Abstract
BACKGROUND:
Ovarian clear cell carcinomas (OCCCs) have been recurrent and refractory among the present treatments, so novel therapeutics are urgently needed.
OBJECTIVE:
The present study accumulates the proof of concept to examine the feasibility of RDH10 as a therapeutic target for treating OCCCs.
METHODS:
Immunohistochemically, RDH10 expression was evaluated in 111 primary epithelial ovarian cancers, including 55 OCCCs, 31 ovarian endometrioid carcinomas and 25 ovarian serous carcinomas. The spherogenecity provoked by RDH10 was evaluated in OCCC cells. To analyze whether RDH10 promotes carbohydrate storage via the vitamin A-gluconeogenesis pathway, phosphoenolpyruvate carboxykinase 1 (PCK1) protein levels and intracellular carbohydrate content were measured in response to modified RDH10 expression.
RESULTS:
Abundant RDH10 was expressed specifically in OCCCs. RDH10 promoted spherogenecity and intracellular carbohydrate storage via modulation of PCK1 expression in OCCC cells.
CONCLUSIONS:
In the present study, abundant RDH10 contributed to cancer cell stemness and intracellular carbohydrate storage in OCCCs. RDH10 is a potentially, new therapeutic candidate for treating OCCC cases.
Introduction
Ovarian clear cell carcinomas (OCCCs) are the second most common epithelial ovarian cancer (EOC) in Japan and East Asia [1, 2]. Frequent recurrence, even after complete reduction with surgical and chemotherapeutic treatments, and a poor response to anticancer agents worsen the prognosis for patients with OCCC [3]. Advanced or recurrent OCCC cases have extremely poor prognoses [3, 4]. OCCC, which arises mostly from endometriosis, exhibits unique pathological features, including abundant cytoplasmic carbohydrate stores in the cancer cells [5]. Regardless of the characteristic findings of OCCCs, the current standard treatment is similar to the treatment for other EOCs, i.e., platinum- and taxane-based chemotherapy. Unfortunately, the therapeutic efficacy is quite insufficient [5]. To overcome the therapeutic insufficiency associated with OCCCs, novel therapeutics are urgently needed, especially for recurrent or refractory OCCC cases.
Cancer stem-like cells (CSCs) are associated with the recurrence and metastasis of malignant tumors [6]. CSCs can survive under the severe oxidative stress induced by radiation therapy or chemotherapy. In OCCCs, molecules regulating the growth and spread of CSCs are not fully understood. CSCs are capable of self-renewal and multilineage differentiation, resulting in tumor initiation, growth, and treatment resistance [7]. The growth of CSCs in OCCC may be associated with both aldehyde dehydrogenase 1 expression and abundant Nrf2-induced antioxidant scavengers, which lower reactive oxygen species relevant to chemoresistance [8].
Through RNA-seq transcriptional analyses of the RXRB (retinoid X receptor beta) axis, we recently demonstrated that retinol dehydrogenase 10 (RDH10), which modulates vitamin A metabolism and gluconeogenesis, affects cancer stemness in OCCCs [9]. In growing cancer spheres, RDH10 was significantly upregulated in the presence or absence of RAB39A (a member of the RAS oncogene family) [9]. RDH10 is involved in retinoic acid (RA), vitamin A, and carotenoid metabolism. RDH10 is required for RA synthesis and contributes to nuclear RXRB function [10, 11]. Insulin regulates RDH expression and RDH subsequently converts retinol to all-trans RA (ATRA), which activates phosphoenolpyruvate carboxykinase 1 (PCK1), resulting in gluconeogenesis [10, 12]. RDH10 upregulation results in the continuous activation of nuclear RXRB, which modulates cancer stemness and increases carbohydrate storage typical in OCCCs. Our preliminary study indicated that OCCC cell lines exhibit elevated RDH10 relative to other types of ovarian cancer cells [9]. Therefore, RDH10 indicates cancer stemness and serves as a novel diagnostic and/or therapeutic target in OCCCs. The objective of the present study was to determine the role of RDH10 in cancer stemness and intracellular carbohydrate storage in OCCCs, and to examine the feasibility of RDH10 as a therapeutic target for treating OCCCs.
Materials and methods
Patient specimens
One hundred eleven histopathological specimens of EOC collected at Shiga University of Medical Science between 1998 and 2017 were analyzed. Patient specimens included 55 OCCCs, 31 endometrial carcinomas (ECs), and 25 serous carcinomas (SCs). This study was carried out in compliance with the Declaration of Helsinki and was approved by the Shiga University of Medical Science Research Ethics Committee (reference number: 29–178).
Immunohistochemistry
Immunohistochemical staining of RDH10 was performed on 5-
Cell lines
Two OCCC cell lines, ES-2 (#CRL-1978) and TOV-21G (#CRL-11730), were obtained from the American Type Culture Collection. The cell lines were cultured in Dulbecco’s modified Eagle’s medium with 10% fetal bovine serum, penicillin (50 units/mL), and streptomycin (50 mg/mL) at 37
Knockdown and overexpression of RDH10
The lentiviral shRNA vector for RDH10 (shRDH) and the non-silencing control vector (shCntl) were purchased from Dharmacon (Horizon Discovery Ltd., UK). The lentiviral cDNA vector for RDH10 (RDH10) was obtained from Applied Biological Materials Inc. (Richmond, British Columbia, Canada). A similar construct of pLenti-GIII-CMV-RFP-2A-puro vector without any gene inserts was used as the overexpression control (Cntl). Lentiviral High Titer Packaging Mix (Clontech, Takara Bio, Japan) was used to prepare lentiviral transferring shRNA and cDNA according to the product manual. ES-2 and TOV-21G cells were transfected with each lentivirus at
Sphere formation culture
ES-2 and TOV-21G with or without lentiviral transfection were seeded in non-adhesive 6-well plates (Coster
Western blot analysis
Cells were lysed in Laemmli-sodium dodecyl sulfate (SDS) buffer with protease inhibitors. Proteins were separated by SDS-polyamide gel electrophoresis and transferred to membrane filters (Trans-Blot Turbo 0.2 micro PVDF Membrane, Bio-Rad Laboratories, Richmond, CA). After blocking with 3% skimmed milk for 60 min, the filters were incubated with primary antibodies for RDH10 (diluted 1:200, #14644-1-AP; Proteintech, IL), PCK1 (diluted 1:200, #sc-271204, Santa Cruz Biotechnology, Dallas, TX), or alpha-tubulin (diluted 1:10,000, #T9026, DM1A, Sigma-Aldrich, MO) in TBS-T containing 1.5% skimmed milk for 120 min. Membranes were then incubated with anti-rabbit or anti-mouse secondary antibodies (Cell Signaling Technology, Danvers, Massachusetts) in TBS-T containing 1.5% skimmed milk for 60 min. Immunoreactivity was detected by ECL SelectTM Western Blotting Detection Reagent (AmershamTM, Cytiva, Tokyo, Japan) and an LAS 4000 bioimage analyzer (Fujifilm, Tokyo, Japan).
Intracellular carbohydrate storage analysis
ES-2 and TOV-21G with or without lentiviral transfection were seeded in chamber slides. After fixing with 10% buffered formalin, periodic acid-Schiff (PAS) staining was performed. To evaluate the intracellular carbohydrate storage, only the red-colored pixel images of the PAS-stained slides were analyzed using Image J (NIH Image, Bethesda, MD) [13].
To examine PCK1 effectiveness for intracellular carbohydrate storage via RDH10-PCK1 pathway, the seeded ES-2 variants were treated with 25
Statistical analysis
Statistical analysis was performed using GraphPad Prism (ver.6.07 for Windows, GraphPad Software, San Diego, CA). RDH10 expression in the histological types was compared using Chi-square tests. Other results were analyzed with Mann-Whitney U or Kruskal-Wallis and Dunnett’s tests for comparison of two or multiple groups, respectively. A
Results
RDH10 is highly expressed in OCCCs
To determine RDH10 abundance in OCCCs rather than other types of EOCs, RDH10 expression was semi-quantitatively evaluated in 111 primary EOCs (55 OCCCs, 31 ovarian ECs, and 25 ovarian SCs). As shown in Fig. 1, significantly more OCCC specimens exhibited highly immunohistochemical staining for RDH10, which were categorized as “high” expression, compared with staining in EC or SC specimens (Fig. 1A). Cases with stronger RDH10 staining of the tumor cells than that of normal ovarian stromal cells were categorized as high RDH10 cases. High RDH10 staining cases were detected in 96.4% (53/55), 45.2% (14/31), and 4.0% (1/25) of OCCCs, ovarian ECs, and ovarian SCs, respectively (Fig. 1B;
Abundant expression of RDH10 in ovarian clear cell carcinomas. A, Representative immunohistochemical images of RDH10 in ovarian cancer samples. RDH10 expression was higher in ovarian clear cell carcinoma than in endometrioid and serous carcinomas. Scale bars indicate 100 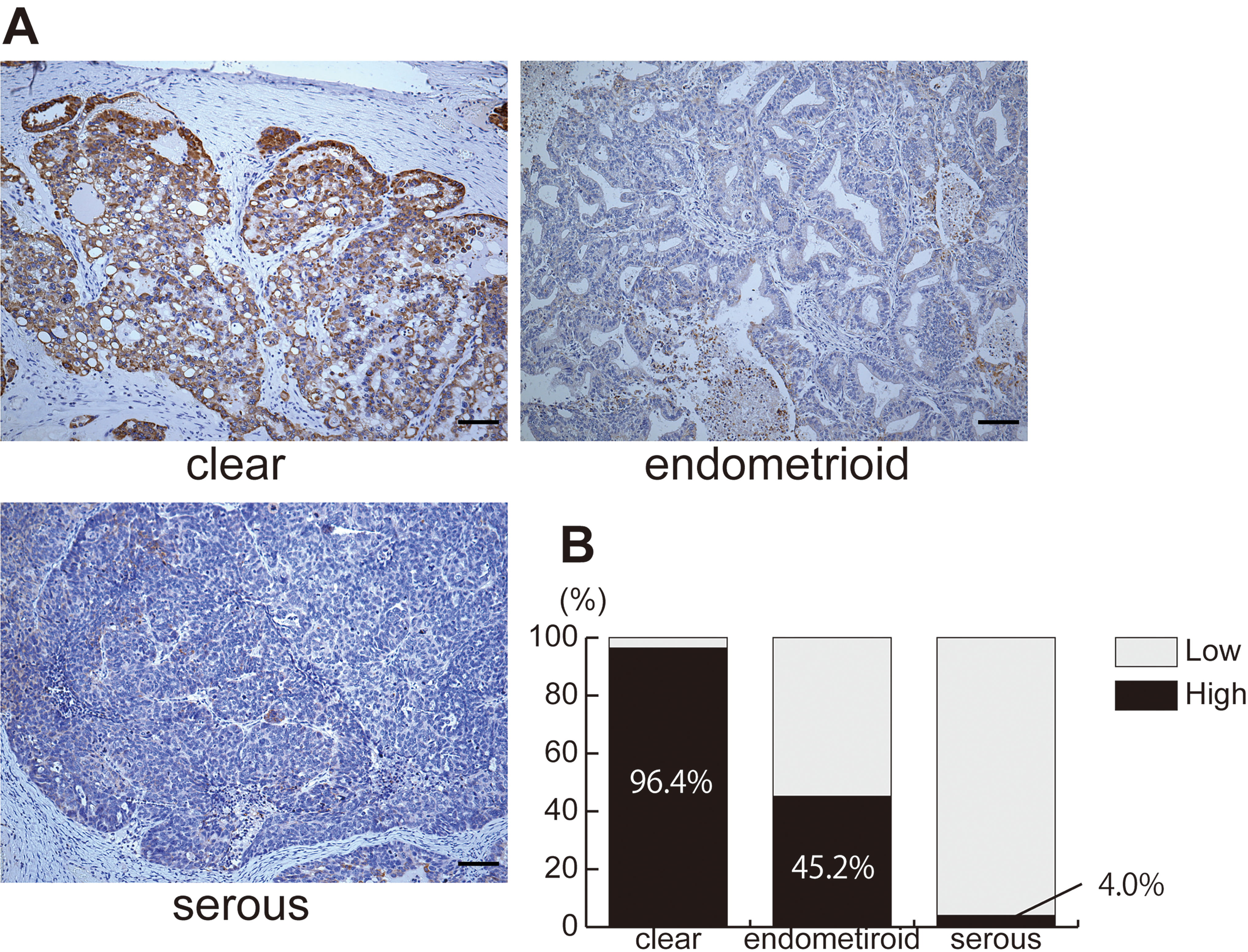
RDH10 affects the spherogenecity of ovarian clear cell carcinoma. A, The sphere-forming ability was enhanced and reduced by RDH10 overexpression and knockdown, respectively. Representative images are shown for ES-2 ovarian clear cell carcinoma cells after RDH10 (RDH10) overexpression, control (Cntl) overexpression parental cells with no modifications (par), control (shCntl), and RDH10 (shRDH) knockdowns. Scale bars correspond to 100 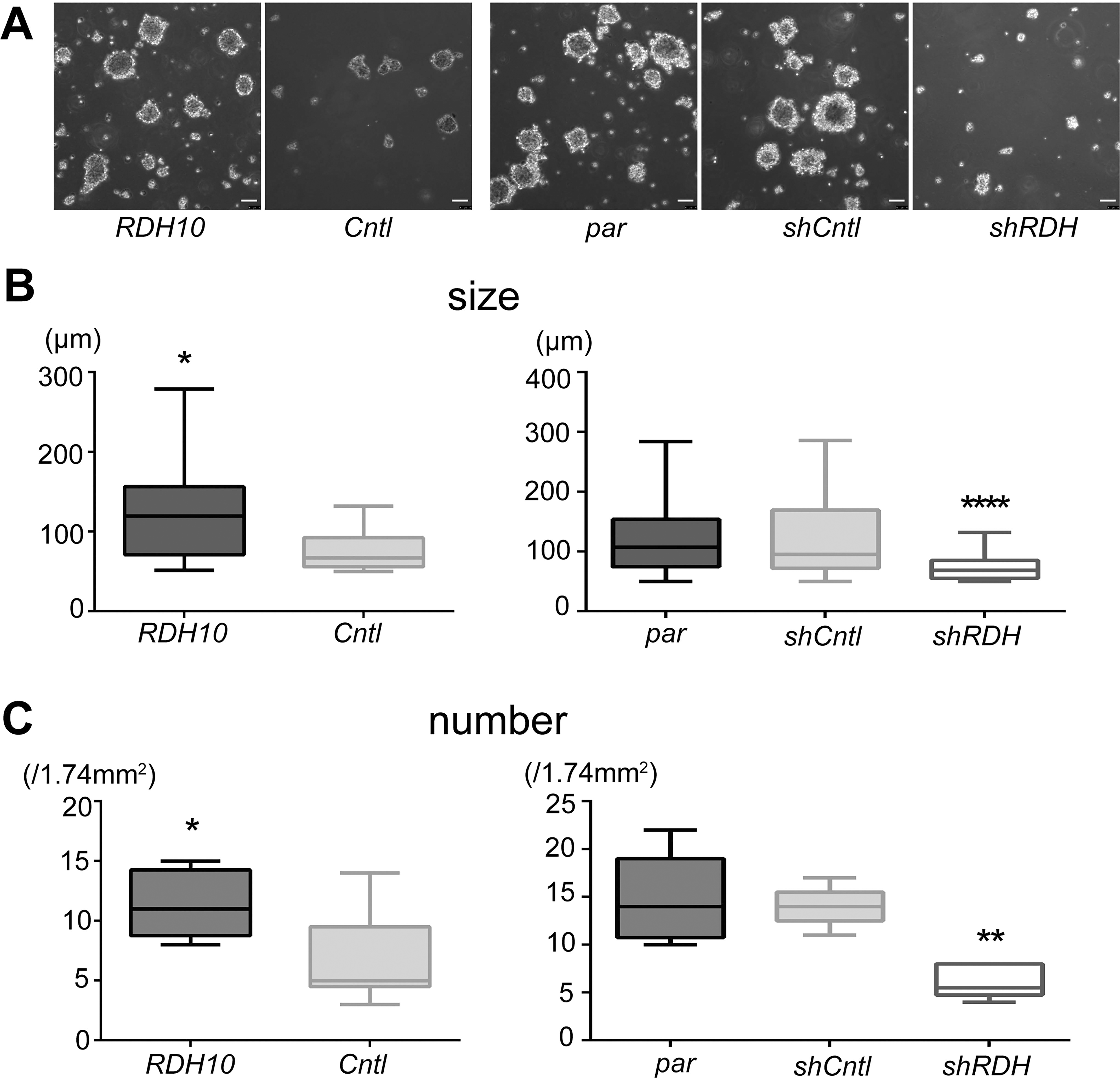
RDH10 affects PCK1 expression in ovarian clear cell carcinomas. A, In ES-2 cell lines (upper rows), RDH10 overexpression induced PCK1 expression, whereas RDH10 knockdown (shRDH) did not affect PCK1 expression, due to low PCK1 expression levels in parental cell lines. In TOV-21G cell lines (lower rows), PCK1 levels were reduced by RDH10 knockdown (shRDH). B, In a schematic diagram of the pathway, RDH10 promotes gluconeogenesis via PCK1 induction.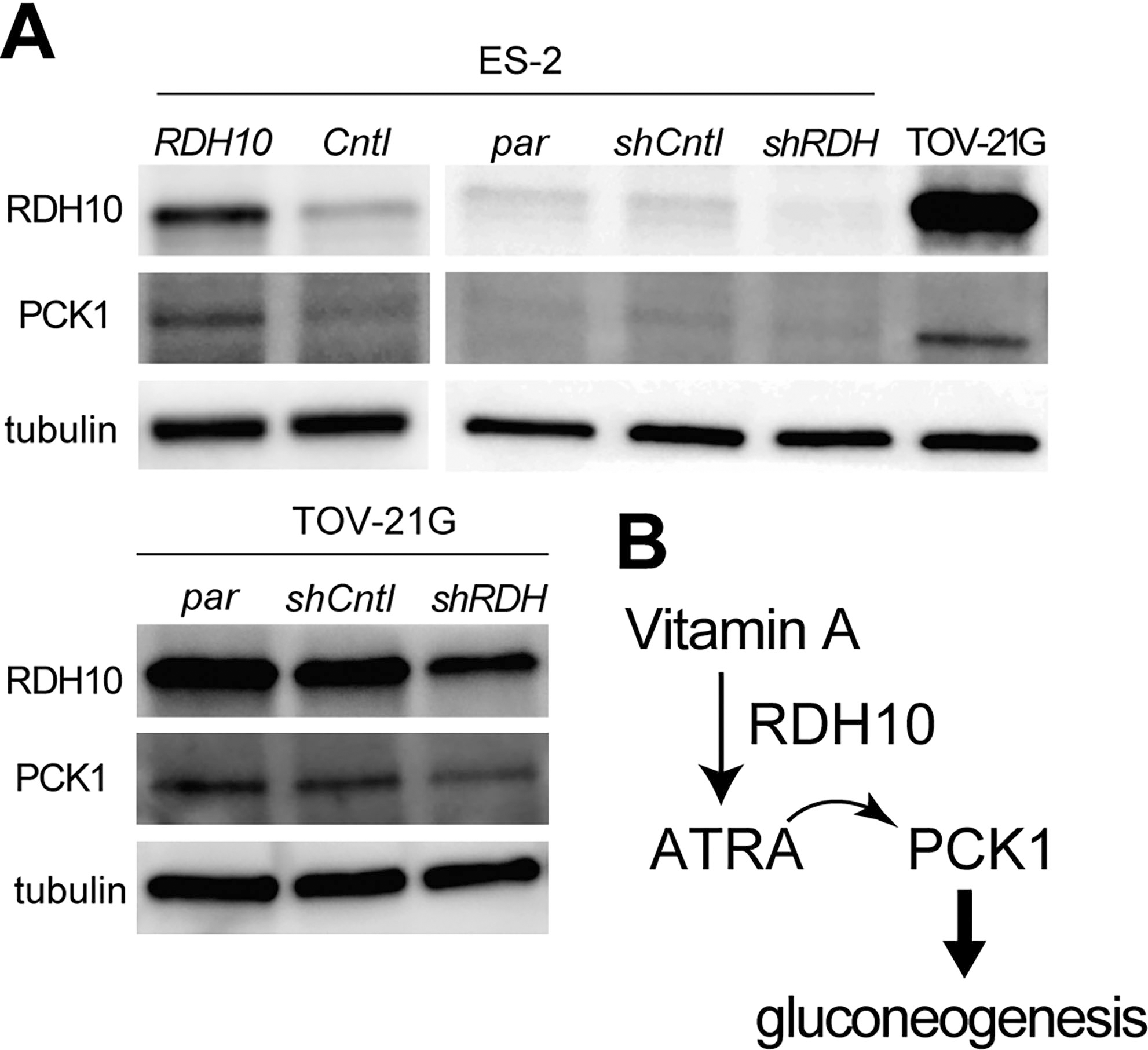
RDH10 promotes gluconeogenesis via RDH10-PCK1 pathway in ovarian clear cell carcinoma. A, The upper and lower rows show the original PAS-staining images and the extracted images with only red color, respectively, in ES-2 ovarian clear cell carcinomas. RDH10 overexpression (RDH10) induced red staining and knockdown (shRDH) reduced red staining. Scale bars correspond to 100 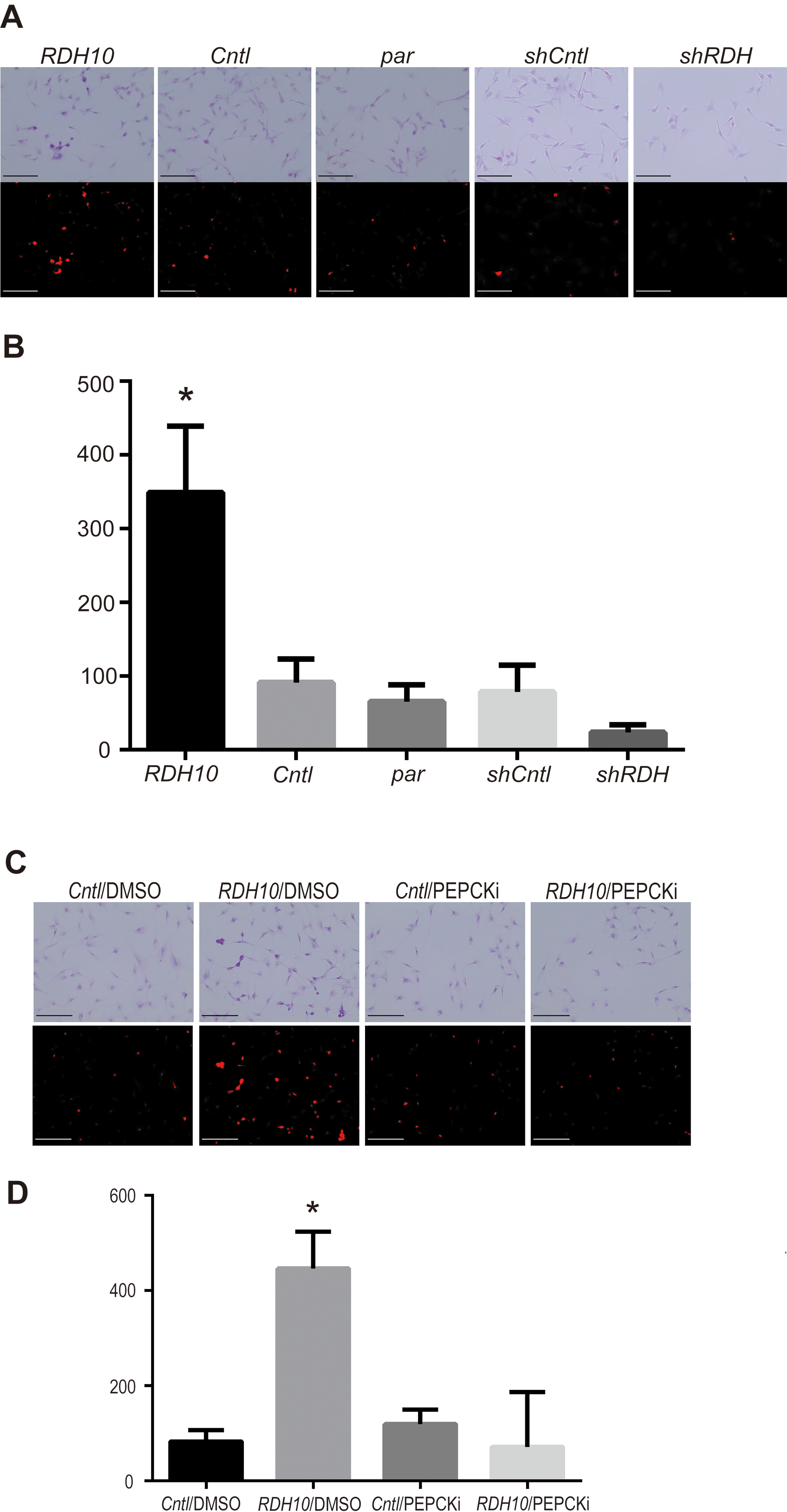
To investigate the role of RDH10 in the cancer stemness of OCCCs, the sphere-forming ability was evaluated in ES-2 OCCC cells after RDH10 overexpression or knockdown. As shown in Fig. 2, spherogenecity was promoted by RDH10 overexpression and was conversely inhibited by RDH10 knockdown (shRDH) in ES-2 cells (Fig. 2A). The diameter and number of spheres were significantly increased after RDH10 overexpression and decreased in shRDH variants of ES-2 (Fig. 2B and C). In TOV-21G cells, which express RDH10 at high levels, RDH10 knockdown significantly reduced sphere diameter and number (Supplementary Fig. 1).
RDH10 positively affects PCK1 expression in OCCCs
To analyze the vitamin A/ATRA and PCK1/gluconeogenesis signaling pathways, RDH10 and PCK1 protein levels were evaluated after modifying RDH10 expression in OCCC cells, as shown in Fig. 3. PCK1 was significantly induced by RDH10 overexpression and reduced after knockdown of RDH10 in ES-2 cells (Fig. 3A, upper rows). RDH10 knockdown also reduced PCK1 expression in TOV-21G (Fig. 3A, lower rows). ATRA, a vitamin A metabolite, promoted gluconeogenesis via PCK1 (Fig. 3B), as shown in a previous study [12].
RDH10 promotes gluconeogenesis and intracellularly carbohydrate storage via PCK1in OCCCs
The red-colored pixel images after PAS staining correspond to carbohydrate contents. To confirm that RDH10 promotes gluconeogenesis and intracellular carbohydrate storage, the red pixels were digitally analyzed and quantified in OCCC cells after RDH10 overexpression or knockdown, as shown in Fig. 4A and B. RDH10 overexpression significantly induced carbohydrate storage (Fig. 4B;
In order to analyze PCK1 effectiveness for intracellular carbohydrate storage via RDH10-PCK1 pathway, PCK1 inhibitor, PEPCKi, were applied into RDH10 overexpressed ES-2 cells. Under PEPCKi condition, carbohydrate storage was significantly reduced even in RDH10 introduction (Fig. 4D;
Discussion
The results of the present study demonstrate that RDH10 is abundant in OCCCs compared with other histological subtypes of EOCs. Furthermore, this retinol metabolizing enzyme plays important roles in OCCC stemness and intracellular carbohydrate storage.
Abundant RDH10 expression was confirmed specifically in OCCCs and RDH10 contributed to specific characteristics of OCCCs. OCCCs arise from the harsh environment of endometriosis and excessive oxidative stress is crucial to carcinogenesis in OCCCs. OCCCs exhibit molecular abnormalities associated with the oxidative stress response and reactive oxygen species (ROS) metabolism [9, 14]. Hepatocyte nuclear function factor 1 homeobox B (HNF1B) is overexpressed in OCCCs and promotes gluconeogenesis and cancer cell survival under hypoxic conditions [15, 16]. Likewise, mitochondrial superoxide dismutase 2 (SOD2) is overexpressed in OCCCs and contributes to oxidative stress tolerance by metabolizing ROS to maintain mitochondrial function [16, 17]. In addition, aldehyde dehydrogenase (ALDH), which metabolizes RA to ATRA and accelerates scavenging of ROS, is abundant in OCCCs [8]. These antioxidant molecules likely allow OCCCs to adapt to hypoxic environments. RDH10 may be induced as a response to oxidative stress, similar to HNF1B, SOD2, and ALDH.
RDH10 promoted spherogenecity in OCCC cells. The RXRB axis plays an essential role in cancer development and stemness in several types of malignancies [18, 19]. RDH10 is crucial for the induction of OCCC stemness following RXRB maintenance regardless of the RAB39A status [9]. RXRB is a member of the RXR family of nuclear receptors that mediates the effects of ATRA and promotes both DNA binding and transcriptional function [20]. In addition, retinol maintains CSCs [21] and RDH10 promotes progression and metastasis of human gliomas [10, 22]. In agreement with these previous studies, the present study shows that RDH10 plays an important role in cancer stemness in OCCCs.
RDH10 abundance activates retinol metabolism to produce ATRA and promotes carbohydrate storage via PCK1 induction in OCCC cells [12, 23]. Cancer cells accumulate carbohydrates as a source of energy to enable survival under adverse conditions, such as glucose deprivation and hypoxia, and to sustain cells during metastasis [24, 25]. PCK1 not only promotes carbohydrate storage, but also processes ROS under hypoxic conditions in OCCCs. Glycogen storage induced by PCK1 can generate abundant NADPH, via the pentose phosphate pathway, to ensure high levels of reduced glutathione. The high levels of reduced glutathione maintain the ROS processing capacity and contribute to the survival of cancer cells under hypoxic conditions [25, 26]. In human OCCCs, a PIK3CA mutation accelerates glycogen storage during hypoxia and the increased storage plays an important role in protecting cancer cells from anticancer agents, hypoxia, and nutrient deficiency [25, 27, 28, 29]. Together, abundant RDH10 followed by PCK1 induction and increased intracellular carbohydrate storage is favorable for OCCC cell survival under nutrient deprivation or hypoxic conditions. Thus, RDH10 is involved in the present therapeutic resistance and poor prognosis in patients with OCCCs. Targeting inhibition of RDH10 followed by carbohydrate reduction is a promising option of near-future therapeutic strategy for treating OCCC.
Conclusion
In conclusion, abundant RDH10 expression supports cancer stemness and intracellular carbohydrate storage via PCK1 in OCCCs. RDH10 contributes to therapeutic resistance and poor prognosis. In near future, therapeutic targeting RDH10-PCK1 pathway will reduce intracellular carbohydrate storage and cancer stemness and improve the prognosis in OCCCs
Author contributions
Conception: T.C.
Interpretation or analysis of data: A.M., T.A, F.Y., H.K., S.M, and T.C.
Preparation of the manuscript: A.M., and T.A.
Revision for important intellectual content: T.A., and T.C.
Supervision: T.M., and T.C.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-210435.
sj-pdf-1-cbm-10.3233_CBM-210435.pdf - Supplemental material
Supplemental material, sj-pdf-1-cbm-10.3233_CBM-210435.pdf
Footnotes
Acknowledgments
This study was partly supported by JSPS KAKENHI, Grant number JP19K07413 (S.M. and T.C.).
Conflict of interest
The authors have no conflicts of interest.
