Abstract
Immune checkpoint inhibitors (ICIs) are commonly used to treat patients with advanced urothelial cancer. However, a significant number of patients do not respond to ICI, and the lack of validated predictive biomarkers impedes the success of the ICI strategy alone or in combination with chemotherapy or targeted therapies. In addition, some patients experience potentially severe adverse events with limited clinical benefit. Therefore, identifying biomarkers of response to ICI is crucial to guide treatment decisions. The most evaluated biomarkers to date are programmed death ligand 1 expression, microsatellite instability/defective mismatch repair phenotype, and tumor mutational burden. Other emerging biomarkers, such as circulating tumor DNA and microbiota, require evaluation in clinical trials. This review aims to examine these biomarkers for ICI response in urothelial cancer and assess their analytical and clinical validation.
Introduction
Bladder cancer is the most common malignancy of the urinary system, with urothelial carcinoma (UC) accounting for 90% of all cases. 1 The disease spectrum ranges from non-muscle invasive to muscle-invasive and metastatic, each with its own clinical behavior, biology, prognosis, and treatment. Patients with non-muscle-invasive tumors are treated with endoscopic resection and adjuvant intravesical therapy, depending on the risk classification. Muscle-invasive tumors are preferably treated with neoadjuvant cisplatin-based chemotherapy followed by surgery, and metastatic tumors with systemic therapies. 2 Up to 70% of patients with metastatic UC (mUC) are ineligible for cisplatin, the main therapeutic option for muscle-invasive or advanced UC, due to poor performance status, impaired renal function, cardiac comorbidities, and hearing loss. 3 The prognosis of these patients remains poor, with a median survival of 13–16 months. 4
Over the last decade, there have been significant advances in the clinical management of UC due to a better understanding of UC biology. 5 In 2017, immune checkpoint inhibitors (ICIs) targeting programmed cell death 1 (PD-1) or programmed cell death-ligand 1 (PD-L1) transformed the treatment of mUC. Early pivotal trials showed that ICIs achieved promising results as second-line therapy and beyond, with a median overall survival (OS) between 8 and 18 months.6–9 As a result, ICI-based regimens were approved for the frontline treatment of cisplatin-ineligible patients and as maintenance among pretreated patients with mUC.10–12 However, developing a predictive biomarker for the response to ICI remains a major challenge. In this article, we aimed to review the biomarkers reported in pivotal trials of ICIs in mUC and discuss their potential clinical implications, focusing on pre-analytical, analytical, clinical validation, and regulatory approval.
Biomarkers related to the tumor
Histology subtypes
The most frequent histology is UC. In around 20% of patients, variant histological features are observed, such as squamous cell differentiation, glandular differentiation, and micropapillary. Pure non-UCs represent 10% of bladder cancers including squamous cell carcinoma, adenocarcinoma, and neuroendocrine tumors. There is limited data evaluating the use of ICIs in UC with histological variants. Epaillard et al. retrospectively collected data on nine patients with variant histological receiving first-line ICI. The overall response rate (ORR) was 22.2%. The median progression-free survival (PFS) was 3.3 months [95% CI: 2.3–not reported (NR)] and the median OS was not reached (95% CI: 13.8–NR). 13 In the SAUL trial which evaluated atezolizumab in 1004 patients with locally advanced or mUC or non-UC, Sternberg et al. reported an ORR of 9% (Complete Response 1%) in non-UC, whereas Mc Gregor et al. noted an ORR of 37% (CR 5%) with the combination of nivolumab and ipilimumab.14,15 In both studies, the median PFS was less than 4 months. In the neoadjuvant PURE trial which evaluated pembrolizumab in patients with muscle-invasive bladder carcinoma, the authors showed a complete pathologic response rate of 16% for patients with a predominant variant. 16 However, all these studies were purely descriptive and the prognostic effect of histological subtype remains uncertain. Similarly, the predictive impact of histological subtypes remains unknown and has not been studied.
Genetic alterations in bladder cancer
The genomic heterogeneity of muscle-invasive bladder cancers is well recognized. Intrinsic UC subtypes were initially identified through transcriptomic analyses using clustering analysis, which yielded luminal papillary, luminal non-specified, luminal unstable, stroma-rich, basal/squamous, and neuroendocrine subtypes. 17 The clusters displayed distinct oncogenic mechanisms, immune stromal phenotypes, and prognoses. The luminal subtype, which accounts for half of UC cases, is characterized by high expression of uroplakins (UPK2 and UPK1A) and urothelial differentiation markers. The luminal-papillary subtype is enriched in fibroblast growth factor receptor 3 alterations in approximately 40%. The basal-squamous subtype represents 35% of patients and is distinguished by high expression of basal and stem-like markers (CD44, KRT5, KRT6A, and KRT14) and squamous differentiation markers (TGM1, DSC3, and PI3). Various exploratory analyses suggested that these molecular classified subtypes respond differently to ICI. In the single-arm multicenter phase II trial IMvigor210, which assessed atezolizumab monotherapy in patients with locally advanced or mUC cisplatin-ineligible, PD-L1 prevalence was higher in the basal subtype. However, responses were observed in all subtypes, with a higher response rate in luminal II subtype patients (34%, p = 0.0017). 18 In the single-arm multicenter phase II trial Checkmate 275, which assessed nivolumab after platinum-based therapy, basal tumors had the highest proportion of responders with an ORR of 30%. Luminal cluster II tumors have an ORR of 25%. 9 These discrepant outcomes may be linked to differences in biopsy tissue sites and a lack of agreement on molecular classification at that time. Recently, Meek et al. identified five transcriptomic subtypes that were associated with clinical and pathological responses to pembrolizumab in patients included in the neoadjuvant PURE trial. They found that tumors with enhanced interferon (IFN) A and IFN signatures had a better response compared to tumors with low IFN expression. In addition, they identified the histone demethylase KDM5B as a repressor of tumor immune signaling pathways in one resistant subtype, known as luminal-excluded. 19 Although the consensus classification suggests possible therapeutic implications, there is currently insufficient clinical validation data to employ molecular classification as a potential predictive biomarker for ICI response. Therefore, a randomized trial is necessary to validate these findings and refine subtype classification as independent predictive biomarkers of ICI response.
Programmed cell death-ligand 1
Concurrently with the development of anti-PD-1/PD-L1 inhibitors, PD-L1 assays have been developed. However, the evaluation of PD-L1 expression by immunohistochemistry varies depending on the prescribed ICI. Different antibodies, cell types (tumor versus immune cell versus both), and cutoff values are used to define positivity. For instance, the use of pembrolizumab requires a combined positive score, defined as the percentage of PD-L1–expressing tumor and infiltrating immune cells relative to the total number of tumor cells, of 10 or more. 10 Trials assessing atezolizumab define positive PD-L1 expression as an expression of ⩾5% in immune cells using the SP142 assay (Roch/Ventana diagnostic) (Table 1). The lack of standardization of PD-L1 expression hampers its use as a reliable biomarker. Furthermore, PD-L1 is a dynamic protein with variable expression over time. Burgess et al. showed that high PD-L1 immunochemistry expression is temporally and spatially discordant between primary and mUC lesions. 20 This dynamic PD-L1 expression is not taken into consideration when clinicians use this biomarker.
Main studies evaluating ICI for mUC.

Ab, antibody; CPS: combined positive score; IC: immune cell; ICI, immune checkpoint inhibitors.; mo, months; mUC, metastatic urothelial carcinoma; N, number of patients; NE, not evaluable; NIVO1-IPI3: nivolumab 1 mg/kg and ipilimumab 3 mg/kg; NIVO3: nivolumab 3 mg/kg; NIVO3-IPI1: nivolumab 3 mg/kg and ipilimumab 1 mg/kg; NR: not reported; ORR: overall response rate; OS, overall survival; PD-L1, programmed cell death-ligand 1; PFS, progression-free survival; Pos.: positive; pts: patients; TC: tumor cells.
Despite its limitations, PD-L1 is the most commonly used ICI biomarker. A recent meta-analysis of 11 studies involving 1697 patients with bladder cancer treated with ICI found that high PD-L1 expression in tumor cells was associated with poorer OS [hazard ratio (HR) 1.83, 95% CI = 1.24–2.71, p = 0.002]. 23 The phase III IMvigor211 trial, which enrolled pretreated patients with platinum-based chemotherapy, showed that atezolizumab had a higher ORR among patients with PD-L1 positive (21% versus 8%) but did not demonstrate significantly longer OS compared to chemotherapy in patients with platinum-refractory mUC overexpressing PD-L1 (IC2/3), failing to meet the primary endpoint. 8 In addition, low PD-L1 expression in immune cells (IC0/1) was found to be a poor prognostic factor in patients receiving atezolizumab or chemotherapy.8,21 Similar results were seen in trials assessing the activity of durvalumab, avelumab, pembrolizumab, and nivolumab, where a higher ORR was observed in the PD-L1-positive subgroup.9,10,12,24 In the adjuvant population, the Checkmate 274 trial has also shown improved outcomes in patients with positive PD-L1 expression. 25 A meta-analysis of eight studies involving 1436 patients evaluated the use of PD-L1 as a biomarker for response to ICI in mUC. 26 High PD-L1 expression was associated with significantly higher ORR than those with low PD-L1 expression [relative risk (RR) 0.53, 95% CI 0.43–0.65] and higher 1-year OS (RR 0.70, 95% CI: 0.54–0.91). The trials cited above also reported a benefit for the PD-L1-negative population but most of these trials were not adequately powered to draw strong conclusions regarding the efficacy of the PD-L1-negative group.
According to the Food and Drug Administration (FDA) and the European Medicines Agency, PD-L1 testing is a mandatory requirement for the use of ICI (pembrolizumab or atezolizumab) as monotherapy in patients with locally advanced or mUC who are unfit for cisplatin-containing chemotherapy and have not received prior therapy. 2 This recommendation is based on exploratory analyses that have shown longer OS in PDL1-positive populations treated with pembrolizumab and atezolizumab. Therefore, in this setting, pembrolizumab or atezolizumab should only be used in patients unfit for cisplatin-containing chemotherapy whose tumors overexpress PD-L1 (i.e. Combined Positive Score ⩾ 10 using the Dako 22C3 assay® for pembrolizumab and tumor-infiltrating immune cells covering ⩾5% of the tumor area using the SP142 assay for atezolizumab).
Tumor mutational burden
Tumor mutational burden (TMB) refers to the number of somatic mutations per megabase, detected by next-generation sequencing, excluding single nucleotide polymorphisms, germline mutations, copy number variations, and structural variations.21,27 High-TMB is associated with an increased expression of tumor-specific neoantigens that are required by the immune system to recognize cells, leading to better response to immunotherapy.28–30 Currently, there is no clear consensus on the TMB cutoff for patient stratification. For instance, Foundation Medicine defines TMB as high, intermediate, and low based on the occurrence of 20 mutations (mut)/Mb or more, 6–19 mut/Mb, and 5 mut/Mb or less, respectively. 31 By contrast, the recent tissue-agnostic FDA approval for pembrolizumab defined TMB-high as the presence of 10 mut/Mb or more according to the KEYNOTE-158 trial (for advanced solid tumors). 32 A large retrospective study of 1936 patients did not support TMB-high as a biomarker for ICI treatment in all solid cancer types and showed discordant outcomes according to tumor type. Cancer types such as melanoma, lung, and bladder cancer where CD8 T-cell levels positively correlated with neoantigen load responded well to ICI treatment, whereas other cancer types without such a relationship failed to achieve a 20% ORR. Specifically, patients with bladder cancer with TMB-high showed a trend for higher ICI response rates (38% versus 15%) and for a better prognosis. 33
UC is a highly complex tumor with a median mutation number of 9.6 mut/Mb. The phase III IMvigor211 trial, comparing atezolizumab versus chemotherapy after platinum-based chemotherapy in mUC, failed to meet its primary end point of OS benefit with atezolizumab. A post hoc analysis of OS according to TMB of 9.65 mut/Mb showed a numerically improved but nonsignificant OS (median OS 11.3 versus 8.3 months; HR 0.68, 95% CI 0.21–0.9).21,34 In the single-arm phase II CheckMate 275 study, which evaluated nivolumab in patients with mUC, analysis of TMB as a continuous variable showed an association with improved ORR (OR 2.13, 95% CI 1.26–3.60), PFS (HR 0.75, 95% CI 0.61–0.92), and OS (HR 0.73, 95% CI 0.58–0.91). 35 The phase III DANUBE study assessed the efficacy of durvalumab alone or in combination with the cytotoxic T-lymphocyte-associated antigen 4 inhibitor tremelimumab versus chemotherapy for mUC. The study did not meet its primary endpoint of OS benefit in monotherapy patients with high tumor PDL1 expression or combination in the intention-to-treat population. In the combination arm, in patients with high blood TMB (>24 mut/Mb), the 12-month OS rates were 76.7% for the combination therapy and 54.3% for chemotherapy, whereas for blood TMB < 24 mut/Mb, the 12-month OS rates were 53.4% for the combination therapy and 51.2% for chemotherapy. 36 In the Javelin Bladder 100 study validating avelumab maintenance after first-line chemotherapy, TMB was also studied, and its median value (7.66 mut/Mb) was the threshold to define TMB as high. Avelumab almost halved the risk of death (HR 0.48, 95% CI 0.332–0.707) in patients with high TMB and did not improve survival in those with TMB-low. 37 The main limitations of this biomarker are a lack of analytical validation, technique standardization, and threshold definition.
Mismatch repair deficiency
Microsatellite instability (MSI) is a well-known predictive biomarker in colorectal cancer and endometrial cancer: in patients with first-line metastatic colon cancer, the phase III trial (KEYNOTE 177) evaluating pembrolizumab as a single agent achieved a PFS of 16.5 months and objective response rates of 43.8% better than standard first-line therapy (PFS of 8.2 months and response rate of 33.1%). 38 The preliminary analysis of mismatch repair status in UC samples suggests that the MSI phenotype is mainly observed in upper tract UC. A prospective translational study evaluated 424 patients with UC using next-generation sequencing and genomic interrogation of microsatellite sites, revealing that 13 (3%) of patients had an MSI score >10, with a median mutation count of 52 (36.5–73.5) versus 85–13 in non-MSI patients, and 10 (71%) had upper tract cancer. 39 Eight of the nine patients had Lynch syndrome, and one had somatic MSH2 mutations. Two patients with low MSI scores had extremely high TMB (213 and 414), and both had POLE mutations. Five patients with mUC who received ICI therapy achieved near-complete or complete responses. These findings suggest that patients with mUC should be screened for MSI, as they are highly likely to respond strongly to ICI therapy. However, it is important to note that these results were based on a small subset of patients and require further validation. Further research is necessary to confirm the clinical utility of MSI testing in mUC and to identify the most effective treatment strategies for this patient population.
DNA damage response and repair genes
The approval of the defective mismatch repair phenotype pathway for predicting response to ICI has led to investigations into the role of other DNA damage response and repair (DDR) defects in predicting the sensitivity of cancer cells to ICI. 40 DDR comprises four major pathways: homologous recombination, non-homologous end joining, base excision repair, and nucleotide excision repair. 41 In patients with mUC treated with ICI, Teo et al. showed that patients with DDR gene alterations had better clinical outcomes than those with wild-type DDR genes. Of the 60 patients analyzed, 28 (47%) had DDR alterations and 15 (25%) with a deleterious or probably harmful mutation in the DDR. Furthermore, the presence of any alteration in DDR was associated with a higher response rate (68% versus 19%; p = 0.001). A higher response rate was observed in patients with known or probably deleterious DDR alterations (80%) in comparison to those with DDR alterations of unknown significance (54%) and wild-type DDR genes (19%).42,43 To date, no data from randomized trials have supported the hypothesis that DDR genes predict the response to ICI among patients with mUC.
Tumor-infiltrating lymphocytes
The tumor microenvironment (TME) is composed of immune and stromal cells that are exploited by cancer cells to facilitate their proliferation. The application of ICI has rekindled interest in the immune component of the TME due to its potential prognostic and predictive value. In addition, some studies have suggested that clusters of differentiation 8 (CD8) lymphocytes infiltrating TME have antitumor functions, as evidenced by their favorable prognosis in various tumors, including bladder cancer.44,45 Few published articles evaluate tumor-infiltrating lymphocytes (TILs), which is not an easy study because it requires iterative biopsies which can be complicated in metastatic patients (non-accessible site, focus on quality of life). In a small study involving 41 patients with localized bladder cancer who underwent neoadjuvant chemotherapy, TIL densities were measured prior to treatment, along with CD8- and Regulatory T cells (Treg)-specific TILs, and the response was evaluated on cystectomy. While CD8 and Treg TIL density did not have any association with response, the ratio of CD8 to Treg TIL was strongly correlated with response to neoadjuvant chemotherapy. Furthermore, high TIL density was strongly correlated with tumor PD-L1 immunochemistry expression (>5%), consistent with the mechanism of adaptive immune resistance in bladder cancer. 46 In another study involving 212 patients with mUC receiving nivolumab as second-line treatment, high CD8+ T cell infiltration was associated with favorable clinical outcomes. Patients with high CD8+ T-cell infiltration showed an ORR of 25% and a median OS of 11.3 months, while those with low CD8+ T-cell infiltration had an ORR of 11% and a median OS of 5.7 months. 35 The IMvigor210 trial, a phase II trial evaluating atezolizumab in mUC, found that CD8+ T-cell infiltration was higher in patients who responded to atezolizumab. 18 Research has demonstrated that a T-effector signature characterized by high immune transcriptional gene expression (IFNG, CD274, and CXCL9) was associated with better ORR outcomes among patients treated with atezolizumab. In addition, a chemokine gene expression signature (CXCL9, CXCL10, CD8, and 12-chemokine) characterizing the inflamed tumor phenotype was strongly observed in responders to nivolumab. 9 In conclusion, TILs show promise, particularly in terms of the balance between immunosuppression and immune activation, as demonstrated by the ratio of CD8+ T cells to Tregs mentioned above, as well as the ratio of dendritic cells to Tregs, among others. The results of the following meta-analysis support this conclusion. With 33 studies and 2559 cancer patients, the meta-analysis showed that high CD8+ TILs were significantly associated with better outcomes in patients treated with ICIs. Subgroup analyses suggested that patients with high CD8+ TILs had a better clinical benefit, regardless of different treatments, cancer types, and CD8+ T-cell locations. 47
Biological peripheral biomarkers
Inflammatory markers
The anticancer immune response is influenced by the interplay between TME and cancer cells. Systemic inflammation can affect the peritumoral inflammatory environment and is a mechanism of immune resistance in patients with cancer. 48 As a result, several scores using peripheral inflammatory parameters have been developed and studied in patients with genitourinary cancer. These include the Lung Immune Prognostic Index score, which is a composite score based on lactate dehydrogenase and derived neutrophil-to-leukocyte ratio, and neutrophil-to-leukocyte ratio >3.49,50 The c-index was 0.66 (95% CI 0.60–0.71) and 0.67 (95% CI 0.59–0.74) for the Lung Immune Prognostic Index (LIPI) score and Bellmunt score, respectively, in this study. When the LIPI and Bellmunt scores were combined in the same multivariable model, the c-index was 0.70 (95% CI: 0.65–0.76). Another score, the systemic immune-inflammation index, defined by neutrophil * platelet/lymphocyte, is a prognostic biomarker linked to poor outcomes in bladder cancer. This score reflects systemic inflammation in a more balanced way and has a higher predictive value than the platelet-to-lymphocyte ratio or neutrophil-to-lymphocyte ratio (NLR). 51 All of these scores have identified systemic inflammatory status as a negative prognostic factor in mUC. Recent meta-analyses have confirmed the prognostic value of such indices based on pretreatment NLR and lactate dehydrogenase (LDH) in patients with mUC at different stages of the disease.52–54 The C-reactive protein (CRP) is a robust surrogate for local and systemic inflammation. Recently, Klümper et al. observed in a prospective study including 154 patients, a correlation between declination C-reactive protein and ICI response: they defined CRP flare responders (at least doubling of baseline CRP within the first month after initiation of ICI followed by a decline below baseline within 3 months), CRP responders (decline in baseline CRP by 30% within 3 months without a prior flare), and the remaining patients as CRP non-responders. Objective response was observed in 57.1% of CRP responders, 45.8% of CRP flare responders, and 17.9% of CRP non-responders (p < 0.001). CRP flare response was associated with prolonged PFS and OS (p < 0.001). 55
However, these scores evaluated in the chemotherapy or ICI cohorts can currently only be used as prognostic biomarkers.
Circulating tumor DNA
Circulating tumor DNA (ctDNA) is an interesting biomarker that has primarily been developed for colorectal and breast cancers and has shown promising clinical implications. 56 In UC, the predictive value of ctDNA was evaluated in a post hoc analysis of the IMvigor010 trial. This randomized phase III trial compared adjuvant atezolizumab with observation after radical cystectomy for resectable urothelial cancer. 57 Among the 214 patients (37%) who had detectable ctDNA after radical cystectomy, atezolizumab improved disease-free survival (DFS) and OS compared to the observation arm (HR 0.58, 95% CI 0.43–0.79 and 0.59, 95% CI 0.41–0.86). However, no difference in DFS and OS between the treatment arms was observed in patients with negative ctDNA. In perspective, adjuvant immunotherapy may be restricted to patients with positive ctDNA and deescalated in patients with negative ctDNA, thus avoiding potentially unnecessary exposure of patients to ICI toxicity. In addition, this study suggests that ctDNA could potentially serve as a useful tool for monitoring response to immunotherapy, but further validation is required in a randomized trial.
Prognostic scores
Prognostic models have been developed that incorporate clinical and inflammatory biological parameters to assess prognosis. Clinical factors such as pretreatment Eastern Cooperative Oncology Group performance status (ECOG PS) and pretreatment liver metastases were integrated into these models as well-known prognostic factors. Sonpavde et al. proposed a model developed as a prognostic model in 405 chemotherapy-pretreated patients with mUC based on ECOG PS, liver metastasis, platelet count, NLR (cutoff 5), and LDH. 58 This model demonstrated good prognostic discrimination with a c-index of 0.692 which compared with 0.635 for the Bellmunt model. Another risk score was developed by Khaki et al. for mUC patients treated with first-line ICIs. The score assigned one point for ECOG PS ⩾2, albumin <3.5 g/dL, NLR > 5, and liver metastases each one point, with a higher score indicating worse OS. 59 Bamias et al. developed a prognosis model in 936 patients receiving atezolizumab in the SAUL trial. They identified additional factors that could improve the Bellmunt score based on hemoglobin, ECOG PS, and liver metastasis. These factors are alkaline phosphatase, NLR, bone metastases, and time from the last chemotherapy. Compared with the Bellmunt score, this model provided enhanced prognostic separation, with a c-index of 0.725 versus 0.685. 60 All these scores were developed to provide better insight into the prognosis of patients receiving immunotherapy.
Microbiota
Gut microbiota plays a crucial role in regulating immune function and may have a negative impact on systemic immune responses and ICI efficacy. 61 Recently, prospective studies have demonstrated that microbiome diversity and composition in patients with metastatic melanoma and non-small-cell lung cancer receiving ICI are, in part, predictive of treatment response.62,63 Exposure to antibiotics may compromise the effectiveness of immune checkpoint blockade of ICI in routine clinical practice by disrupting the ecological balance of the microbiome, which is essential for immune activation. A meta-analysis explored the impact of antibiotics on OS and PFS in patients with solid malignancies treated with ICI. The analysis included 2889 patients, of whom 16% had UC. OS and PFS were longer in those who did not receive antibiotics (pooled HR 1.92, 95% CI 1.37–2.68 and 1.65, 95% CI 1.3–2.1). 64 A post hoc analysis of the IMvigor210 and 211 trials showed that antibiotic use among patients receiving atezolizumab was significantly associated with worse OS (HR = 1.44, 95% CI 1.19–1.73) and PFS (HR = 1.24, 95% CI 1.05–1.46), in contrast to patients treated with chemotherapy. 65 The use of proton pump inhibitors may have an impact on the survival outcomes of patients treated with atezolizumab, as opposed to chemotherapy. This is probably due to the significant changes in the gut microbiota induced by proton pump inhibitors, both through altering stomach acidity and direct compound effects. Studies have shown that proton pump inhibitors can decrease the alpha diversity of the gut microbiota and increase the relative abundance of Actinomycetales, Micrococcaceae, Enterobacteriaceae, and Streptococcaceae. 66
Insufficient data are available regarding the urine microbiome and response to ICI. However, preliminary findings have indicated differences in the urinary microbiota between patients with bladder cancer (n = 29) and those without cancer (n = 26). Specifically, patients with bladder cancer had a higher abundance of Actinomyces, while other strains were more enriched in the control group, particularly Actinomyces European. 67
Conclusion and future perspectives
The field of immunotherapy in UC is rapidly evolving, with the identification of various biomarkers that hold promise for predicting responses to ICIs. Although molecular subtype categorization, TMB, immune gene and stromal signatures, and gut and urinary microbiota may prove useful in predicting response to immunotherapy, they are not yet widely available. On the other hand, PD-L1 expression is routinely measured, but its predictive value in patients with UC is limited. The strengths and limitations of the biomarkers discussed in this review are outlined in Table 2. This review discusses each biomarker separately, but it is important to recognize that these biomarkers interact with one another as part of a delicate balance between proinflammatory antitumor immune response and the tumor immune evasion mechanisms that counteract these responses. Subsequently, using a composite algorithm that combines these biomarkers, rather than relying on a single biomarker, may lead to improved prediction of the response to ICI. The emerging field of artificial intelligence offers a promising strategy for comprehensively analyzing datasets combining radiomic and molecular features, to provide a comprehensive approach to this topic. Prospectively validated prognostic and predictive biomarkers will be valuable adjuncts to clinical and pathological data. However, validation trials with extended follow-up periods will be needed to resolve the numerous unanswered questions.
Strengths and limitations for each biomarker discussed in this review.
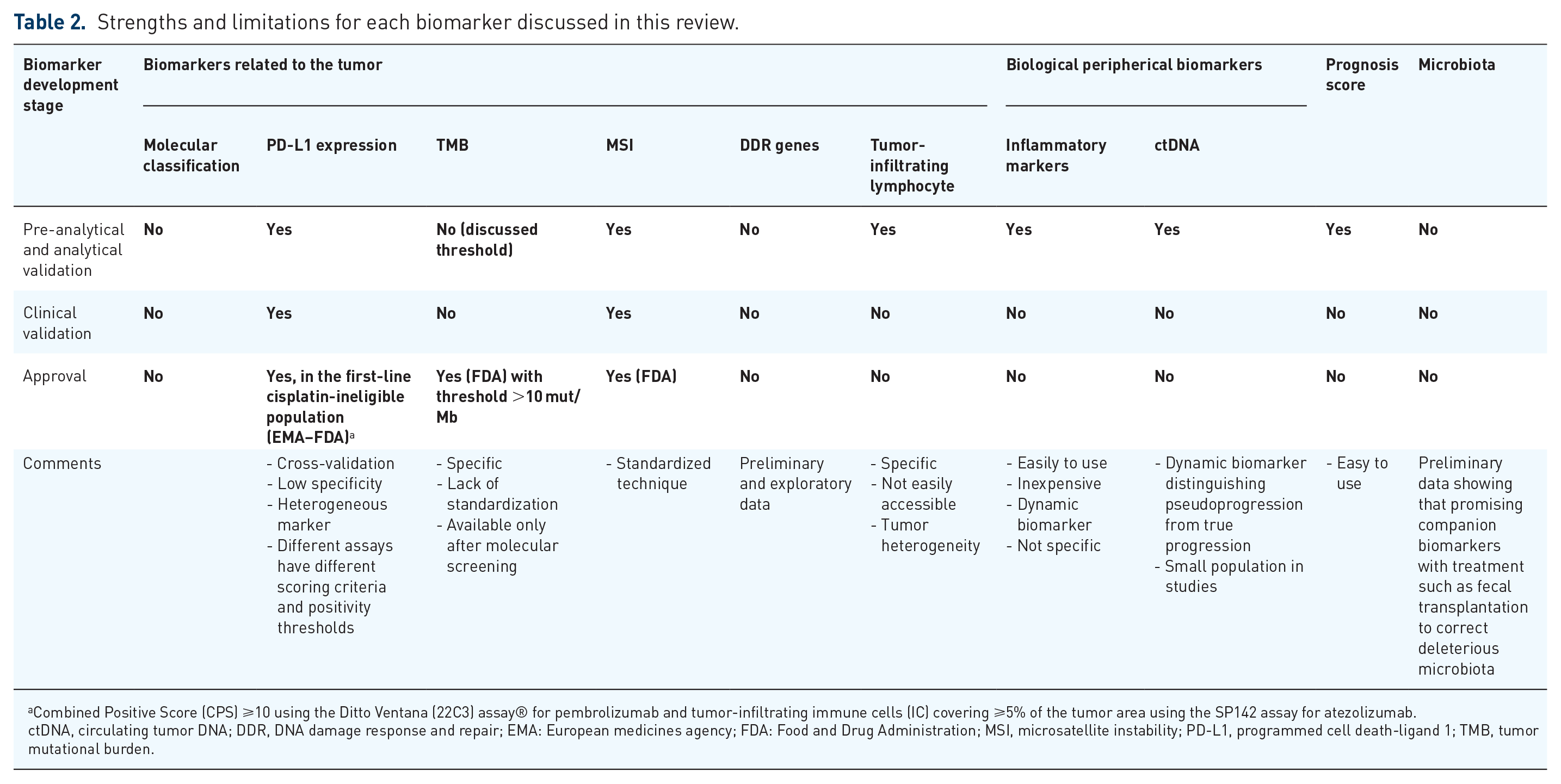
Combined Positive Score (CPS) ⩾10 using the Ditto Ventana (22C3) assay® for pembrolizumab and tumor-infiltrating immune cells (IC) covering ⩾5% of the tumor area using the SP142 assay for atezolizumab.
ctDNA, circulating tumor DNA; DDR, DNA damage response and repair; EMA: European medicines agency; FDA: Food and Drug Administration; MSI, microsatellite instability; PD-L1, programmed cell death-ligand 1; TMB, tumor mutational burden.
In conclusion, while significant progress has been made in identifying potential biomarkers for predicting response to immunotherapy in UC, further research and validation are needed to translate these findings into clinical practice. Continued efforts in this field will enhance our understanding and management of UC, ultimately leading to improved patient outcomes.
