Abstract
BACKGROUND:
Endothelial activation and insult may contribute to the aggressive clinical course of small-cell lung cancer (SCLC); however, no predictive biomarker for this pathogenesis has been identified.
OBJECTIVE:
To evaluate the clinical impact of the endothelial activation and stress index (EASIX) in SCLC.
METHODS:
In this retrospective study, the EASIX was calculated from measurements of serum lactate dehydrogenase, creatinine, and platelet levels. A total of 264 patients with SCLC treated with platinum-based chemotherapy were stratified into high and low EASIX groups.
RESULTS:
Complete and objective response rates in the limited-stage (LD) were 19.5% vs. 33.3% (
CONCLUSION:
EASIX may be a potential predictive biomarker of SCLC.
Introduction
Small-cell lung cancer (SCLC) is a highly aggressive tumor. Over the past 15 years, the 2-year relative survival of SCLC has remained low, ranging from 14–15% overall, and from 7–8% in patients with distant metastasis [1]. Its poor prognosis is explained by the rapid growth of primary tumors, early invasion, and metastasis [2]. Although the molecular and biological characteristics of SCLC remain insufficient to affect diagnostic methods, several mechanisms explaining the aggressiveness of SCLC, such as systemic inflammation, cancer cachexia, heterogeneity of tumor cells, and angiogenesis, have been suggested to be associated with a poor prognosis [3, 4, 5, 6, 7, 8, 9]. A previous study suggested that vasculogenic mimicry, a process in which cancer cells acquire endothelial cell-like behavior, is present in human SCLC tumor specimens and is associated with decreased tumor latency, reduced efficacy of cisplatin, and reduced overall survival (OS) [10]. Another clinical study reported that baseline levels of circulating endothelial cells, indicating profound vascular damage, were higher in patients with SCLC than in healthy subjects, and correlated with a lower treatment response rate and worse progression-free survival (PFS) [11, 12]. Despite such theoretical developments, there is little evidence of a clinically useful biomarker of endothelial activation and insult in SCLC.
Recently, the endothelial activation and stress index (EASIX) was introduced as a novel biomarker for endothelial dysfunction, predicting OS and non-relapse mortality after the onset of acute graft-versus-host disease in patients who underwent allogeneic stem cell transplantation [13]. The EASIX is easily calculated from routine laboratory tests, including those for serum lactate dehydrogenase (LDH), creatinine, and platelet count. Subsequently, EASIX and its modified form have been suggested as prognostic factors in lower-risk myelodysplastic syndromes and multiple myeloma [14, 15]. In addition, it has been proposed as a predictive marker for chimeric antigen receptor T-cell-related toxicities and disease severity in severe acute respiratory syndrome coronavirus-2 [16, 17]. The clinical implications of the EASIX in solid tumors, including SCLC, have not yet been reported. In this study, we evaluated the predictive ability of the EASIX for treatment response, treatment-related toxicity, and survival in patients with SCLC.
Materials and methods
Patients
The electronic medical records of 264 consecutive patients who were histologically diagnosed with SCLC between July 2006 and August 2021 at a single institution were retrospectively reviewed. The eligibility criteria were as follows: (1) treatment with concurrent chemoradiotherapy (CCRT) or palliative chemotherapy with etoposide or irinotecan plus platinum-based regimens as first-line therapy; (2) records of pretreatment serum LDH, creatinine, and platelet levels; and (3) no evidence of double primary malignancy. Consent to participate was not applicable due to the retrospective nature of this study in which the authors reviewed the electronic medical records without any intervention. The study was approved by the Institutional Review Board of Gyeongsang National University Hospital.
Assessments
EASIX was calculated as serum LDH (IU/L), serum creatinine level (mg/dL), and platelet count (
Time-dependent receiver operating characteristic (ROC) curve of the Endothelial Activation and Stress Index (EASIX) for prediction of (A) 18-month overall survival in limited-stage disease (LD) and (B) 10-month overall survival in extensive-stage disease (ED). Black circles indicate EASIXs of (A) 0.73 in LD and (B) 1.27 in ED. These cut-off points were determined by maximizing the Youden index.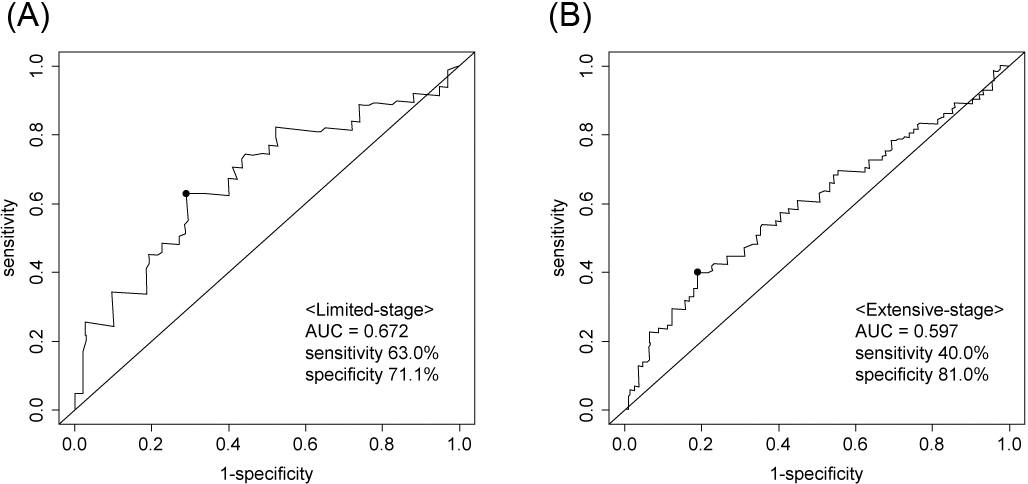
The correlations between variables were tested using the Mann-Whitney U test and the chi-squared test for continuous and categorical variables, respectively. Time-to-progression (TTP) was calculated as the time from the first day of treatment to the progression or last follow-up date. The definition of PFS was the same as that of TTP; however, death from any cause was also considered an event. OS was defined as the time from the first day of treatment to death or the last follow-up date. The Kaplan-Meier method, log-rank test, and Cox regression analysis were used to estimate survival. All variables with a
Results
Patient characteristics
In total, 264 patients were included in the analysis. The cut-off values of EASIX were 0.73 (area under curve [AUC] 0.672, sensitivity 63.0%, and specificity 71.1%) and 1.27 (AUC 0.597, sensitivity 40.0%, and specificity 81.0%) in LD and ED, respectively. The number of patients with high and low EASIX was 41 and 48 in the LD group and 51 and 124 in the ED group, respectively (Fig. 1).
Comparison of baseline characteristics between the low- and high-EASIX groups
Comparison of baseline characteristics between the low- and high-EASIX groups
Abbreviations: EASIX, Endothelial Activation and Stress Index; IQR, interquartile range; ECOG PS, Eastern Cooperative Oncology Group performance status.
The patient characteristics were compared between the high and low EASIX groups (Table 1). In the LD group, the proportion of elderly (median age, 69 vs. 65 years) and male (100% vs. 72.9%) patients was higher in the high EASIX group than in the low EASIX group. Prophylactic cranial irradiation was performed less frequently in the high EASIX group than in the low EASIX group (46.3% vs. 68.8%) because of a lower treatment response rate in the high EASIX group. In the ED, male sex (96.1% vs. 85.5%) and poor PS (35.3% vs. 18.6%) were more common in the high EASIX group than in the low EASIX group. Gender differences between the high and low EASIX groups in both stages could result from the essential differences in serum creatinine level by sex (mean
Treatment responses
Abbreviations: EASIX, Endothelial Activation and Stress Index; N/E, not evaluable.
Treatment-related toxicity and treatment compliance
Abbreviations: EASIX, Endothelial Activation and Stress Index; IQR, interquartile range.
The treatment response rates according to EASIX are presented in Table 2. In the LD group, the CR rates were 19.5% and 33.3% (
Kaplan-Meier curves for (A, D) time to tumour progression, (B, E) progression-free survival, and (C, F) overall survival according to the Endothelial Activation and Stress Index (EASIX) and tumour stage.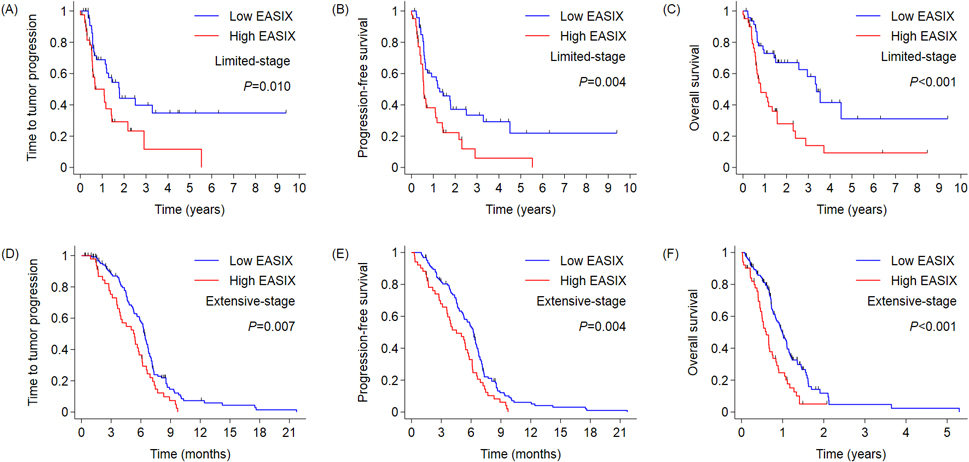
There were no differences in hematologic toxicities between the two groups (Table 3). The median treatment cycles were 4 and 5.5, respectively, in the high and low EASIX groups in both stages. Because a total of four cycles of chemotherapy (with two optional cycles in the case of ED) are currently recommended [21], it is difficult to determine whether the high EASIX group was undertreated. The frequency of early discontinuation and treatment-related mortality was not statistically different between the two groups, regardless of the stage.
Survival
The median follow-up durations were 27 and 18 months in the LD and ED groups, respectively. In LD, the median TTP was 13.1 and 21.4 months in the high and low EASIX groups, respectively (
Cox regression analysis for PFS and OS
Cox regression analysis for PFS and OS
Abbreviations: PFS, progression-free survival; OS, overall survival; HR, hazard ratio; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; EASIX, Endothelial Activation and Stress Index.
Multivariate analysis was performed to determine the PFS and OS (Table 4). The common independent prognostic factors in all stages and dependent variables were ECOG PS and EASIX. In LD, high EASIX was an independent prognostic factor for worse PFS (hazard ratio [HR] 1.967, 95% confidence interval [CI] 1.176–3.290,
This is the first study to report the role of EASIX as a predictive and prognostic factor in patients with SCLC. Individuals in the high EASIX group had a significantly worse prognosis than those in the low EASIX group. Given the small difference in the treatment response rate between the two groups, the difference in survival was larger than expected. Such a difference was more pronounced in the LD. The low EASIX group in LD had a median OS longer than 3 years, and approximately one-third of the patients were long-term survivors. The median TTP, PFS, and OS of the high EASIX group in LD were slightly greater than those in patients with ED, and none of the patients remained disease-free. Treatment-related toxicities and treatment compliance were not significantly different between the two groups. These findings suggest that a non-durable response to treatment coupled with the aggressiveness of tumor cells may result in poor prognosis in the high EASIX group.
The clinical impact of each EASIX component in SCLC has been evaluated in many previous studies. Elevated serum LDH levels indicate the release of the enzyme by inflammation and tissue turnover, which reflects a high tumor burden and metabolic activity [22, 23]. High serum LDH levels are associated with a low treatment response rate and worse PFS and OS in patients with SCLC treated with first-line platinum-based chemotherapy [22, 24, 25]. A pooled analysis of 14 trials found that an elevated serum creatinine level was a negative prognostic factor in ED-SCLC but not in LD-SCLC [26]. Thrombocytopenia, confirmed by bone marrow biopsy, is an indication of bone marrow involvement in SCLC, which negatively affects the prognosis [27]. These findings support the concept that the measurements used to calculate EASIX can be used synergistically to predict the prognosis of SCLC.
Rapid and widespread metastasis is a challenging problem in the treatment of SCLC. Vascular inflammation, increased endothelial permeability, and extravasation of tumor cells through the VCAM1 and Notch signaling pathways contribute to cancer metastasis [28, 29, 30]. Many studies have investigated the role of angiogenesis inhibitors in SCLC; however, most have failed to demonstrate their efficacy. Bevacizumab, a monoclonal antibody for vascular endothelial growth factor (VEGF), as triplet induction therapy with etoposide and a platinum-based regimen, has resulted in modest improvement in treatment response, PFS, or no OS benefit, and more frequent adverse events in several randomized trials for ED-SCLC [28, 31, 32]. Small molecular weight tyrosine kinase inhibitors for the VEGF receptor, such as sunitinib, sorafenib, vandetanib, and apatinib, also did not show significant clinical benefit regardless of stage and treatment setting [29, 33, 34, 35]. The main problem with angiogenesis inhibitor trials is the lack of predictive biomarkers. Although elevated VEGF levels have been reported to be associated with worse prognosis in SCLC [36], assessment of serum VEGF and soluble VEGF receptor levels has failed to identify patients who may benefit from bevacizumab therapy [32]. In contrast, EASIX has several attributes that provide a theoretical background that supports its role as a predictive biomarker of angiogenesis inhibition. LDH-A, a subunit of LDH, is required to sustain rapid angiogenesis in pulmonary microvascular endothelial cells [37]. Renal impairment is accompanied by endothelial activation [38, 39]. Thrombocytopenia is induced by thrombotic microangiopathy [40, 41]. Unfortunately, the role of EASIX as a predictive biomarker for angiogenesis inhibitor-based therapy could not be analyzed in our study, because no patient was treated with angiogenesis inhibitors. This is a limitation of our study that should be assessed in future studies.
The retrospective design is the main limitation of our study. This might lead to a potential selection bias due to the imbalance in baseline characteristics, including age, sex, and performance status, between the two groups. Statistical significance of the survival difference between the high and low EASIX groups was demonstrated on multivariate analyses adjusted for these factors. The small sample sizes in each stage made it difficult to determine the adverse effects of treatment-related toxicity on the patients’ prognosis. In the LD group, hematologic toxicities and early discontinuation of treatment were numerically higher in the high EASIX group than in the low EASIX group, but the difference was not statistically significant. Another limitation is that a minority of patients (10.9%, 19 of 175) received immune checkpoint inhibitor-based therapy for ED-SCLC due to the long period of data collection. Pivotal trials for atezolizumab- or durvalumab-based triplet induction therapy followed by maintenance therapy using these agents were reported in 2018 and 2019 [42, 43]. The number of female patients was too small to stratify our findings into sex-specific EASIX groups, even though there was a difference in serum creatinine levels between male and female patients. Therefore, our results should be validated by a prospective study with a larger sample size and expanded to patients who are treated with novel therapeutic options, such as immunotherapy and angiogenesis inhibitor-based combination regimens.
In conclusion, this study showed a close relationship between EASIX and prognosis in LD- and ED-SCLC patients. Pretreatment with EASIX may help to identify patients who need to be treated with alternative therapeutic strategies for SCLC. We also suggest further evaluation of the validity of EASIX as an easily available predictive biomarker for angiogenesis inhibitor-based therapies.
Footnotes
Acknowledgments
This study was supported by the Ministry of Education of the Republic of Korea and the National Research Foundation of Korea (NFR 2019S1A5A2A03041296).
Author contributions
Conception: Se-Il Go and Gyeong-Won Lee.
Interpretation or analysis of data: Se-Il Go, Sungwoo Park, Myoung Hee Kang, and Gyeong-Won Lee.
Preparation of the manuscript: Se-Il Go, Hoon-Gu Kim, Jung Hun Kang, Jung Hoon Kim, and Gyeong-Won Lee.
Revision for important intellectual content: All authors.
Supervision: Gyeong-Won Lee.
