Abstract
BACKGROUND:
The relationship of the ABO blood group system with the immune response is known, but its relationship with immune checkpoint inhibitors (ICIs) has not been clearly investigated until now.
OBJECTIVE:
In this study, the relationship between different blood groups and nivolumab treatment response in patients with advanced malignant melanoma was investigated.
METHODS:
The data of patients who used nivolumab for advanced malignant melanoma between April 2018 and April 2021 were retrospectively reviewed.
RESULTS:
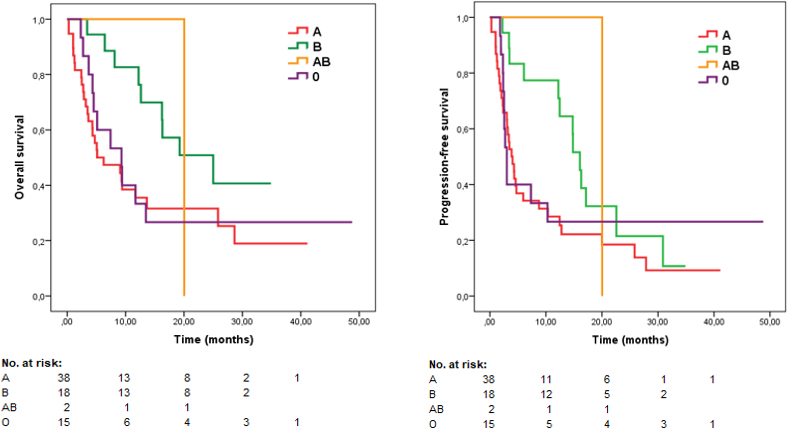
A total of 73 patients were included in the study. In the progression-free survival (PFS) analysis according to blood groups, it was 3.9 months, 16.1 months, 20.0 months and 3.0 months for A, B, AB and O, respectively (
CONCLUSIONS:
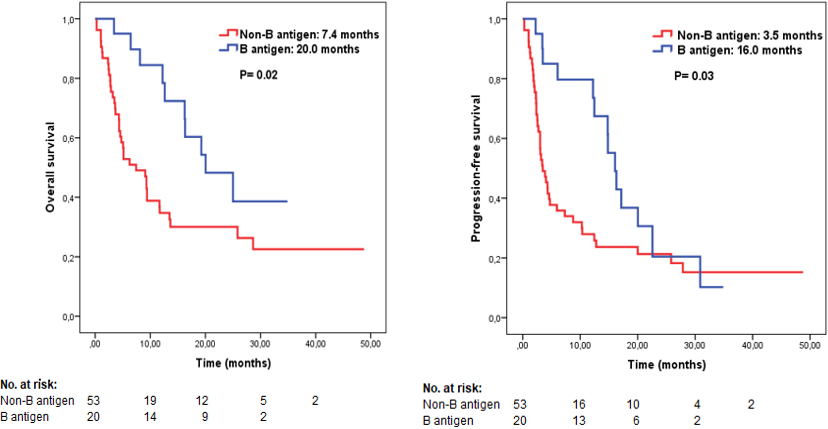
The presence of B antigen provides a significant advantage in terms of survival in patients using ICIs for advanced melanoma.
Introduction
Melanoma is a skin cancer with one of the worst prognoses. It is the fifth most common cancer in men and women in the USA and its incidence increases with age [1]. Most cases of malignant melanoma are diagnosed at an early stage, when surgical excision can be curative. However, immunotherapies, targeted agents, and cytotoxic chemotherapy are needed in the treatment of advanced melanoma. Immune checkpoint blockade induces response and provides unique clinical benefits in advanced melanoma treatment. Promising results have been obtained in treatment with immune checkpoint inhibitors (ICIs) such as programmed cell death-1 (PD-1) inhibitors (pembrolizumab and nivolumab) and cytotoxic T lymphocyte-associated protein-4 (CTLA-4) inhibitors (ipilimumab). In addition, targeted therapies (BRAF plus MEK inhibitors) also prolong progression-free and overall survival compared to chemotherapy. However, treatment success is prevented by the high resistance to anti-PD-1 agents seen in advanced metastatic melanoma.
ABO blood group antigens are expressed in various tissues. Specific ABO blood groups are easily observed parameters that have been reported to be associated with the increased incidence of different tumor types. Previous studies showed a relationship between ABO blood groups and malignancies such as hepatocellular carcinoma and gastric and pancreatic cancer [2, 3, 4]. The relationship between blood types and the incidence of malignant melanoma has been investigated before and there are conflicting data. However, the relationship between ABO blood groups and immunotherapy response in cancer treatment is full of unknowns. To the best of our knowledge, the relationship between blood groups and ICIs’ response in malignant melanoma has not been investigated so far. In the present study, the relationship between different blood groups and nivolumab treatment response in patients with advanced malignant melanoma was investigated.
Materials and methods
The data of patients using nivolumab with a diagnosis of advanced stage, unresectable malignant melanoma and followed up in 5 different oncology centers in Turkey between April 2018 and April 2021 were reviewed retrospectively. In accordance with the health policies in this country, in order to use nivolumab in the treatment of malignant melanoma, there is a prerequisite to receive one line treatment (chemotherapy or BRAF/MEK inhibitors). It can also be used as a single agent only. Therefore, our patient population consists of patients who received at least one line of treatment and then used nivolumab as a single agent.
Age 18 years or older, histologically proven cutaneous malignant melanoma, Eastern Cooperative Oncology Group (ECOG) performance status 0–1 or 2, measurable unresectable metastatic disease according to the Immune Response Evaluation Criteria in Solid Tumors (iRECIST), and ABO blood group known patients were included in the study. In addition to those who did not meet the inclusion criteria, patients with brain metastases and second primary cancer were excluded from the study.
Age, sex, smoking status, ECOG performance status, BRAF mutation status, number of metastases, metastatic site localization, treatment line using nivolu-mab, ABO and RH blood groups, and hemogram and biochemical parameters were recorded from the patient files. Toxicity analysis could not be performed due to insufficient toxicity data.
Nivolumab was given at a dose of 3 mg/kg (max dose 240 mg) every 2 weeks. All patients underwent a physical examination and hematological and biochemical evaluations every 2 weeks in the first cycle and then on the first day of each cycle. Tumor response was assessed by computed tomography (CT) or positron emission tomography (PET)/CT every 12 weeks according to iRECIST criteria.
Prognostic and predictive factor analysis
Nine variables that may have an impact on survival were selected. The variables were divided into two categories: age (
Statistical analysis
Baseline characteristics of all patients
Baseline characteristics of all patients
ECOG: Eastern Cooperative Oncology Group, LDH: Lactate Dehydrogenase.
The statistical analysis was performed using the Statistical Package for the Social Sciences Version 22.0 for Windows (SPSS Inc., Chicago, IL, USA). Comparison of the 2 groups was performed by Mann-Whitney U test and Pearson chi-square or Fisher test for continuous and categorical variables, respectively. Survival analysis was analyzed by the Kaplan-Meier method using the log-rank test. Survival times were determined in the 95% confidence interval (CI) range.
Overall survival (OS) was calculated from the day of receiving analyzed treatments to death or the last visit. The time to disease progression or death from any cause was calculated as progression-free survival (PFS).
Patient characteristics by B antigen status