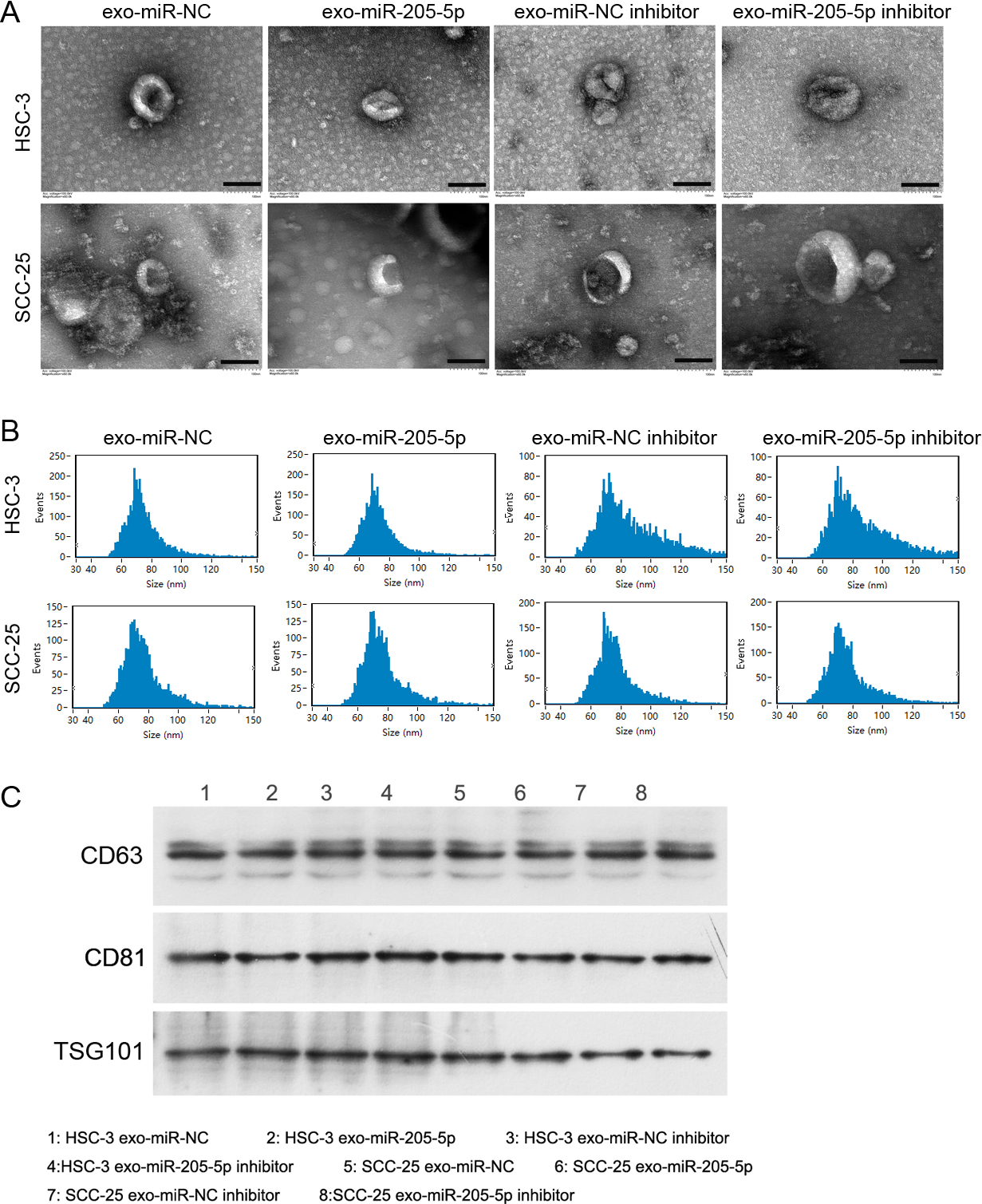
The quality of the exosomes is the key to the experiment; therefore, we initially identified the extracted exosomes. Transmission electron microscopy images showed that the extracted vesicles appeared exosome-like (Fig. 1A). They were 40–100 nm in diameter (Fig. 1B). Moreover, the expression levels of exosome-specific marker proteins CD63, CD81, and TSG101 were detected in these vesicles (Fig. 1C). These experiments indicated that the exosomes were successfully extracted.
Effect of exo-miR-205-5p extracted from HSC-3 and SCC-25 cells on HUVEC function. HSC-3 and SCC-25 cells were transfected with miR-NC, miR-205-5p, miR-NC inhibitor, and miR-205-5p inhibitor. Next, the exosomes were extracted from the exo-miR-NC, exo-miR-205-5p, exo-miR-NC inhibitor, and exo-miR-205-5p inhibitor. The miR-205-5p level in these exosomes was assessed using quantitative reverse transcription-PCR (RT-PCR) (A). After incubation with different exosomes, cell proliferation was determined using the CCK-8 assay (B), cell migration was evaluated using the Transwell assay (C), cell invasion was detected using the Transwell-Matrigel assay (D), tube formation capability was assessed using the Matrigel-based tube formation assay (E), and the VEGFR2 protein level in HUVECs was measured using western blot analysis (F). 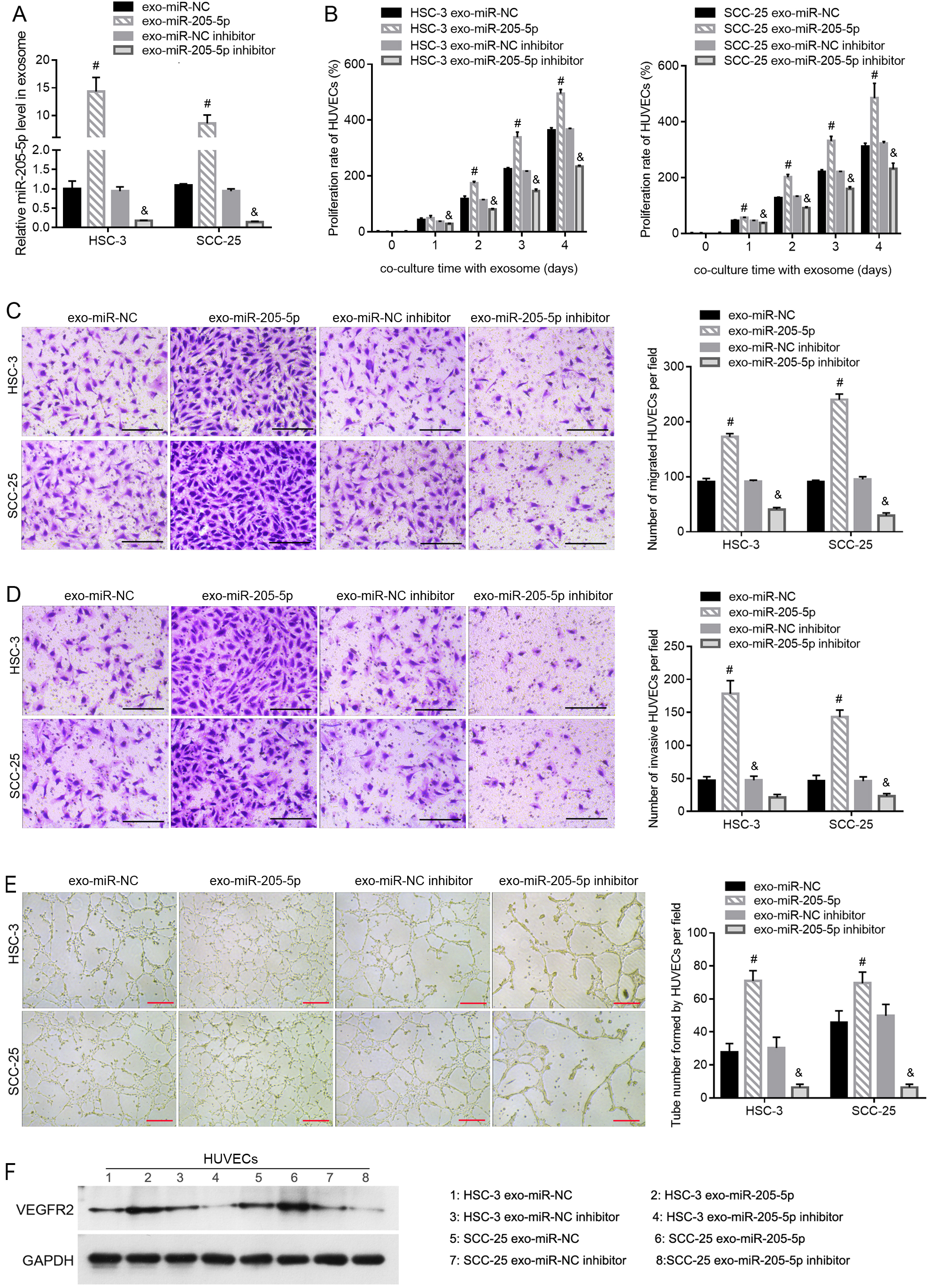
The miR-205-5p levels were higher in exo-miR-205-5p than in exo-miR-NC and were lower in the exo-miR-205-5p inhibitor than in the exo-miR-NC inhibitor (Fig. 2A). To investigate the role of exosomal miR-205-5p in HUVECs, the exosomes extracted from HSC-3 and SCC-25 cells were added to the medium of HUVECs. As shown in Fig. 2B, the proliferation rate of HUVECs was higher after incubation with exo-miR-205-5p compared with that with exo-miR-NC and was lower after incubation with the exo-miR-205-5p inhibitor compared with that with the exo-miR-NC inhibitor. Exo-miR-205-5p incubation increased the number of migrated and invasive HUVECs and the number of tubes formed by HUVECs (Fig. 2C–E). Incubation with the exo-miR-205-5p inhibitor decreased the number of migrated and invasive HUVECs and the number of tubes formed by HUVECs (Fig. 2C–E). Moreover, exo-miR-205-5p incubation upregulated the level of VEGFR2, while incubation with exo-miR-205-5p inhibitor incubation downregulated the level of VEGFR2 in HUVECs (Fig. 2F).
Verification of binding between miR-205-5p and 3
UTR of AMOT
Through bioinformatic analysis, we identified the potential target genes related to angiogenesis. The target genes that inhibit angiogenesis included
Verification of binding between miR-205-5p and the 3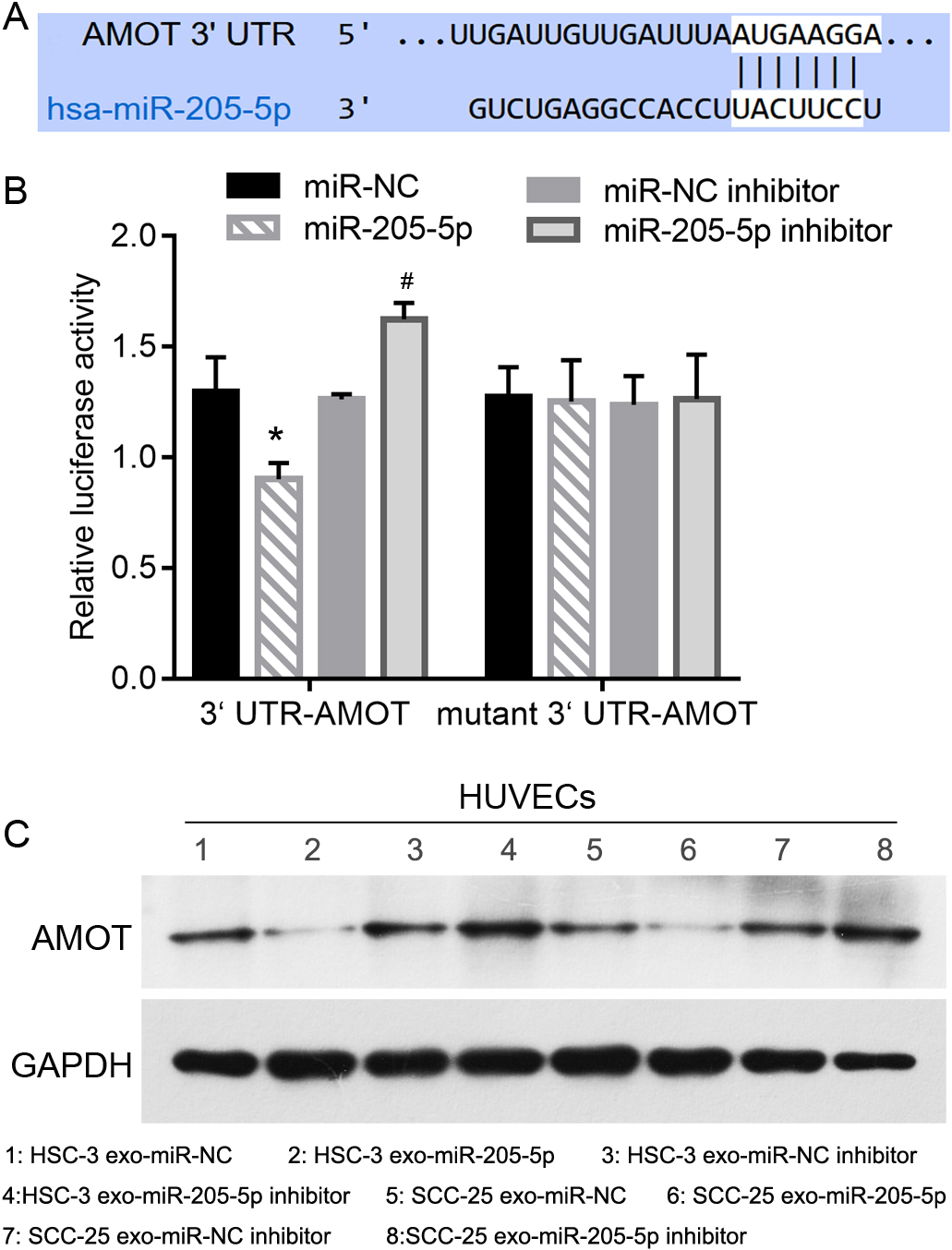
The predicted binding information between miR-205-5p and
AMOT overexpression blocks the effect of exo-miR-205-5p on HUVEC function. HSC-3 and SCC-25 cells were transfected with miR-NC, pcDNA-AMOT 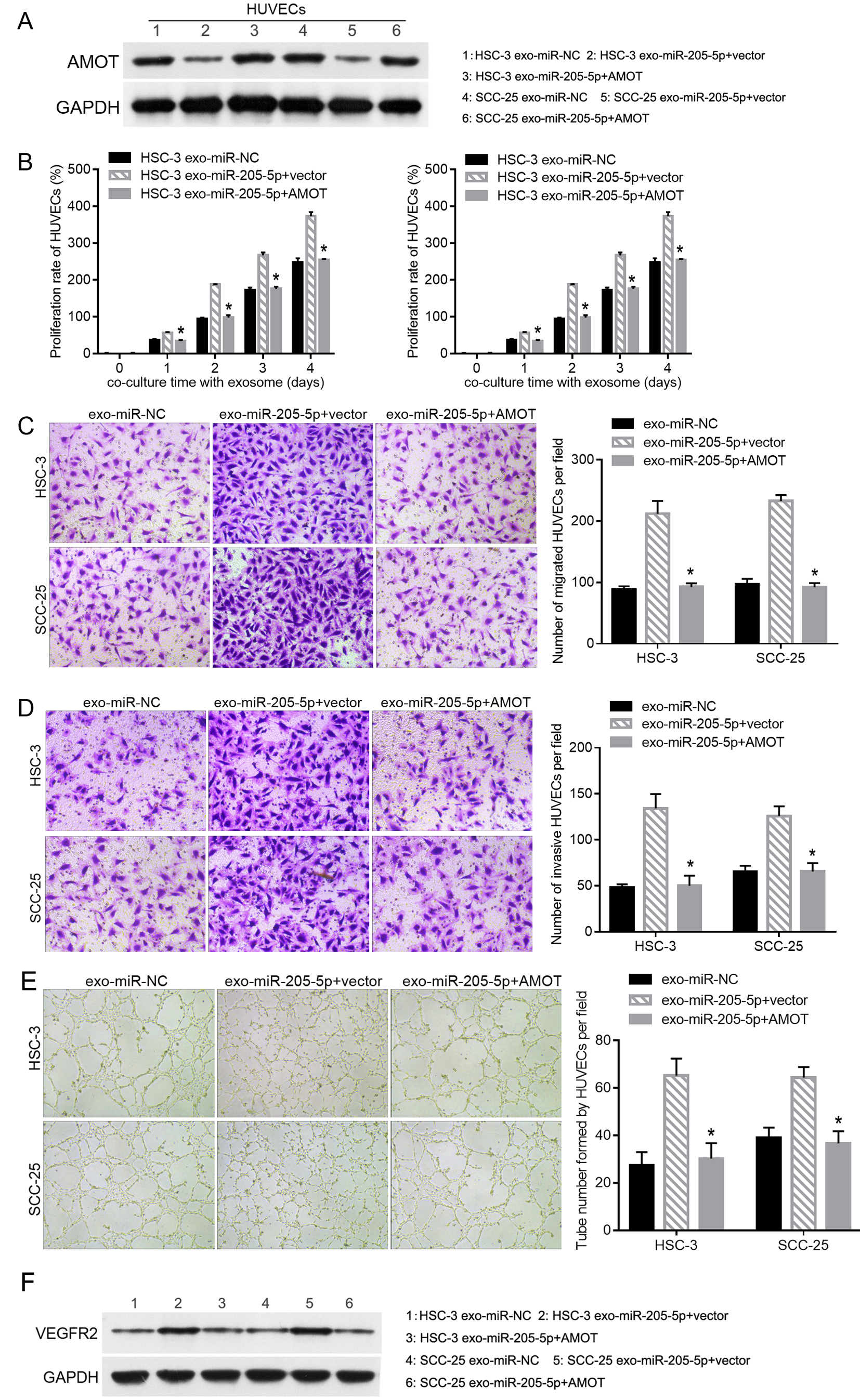
To rescue the inhibition of exo-miR-205-5p on AMOT expression, pcDNA-AMOT was transfected into HUVECs incubated with exo-miR-205-5p to upregulate AMOT expression. The results of western blot showed that AMOT expression was successfully increased in the exo-miR-205-5p
Discussion
A higher level of miR-205-5p expression is associated with inferior progression-free survival in patients with p16-positive oropharyngeal SCCs [19]. miR-205-5p promotes genomic instability in head and neck SCC by targeting BRCA1 and RAD17 [20]. In addition, miR-205-5p promotes cell growth and metastasis in the head and neck SCC cell lines HN4 and CAL27 [21]. These evidences show that miR-205-5p is an important miRNA that is associated with the progression of head and neck SCC. TSCC is the most common type of head and neck SCC, and miR-205-5p might play a role in regulating TSCC. The role of miR-205-5p as an exo-miRNA in regulating the angiogenesis of nasopharyngeal carcinoma has been reported [16]. To further clarify the role of exo-miR-205-5p in regulating tumor angiogenesis, we analyzed the effect of exo-miR-205-5p derived from the TSCC cell lines HSC-3 and SCC-25 in regulating the angiogenic activity of HUVECs.
The results of the present study show that exo-miR-205-5p can enhance the proliferation, migration, invasion, and tube formation capabilities of HUVECs. These results suggest that exo-miR-205-5p plays a role in regulating the angiogenic activity of endothelial cells. These roles of exo-miR-205-5p are consistent with the effects of nasopharyngeal carcinoma-derived exo-miR-205-5p on HUVECs [16]. Nonetheless, a previous study showed that miR-205-5p overexpression suppresses angiogenesis in gastric cancer cells [22]. Based on these reports, we hypothesized that miR-205-5p in cancer cells and exo-miR-205-5p derived from cancer cells might play different roles in the regulation of angiogenesis. Moreover, the exo-miR-205-5p inhibitor could alleviate the proliferation, migration, invasion, and tube formation capabilities of HUVECs. Thus, reducing miR-205-5p levels in the exosomes of cancer cells may be a potential method to inhibit angiogenesis in TSCC. This possibility should be further supported by
Several studies have shown that miRNAs exert their biological functions by participating in the regulation of the translation of their downstream genes [23]. Therefore, target gene identification is important to elucidate the mechanism by which exo-miR-205-5p regulates the angiogenic activity of endothelial cells. The present study reveals that
In conclusion, exo-miR-205-5p derived from TSCC plays a role in regulating the angiogenic activity of HUVECs by targeting AMOT. The exo-miR-205-5p/ AMOT axis may be a new molecular target for the development of anti-vascular drugs for TSCC. This study is a preliminary exploration of the role of the exo-miR-205-5-p/AMOT axis in the angiogenesis of TSCC. Further experiments are warranted to confirm the function of this signaling axis, including those investigating the role of the exo-miR-205-5-p/AMOT axis in other vascular cell models and their role in animal models.
Funding
This work was supported by grants from the Foshan Municipal Health Bureau Scientific Research System (No.20220256).
Author contributions
Conception: Wenxi Huang.
Interpretation or analysis of data: Wenxi Huang, Zanwen Zeng, Zhibin Mai.
Preparation of the manuscript: Wenxi Huang, Yonghui Xu, Zhibin Mai.
Revision for important intellectual content: Wenxi Huang, Zanwen Zeng.
Supervision: Wenxi Huang.
