Abstract
BACKGROUND:
Endometrioid ovarian carcinoma and clear cell ovarian carcinoma are both classified as endometriosis-associated ovarian cancer (EAOC). Despite the high rates of recurrence and mortality of EAOC, no prognostic biomarkers have been determined. ADP-ribosylation factor-like protein 4C (ARL4C) has been reported to be involved in various tumor progression processes, but its clinical significance for predicting prognosis in EAOC cases has never been studied.
OBJECTIVE:
The present study aimed to determine the clinical significance of ARL4C expression in EAOC prognosis.
METHODS:
ARL4C expression was semi-quantitatively evaluated via immunohistochemistry in 61 EAOC patients, and the correlations between ARL4C expression and clinicopathological data and survival were statistically analyzed.
RESULTS:
Thirty-six (59%) cases had high levels of ARL4C, which was related to worse 5-year overall survival (OS) (log-rank test,
CONCLUSIONS:
ARL4C is a biomarker for worse prognosis and a novel therapeutic target in EAOC.
Keywords
Introduction
ADP-ribosylation factor-like protein 4C (ARL4C) belongs to the family of small GTP-binding proteins (small G proteins) and is widely expressed in the human bodies including in ovarian follicular cells. ARL4C is involved in several physiologic processes, such as the transport of intracellular cholesterol [1]. The simultaneous activation of Wnt/
Epithelial ovarian cancer (EOC) is the fourth leading cause of cancer death among women in Western countries [5]. EOC is asymptomatic in the early period. Early detection of EOC is difficult; so many patients with EOC are diagnosed at progressively advanced stages and thus have poor prognoses. In addition, patients affected by EOC usually undergo satisfactory response to the initial surgical cytoreduction and chemotherapy, but considerable numbers of them have drug-resistant recurrence later in time, that is conceivably due to the ability of ovarian cancer drug-resistant cells to escape first-line chemotherapy. In difficulty, ovarian cancer drug-resistant cells and EOC progressions may be based on inherent resistance rather than acquired resistance for the therapeutics [6, 7]. New diagnostic approaches such as gene chips, proteomics, and immunohistochemistry have been gathering attention for early detection of EOC [8]. If we can detect EOC at an early stage, we will not only be able to treat it with better prognosis, but also to perform it under minimally invasive procedures such as laparoscopic and robot-assisted laparoscopic surgeries [9].
Among EOCs, the most common type is serous adenocarcinoma, but its incidence is lower in Asia than in Western countries [10]. In Japan, the incidence of EOC comprises 35.7% serous carcinoma, 24.8% clear cell carcinoma (CCC), 17.5% endometrioid ovarian carcinoma (EnOC), and 9.6% mucinous carcinoma [11]. Both CCC and EnOC are associated with endometriosis and are thus classified as endometriosis-associated ovarian cancers (EAOC). Despite the high recurrence and mortality rates of EAOC, prognostic biomarkers haven’t been well estimated yet [12].
Moreover, the association between ARL4C and EOC has not been investigated, and no study has evaluated the relationship between ARL4C expression and the clinical prognosis of EAOC. The present study aimed to clarify the significance of ARL4C as a prognostic biomarker and therapeutic target in EAOC.
Material and methods
Patients and tumor samples
Sixty-one patients with EAOC who were treated at the Department of Obstetrics and Gynecology of Shiga University of Medical Science Hospital between 1998 and 2017 were examined in this study. All patients underwent initial surgery, and staging was according to the International Federation of Gynecology and Obstetrics (FIGO) 2014 classification. The patients’ samples were selected from primary surgery specimens including normal ovarian stroma. Clinical and pathological data; age at diagnosis; FIGO stage; blood markers including serum carbohydrate antigen 19-9, serum carbohydrate antigen 125, or serum carcino-embryonic antigen, histological type; completion of treatment; and follow-up data were retrieved from the medical records. The primary endpoints for the 5-year observation period were progression-free survival (PFS) and overall survival (OS). PFS was defined as disease recurrence or progression and was calculated from the start of treatment until the last visit to the hospital, while OS was defined as cancer death and was calculated from the start of treatment until the last hospital visit. The secondary endpoints were not set up. The present study has been approved by the Shiga University of Medical Science Research Ethics Committee (reference number: 29-178), and all patients provided written informed consent.
Immunohistochemical analysis
Formalin-fixed paraffin embedded tissue was used for ARL4C immunohistochemistry. Samples were sliced into 5-
Samples of specimens with ARL4C expression on immunohistochemistry in endometriosis-associated ovarian cancers. Cases with high and low ARL4C expressions are demonstrated in A–C and D–F, respectively. Hematoxylin and eosin staining of both high and low cases ARL4C expressions are shown in A and D. High magnifications of each case are indicated in C and F with the scale bars corresponding to 100 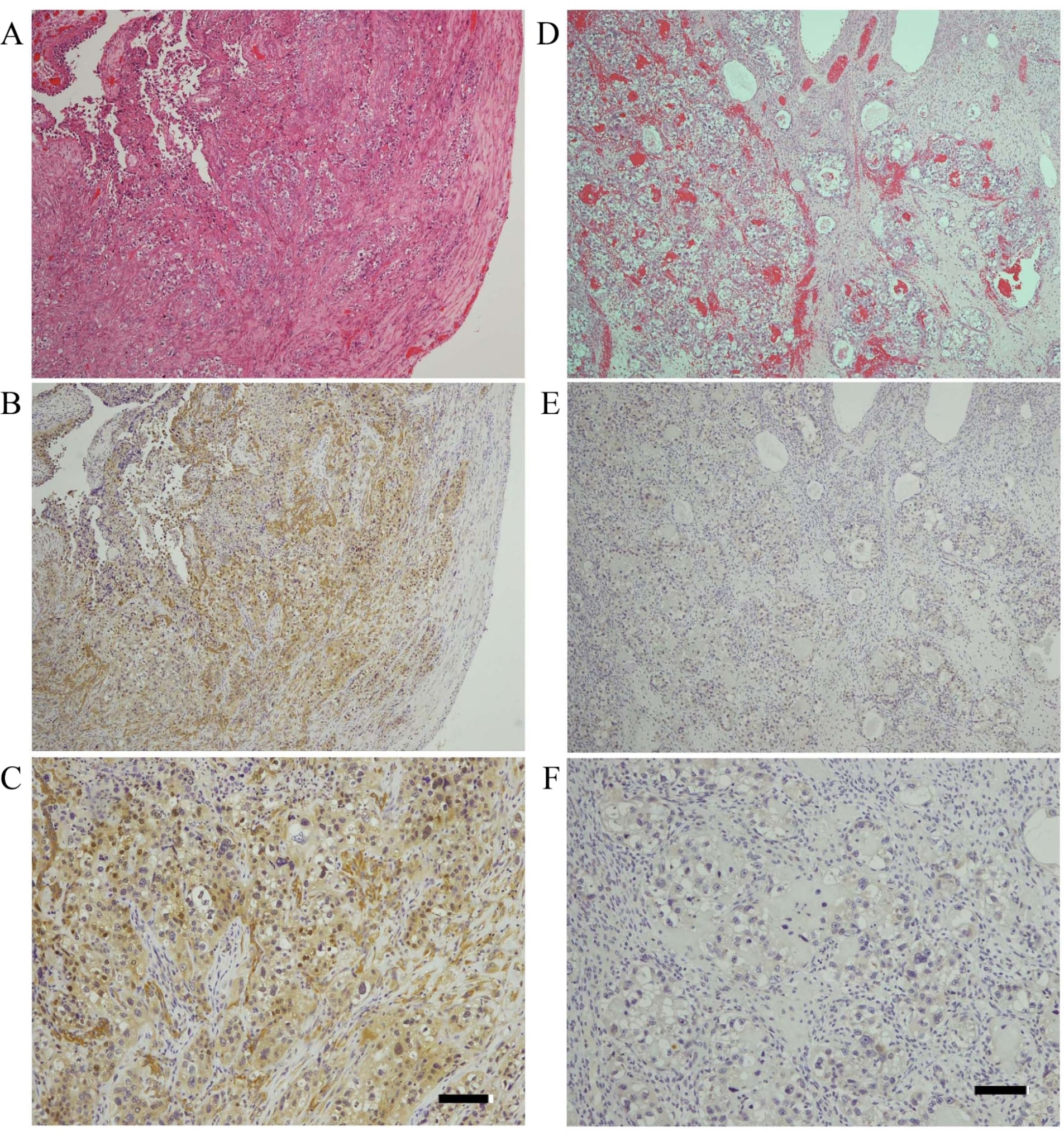
The chi-square test or Fisher’s exact test was used to evaluate the correlations between ARL4C expression and the clinicopathological characteristics of the patients. The Kaplan-Meier survival curves and the log-rank and chi-square test were used to evaluate differences in OS and PFS. Prognostic factors for OS and PFS were analyzed via univariate and multivariate analyses using Cox proportional hazards regression models. OS and PFS intervals were used as the indicator for the hazard ratio (HR) and 95% confidence interval (CI). The Kaplan-Meier analysis for OS and PFS were performed using GraphPad PRISM 7 software. All other statistical analyses were performed with SPSS statistical analysis software (version 22.0; SPSS Inc., Chicago, IL, USA).
Correlation between ARL4C expression and clinicopathological characteristics in endometriosis-associated ovarian cancers [
61]
Correlation between ARL4C expression and clinicopathological characteristics in endometriosis-associated ovarian cancers [
*Chi-square and Fisher’s exact tests.
Prognostic evaluation of clinicopathological factors affecting 5-year overall survival in endometriosis-associated ovarian cancers [
*Cox proportional hazards regression models. HR: hazard ratio; CI: confidence interval.
Prognostic evaluation of clinicopathological factors affecting 5-year progression-free survival in endometriosis-associated ovarian cancers [
*Cox proportional hazards regression models. HR: hazard ratio; CI: confidence interval.
Five-year overall (A) and progression-free (B) survivals of the groups categorized according to ARL4C expression in a cohort of endometriosis-associated ovarian cancers. High ARL4C expression was significantly associated with poor overall survival (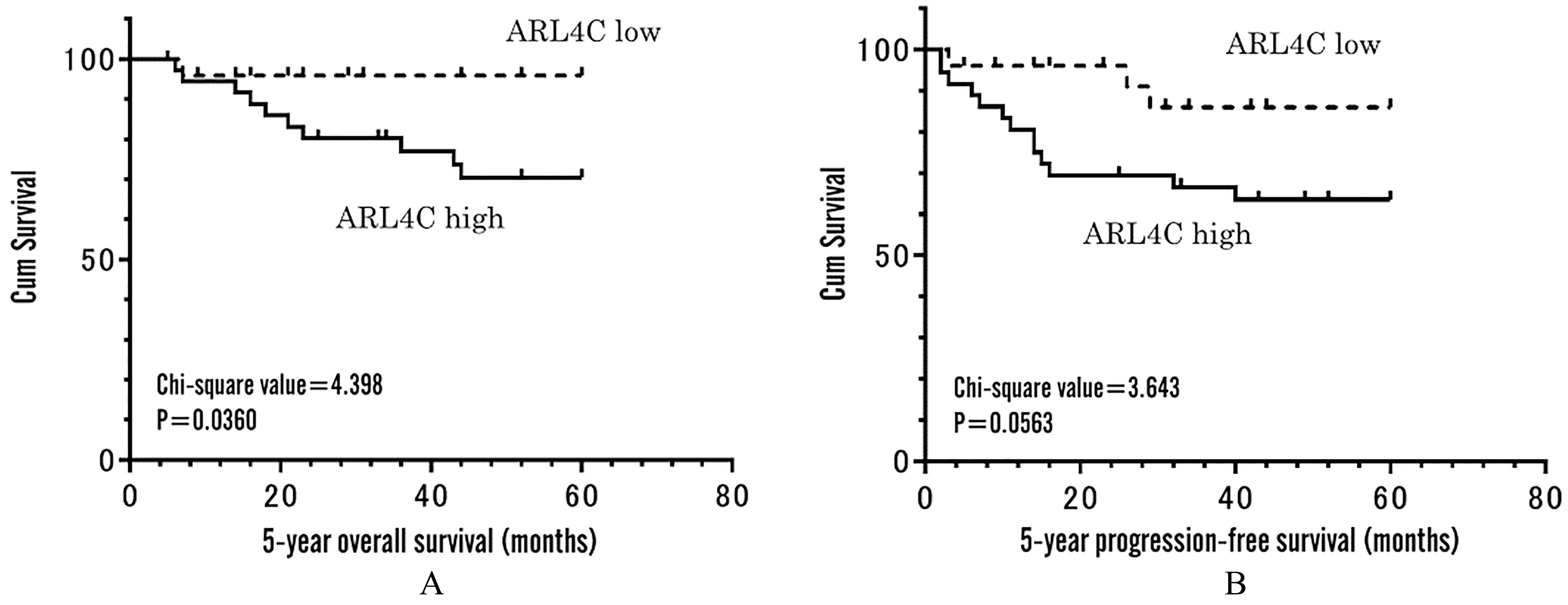
Patients’ characteristics
The median age of the patients at surgery was 48 years (range, 30–80 years). The median follow-up periods were 58 months (range, 1–156 months) for PFS and 65 months (range, 5–156 months) for OS, respectively. In terms of tumor stage, 51 and 10 patients had FIGO stages I–II (early stage) and stages III–IV (late stage), respectively. Histologically, 41 EAOC were clear cell and 20 were endometrioid carcinomas. Eight of the 61 patients did not receive platinum-based adjuvant chemotherapy due to patients’ preference or treatment complications. The other 53 patients completed standard treatment of platinum-based chemotherapy followed by maximal surgical debulking. Therefore, all patients were chemotherapy-naive at the time of surgery.
ARL4C expression in endometriosis-associated ovarian cancers and its relation to clinicopathological characteristics
ARL4C expression was semi-quantitatively evaluated in 61 primary EAOC tumors. The results of hematoxylin-eosin staining of the EAOC specimens and examples of stained sections of high and low ARL4C expressions are illustrated in Fig. 1A–F. Of the 61 patients with EAOC, 36 and 25 were categorized into the high (59.0%) and low (41.0%) ARL4C expression, respectively. The patients’ age (
ARL4C expression can predict poor prognosis in endometriosis-associated ovarian cancers
Kaplan-Meier analysis and log-rank test showed that the 5-year OS of the patients in the ARL4C high expression group indicated significantly worse survivals than that of the low expression group (chi-square value
In the univariate analysis of Cox proportional hazard regression model, higher FIGO classification, incomplete treatment, and high ARL4C expression were correlated with poor prognosis of both 5-year OS and 5-year PFS (Tables 2 and 3). To determine whether high ARL4C expression was an independent predictive factor for 5-year OS and 5-year PFS of EAOC patients, multivariate analysis was additionally performed using the variables of FIGO classification and treatment. The results showed that high ARL4C expression was a significant independent predictive factor for worse 5-year OS (HR
Discussion
This is the first study to demonstrate that ARL4C is highly expressed in EAOC. In a previous study, a high expression of ARL4C was noted in 55 of 117 cases (47.0%) of colorectal cancer and in 51 of 65 cases (78.5%) of lung adenocarcinoma [4]. These rates were similar to those found for EAOC in the current study, indicating that high ARL4C expression is also a useful biomarker for the clinical prediction of prognosis of EAOC similar to that in other carcinomas. Both univariate and multivariate analyses for clinicopathological variables showed that high ARL4C expression is an independent prognostic factor for worse 5-year OS and 5-year PFS in EAOC patients.
In EAOC, AT-rich interactive domain 1A (ARID1A), a tumor suppressor gene was frequently mutated and inactivated, and these atypical changes were considered as initial malignant transformations of endometriosis that may lead to ovarian cancer [13, 14, 15]. Early decrease in ARID1A expression has been occurred, especially in endometriotic tissue. Oxidative stress suppresses ARID1A expression in endometriotic cells; conversely, the low ARID1A activity occurring in endometriosis could increase the susceptibility of these lesions to malignant transformation [16, 17]. As with ARID1A repression, high ARL4C expression can be a valuable biomarker in EAOC.
Abundant ARL4C may contribute to infiltration, metastasis, and chemotherapeutic resistance in EAOC via molecular mechanisms such as epithelial-to-mesenchymal transition (EMT), and high ARL4C expression in EAOC has been related to poor prognosis in the present study. ARL4C activates ADP-ribosylation factor 6 (Arf6) through the Arf nucleotide-binding site opener [18]. Arf6 is involved in cancer cell invasion and metastasis, and it plays a key role in promoting EMT [19]. EMT is an important mechanism causing invasion and metastasis of epithelial tumors and is also related to radio- and chemo-resistance [20]. Moreover, EMT is a key factor that promotes malignancy and is associated with poor prognosis in EOC [21].
In a cohort of EAOC, both univariate and multivariate analyses based on Cox proportional hazard regression model indicated that ARL4C was a prognostic factor for 5-year OS and 5-year DFS. High ARL4C transcription was found to be a predictor of poor prognosis in colorectal and gastric cancers [22, 23]. Meanwhile, in contrast to our results, Su et al. [24] reported that high ARL4Cgene expression was related to favorable prognosis in ovarian cancers. The discrepancy between their study and our presented study may be caused by pathological differences of ovarian cancer types in each cohort. In a cohort of ovarian cancer cases analyzed by Su et al., 85 of the 212 patients had high-grade serous ovarian carcinoma, and the other 127 cases had non-serous carcinomas, whose classification was precisely unidentified. Their study verified that most cases of high-grade serous ovarian carcinomas originate from the fallopian tube [25]. Meanwhile, EAOC has different biological and pathological properties from serous ovarian carcinomas [25, 26]. Moreover, there are possibly analytical differences between gene and protein expressions. In the study of Su et al. [24], ARL4C expression was investigated via quantitative real-time polymerase chain reaction (qRT-PCR) using the cell lines of serous carcinoma type. As revealed in this study, ARL4C is also expressed in both normal cells of ovarian stroma and degenerative endometriosis, and a complex mixture of tumorous and normal tissue may cause differences in qRT-PCR results. Our analysis using stromal tissue as an internal control and assessing tumor tissue-specific expression offers a semi-quantitative advantage to evaluate the ARL4C expression of cancer cells.
The treatments of ovarian cancers remain challenging, particularly in the cases of EAOC, which is common among Asian populations including the Japanese. The standard treatment protocol for ovarian cancer is a multidisciplinary approach of surgery combined with chemotherapy that targets high-grade serous carcinomas. Despite the slow rate of progression, EAOC, particularly CCC, has high recurrence and mortality [27]. Many cases of advanced CCC are chemotherapy resistant and are frequently difficult to treat adequately; thus, novel therapeutic strategies are needed to improve treatment. In this regard, a therapeutic strategy targeting ARL4C seems feasible. In xenotransplanted tumor model derived from HCT116 cancer cells, the direct injection of ARL4C siRNA had a significant antitumor effect (
Drug repositioning targeting ARL4C may be a useful treatment strategy, and statins and bisphosphonates are likely candidates for ARL4C-targeted treatment. Activation of the small G proteins including ARL4C requires binding to cell membrane. As such, prenylation of G-proteins with farnesyl or geranylgeranyl is important [28]. Cys residues on the C-terminal prenylation are required for the binding of many small G proteins to the cell membrane. ARL4C also has Cys residues on the C-terminal, and its prenylation is essential for its activation [29]. The main synthetic pathway of isoprenoid intermediates such as farnesyl pyrophosphate and geranylgeranyl pyrophosphate is the mevalonate pathway [30]. Statins and bisphosphonates are strong candidates for inhibiting the mevalonate pathway and decreasing the downstream prenylated substrate. Statins inhibit 3-hydroxy-3-methylglutaryl coenzyme A reductase located upstream of the mevalonate synthesis pathway, and bisphosphonates inhibit farnesyl pyrophosphate synthase producing farnesyl pyrophosphate downstream of the mevalonate synthesis pathway. The antitumor effects of statins and bisphosphonates have been reported in cohort studies of ovarian cancers [31, 32, 33, 34, 35]. Both can inhibit the synthesis of isoprenoid intermediates involved in the prenylation of G-proteins including ARL4C, consequently inducing an antitumor effect. Statins are anti-dyslipidemic, and bisphosphonates are anti-osteoporotic drugs, and they are commonly and safely used worldwide. Thus, they are appropriate for therapeutic drug repositioning in EAOC treatment.
In conclusion, ARL4C is a biomarker for worse prognosis and a therapeutic target in EAOC. To the best of our knowledge, we are the first to report such results. Statins and bisphosphonates have a potential in ARL4C-targeted therapy in EAOC, but further studies are needed to verify its applicability.
Footnotes
Acknowledgments
The authors thank Takefumi Yamamoto and Hiroko Kita (Shiga University of Medical Science) for their assistance in the experiments. This work was supported by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan (Grant-in-Aid for Scientific Research (C) 18K09192).
Conflict of interest
None declared.
