Abstract
BACKGROUND:
Long non-coding RNA (LncRNA) TUG1 plays a critical role in the development of human cancers. This study explored whether TUG1 is involved in the cytotoxicity of dendritic cells and cytokine-induced killer cells (DCs-CIK), an immunotherapy approach, in neuroblastoma.
METHODS:
A TUG1 expression plasmid was transfected into DCs. Neuroblastoma SK-N-SH cells were incubated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation. SK-N-SH cell viability, colony formation, migration, and apoptosis were analyzed using CCK-8, colony formation assay, transwell assay, and flow cytometry, respectively. Production of IL-12, IL-2 and IFN-
RESULTS:
Compared to CIK alone or DC-CIK therapy, overexpression of TUG1 significantly suppressed tumor cell proliferation, colony formation, and migration of neuroblastoma cells. Moreover, upregulation of TUG1 robustly induced apoptosis and altered key molecules associated with apoptosis and epithelial-mesenchymal transition. Contents of IL-12, IL-2 and IFN-
CONCLUSION:
Overexpression of TUG1 promotes DC maturation and enhances CIK cytotoxicity, suggesting that TUG1 may be a novel target for enhancing DC-CIK based immunotherapy for neuroblastoma.
Introduction
Neuroblastoma is one of the most common cancers during childhood. The incidence of neuroblastoma has increased rapidly in the past 20 years and the 5-year survival rate remains dismal [1, 2]. Current available therapeutic modalities for this disease include surgery, radiotherapy, and chemotherapy. Given the unsatisfactory outcomes of these treatment regimens, a more effective and safer approach is urgently required for neuroblastoma.
In recent years, immunotherapy is considered as a promising approach in addition to surgery, radiotherapy, and chemotherapy [3]. Various immune agents have been developed for cancer immunotherapy, such as checkpoint inhibitors and kinase inhibitors [4]. However, their applications have been confined to patients carrying specific antigen-expressing cells. Notably, many researchers have paid much attention on the cellular immunotherapy by utilizing dendritic cells (DCs), natural killer (NK) cells, and cytokine-induced killer (CIK) cells [5].
Amounting evidences indicate the unique property of CIK cells in cancer immunotherapy, such as higher proliferation capacity, stronger antitumor activity, and broader antitumor spectrum. The tumoricidal ability of CIK cells is induced by direct contact and secretion of cytokines such as IL-2 and IFN-
Long non-coding RNAs (lncRNAs) are a group of non-protein coding RNAs with lengths longer than 200 nucleotides. Growing evidences have indicated that lncRNAs are key regulators in physiological and pathological events [10]. LncRNA taurine upregulated gene 1 (TUG1) is recognized to be a key player in the initiation and progression of human cancers [11]. In addition, TUG1 is actively involved in tumor malignant behaviors and acquired drug resistance to chemotherapy in human cancers [12, 13, 14]. However, the role of TUG1 in the efficacy of cellular immunotherapy based on DCs-CIK cells has not been explored. In the present study, we investigated the potential value of TUG1 in DC-CIK cell immunotherapy both in vitro and in vivo.
Materials and methods
Ethics
The study was approved by the Ethics Committee of the local Hospital and conducted in accordance with the Declaration of Helsinki. Peripheral blood was donated by healthy volunteers, and informed consent from all volunteers was obtained. Animals were housed in a pathogen-free (SPF) condition with free access to food and water.
Cell culture
Human neuroblastoma SK-N-SH cells were obtained from the Shanghai Institute of Biological Sciences, Chinese Academy of Sciences (Shanghai, China). Cells were cultured in RPMI-1640 (Gibco, Grand Island, NY, USA) containing 10% FBS, 100
Preparation of DCs and CIK cells
For preparation of DCs, the monocytes were purified from the PBMCs using CD14 microbeads (Miltenyi, Germany), cultured in RPMI-1640 culture medium supplemented with 10% FBS, 1000 U/ml human rGM-CSF (PeproTech, USA), and 1000 U/ml human rIL-4 (PeproTech, USA). For preparation of CIK cells, PBMCs were cultured in RPMI-1640 culture medium supplemented with 10% FBS, 1000 U/ml IFN-
Cell viability assay
The cell viability was determined using CCK-8 assay according to the manufacturer’s instruction. In brief, neuroblastoma SK-N-SH cells were seeded onto 96-well plates, incubated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation. After removing DCs-CIK cells with PBS, the cell viability of SK-N-SH cells in each well was determined by adding 10
Colony formation assay
Neuroblastoma SK-N-SH cells were incubated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation. Cells were seeded on 60 mm dish and cultured for two weeks. The colonies were stained with crystal violet (0.1%) and the number of surviving colonies was counted.
Transwell cell migration assay
Neuroblastoma SK-N-SH cells (2
Cell apoptosis
Neuroblastoma SK-N-SH cell apoptosis was analyzed using annexin V/PI apoptosis kit (Invitrogen, USA) according to the manufacturer’s instruction. In brief, cells were seeded in a 6-well plate and incubated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation. The cells from each group were washed with ice-cold PBS, and were resuspended in 100
Luciferase reporter assay
Neuroblastoma SK-N-SH cells were seeded in triplicate in 24-well plates and incubated overnight. The wild type 3’-UTR of TUG1 was cloned and amplified. Mutation in 3’-UTR of TUG1 gene with miR-204 putative target binding site was generated with the QuickChange Site-Directed Mutagenesis kit (Stratagene, CA, USA). The wild and mutant TUG1 sequences were cloned into the pGL-3-vector (Promega, Wisconsin, USA) downstream of the firefly luciferase gene. Cells were co-transfected with luciferase reporter plasmids, firefly luciferase reporter, and miR-204 mimics or miR-204 negative control using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). Then, firefly luciferase activities were measured by the Dual-Luciferase Reporter Assay System Kit (Promega, Wisconsin, USA).
Enzyme-linked immunosorbent assay (ELISA)
The contents of IL-12, IL-2 and IFN-
Western blot analysis
Neuroblastoma SK-N-SH cells were harvested in ice-cold PBS, and the protein concentration was determined using the BCA Protein Assay Kit. Equal amounts of protein were subject to SDS-PAGE, transferred onto nitrocellulose membranes (Millipore, Wisconsin, USA), and blocked with 5% non-fat dry milk for 2 h. Then, the membranes were incubated at 4
Xenograft mouse
Animal xenograft model studies were performed according to institutional guidelines. Six-week-old male athymic BALB/c nude mice were maintained in special pathogen-free (SPF) conditions, Prior to the experiment, 4
Statistical analysis
Statistical analysis was carried out using SPSS 15.0 software. Data are expressed as mean
Results
Overexpression of TUG1 in DC enhances CIK cytotoxicity in neuroblastoma
Overexpression of TUG1 in DCs enhances CIK cytotoxicity in SK-N-SH cells. (A) Detection of CD80, MHC-II, and CD11C expressions in DCs using flow cytometry. (B) CCK-8 assay was used to detect the cell viability in SK-N-SH cells treated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation. (C) Detection of colony formation in SK-N-SH cells treated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation. (D) Migration capability in SK-N-SH cells treated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation. 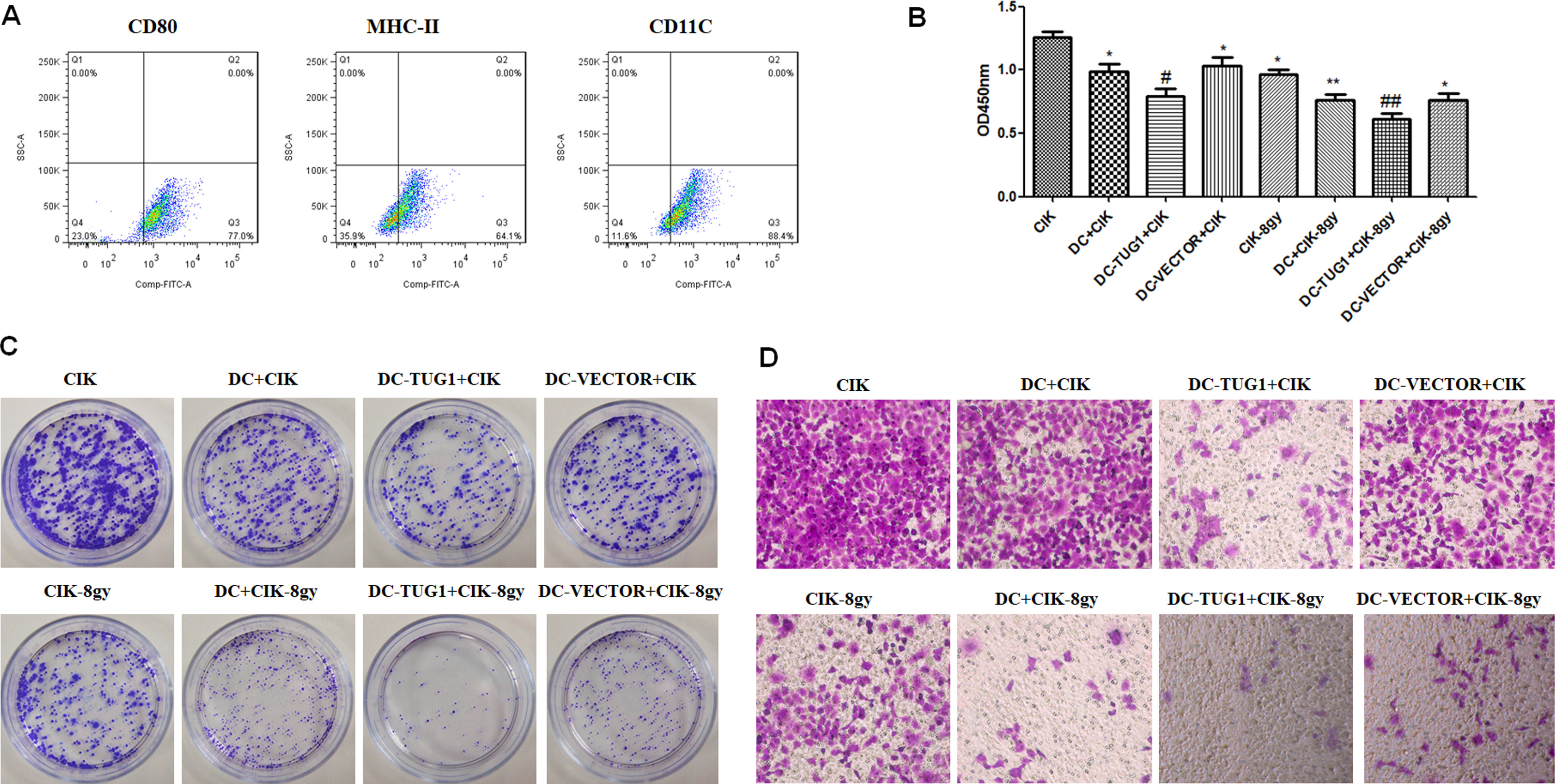
Firstly, monocytes were purified from the PBMCs, stimulated with human rGM-CSF and human rIL-4, and identified by examining the expression of CD80, MHC-II and CD11c (Fig. 1A). In order to evaluate the immunotherapy of DC-CIK, human neuroblastoma SK-N-SH cells were co-cultured with CIK alone or DC-loaded CIK cells. As a result, SK-N-SH cells showed decreased cell viability in DC-CIK group compared to CIK group. In addition, overexpression of lncRNA TUG1 in DC further decreased SK-N-SH cell viability, as compared to DC-CIK treated tumor cells (Fig. 1B,
We then explored whether TUG1 could induce apoptosis in neuroblastoma exposed to the DC-CIK immunotherapy. Flow cytometry analysis showed that combined treated with DC and CIK cells dramatically induced apoptosis in neuroblastoma cells; and overexpression of TUG1 in DC further potentiated DC-CIK induced apoptosis (Fig. 2,
Overexpression of TUG1 in DCs induces apoptosis of CIK-loaded SK-N-SH cells. Flow cytometry was performed to detect the apoptosis in SK-N-SH cells treated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation.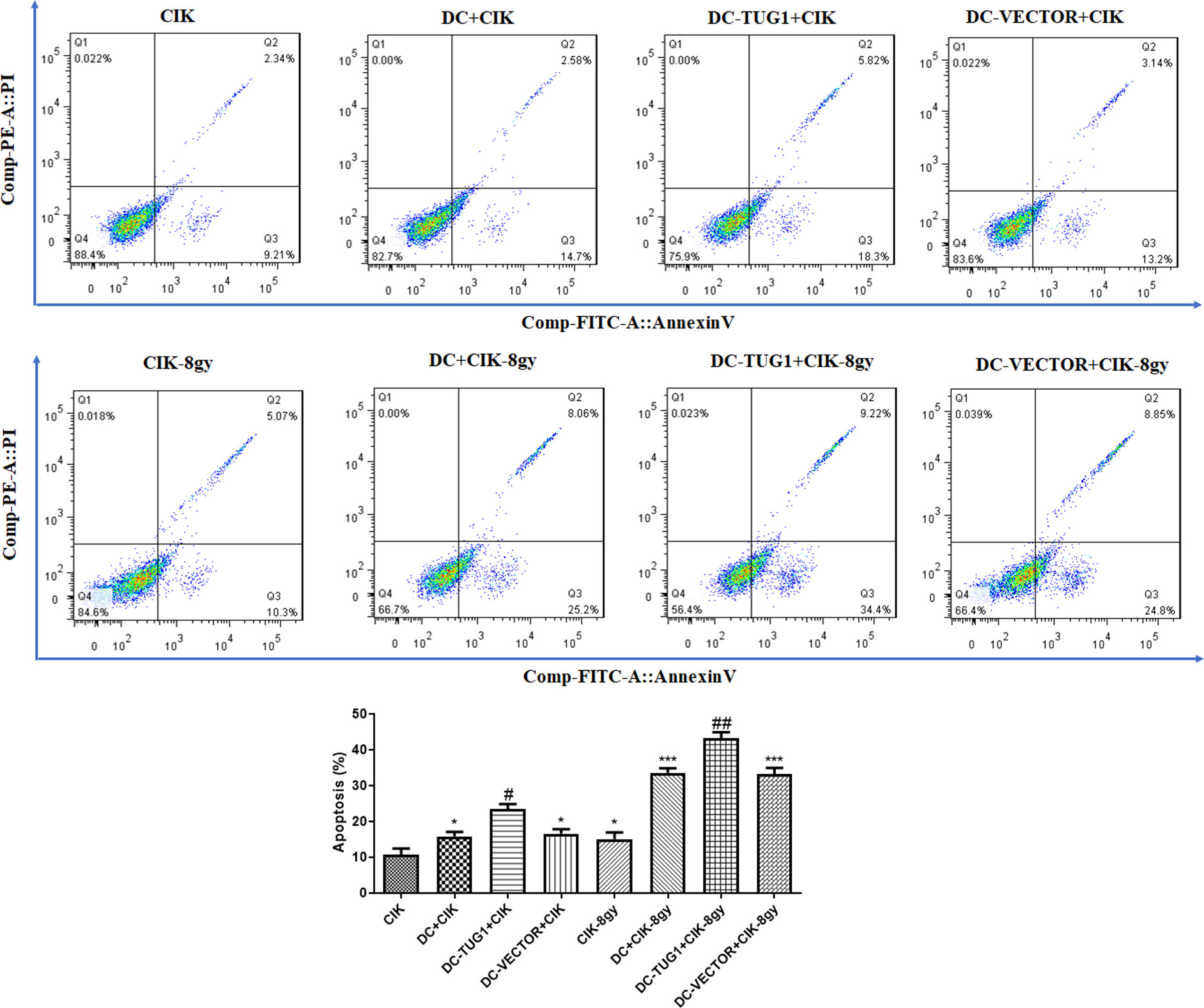
Given that TUG1 could regulate apoptosis and migration of SK-N-SH cells, we asked whether overexpression of TUG1 alters the molecular events in neuroblastoma under immunotherapy. As shown in figure, western blot analysis suggested that combined immunotherapy with DC-CIK cells obviously promoted the protein expression of activated-caspase-3, and inhibited the anti-apoptotic Bcl-2, in SK-N-SH cells with or without irradiation therapy. Particularly, overexpression of TUG1 in DC further increased activated-caspase-3 and decreased Bcl-2, suggesting the regulatory role of TUG1 on apoptosis-related molecules in neuroblastoma (Fig. 3). In addition, we detected levels of molecular events involved migration by western blot. We found that DC-CIK immunotherapy promoted E-cadherin, an epithelial marker, and inhibited the mesenchymal marker N-cadherin, in SK-N-SH cells with or without irradiation treatment. Importantly, overexpression of TUG1 further promoted the protein expression of E-cadherin, and inhibited N-cadherin (Fig. 3), suggesting that TUG1 alters the molecular events associated with migration in neuroblastoma.
Effects of TUG1 on the protein expression of key factors in SK-N-SH cells. Western blot was performed to detect the apoptosis-related proteins (activated-caspase-3 and Bcl-2) and migration-associated factors (E-cadherin and N-cadherin) in SK-N-SH cells treated with CIK cells, DCs-CIK cells, and TUG1-overexpressing DCs-CIK cells, with or without irradiation.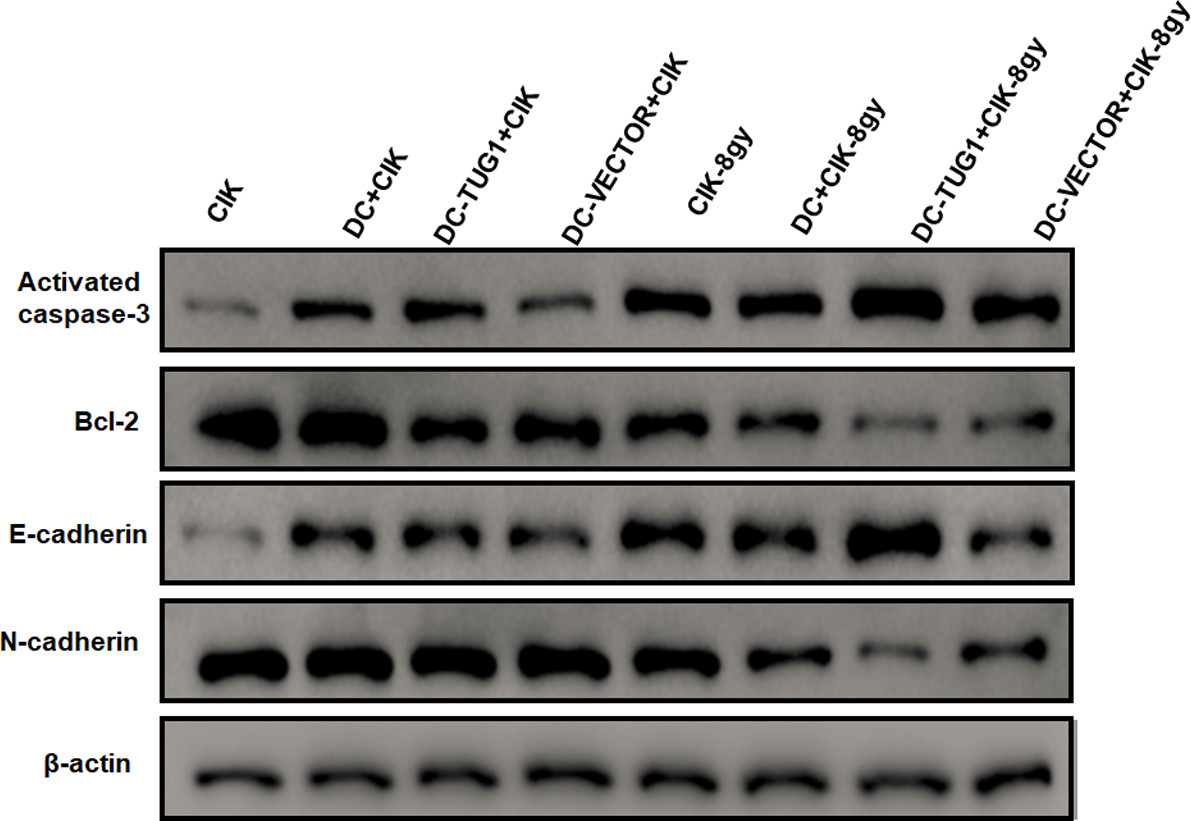
To test the hypothesis that TUG1 acts as a ceRNA, we searched for potential miRNA that could interact with TUG1. We found that TUG1 sequence had miR-204 binding sites (Fig. 4A). To verify this prediction, we generated wild type (Wt) TUG1 luciferase plasmids containing potential miR-204 binding sites, as well as mutant variants of each site. Luciferase assays verified that transfection with Wt-TUG1 and miR-204 mimic significantly suppressed the luciferase activity, as compared to Wt-TUG1+miR-204 negative control-transfected cells. However, there were no obvious differences of the luciferase activity between mutant TUG1+miR-204 mimic and mutant TUG1+ miR-204 negative control groups (Fig. 4B). Consistently, luciferase activity assay also revealed that TUG1 targeted miR-204 in DCs (Fig. 4C). Moreover, overexpression of TUG1 significantly suppressed the expression of miR-204 in DCs (Fig. 4D). Collectively, these data suggest that miR-204 could serve as a target of lncRNA TUG1.
Overexpression of TUG1 in DC promotes cytokine production by targeting miR-204
Given that TUG1 could regulate the cytotoxicity of CIK cells, we asked whether overexpression of TUG1 could regulate the production of cytokines, such as IL-12, IL-2 and IFN-
TUG1 targets miR-204 and regulates cytokine production. (A) Bioinformatics analysis of potential lncRNA TUG1/miRNA interactions. (B) A dual luciferase reporter plasmid containing TUG1-Wt or TUG1-Mut was co-transfected into SK-N-SH cells along with miR-204 mimics or miR-204 NC, and luciferase activities were determined. (C) Luciferase activity assay was performed to explore the relationship between TUG1 and miR-204 in DCs. (D) The expression level of miR-204 was determined in DC overexpressing TUG1. ELISA was used to detect the production of cytokines including IL-12 (E), IL-2 (F) and IFN-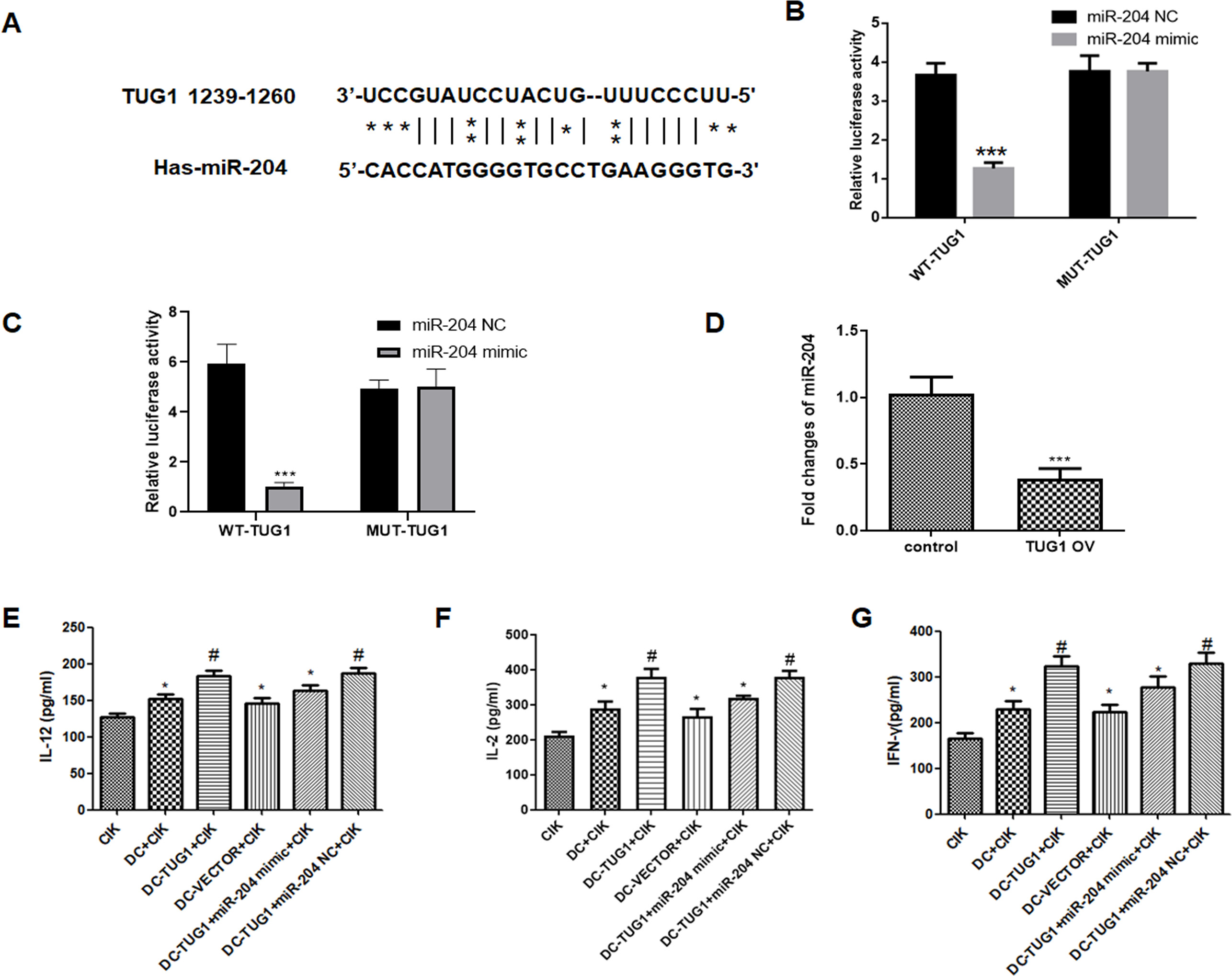
In order to evaluate the therapeutic efficacy of TUG1 in vivo, we introduced CIK cells or TUG1-loaded DCs and CIK cells into nude mice bearing neuroblastoma followed by irradiation at 8 Gy. Compared to CIK group, combined immunotherapy with DC+CIK cells significantly decreased the average tumor volume. Furthermore, overexpression of TUG1 dramatically suppressed the tumor growth compared to negative-vector loaded DCs (Fig. 5A and B). Additionally, western blot analysis indicated that combined immunotherapy with DC-CIK cells obviously promoted the protein expression of activated-caspase-3, and inhibited the anti-apoptotic Bcl-2, in tumor tissues. Particularly, overexpression of TUG1 further increased activated-caspase-3 and decreased Bcl-2, suggesting the apoptotic role TUG1 in neuroblastoma (Fig. 5C). Collectively, these data suggest that TUG1-loaded DCs could enhance the therapeutic efficacy of CIK in vivo.
TUG1-loaded DCs enhance the therapeutic efficacy of CIK in vivo. A mouse model bearing neuroblastoma was established using SK-N-SH cells. The average tumor volume in different group was determined (A and B). Tumor tissues from each group were further subjected to western blot assay (C). 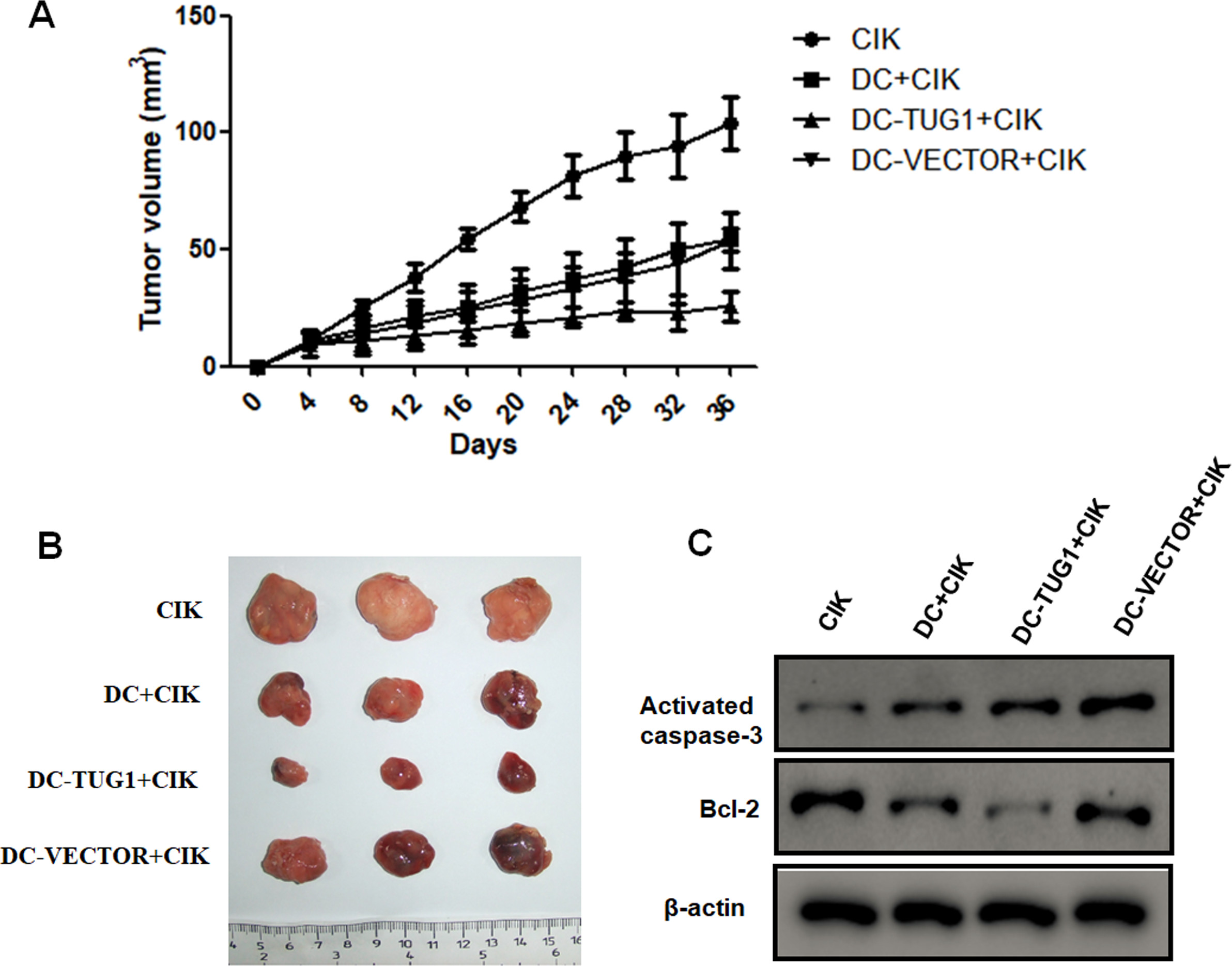
Neuroblastoma is one of the most frequent gastrointestinal malignancies with no effective treatment approaches. In addition to the traditional surgery, radiotherapy and chemotherapy, cellular immunotherapy is considered as an effective approach [15]. Recent studies have revealed that lncRNAs are actively involved in tumorigenesis, progression and acquired resistance to chemo/radiotherapy, representing novel biomarkers and therapeutic targets for human malignancies [16]. In the present study, we investigated the role of lncRNA TUG1 in DC-CIK based cellular immunotherapy for human neuroblastoma. Our in vitro and in vivo data suggest that upregulation of TUG1 could enhance the efficacy of DC-CIK immunotherapy in neuroblastoma.
Currently, CIK therapy or DC-CIK cell co-culture has been extensive applied to treat human malignant tumors, including gastric cancer, hepatocellular carcinoma, and neuroblastoma [7]. The therapeutic efficacy and cytotoxic effects of CIK or DC-CIK immunotherapy against tumor progression have been demonstrated both in vitro and in vivo[17, 18]. Many studies have suggested that the proliferation and antitumor activity of CIK cells are dramatically potentiated following coculture with DCs. For instance, the cytotoxic effect of DC-CIK co-culture system exhibited a much stronger antitumor activity than that of CIK cells alone on various human malignancies, including leukemia, cervical cancer, and liver cancer [19]. In addition, coculturing with the DCs significantly promoted CIK proliferation, differentiation, and cytotoxicity. On the molecular level, DC-CIK based immunotherapy dramatically induced tumor cell apoptosis, by inhibiting proliferating cell nuclear antigen (PCNA) and promoting caspase-3 protein expression [5].
In the present study, combination with DC-CIK based immunotherapy exhibited a much stronger cytotoxicity against neuroblastoma cells with or without irradiation, as evidenced by decreased cell proliferation and colony number. Moreover, coculturing with DCs and CIK cells obviously suppressed the migration capability by elevating E-cadherin and suppressing N-cadherin, and induced a robust apoptosis by increasing activated-caspase-3 and decreasing Bcl-2 levels. LncRNA TUG1 has been recognized as a key player in human cancers. Accumulating evidences have shown that TUG1 is closely associated with cell proliferation, differentiation, and apoptosis by altering its downstream targets, such as Nrf2, KLF2, miR-144, miR-197, etc. [20, 21, 22, 23]. In the current study, overexpression of TUG1 in DCs dramatically enhanced the antitumor activity of CIK cells on human neuroblastoma cells. Furthermore, upregulation of significantly enhanced the levels of IL-12, IL-2 and IFN-
It has reported the critical role of TUG1/miRNA interactions in tumor initiation and progression [11]. Thus, we searched for potential binding sites between miRNA and TUG1, and miR-204 was found to be targeted by TUG1, which was further validated by dual luciferase activity assay, suggesting that TUG1 serves as miR-204 sponge to mediate cellular immunotherapy. Several studies have demonstrated that miR-204 plays a critical role during the pathogenesis of neuroblastoma [26, 27]. Particularly, miR-204 has been reported to transcriptionally regulate different targets, such as MYCN and PHOX2B, in neuroblastoma tumorigenesis [28, 29]. A recent study has verified a mechanism in which miR-204 regulated dendritic cells and played a critical role in thyroid carcinoma [30]. Indeed, combination with DC-CIK cells enhanced the levels of IL-12, IL-2 and IFN-
In conclusion, both in vitro and in vivo assays demonstrate that overexpression of TUG1 contributes to DC maturation and enhances CIK cytotoxicity against neuroblastoma. These data suggest that TUG1 may be a novel target for enhancing DC-CIK based immunotherapy for neuroblastoma.
Author contributions
Conception: Wei-Qiang Tan.
Interpretation or analysis of data: Li Yuan, Xu Cao.
Preparation of the manuscript: Xiao-Yan Wu.
Revision for important intellectual content: Yi-Qun Xing.
Supervision: Ming Ye.
Funding
The study was supported by Natural Science Foundation of Jiangsu Province of China (No: BK20191176); Gusu Health Talent Project of Suzhou City (No: GSWS2019052, GSW2020049).
