Abstract
BACKGROUND:
Fibroblast growth factor receptors (FGFRs) are frequently altered in cancers and present a potential therapeutic avenue. However, the type and prevalence of FGFR alterations in infiltrating gliomas (IGs) needs further investigation.
OBJECTIVE:
To understand the prevalence/type of FGFR alterations in IGs.
METHODS:
We reviewed clinicopathologic and genomic alterations of FGFR-mutant gliomas in a cohort of 387 patients. Tumors were examined by DNA next-generation sequencing for somatic mutations with a panel interrogating 205-genes. For comparison, cBioPortal databases were queried to identify FGFR-altered IGs.
RESULTS:
Fourteen patients (3.6%) with FGFR-mutant tumors were identified including 11 glioblastomas, Isocitrate dehydrogenase (IDH) – wildtype (GBM-IDH-WT), 2 oligodendrogliomas, and 1 astrocytoma IDH-mutant. FGFR-altered IGs showed endocrinoid capillaries, microvascular proliferation, necrosis, oligodendroglioma-like cells, fibrin thrombi, microcalcifications, and nodular growth. FGFR3 was the most commonly altered FGFR gene (64.3%). The most common additional mutations in FGFR-altered IGs were TERTp, CDKN2A/B, PTEN, CDK4, MDM2, and TP53. FGFR3 alterations were only observed in GBM-IDH-WT. EGFR alterations were rarely identified in FGFR3-altered gliomas.
CONCLUSIONS:
Histologic features correlate with FGFR alterations in IGs. FGFR3-TACC3 fusion and FGFR3 amplification are the most common FGFR alterations in IGs. FGFR alterations are a rare, but potentially viable, therapeutic target in asubset of IGs.
Introduction
Approximately 100,000 individuals are diagnosed with infiltrating gliomas (IGs) annually [1] and, while this number only amounts to
The fibroblast growth factor family of receptors (FGFRs) are frequently altered in different human cancers (bladder, esophageal carcinomas, lung adenocarcinoma, among others) [14] and have been garnering attention with regards to their role in IGs. Four FGFR genes have been identified: FGFR1-4, which encode for transmembrane receptor tyrosine kinases (TK) that are involved in a myriad of critical cellular processes including cell proliferation, angiogenesis, cell survival, migration, and differentiation [14]. In glioblastoma (GBM), multiple genetic alterations in FGFR genes have been identified, including fusions between FGFR1, -2, and -3 with TACC1 and TACC3[15]. The potential therapeutic value of FGFR inhibitors is supported by an ongoing phase 1 clinical trial evaluating the pan-FGFR inhibitor Erdafitinib (JNJ-42756493) for the treatment of solid tumors (Clinical Trial NCT02365596). Evidence of clinical benefit in patients with recurrent GBMs harboring the FGFR3-TACC3 fusion has been reported [16, 17, 18]. Moreover, several clinical trials evaluating FGFR inhibitors as therapy for gliomas are in progress or have been recently completed (
Material and methods
Patients and tumor samples
We performed a retrospective review of an institutional database of 387 patients diagnosed with IGs between 2010 to 2019. Patients were included in the study if they had 1) histologic diagnosis of IG; 2) analysis by next-generation sequencing; 3) alteration in an FGFR gene. This study was approved by the institutional review board of the University of Texas Health Science Center at Houston (UTHealth), protocol HSC-MS-17-0917. Clinical data were collected from the electronic medical record and compiled using a REDCap database [19]. These included age, gender, histologic diagnosis, imaging studies, recurrence, and survival. Histologic diagnosis was performed following the 2016 WHO Classification of CNS Tumors by a board-certified neuropathologist. A group of 210 patients diagnosed with GBM with wildtype (WT) IDH1, IDH2, and FGFR1-4 genes from our database were used for comparison of clinical, demographic, and genomic characteristics. Patients diagnosed with diffuse astrocytic gliomas, IDH-WT, with molecular features of GBM, WHO grade IV were considered as GBM-IDH-WT for further analysis (Supplementary Fig. 1) [20].
Histologic features
All slides prepared as part of routine initial diagnostic workup [Hematoxylin & Eosin-stain, GFAP (Agilent-GA524-Polyclonal), IDH1-R132H (Dianova-DIA H09-H09), and p53 (Agilent-M7001-DO7)] were reviewed by microscopic examination by a board-certified neuropathologist (LYB).
Targeted sequencing
Tumor samples were analyzed for genetic alterations by a targeted next-generation sequencing assay (NGS) DNA-based assay interrogating 205 genes for somatic mutations and 26 genes for rearrangements, as well as genomic signatures including microsatellite instability and tumor mutational burden (FoundationOne, Foundation Medicine Inc., Cambridge, MA, USA). The details of the FoundationOne assay have been described elsewhere [21, 22, 23].
cBioPortal cohort
We searched for FGFR-altered IG analyzed for single-nucleotide variant (SNV) and copy-number alterations (CNA) in cBioPortal (
Statistical analyses
The clinical and demographic characteristics and genomic alterations between FGFR-altered and FGFR-WT IGs were evaluated by Fisher’s exact test or Mann-Whitney
Results
Clinical and demographic characteristics
There were 387 patients with IGs in the institutional database with a median age of 57 years (range 13–87 years). There were 231 males (59.7%) and 156 (40.3%) females. Ethnicities included 275 Caucasians (71.1%), 52 Hispanics (13.4%), 37 African Americans (9.6%), 17 Asians (4.4%), and 6 of other ethnic backgrounds (1.5%). The most common histologic diagnoses were GBM-IDH-WT (276, 71.3%), followed by GBM-IDH-mutant (IDH-Mut) (26, 6.7%), oligodendroglioma IDH-mutant and 1p/19q co-deleted (OD) (23, 6.0%), anaplastic astrocytoma (AA) IDH-Mut (16, 4.1%), diffuse astrocytoma (DA) IDH-Mut (16, 4.1%), anaplastic oligodendroglioma IDH-mutant and 1p/19q co-deleted (AO) (15, 3.9%), AA IDH-WT (9, 2.4%), DA IDH-WT (6, 1.5%).
Alterations in FGFR genes (FGFR1, FGFR2, FGFR3, and FGFR4) were identified in 14/387 (3.6%) patients: 11/276 (4%) GBM-IDH-WT, 1/23 (4.3%) OD, 1/16 (6.3%) DA IDH-Mut, and 1/15 (6.7%) AO. The clinical characteristics of these 14 patients are depicted in Table 1. Eight patients were females, and the median age of FGFR-mutant patients was 56 years (range 15–75 years). Within the group of FGFR-mutant gliomas, patients with GBM-IDH-WT had a median age of 58.5 years (range 32–70 years). There were no statistically significant differences in clinical characteristics between patients with GBM-IDH-WT, FGFR-altered (
Histologic features of FGFR-altered gliomas
Microvascular proliferation was observed in 10/11 (91%) and pseudopalisading necrosis in 8/11 (72.7%) GBM-IDH-WT tumors, respectively. These histologic findings are well-known features associated with high-grade gliomas. Interestingly, other histologic features were observed in the FGFR-altered tumors independent of their WHO grade.
Oligodendroglia-like features (honeycomb-like arrangement of evenly spaced tumor cells with uniformly rounded nuclei and clear haloes imparting a “fried egg” appearance) were observed in 71% of cases; interestingly, this finding was observed in 67% of non-oligodendroglial tumors. 50% of all FGFR-altered patients exhibited evidence of vascular fibrin thrombi, all of which were GBM-IDH-WT. Additionally, an endocrinoid capillary network was identified in 57.1% of the 14 patients. Extensive microcalcifications were present in 35.7% of patients (all of them GBM-IDH-WT). Finally, a nodular growth pattern was identified in 14.3% of FGFR-altered patients (both were GBM-IDH-WT). Representative cases of these histopathologic features are seen in Fig. 1. The histopathologic features of FGFR-altered IG are summarized in Fig. 2.
Demographic, clinical characteristics, and mutations in FGFR-altered gliomas
Demographic, clinical characteristics, and mutations in FGFR-altered gliomas
Abbreviation: WHO: World Health Organization; PFS: progression-free survival; OS: overall survival; VUS: variants of unknown significance; GBM: glioblastoma; AA: Anaplastic Astrocytoma; DA: Diffuse Astrocytoma; OD: Oligodendroglioma IDH mutant 1p/19q Co-deleted; AO: Anaplastic Oligodendroglioma IDH mutant 1p/19q Co-deleted; Amp: amplification; TMZ: Temozolomide; XRT: Radiotherapy; Rec: At Recurrence; BCZ: Bevacizumab; CPT-11: Irinotecan; GK: Gamma Knife.
Microscopic characteristics of FGFR-altered gliomas are diverse. (A) Tumor cells with perinuclear clearing (indicated by arrows) are present in this oligodendroglioma, IDH-mutant, 1p/19q-codeleted with an FGFR1 mutation (case #11). (B–D) Tumor cells with perinuclear clearing, nodular growth pattern, and vacuolated appearance of the neuropil (indicated by arrows) are evident in this case with FGFR2 mutation (case #9). (E) Tumor cells with perinuclear clearing (horizontal arrow) and microcalcifications (vertical arrows), (F) nuclear palisading, and nodular growth (indicated by arrows) (G) are present in this case with the FGFR3-TACC3 fusion. (H) This other case with an FGFR3-TACC3 fusion shows characteristic features of glioblastoma including vascular proliferation (arrow) and pseudopalisading necrosis (star) (case #2). (I) FGFR4 altered case (#13) with palisading necrosis (stars). (J) Case #14 showed characteristic features of diffuse astrocytoma, IDH-mutant, including an increased number of mildly atypical glial cells. Bar 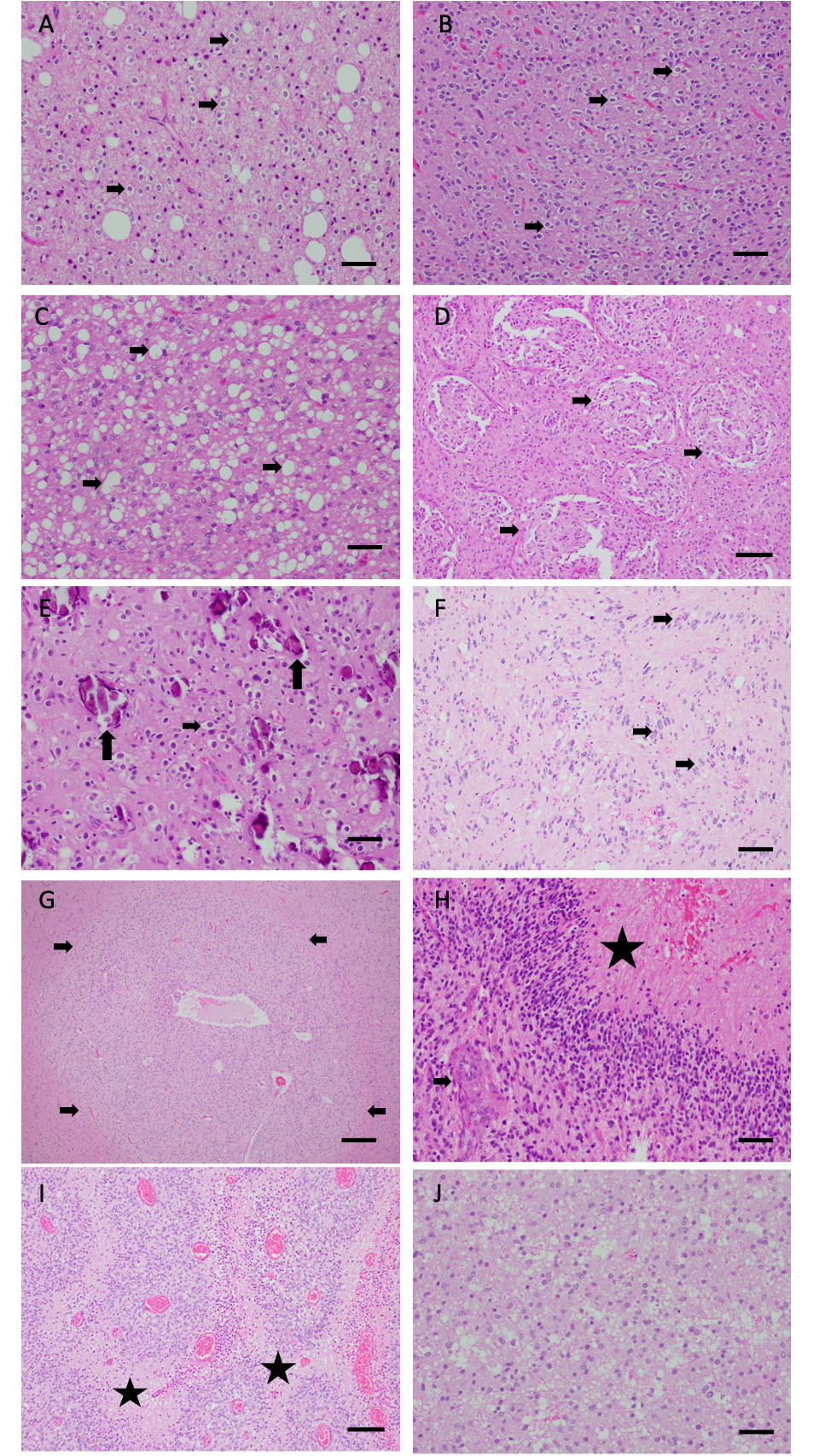
Histological and molecular features of diffuse gliomas with FGFR alterations in the study cohort. All TERT mutations were promoter mutations; PTEN, IDH1/IDH2, TP53, PIK3CA, CIC, and ATRX alterations were point mutations; CDKN2A/B were loss of the gene; and CDK4, MDM2, and EGFR were amplifications of the gene. 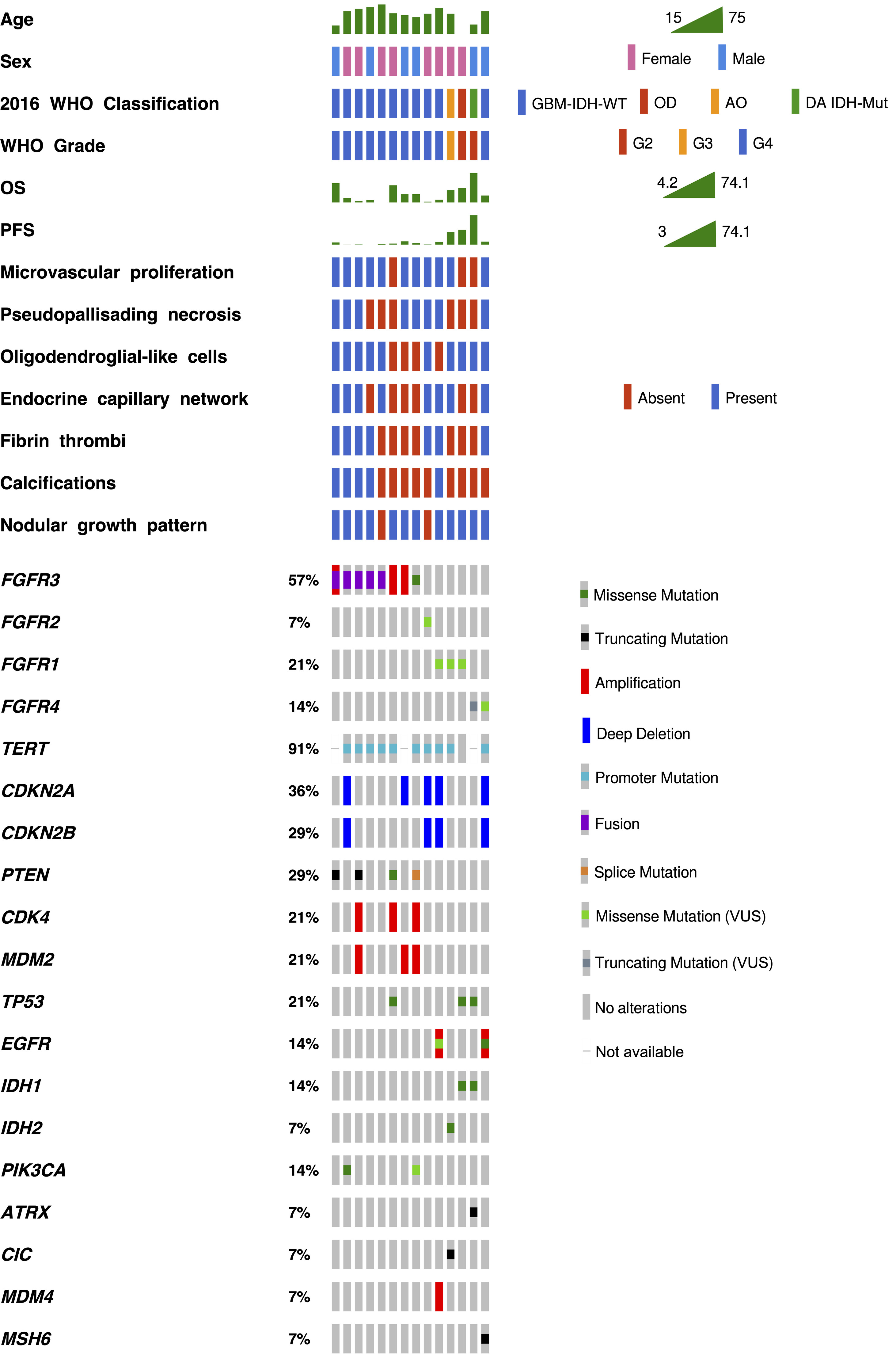
Demographic, clinical, and genomic characteristics of GBM IDH-WT by FGFR alterations
GBM: glioblastoma; WT: wildtype; Mut: mutant.
NGS analysis identified 134 alterations involving 93 genes, approximately half (70/134; 52.2%) were reported as variance of unknown significance. There were 64 alterations in 30 genes that were classified as mutations (SNV, fusions, and CNA). The median number of genetic alterations per patient was 3 (range 1–7). The genetic alterations observed in FGFR-altered cases are described in Table 1. FGFR1-4 alterations were mutually exclusive between FGFR genes but can occur within the same gene (e.g., FGFR3-TACC3 fusion and FGFR3 amplification), as previously described [32]. As demonstrated in Fig. 2, 5/14 (35.7%) tumors had the FGFR3-TACC3 fusion, while 3/14 (21.4%) showed FGFR3 amplification, and one patient had a tumor with an FGFR3 p.K650E SNV. One patient showed both FGFR3-TACC3 fusion and FGFR3 amplification. The remaining FGFR alterations were SNVs, each was observed in a single patient and included FGFR1 (p.T141R, p.N546K, p. K656E), FGFR2 (p.K659), and FGFR4 (p.R80W, H359fs*5).
In addition to FGFR alterations, FGFR-altered gliomas frequently showed alterations in telomerase reverse transcriptase promoter (TERTp) 10/11, 90.1%. CDKN2A/B was altered in 5/14 (35.7%), PTEN in 4/14 (28.6%)
The Fischer-exact test was used to compare the most common genetic alterations between GBM-IDH-WT, FGFR-altered (
FGFR-altered infiltrating gliomas in cBioPortal
In cBioPortal, 104/1884 (5.5%) IG with FGFR alterations were identified. Fifty-eight of these 104 patients were included in the study (3.1% of the cohort), as some patients were incompletely characterized, had an unclear histologic diagnosis, or had FGFR variants of unknown significance. Most FGFR alterations 44/58 (76%) involved the FGFR3 gene. The FGFR3 alterations included 24 cases with amplification (10 co-occurred with an FGFR3 fusion), 19 FGFR3-TACC3 fusion, 4 p.K650E SNV, 1 p.R248H SNV, and 6 other FGFR3 fusions that occurred in one case each (FGFR3-ELAVL3, FGFR3-FBXO28, FGFR3-AMBRA1, FGFR3-ST7L, FGFR-PTBP1, FGFR-NBR1). Most cases were IDH-WT infiltrating gliomas (43/44, 97.7%), except for one AA-IDH-mutant that showed FGFR3 amplification. The 5 cases with FGFR1 alterations showed amplification of the gene. Of the 5 patients with FGFR1-amplified tumors, 3 were GBM-IDH-WT, while 2 were DA-IDH-mutant. Five patients harbored alterations in the FGFR2 gene, including 2 amplifications, 1 FGFR2-SPRNP1 fusion, 1 p.R203H SNV, and 1 p.E731K SNV. FGFR2 altered patients included 2 GBM-IDH-WT, and 3 astrocytomas-IDH-mutant (grade 2–4). Finally, FGFR4 alterations in this cohort included 3 amplifications and 1 p.Y367C SNV. Two GBM-IDH-WT, 1 DA IDH-mutant, and 1 OD harbored FGFR4 alterations. Additionally, the evaluation of other genetic alterations of FGFR-altered IG revealed that FGFR3 alterations were mutually exclusive with ATRX (
Prognosis and survival
There was no significant difference in the overall survival of patients with GBM-IDH-WT, FGFR-altered (
Discussion
The improvement of molecular diagnostic assays has contributed to elucidating a diversity of genomic alterations in IGs. This has improved the classification of gliomas and has also uncovered several potential therapeutic targets including FGFR genes [33]. In this study, we identified 14 IGs with FGFR alterations (out of 387 cases) and reviewed their histologic features and additional genetic alterations.
We identified 3 cases with FGFR1 alterations, p.T141R, p.K656E, and p.N546K. FGFR1 p.T141R was previously described in lung squamous cell carcinoma (lung SCC) [33]. FGFR1 p.K656E and p.N546K are known to occur in GBM [33], however, both SNVs were identified in oligodendrogliomas (with IDH-mutation and 1p/19q codeletion) in our cohort. The same two SNVs have also been identified in pilocytic astrocytomas and rosette-forming glioneuronal tumors [34]. Both FGFR1 p.K656E and p.N546K affect the
Overall Survival of GBM-IDH-WT with and without FGFR alterations. No difference in OS between GBM-IDH-WT, FGFR-Altered (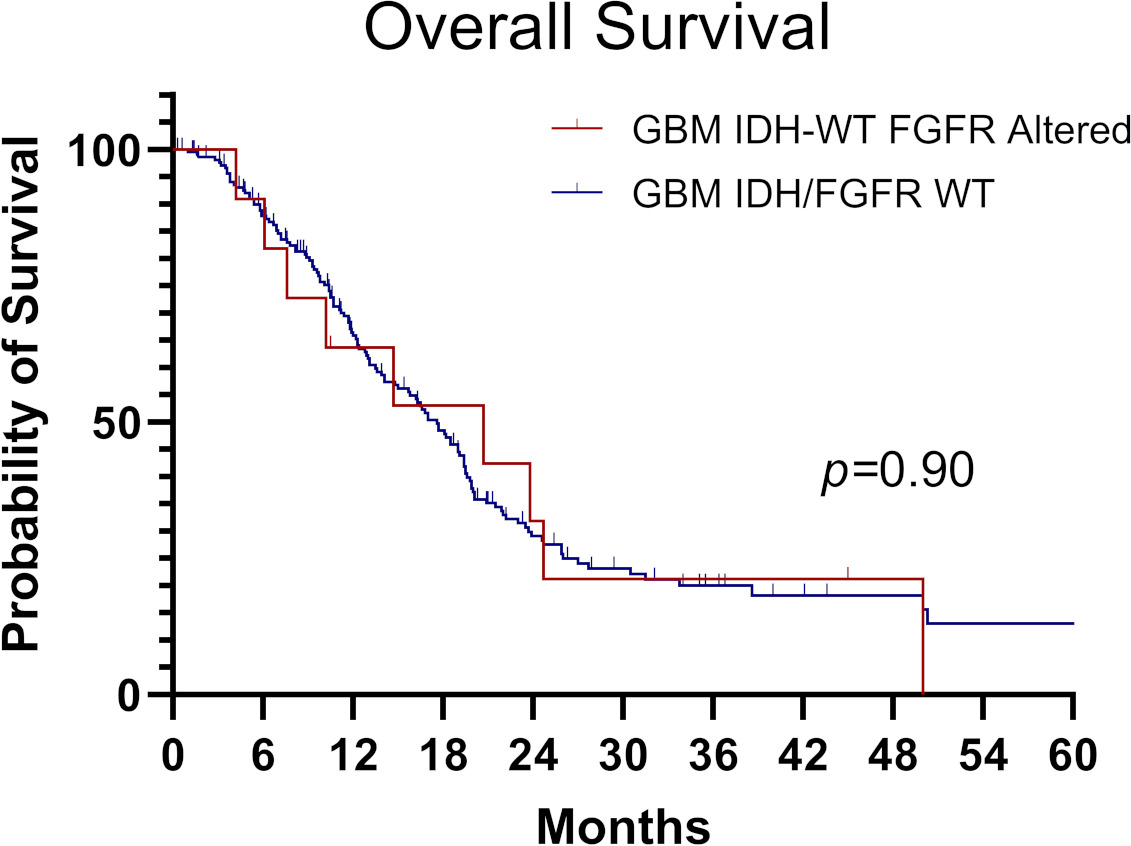
FGFR2 is critical for astrocytes and its overexpression has been previously reported to be associated with lower tumor grade and better survival in gliomas [37]. However, we identified the FGFR2 p.K659E missense SNV, affecting the intracellular tyrosine kinase domain, in one patient with GBM-IDH-WT. This SNV has been described in multiple neoplasms including GBM, pilocytic astrocytoma, lung SCC, endometrial carcinoma, medulloblastoma, and spermatocytic seminoma [33]. The cBioPortal dataset showed 2 cases with FGFR2 amplification, one case with FGFR2-SPRNP1 fusion, and two cases with FGFR2 SNV (p.R203H and p.E731K). The p.R203H SNV has been previously described in colon and endometrium carcinomas, meanwhile, the p.E731K SNV involves the tyrosine kinase domain. Various FGFR2 fusions have been associated with OD in the pediatric population [38]. However, in cBioPortal, FGFR2-mutant tumors were astrocytoma-IDH-mutant or GBM-IDH-WT. In addition, FGFR2 and FGFR3 alterations have recently been described in the Polymorphous low-grade neuroepithelial tumor of the young (PLNTY) [39]. PLNTY is characterized by mutually exclusive genetic alterations involving either BRAF or FGFR2/FGFR3 genes; both pathways are involved in MAPK signaling activation [39].
Summary findings if FGFR-altered infiltrating gliomas
Five patients had a fusion between FGFR3 and the transforming acidic coiled-coil containing 3 (FGFR3-TACC3 fusion). This fusion has been recently discovered in GBM, bladder cancer, lung cancer, head, and neck SCC, and gallbladder cancer [40]. In addition to the FGFR3-TACC3 fusion, we identified two patients with FGFR3 amplification, which has been identified as the most common FGFR3 alteration in human cancers (
We identified 2 cases with FGFR4 alterations (p.R80W and p.H359fs*5). FGFR4 p.R80W has been rarely detected in cancer [32] and the functional consequences of this mutation remain unknown. The p.H359fs*5 mutation is a truncating mutation that has not been reported in COSMIC or TCGA, we predict that it leads to FGFR4 inactivation. Therefore, we interpret these FGFR4 mutations as likely incidental findings (or passenger mutations). The cBioPortal dataset showed 3 cases of FGFR4 amplification and one case with a p.Y367C SNV, which has been suggested to drive tumor growth in a breast cancer cell line [46]. However, FGFR4 alterations are rare events in IG and their significance needs to be further studied.
Previous studies have focused on gliomas with FGFR3-TACC3 fusion [47]. Bielle et al. reported recurrent histologic features including monomorphous ovoid nuclei, nuclear palisading, thin parallel cytoplasmic processes, an endocrinoid network of thin capillaries associated with frequent microcalcifications and desmoplasia in patients harboring the FGFR3-TACC3 fusion [47]. Our findings confirm histologic features that are associated with FGFR-TACC3 fusion including microcalcifications, oligodendroglia-like cells, endocrinoid capillary network, and a nodular growth pattern of tumor cells. We recognize that these features are not specific to FGFR-altered gliomas, however, the presence of some of these features in an IGs could alert the pathologist to the possibility of an FGFR alteration.
The overall survival of the patients harboring the FGFR3-TACC3 fusion ranged from 4.2 to 14.7 months. Interestingly, 1 of the 5 FGFR3-TACC3 positive cases also had coexisting FGFR3 amplification. The overall survival for this case was 50 months, longer than the other FGFR3-TACC3 positive cases. Moreover, 2 additional cases with FGFR3 amplification (#6 and #7) showed OS longer than the average for GBM, IDH-WT, namely 24.7 and 45 months, respectively. These findings raise the question of whether GBM, IDH-WT with FGFR3 amplifications have a better OS. Even though small case series have anecdotally associated FGFR alterations with an aggressive phenotype [48], recent large studies evaluating FGFR3-TACC3 fusions in GBM have identified that patients harboring this fusion had improved survival compared to FGFR3-TACC3 WT patients [44, 45]. The survival benefit on FGFR3-TACC3 fused patients might not be exclusive from this fusion but rather a common feature among patients with FGFR3 alterations that increase the tyrosine domain activity (amplification and SNV). Further studies with a larger number of FGFR3 alterations other than FGFR3-TACC3 fusion are needed to elucidate this question.
This manuscript has the inherent limitations of retrospective cohorts, as well as a cohort with a relatively small sample size. However, the rare frequency of FGFR alterations (
In summary (Table 3), there are characteristic histologic features that can alert the pathologist about the possibility of FGFR alterations in IGs. Alterations in FGFR1-4 are a rare event in adult IGs and frequently co-occur with TERTp mutations in GBM-IDH-WT. FGFR3-TACC3 fusion and FGFR3 amplification are the most common FGFR alterations in adult IDH-WT-IGs. In addition, FGFR3 p.K650E is the most common FGFR3 SNV in IDH-WT astrocytomas. EGFR amplification is infrequent in FGFR3-altered GBM-IDH-WT. No significant difference in OS was observed between FGFR-altered and FGFR-WT GBM-IDH-WT. It is important to identify FGFR alterations in IGs, given the potential implications for diagnosis and targeted therapies.
Funding
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under Award Number K08CA241651 (LYB). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author contributions
Antonio Dono M.D.: Interpretation and analysis of data, preparation of the manuscript.
Hanadi El Achi M.D.: Preparation of the manuscript.
Bethany E. Bundrant B.A.: Interpretation and analysis of data, preparation of the manuscript.
Puneetha S. Goli: Interpretation and analysis of data, preparation of the manuscript.
Ping Zhu M.D., MMed., Ph.D.: Interpretation and analysis of data.
Hanim Istem Ozkizilkaya M.D.: Preparation of the manuscript.
Yoshua Esquenazi M.D.: Conception, interpretation and analysis of data and preparation of the manuscript.
Leomar Y. Ballester M.D., Ph.D.: Conception, interpretation and analysis of data and preparation of the manuscript.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-220041.
Footnotes
Acknowledgments
We would like to thank Kimberly Fontenot for her assistance with this project.
Conflict of interest
The authors declared no conflict of interest.
